Abstract
Chylothorax is a pleural effusion of >110 mg/dL of triglycerides with a milky appearance with transudative being rare. In this article, we present a case of transudative chylothorax with concurrent chylous ascites that is secondary to congestive heart failure (CHF). A 70-year-old male with CHF with ejection fraction of 10%, coronary artery disease status post coronary artery bypass graft, sleep apnea, chronic kidney disease stage 3, and chronic obstructive pulmonary disease presented with worsening abdominal distention, shortness of breath, and increased lower extremities edema. He denied any cough or fever but had orthopnea and paroxysmal nocturnal dyspnea. He requires monthly paracentesis with drainage of 5 to 9 L each time. On physical examination, he had crackles bilaterally with no wheezes or jugular venous distension. His cardiac examination was unremarkable. He did have abdominal distension with dullness to percussion and a positive fluid wave. There was +2 bilateral pitting edema of lower extremities. He had a diagnostic paracentesis where 9.2 L of cloudy milky fluid was drained and therapeutic thoracentesis where 1.1 L of milky fluid was drained. Pleural fluid for triglycerides was 280. His peritoneal fluid had triglycerides of 671 confirming chylous ascites. CHF can lead to chylous ascites due to the increased lymph production in the abdomen, which flows to the thoracic duct. Due to the stiffness at the lymphatic junction, there is high pressure for less flow. The diaphragm plays a role allowing the chylous ascites to be absorb into the thorax.
Background
Chyle is transported by the lymphatic system. In the intestines, fat, chylomicrons, and other digestive products are broken down to chyle. 1 When chyle leaks out from the lymphatic system into the pleural cavity, it is known as chylothorax. Chylothorax is a pleural effusion containing >110 mg/dL of triglycerides often with a milky appearance. According to Maldonado’s study, the majority of patients studied with chylothorax had a milky appearance to the pleural fluid; however, it can also be serous, serosanguinous, or bloody. 2 Pleural effusions are classified by the Light criteria into transudative and exudative. In the same study, the majority of chylothorax studied was exudative. 2 It is rare to classify chylothorax as transudative. Transudative chylothorax is secondary to cirrhosis, nephrosis, and heart failure. 3 The most common presenting symptom is often dyspnea. There are only a few cases of chylothorax that is secondary to congestive heart failure (CHF) and pulmonary hypertension even more rarely due to ischemic cardiomyopathy. Due to the composition of chylothorax, complications, if left untreated, are malnutrition, immunosuppression, and respiratory distress. 1 We now present a case of transudative chylothorax with concurrent chylous ascites that is secondary to CHF and pulmonary hypertension.
Case Presentation
A 71-year-old male with a medical history of CHF with an ejection fraction of 10%, pulmonary hypertension with right ventricular systolic pressure (RVSP) of 59, coronary artery disease status post coronary artery bypass graft (CABG) ×6 in 2013, sleep apnea, chronic kidney disease stage 3, and chronic obstructive pulmonary disease presented to the hospital with worsening abdominal distention, shortness of breath, and increased lower extremities edema within the last couple of days. He uses 3 L of oxygen at home with BiPAP (bilevel positive airway pressure) at night. The patient admitted to orthopnea and paroxysmal dyspnea. He notes adherence to a strict oral fluid intake and takes lasix 40 mg daily in addition to bumex 2 mg twice a day. He first developed ascites and pleural effusion in 2019 after he presented with worsening shortness of breath and bloating with abdominal ultrasound showing ascites. He underwent diagnostic paracentesis with drainage of straw-colored fluid. Computed tomography (CT) abdomen from June 2019 showed small pleural effusion and was not drained. On further review, he required monthly paracentesis for his ascites with drainage of straw colored or light green fluid of 5 to 9 L each time. With his recurrent ascites, he had at least 5 different surgeons performing his therapeutic paracentesis for varying record of either straw colored or cloudy fluid.
On physical examination, the positive findings noted were normal respiratory effort with crackles noted bilaterally with diminished breath sounds in the right base and jugular venous distension noted. His cardiac examination was unremarkable. He did have abdominal distension with dullness to percussion with a positive fluid wave. There was also +2 pitting edema to bilateral lower extremities. Vitals were stable. Laboratory tests done on admission was unremarkable with blood urea nitrogen of 41 mg/dL and creatinine of 1.59 mg/dL, which is his baseline. His serum lactate dehydrogenase was 152 IU/L, albumin was 3.0 g/dL, and total protein of 7.2 g/dL. His chest X-ray showed moderate pleural effusion with atelectasis or infiltrate in the right lower lobe. He then had a chest CT that showed moderate right pleural effusion with compression atelectasis, no infiltrate, and moderate ascites. Review of his abdominal CT scan done in December 2019 had shown mild hepatomegaly with possible hepatocellular disease, which could be secondary to congestive hepatomegaly. There were no masses seen in either the chest or abdomen CT. Bedside ultrasound of the lung showed moderate pleural effusion.
For this visit, he had a diagnostic paracentesis in which 9.2 L of cloudy milky fluid was drained. His peritoneal fluid analysis workup as seen in Table 2 was unremarkable with negative culture. His serum ascites albumin gradient was 1.6 g/dL. However, he does not have a history of any cirrhosis. His peritoneal fluid sent for flow cytometry was negative for any B- or T-cell lymphoma. He had a trial of intravenous laxis, but the next day, he still had a moderate-sized pleural effusion as seen on ultrasound. He then elected for a therapeutic thoracentesis in which 1.1 L of milky fluid was drained. Due to the milky appearance of the fluid, the decision was made to test the pleural fluid for triglycerides, which came back as 280 mg/dL and cholesterol of 140 mg/dL. His pleural fluid analysis as seen in Table 1 had shown a total protein of 2.8 g/dL, lactate dehydrogenase (LDH) of 82 IU/L, and lymphocytic predominance. Based on the Light criteria, it was a transudative pleural effusion. After thoracentesis and paracentesis, his dyspnea improved to where he was discharged home.
Characteristics of Pleural Fluid Drained.

Abbreviations: WBC, white blood cell; RBC, red blood cell; LDH, lactate dehydrogenase.
Characteristics of Peritoneal Fluid Drained.
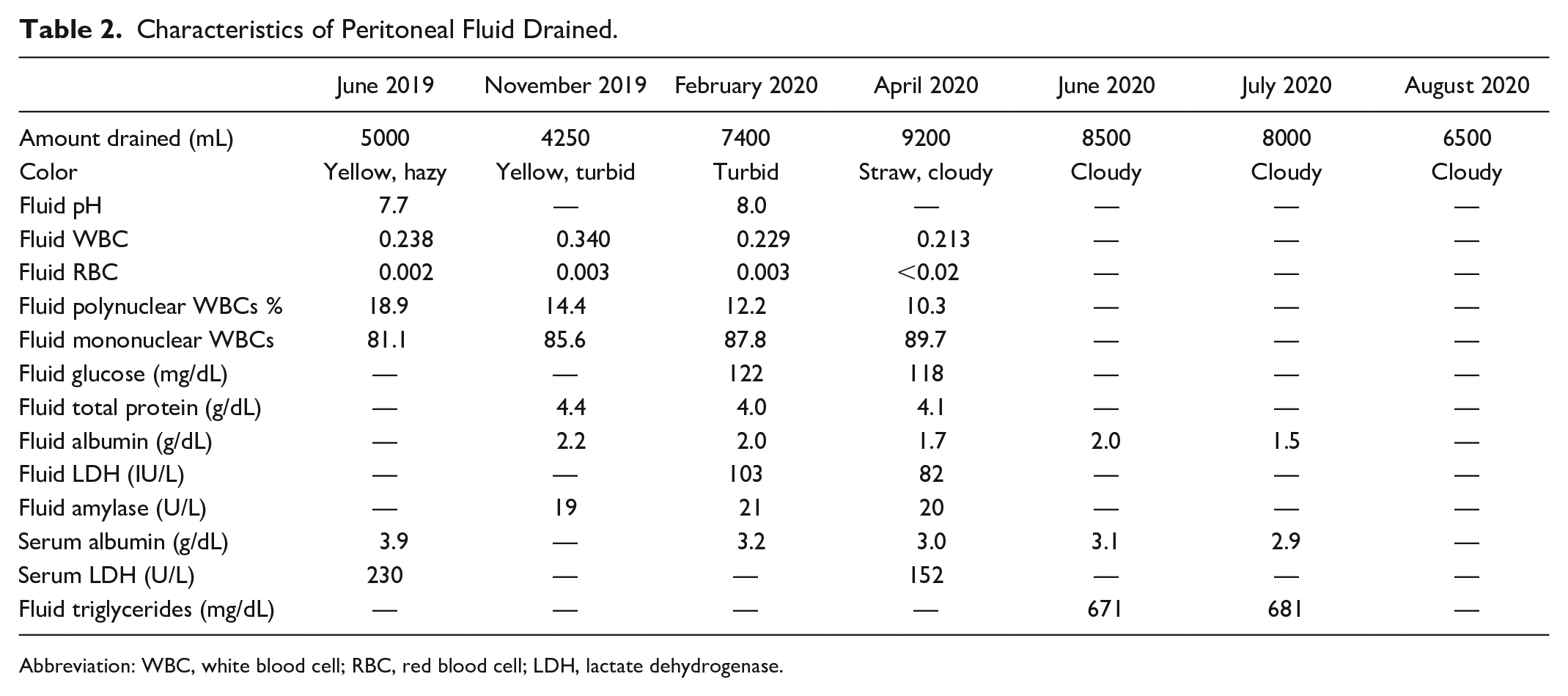
Abbreviation: WBC, white blood cell; RBC, red blood cell; LDH, lactate dehydrogenase.
After his hospitalization, he had further workup for chylothorax. He did need another paracentesis 2 months later in which the peritoneal fluid was tested for triglycerides, which was 671 mg/dL confirming chylous ascites. He had a lipid panel done which showed triglycerides of 125, total cholesterol of 143, low-density lipoprotein of 97, and high-density lipoprotein of 21. A liver ultrasound showed no focal abnormality with no dilated ductal structures visible. Repeat echocardiogram was obtained which showed a severely dilated left ventricle with an ejection fraction <15% with severely reduced left ventricular systolic function. There was also severe global hypokinesis of the left ventricle. Right ventricle (RV) was moderately dilated with severely reduced function with TAPSE of 9.7 mm and RVSP of 55 indicative of moderate pulmonary hypertension.
He was recommended to adapt to a medium chain triglyceride diet in which our dietitian here educated him on. Unfortunately, no centers close to us have lymphoscintigraphy available. We did find a center that was able to do the test; however, the patient has no means of transportation to get that far, so he had elected to not pursue this option at this time. Liver cirrhosis was ruled out, and due to the low percentage rate of thoracic surgery causing chylothorax and with CABG in 2013 and his ascites and pleural effusion started in 2019, we determined that his biventricular failure was likely contributing to his chylous ascites and chylothorax. He is currently on conservative management with monthly paracentesis, medium chain triglyceride diet, lasix, and bumex for fluid diuresis. Even though his blood pressure, which stays on the low side and history of chronic kidney disease, is limiting his diuresis management, he is maintaining an overall negative balance daily.
Discussion
Our patient did have a history of ascites with small pleural effusions since 2019. However, he was being treated for CHF exacerbations with diuresis improving his dyspnea and regular paracentesis. He noted improvement with his dyspnea after paracentesis. In April 2020, after paracentesis, his dyspnea still was not improved, and he had moderate pleural effusion on bedside ultrasound electing for therapeutic thoracentesis.
Based on the Light criteria, a pleural effusion can be labeled as exudative if it meets at least one of the following: pleural fluid protein/serum protein level >0.5, pleural fluid LDH/serum LDH level >0.6, or pleural fluid LDH level more than two thirds of upper limit of normal serum LDH value. If none is met, it is classified as transudative. In our patient, his pleural fluid was transudative due to him not meeting any criteria since his pleural protein to serum protein was 0.39 and pleural fluid LDH to serum LDH was 0.54. With his triglyceride level being 280 mg/dL, chylothorax is his diagnosis. Based on the study by Maldonado et al, 2 transudative chylothorax is rare and has been reported between 15% and 32%.
Transudative chylothorax most likely is caused by cirrhosis, nephrosis, and heart failure. Two cases of transudative chylothorax are attributed to pulmonary hypertension 4 in the past. Our patient is a case of transudative chylothorax due to biventricular failure (ejection fraction <15% with moderate pulmonary hypertension and severely reduced RV function).
Chylothorax is the accumulation of triglycerides within the pleural space due to chylomicron. Chylomicrons in addition to fat and other digestive products are broken down within the intestines and then transported as lymphatic fluid by the thoracic duct to the venous system. Of note, fatty acids that are a short chain of just 10 carbon atoms can be absorbed through the portal venous directly allowing the chyle leak to heal. 1 Due to this nature, our patient was started on medium chain triglycerides and high protein diet. He is responding well to the diet although he is still requiring monthly paracentesis. However, he has not needed any further thoracentesis since his dyspnea improves after paracentesis. Long-standing chylothorax that is not managed well can lead to respiratory distress, malnutrition, and immunosuppression due to the loss of nutrition that is found with chyle. 3
The majority of chylothorax is due to a disruption of the thoracic duct secondary to trauma, especially with any thoracic surgery.1,5 Our patient did have a history of CABG ×6. In Doerr’s study, they linked 4 cases to CABG making it only a 0.5% risk. 5 However, due to the long distance of a facility with the capability to do lymphoscintigraphy, we cannot rule out a thoracic duct injury as a factor, but due to the low rate of occurrence, it is highly unlikely since it has been years since his CABG before the onset of his ascites. This study only recommends lymphoscintigraphy if the patient is considering thoracic duct ligation. 3 At this time, the patient is electing conservative management with monthly paracentesis and diuresis. Since he does not want to pursue any surgical intervention, lymphoscintigraphy is not urgently needed.
Chylous ascites is associated with heart failure in only 3% of cases with triglycerides >200 mg/dL in ascitic fluid. 6 Based on his paracentesis, his triglyceride level was 671 mg/dL, confirming chylous ascites. It is unknown as to when he first developed chylous ascites due to him seeing 6 different surgeons for therapeutic paracentesis. In June 2019, it was documented as straw-colored fluid. Then in September 2019, it was light green colored fluid. Cloudy fluid was then documented in October 2019. After he was diagnosed with chylorthorax with pulmonology recommendation, his peritoneal fluid was sent for a triglyceride level. Biventricular heart failure can lead to chylous ascites secondary to increased abdominal lymph production which flows to the thoracic duct; however, flow from the neck area have stiffness at the lymphatic junction making the flow less pressurized. It is noted that the left subclavian has a high pressure, which contributes to reduced lymphatic drainage. 6 Due to the reduced lymphatic flow, the fluid will leak outside of the lymphatic system and into the open space, which is often the peritoneal and thoracic cavity. This is likely what is happening with our patient. His dyspnea is related to his abdominal distension, which improves after paracentesis. As seen in a case of cirrhosis, the chylous ascites can absorb through the diaphragm crossing into the thorax. 6 Through this mechanism, with our patient having moderate ascites, the chylous ascites is absorbed through the diaphragm for his chylothorax presentation. He is responding well to paracentesis with no further need for thoracentesis at this time.
In summary, we presented a case of transudative chylothorax and chylous ascites secondary to bi-ventricular heart failure (low ejection fraction and moderate pulmonary hypertension with severely reduced RV function). Transudative chylothorax is rare.
In this case, we could not entirely rule out a thoracic duct injury causing the recurrence secondary to the patient not willing to travel a long distance to get the lymphoscintigraphy done. At this time, the patient elects for conservative management with monthly paracentesis, strict adherence to medium chain triglycerides and high protein diet, and negative fluid balance with lasix and bumex. In conclusion, transudative chylothorax is rare and would need further workup to evaluate the cause of effusion.
Footnotes
Authors’ Note
This case was presented as an abstract at the Southern Regional Meeting, virtual, on February 25, 2021.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Informed consent for patient information to be published in this article was not obtained because patient had expired.
