Abstract
Chylous ascites may result from a variety of pathological conditions, most of them from nontraumatic causes, such as congenital defects of the lymphatic system, infections, liver cirrhosis, and malignancy. Rarely, chylous ascites occurs as an iatrogenic complication after left-sided laparoscopic donor nephrectomy (LDN). Injury to the cisterna chyli and its main lymphatic tributaries around the para-aortic region intraoperatively can cause the lymphatic fluid to accumulate. There is currently no standardized treatment for chylous ascites as there have only been 54 cases documented to date. Most patients can be managed with conservative therapy. Recommended guidelines include high-protein and low-fat diet with medium-chain triglycerides. Paracentesis is often used as a diagnostic and therapeutic first-line measure with total parenteral nutrition (TPN), bowel rest, and somatostatin analogue as adjunct therapies. We present a case of massive chylous ascites refractory to conservative therapy. The patient had progressive abdominal distention and unintentional weight gain 2 weeks postoperatively warranting multiple paracenteses of >7 L of chylous fluid. Ultimately, the patient was successfully treated with lymphatic embolization using N-butyl cyanoacrylate glue.
Introduction
Laparoscopic donor nephrectomy (LDN) for organ procurement has been the standard procedure for treating end-stage renal disease with living kidney transplantation. Since the first LDN performed by Ratner et al 1 in 1995, more donors are consenting to surgery as the benefits include shorter hospital stay and decreased postoperative pain.
Chylous ascites may result from a variety of pathological conditions, most of them from nontraumatic causes, such as congenital defects of the lymphatic system, infections, liver cirrhosis, and malignancy. Classically, the 2 most common iatrogenic causes of chylous ascites include abdominal aortic surgery and oncologic surgery requiring retroperitoneal lymph node dissection. 2 Disruption to the cisterna chyli and its main lymphatic tributaries around the para-aortic region intraoperatively leads to a collection of chylous fluid within the peritoneal cavity.3-5 Chylous ascites after LDN, first reported by Shafizadeh et al 6 in 2002, is a rare and problematic complication that can result in significant morbidity, prolonged hospital stays and thus increased health care costs. It appears that the incidence of chylous leakage/ascites after LDN is higher than previously reported, with the most recent data from Dale et al suggesting incidence ranges from 0.6% to 5.9%.5,7,8 This is a notable increase from a previous 2009 report by Aerts et al 9 who estimated incidence to range from 0% to 1.8%. Current literature review shows that to the best of our knowledge, only 54 cases have been documented, with most of them occurring after left, rather than right, LDN.7,10,11
Continuous chylous leakage can lead to malnutrition from protein loss, electrolyte abnormalities, and immunodeficiency from lymphocyte depletion.3,12,13 There is no standardized treatment for chylous ascites. Current treatment recommendations include conservative measures with dietary modifications and supplementation followed by medical, interventional radiology, and/or surgical treatment should chylous ascites fail to resolve with conservative management. We present a case of massive chylous ascites where conservative therapy failed and thus required radiological intervention with successful percutaneous embolization of lymphatic leak.
Case Report
Our donor was a 48-year-old caucasian male who underwent a left-hand-assisted laparoscopic donor nephrectomy (HALDN) in August 2019. His past medical history included childhood asthma, and he had no past surgical history. His immediate postoperative course was uncomplicated, and he was discharged home on postoperative day 3. He was seen in routine postoperative outpatient follow-up clinic 2 weeks later and reported slight increased abdominal distention with some discharge from his infraumbilical midline hand port incision site. Staples were removed from the superior aspect of the incision to allow drainage, and he was advised to perform daily dressing changes and started on oral antibiotics for suspicion of superficial skin infection (SSI). In clinic, we were not able to express any discharge from wound but we advised patient to send us a picture in case of more drainage. Almost 24 hours after clinic visit patient sent us a picture (Figure 1). At that time, chylous ascites was high on the list and we asked patient to start low*fat diet (<25 g/day).
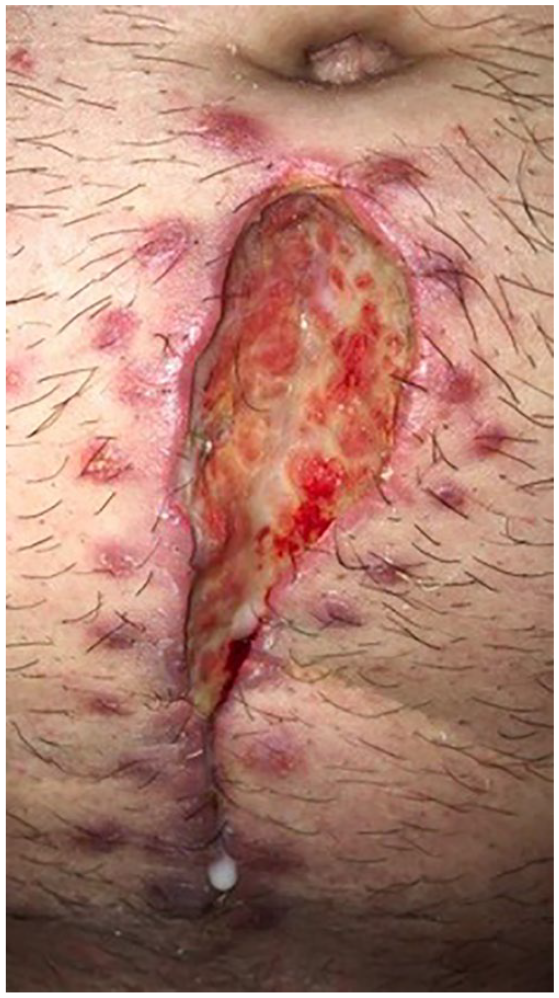
Milky white discharge (chyle) from the surgical wound.
On postoperative day 21, he presented to our institution with significant increasing abdominal distention, bilateral lower extremity edema extending up to the distal thighs, and shortness of breath prompting further investigation. A CT of the abdomen/pelvis demonstrated large-volume ascites (Figure 2). The patient underwent paracentesis with 9.2 L of milky fluid removed. Fluid analysis revealed 888 white blood cells with 84% lymphocytes and 838 mg/dL triglycerides, confirming diagnosis of chylous ascites. Fluid amylase, creatinine, total protein, and lactate dehydrogenase were normal. He had a normal fasting lipid profile with a very low vitamin D level. Ascitic fluid cytology was negative for malignancy. Abdominal ultrasonography with duplex showed normal liver and spleen size with normal hepatic and splenic vessels without portal vein dilatation. During his 10-day hospitalization at our institution, he had rapid re-accumulation of ascites requiring additional paracentesis with removal of 7 L of chylous fluid. During this time, he was also managed with high-protein, low-fat, medium-chain triglyceride diet in addition to octreotide 50 mg subcutaneously every 8 h and albumin replacement. To help localize site of lymph leak, lymphoscintigraphy was performed which demonstrated lymphatic activity reaching the lower right iliac and left common iliac lymph nodes, with obstruction noted below the level of the para-aortic lymph nodes (Figure 3). Due to rapid reaccumulation of ascites requiring intervention (paracentesis), we decided to go for radiological intervention. At this time, we discussed the case with interventional radiology team and decided to consult University of Pennsylvania interventional radiology lymphatic center for assistance in care of this patient.
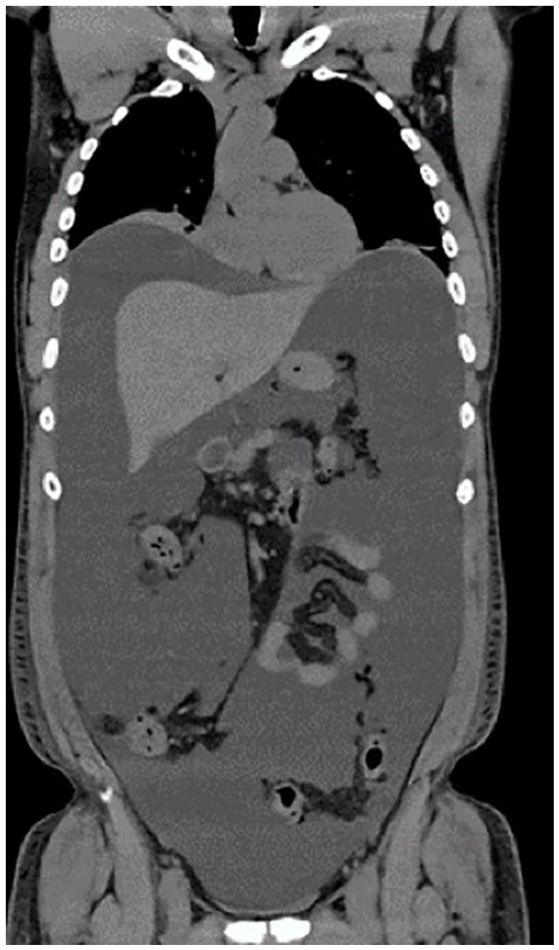
Computed tomography scan showing large-volume ascites.

Lymphoscintigraphy demonstrated lymphatic activity reaching the lower right iliac and left common iliac lymph nodes, with obstruction noted below the level of the para-aortic lymph nodes.
He continued to have reaccumulation of fluid, and at the outside institution, he underwent 2 additional paracenteses, each aspirating about 7 L of chylous fluid. On postoperative day 41, he underwent MRI lymphangiogram. Results showed contrast leakage around the left kidney resection site, normal retroperitoneal lymphatics, and thoracic duct. The following day, he underwent an intranodal lymphangiography with N-butyl cyanoacrylate embolization (N-BCA) of the left retroperitoneal chylous leak under ultrasound guidance. At this time, an abdominal drain was placed which was productive of high fluid output necessitating repeat lymphangiography with embolization 7 days later for persistent leakage. The second embolization was performed superior to the previous embolization site, which was successful in resolution of any active extravasation. Drain output diminished, and the drain was removed 3 days after the repeat embolization, on postoperative day 52.
The patient remained on high-protein, low-fat, medium-chain triglyceride diet for 8 weeks. His infraumbilical port site wound completely healed, post intervention (Figure 4). No recurrence or any additional complications were noted at his 3-month follow-up.

Patients wound healed after N-butyl cyanoacrylate embolization (N-BCA).
Discussion
Chylous ascites is a rare iatrogenic complication that most commonly occurs from disruption of the abdominal lymphatics after abdominal surgery. Paralumbar lymphatic trunks in the retroperitoneum transport most of the lymphatic fluid from the pelvis and lower extremities. These paralumbar trunks join the intestinal trunks and cisterna chyli near the renal hilum (L1-L2 vertebrae) alongside the aorta to drain into the thoracic duct cranially. 14 Injury to the retroperitoneal para-aortic lymphatics, thoracic duct, or cisterna chyli can lead to a lymphatic leak.5,12 Lymphatic dissection is inevitable along the lymphatic rich para-aortic region while dividing out the necessary length of renal vessels for a successful transplantation. 12 Different intraoperative techniques have been investigated to help control chylous leaks with mixed results. Malik et al 15 explored the use of LigaSureTM and tissue glue during nephrectomy to control chylous leak, yet there was no significant change in incidence. However, decreased incidence of chylous leak was observed with intraoperative application of surgical clips and use of energy sealing devices.15,16
Most nephrectomies are performed on the left side due to the longer renal vein. The shorter renal vein and liver retraction are some of the reasons that make right-sided LDN less preferable. 10 In a retrospective review by Seth et al 7 of complications from a single-center study of 253 patients undergoing LDN (51 right and 202 left) from 2003 to 2012, there were 15 noted cases of chylous ascites after left nephrectomy (7.4%) compared to 1 case after right nephrectomy (1.9%). Similar findings were noted in a large retrospective review in India by Kumar et al. 11 Other anatomic factors may also contribute to decreased chylous leak after right-sided LDN compared to left. The right renal artery is usually considerably longer than the left and therefore requires a less-extensive dissection with respect to near the aorta. 5 In addition, the right renal artery is positioned latero-caudal and posterior to the inferior vena cava, thus it is farther away from the para-aortic lymph nodes that are left undisrupted. 14 During donor evaluation, both kidneys are screened, and the kidney ultimately chosen for donation should be the one with either decreased function in GFR and/or morphological abnormality (cysts, stones, lipomas, and number of vessels) with the intent of minimizing donor morbidity.11,17
Patients commonly become symptomatic from iatrogenic chylous ascites in the second postoperative week. 7 Similar to our patient, chylous ascites will present with abdominal distention, weight gain, and dyspnea from the increased abdominal pressure. Patients may also complain of early satiety, anorexia, nausea, vomiting, edema, abdominal pain, malaise, and fever.13,18
During diagnostic workup, CT imaging can be difficult to distinguish chylous ascites from other causes of ascites because the CT density of chylous fluid is similar to that of urine, bile, and bowel secretions. 13 Nonetheless, it is useful in surveying fluid accumulations. Abdominal paracentesis is the diagnostic test of choice in confirming chylous ascites. The ascitic fluid will be milky, odorless, alkaline, and sterile.13,18 It should be analyzed for cell count, culture, gram stain, total protein, albumin, triglyceride levels, glucose, lactate dehydrogenase, amylase, and cytology to rule out other disease processes in the right clinical setting. 13 When sending ascites fluid for culture, at least 10 mL of fluid should be sent in blood culture bottles to increase sensitivity of the test. 19 Triglyceride levels will be greater >200 mg/dL. 13 Paracentesis is also therapeutic in providing symptomatic relief by removing the source of intra-abdominal pressure. Repeat paracentesis is not a long-term solution as it can prolong leakage, deplete nutritional levels, lower immunity, and increase the risk of infection. 18
After diagnosis is confirmed, different imaging modalities such as lymphoscintigraphy and lymphangiography can be used to detect area(s) of lymphatic leak. Ingesting milk or a high-fat diet prior to imaging promotes lymphatic flow, thereby increasing the chances of identifying the source of leak.2,7 Lymphoscintigraphy is a noninvasive technique with no known adverse side effects. However, facilities may not be equipped to routinely perform this test. Lymphangiography is considered the gold standard for detecting lymphatic leaks. 13 Conventional lymphangiography uses lipiodol, an ethiodized oil, to inject into the lymphatic vessels either bipedally or intranodal to determine the exact location of the leak and facilitate surgical intervention.13,18 A recent advancement in lymphangiography includes dynamic contrast enhanced MR Lymphangiogram (DCRML) which is performed using MR after intranodal injection of gadolinium contrast agents and also may have therapeutic benefit. 20 However, lymphangiography can be painful and can precipitate contrast emboli.
There is no standardized treatment for chylous ascites. Current treatment guidelines include conservative measures with dietary modifications and supplementation as first-line therapy. More than half of patients are managed conservatively with diet and paracentesis for symptomatic relief.3,7,13,21,22 Replacing albumin after large volume paracenteses (>5 L) is recommended as it can reduce mortality. 23 Patients who either fail conservative measures or have massive ascites requiring multiple large volume paracentesis, like the patient in this case, necessitate early intervention. 7 Dietary modifications include a high-protein and low-fat diet with medium-chain triglycerides to reduce the production of chyle.13,18 While long-chain fatty acids must travel through the lymphatics to enter into the venous system and consequently increase lymph flow, medium-chain triglycerides are absorbed directly into the portal venous system and bypass lymphatic glands.12,18 Thus a medium-chain fatty acid diet is recommended because it does not contribute to increased lymph flow. Total parenteral nutrition (TPN) and bowel rest to further reduce lymphatic flow are second-line conservative measures. Support for these measures lies in the fact that fasting baseline lymphatic flow through the thoracic ducts/cisterna chyli is typically less than 1 mL per minute which is dramatically increased to greater than 200 mL per minute after a fatty meal.18,24 Several authors recommend patients continue conservative therapy for 4 to 8 weeks.3,7,12 Octreotide, a somatostatin analogue, can be used as an adjunct to dietary modifications in reducing lymphatic flow. It decreases intestinal fat absorption by suppressing pancreatic secretions and lowers overall triglyceride levels in the thoracic duct.12,13,18,25
Patients refractory to conservative measures are now often managed with interventional radiology. Many authors suggest that early intervention is warranted in severe cases, such as the patient in this case with massive ascites requiring multiple large-volume paracentesis.21,26 Prolonging intervention in these severe cases for an extended trial of conservative therapy often can be debilitating to the patient and increase malnutrition and immunodeficiency. Sinha and Mamode 26 asserts that early intervention should be considered as opposed to conservative measures for severe cases, although almost all documented cases of severe chylous ascites are first managed conservatively. Conversely, Dale et al 5 believe that intra-abdominal drain placement should be considered as a first-line therapy in select patients for treatment of chylous ascites as a reasonable alternative to both conservative and surgical management. Interventional radiology is the next step if conservative management fails with surgical intervention as last resort. Surgical identification of leak area is extremely difficult. Various surgical techniques that have been successful for refractory cases include application of clips and/or fibrin glue sealants, coagulation, suture ligation, embolization, and peritoneo-venous shunting.7,20,21,22,24,26
Chylous ascites in our case was managed with intervention radiology embolization using n-BCA glue into the leakage site and surrounding lymph nodes which were identified using DCRML. A 2017 retrospective review by Nadolski et al 20 assessed the effectiveness of lymphangiography, DCRML, and lymphatic embolization in 31 patients presenting for diagnosis and treatment of refractory chylous ascites. Using a combination of conventional lymphangiography and DCRML, they were able to visualize lymphatic leak in 17/31(55%) patients. 20 Eleven of those 17 patients underwent lymphatic embolization with n-BCA glue, and 9/11 (82%) had reported resolution of chylous ascites. 20 Our patient’s outcome supports these findings in that embolization lead to ultimate resolution.
Conclusion
Laparoscopic donor nephrectomy has been a standard procedure and chylous ascites is a rare complication. We believe that management should be individualized and tailored to the severity of chylous leak and presentation. To best prevent chylous leak, consideration should be given to routinely clipping lymphatics in addition to cautery with energy devices to safely secure lymphatic channels. Early intervention with lymphatic embolization is warranted in refractory cases with significant re-accumulation of chylous fluid after diagnostic paracentesis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases.
Informed Consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
