Abstract
Aortic dissection (AD) is a life-threatening emergency involving a tear in the aortic intima, leading to a false lumen. Atrial fibrillation (AF) can complicate AD, increasing management challenges and mortality risks. We report a 67-year-old male with no known past medical history who presented with a 1-day history of abdominal pain. Initial examination showed mild hypertension, elevated bilirubin level, and leukocytosis. Imaging studies indicated gallbladder distension with cholelithiasis. Discharged with outpatient follow-up for elective cholecystectomy, the patient returned 2 days later for preoperative clearance, was hypertensive and tachycardic, presented with atrial flutter, and was referred to the emergency department. A 2D echo showed left ventricular ejection fraction (LVEF) 35% to 40% and pericardial effusion. Transesophageal echocardiography (TEE) revealed LVEF 50% to 55% and no thrombus, converting to sinus rhythm postcardioversion. The TEE also suggested type A AD, confirmed by computed tomography (CT) angiography, showing dissection from the aortic valve to the left external iliac artery with pericardial effusion. Emergent surgical repair was performed, and the patient was stabilized and discharged with follow-up. This case illustrates the complexity of diagnosing and managing AD, especially with concurrent AF. Recognizing the association between AD and AF is essential as AF increases in-hospital mortality in AD patients.
Keywords
Introduction
Aortic dissection (AD) is a life-threatening emergency occurring when a tear in the aortic intima allows blood to enter the media layer, creating a false lumen leading to its separation from the adventitia. In 1970, a group of surgeons at Stanford University proposed the Stanford Classification system for AD, which consists of 2 principal types defined by the origin of the false lumen: type A, which affects the ascending aorta, and type B, affecting the aorta distal to the left subclavian artery. 1 If the dissection involves the aortic arch without affecting the ascending aorta, it is classified as non-A non-B AD. 2 Patients typically present with acute tearing chest pain radiating to the back or abdomen. 2
The incidence of AD in North America is 4.4 per 100 000 per year, whereas the worldwide incidence is 4.8 per 100 000 per year, with type A at 3 per 100 000 per year and type B at 1.6 per 100 000 per year. The global mortality rate is quite high, with an overall mortality of 51.1% for all ADs, 43.2% for type A, and 26.4% for type B. 3 However, a new classification model proposed by Sievers et al supplements the Stanford Classification. Known as the TEM Classification, it classifies the dissection based on its entry point in conjunction with the Stanford Classification, denoting the Type, Entry, and Malperfusion associated with the dissection. This model introduces the characteristic of entry denoted by E0, E1, E2, and E3; where E0 is a dissection with an unclear entry which can also be referred to as an intramural hematoma, E1 for entry in the ascending aorta, E2 for entry within the aortic arch, and E3 for entry in the descending aorta. 4
Aortic dissection can present with various electrocardiogram (EKG) changes, laboratory abnormalities, and diagnostic challenges. The EKG findings in AD are often nonspecific but can include ST-segment changes, such as elevation or depression, which may mimic acute coronary syndrome. Patients can also have sinus tachycardia, third degree atrioventricular block and atrial fibrillation (AF). Laboratory tests may reveal elevated D-dimer levels, which, while nonspecific, can be a useful screening tool when combined with clinical suspicion and imaging findings. The gold standard for diagnosing AD is imaging, with computed tomography angiography (CTA) being the preferred modality due to its high sensitivity and specificity. Transesophageal echocardiography (TEE) is also highly effective, especially in hemodynamically unstable patients or when rapid bedside diagnosis is needed. Magnetic resonance angiography (MRA) offers excellent visualization without radiation exposure but is less commonly used due to longer acquisition times and limited availability. 5
Atrial fibrillation, as an initial presentation of AD, has been reported and studied. Hu et al reported a bidirectional association between AF and AD with a 1.32 times incidence rate of AF in AD patients. 6 Similarly, Koracevic et al 7 reported AF in 17.4% of patients with AD. The data relationship between AD and atrial flutter are scarce. Here, we present the first case of type A AD presenting as atrial flutter.
Case Presentation
We present a case of a 67-year-old male patient with no known past medical history who presented to the emergency department complaining of abdominal pain of 1-day duration. Upon initial presentation, the patient was mildly hypertensive with a blood pressure (BP) of 134/76 mm Hg, a heart rate (HR) of 76 beats per minute (bpm), afebrile, and saturating 98% on room air. On physical exam, the abdomen was mildly distended with epigastric tenderness but without rebound, rigidity, or guarding. Laboratory results were significant for an elevated total bilirubin of 1.7 mg/dL (reference range: <1.1 mg/dL) and leukocytosis of 11.7 × 103/mm³ (reference range <11 × 103/mm³). The initial EKG showed a normal sinus rhythm with a ventricular rate of approximately 118 bpm, without abnormal ST or T wave changes. A CT scan of the abdomen and pelvis with contrast showed gallbladder distension with cholelithiasis and possible trace gallbladder wall or pericholecystic edema at the interface between the gallbladder and the liver. A right upper quadrant ultrasound revealed sludge/stones versus polyps in the gallbladder lumen, which was only partially visualized. Surgery was consulted, and the patient was cleared for discharge after symptomatic control, with a recommendation for outpatient follow-up for elective cholecystectomy.
Two days later, the patient presented to his primary medical doctor for medical clearance for elective cholecystectomy. During this visit, the patient was found to be in atrial flutter and was referred to the emergency department. Upon presentation to the ED, the patient was still complaining of epigastric abdominal pain associated with decreased oral intake. The patient was hypertensive with a BP of 156/91 mm Hg, tachycardic with an HR of approximately 157 bpm, afebrile, and saturating 99% on room air. Laboratory tests showed mildly elevated troponin level of 39 pg/mL (reference range = 3-23 pg/mL), mildly elevated brain natriuretic peptide of 111 pg/mL (reference range = <100 pg/mL), an elevated total bilirubin of 3.3 mg/dL, and leukocytosis of 12.2 × 103/mm³. The EKG showed typical 2:1 atrial flutter with an atrial rate of around 300 bpm and a ventricular rate of around 150 bpm (Figure 1). The patient was given diltiazem 20 mg IV push and started on a diltiazem drip. Despite these interventions, the patient’s HR was still not controlled. The patient was transferred to the Cardiac Care Unit. The 2D echo showed a left ventricular ejection fraction (LVEF) of 35% to 40% and a small pericardial effusion. Transesophageal echocardiography showed an LVEF of 50% to 55% with no left atrial or left atrial appendage thrombus, so cardioversion at 200 joules was performed, converting the rhythm to sinus. During TEE, a type A AD was suspected, so the heparin infusion was held (Figure 2). The CT angiogram of chest abdomen pelvis confirmed Stanford type A AD extending from the aortic valve region to the left external iliac artery, associated with a pericardial effusion (Figure 3).

EKG showing typical 2:1 atrial flutter with an atrial rate of around 300 bpm and a ventricular rate of around 150 bpm.
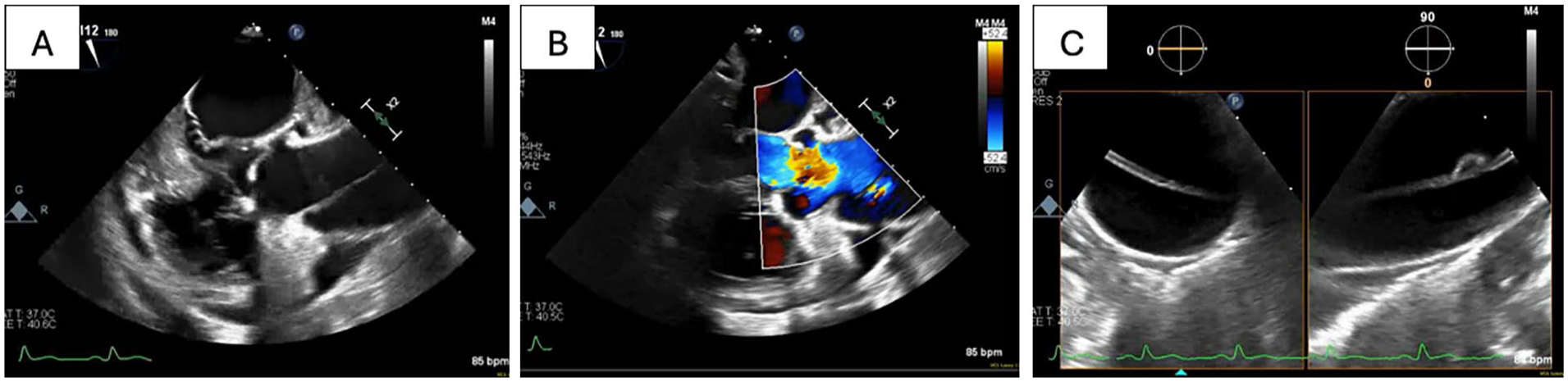
TEE (A) mid esophageal long axis. (B) Mid esophageal long axis with color Doppler showing ascending aorta dissection. (C) Biplane descending aorta dissection.
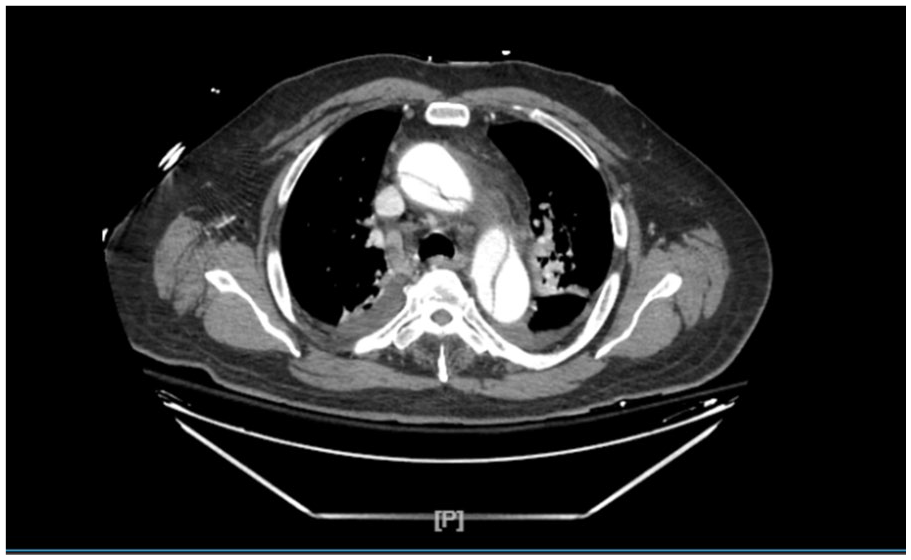
CT angiogram of chest abdomen pelvis showing Stanford type A aortic dissection extending from the aortic valve region to the left external iliac artery.
The patient was prepped for emergent repair of the type A AD under deep hypothermic circulatory arrest with aortic valve resuspension and replacement of the ascending aorta. Postoperatively, the patient was transferred to the surgical intensive care unit, then downgraded to the medical floor once he was deemed stable, and subsequently discharged home with outpatient follow-up. Postsurgical repeat echocardiogram showed LVEF of 55% to 60%, mild aortic root dilatation. Our patient’s atrial flutter was deemed to right coronary artery involvement secondary to AD, which did not re-occur after the surgery. On cardiology and cardiothoracic surgery follow-up, the patient remained asymptomatic.
Discussion
Incidence of AD is a complication of risk factors such as hypertension, genetic conditions such as Marfan syndrome, drug use, and increased aortic diameter.8,9 Patients typically feel an abrupt, tearing sensation of chest pain that radiates to the back.2,5 However, it can present in various other ways, spanning a spectrum of different symptoms. These atypical presentations can manifest with features of acute coronary syndrome, stroke, pulmonary embolism, or even be asymptomatic. Although cases of pulmonary embolisms would warrant a CT angio of the chest that can help identify the presence of a dissection, other presentations make it challenging to diagnose without a high suspicion. 8
Upon literature review, many cases of AD with varying presentations have been reported. Carrillo-Alfonso et al described a patient presenting with dizziness and imbalance progressing to stroke symptoms with focal neurological deficits. Later complaints of dorso-lumbar pain and an ischemic stroke, along with risk factors such as hypertension and AF, raised concerns for AD. Appropriate workup confirmed a type A AD. 10 This case highlights the diagnostic challenges of AD and the potential detrimental consequences if misdiagnosed, such as administering thrombolytics, which could increase the risk of rupture. Filip et al reported a patient with chronic abdominal pain for 30 days, who had no history of cardiovascular or connective tissue diseases. A type B AD was identified by CT after an emergent esophagogastroduodenoscopy revealed signs of small bowel infarction, illustrating a rare complication of type B AD. 11
Atypical painful presentations can lead to missed diagnoses of AD in patients with atypical symptoms. Conversely, painless AD also presents diagnostic challenges. Navari et al reported a patient with no significant medical history presenting with loss of consciousness and large pericardial effusion causing cardiac tamponade. Emergent surgery and subsequent CT scan revealed an intramural hematoma of the aorta with a type A dissection. 12
Although data on incidence of atrial flutter are scarce, there are 6 cases of AF have been reported in the setting of AD to date. 13 The association between AF and AD is underappreciated, and this co-presentation complicates management. Acknowledging this co-occurrence is vital, as Campia et al 14 detected higher in-hospital mortality in patients presenting with both AF and AD compared to patients without AF, with no significant difference in stroke risk. The pathophysiology behind this relationship is unknown; however, it could be secondary to left atrial compression due to dissection induced hematoma, severe pain, acute heart failure due to aortic regurgitation, or myocardial ischemia disrupting the supply of sinoatrial node.
The current recommendations of management of AD are as follows: it is recommended that all patients with type A AD undergo emergent surgical repair of the dissection with adjunct therapy with beta-blockers, meanwhile the mainstay management for non-complicated type B AD is optimal medical therapy with beta-blockers, pain relief, and appropriate BP management. Based on the current 2022 guidelines, in the presence of suitable anatomy, it is recommended that patients with complications of type B AD such as rupture, endovascular intervention. 8 In a study analyzed by Hameed et al, it was found that the use of beta-blockers in patients who are undergoing a type A AD repair, in-hospital mortality with emergent surgery is 19.7% compared to 57.1% for patients who are managed solely with medication. This is reinforced by another study that showed a 48-hour mortality rate to be 4.4% for patients who have undergone emergent repair compared with 23.7% for those managed medically. For patients with type B AD, thoracic endovascular aortic repair (TEVAR) has been shown to have a 30-day in-hospital mortality rate of 9.1% compared with 14.7% for open surgical repair as well as a lower 5-year mortality with an odds ratio of 0.46. 15
Conclusion
The presence of AF in patients with AD not only complicates the clinical picture but also poses challenges in management, particularly regarding the use of anticoagulation therapy. Higher in-hospital mortality rates associated with this co-occurrence, as highlighted in studies, emphasize the need for vigilance and prompt diagnosis. Advanced imaging techniques and comprehensive diagnostic evaluations are essential in identifying AD in patients presenting with AF, especially when symptoms are atypical.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient for their anonymized information to be published in this article.
