Abstract
Genomic variants of the hepatitis B virus (HBV) preS/S protein are well-known to occur. Typically, immunity is gained through recovered HBV infection or by immunization. Very rarely, there are certain mutations that may enable HBV escape from the immune detection. PreS/S mutants may present with unpredictable pathobiologic, clinical, and transmittable implications. Standard laboratory testing for genomic HBV variants is not routinely performed by reference guidelines. s-variant HBV management remains challenging. Herein is a case of s-variant chronic HBV infection in a 55-year-old man. Diagnosis and treatment are described.
Introduction
Approximately 350 million people worldwide and 1.2 million people in the United States have been diagnosed with hepatitis B virus (HBV) infection. Many people with acute and chronic HBV infection have few or no clinical manifestations and are unaware. 1
Hepatitis B virus incidence is worldwide with particularly high incidence in Asia and Africa. See Figure 1 for global distribution of HBV. 1
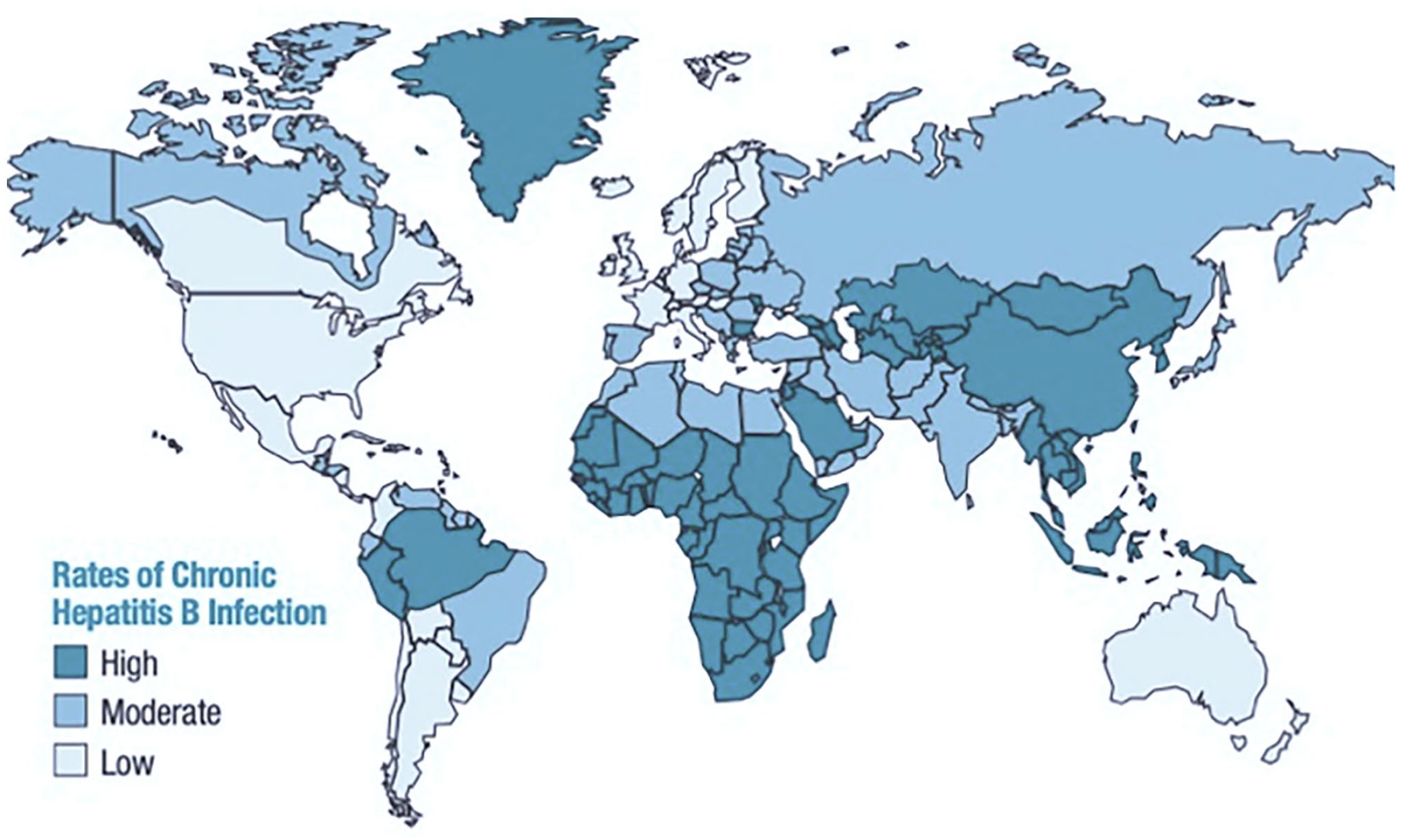
Geographical distribution of HBV, rate of chronic infection. 1
Increased rates of disease are attributable to vertical and horizontal transmission. Hepatitis B virus replication carries a more than 10-fold mutation rate as compared with other DNA virus counterparts. 2 This is typically due to the viral polymerase of HBV which lacks proofreading during transcription of the pre-genomic RNA. Consequently, mutations are common and account for genetic heterogeneity of HBV.
Antigenic/immune escape occurs when the immune system is unable to recognize or respond adequately to an infectious agent (ie, HBV). In this circumstance, the host fails to generate immunity against HBV. Antigenic escape should be suspected when there is persistent serologic coexistence of hepatitis B surface antigen (HBsAg) and hepatitis B surface antibody (HBsAb). Serologic coexistence of HBsAg and HBsAb has a high sensitivity but low specificity for immune escape variants of HBV.2-4 Quantification of HBsAg is not currently recommended per American Association for the Study of Liver Disease (AASLD) guidelines. 3
S-variant HBV, like all HBVs, are transmittable through blood and body fluids. There is an increased risk for progression to malignancy. Transmission rates in immune escape variants remain unknown. 2 The illness produced by s-variant HBV is synonymous to other HBV infections. Three outcomes exist: 90% of acute HBV infections are cleared and may be symptomatic or asymptomatic, 10% result in chronic disease based on sero-persistence of HBsAg for more than 6 months to 10 years, and rarely, fulminant disease and related death may occur. 5 Serologic screening for coinfection with hepatitis C or D, and HIV is recommended. 3
Diagnosis of s-variant HBV is typically based on measuring the presence of surface antigen and IgG, IgM core antibody, and surface antibody. There is antigenic and serologic evidence of disease but due to inherent testing limitations, s-variants are specifically not revealed. It is also important to distinguish between heterologous antibody versus HBV active disease as false positives are known to occur (Chart 1).

Serologic proteins or “markers” used to identify hepatitis B status: acute HBV, chronic HBV, immune by vaccination, immune by recovered HBV, or susceptible to infection. 2
Therapeutic intervention is provided for severe or protracted cases (based on INR > 1.5, total bilirubin > 3 mg/dL, deep jaundice > 4 weeks, encephalopathy or ascites). Treatment is indicated in these cases using antiviral therapy with entecavir, tenofovir alafenamide (TAF), or tenofovir disoproxil fumarate (TDF), since the 2006 National Institutes of Health HBV Meeting. 3 Pegylated interferon gamma (Peg-IFN) is contraindicated due to its increased risk of bacterial infections and hepatic necro-inflammation. Seroconversion of HBsAg typically indicates a response to therapy. 3 Clinical decisions are continued based on liver enzymes and monitoring of disease activity (ALT in men < 29-33 U/L, in women < 19-25 U/L, HBV antigens, and HBV DNA). 3 Ultrasound imaging is recommended. For patients receiving antiviral therapy, lactic acidosis monitoring is advised. Treatment can be discontinued after 2 consecutive serologic tests, 4 weeks apart, which confirms sero-clearance of HBsAg. 3
Chronic HBV in previously undiagnosed patients can be difficult to differentiate from acute HBV. Tenofovir or entecavir is preferred. Adefovir is not usually preferred due to its weak antiviral activity. 3 Diagnosis and treatment recommendations are not available specifically for s-variant HBV chronic infections.
Methods
Approval was obtained from the Institutional Review Board of Kern Medical. Retrospective review of the patient’s record was performed. A literature search was conducted on PubMed, ResearchGate, Google Scholar, Centers for Disease Control and Prevention (CDC), Infectious Diseases Society of America’s (IDSA) Clinical Infectious Diseases Journal database, AASLD, and
Case Presentation
A 59-year-old Filipino man was diagnosed with HBV infection in 1994 in the Philippines. He remained without symptoms until 25 years later, when he presented to our institution sent by his primary care provider for positive HBsAg. His only pertinent history was heavy alcohol intake years prior to presentation which was reduced to 1 to 3 glasses of wine daily.
His work up revealed AST 29 U/L, ALT 47 U/L, hemoglobin of 16.8 g/dL, and platelets of 289 cells/UL. Blood chemistry demonstrated a creatinine of 0.89 mg/dL, BUN of 10 mg/dL, and sodium of 145 mEq/L. Alpha fetoprotein (AFP) was 2.9 ng/mL. Initial hepatitis work up revealed HBsAg reactive, HBcAb reactive, and HBeAg and HBeAb non-reactive with HBV DNA viral load of 36 IU/mL indicating chronic active hepatitis B, but also HBsAb 590 mIU/mL. Given the contradictory nature of the results, a repeat testing was obtained, which again showed HBsAg reactive, HBcAb reactive, HBeAg and HBeAb non-reactive, HBV DNA 73 IU/mL, and HBsAb 706 mIU/mL. The confirmed coexistence of HBsAg and high titers of HBsAb indicated immune escape variants.
Ultrasound imaging of the abdomen revealed irregular liver margins suggestive of cirrhosis. In accordance with AASLD guidelines, hepatic cirrhosis was diagnosed by imaging. Routine screening for HIV returned non-reactive and the clinical concern for lactic acidosis was low. 3 Co-infection with hepatitis C or D was ruled out by nonreactive serologies. Hepatitis B virus genotype and basal core promoter (BCP)/precore mutation were sent to reference laboratory and came back as “not detected” perhaps due to inadequate viral load. Repeat BCP was deferred due to viral suppression and minimal evidence of clinical impact.6-11
Treatment of naïve s-variant chronic HBV and immune active disease, was initiated with entecavir 1 mg daily. 3 Viral suppression was achieved within 2 months of therapy with undetectable HBV DNA <10 IU/mL and sustained to the time of this report at 18 months. He remains asymptomatic and adherent (Table 1).
Summary of HBV Disease and Treatment Course.

HBV DNA: polymerase chain reaction test. Reportable range is 10 to 1 000 000 000 IU/mL. AFP: reference range < 8.0 ng/mL, not to be interpreted as the presence or absence of disease.
Abbreviations: AST, aspartate transaminase; ALT, alanine transaminase; HIV, human immunodeficiency virus; BCP, basal core promoter; (—), data not collected; HBV, hepatitis B virus; HBVeAg, hepatitis B virus enveloped antigen; NR, nonreactive; HBVeAb, hepatitis B virus enveloped antibody; HBVsAg, hepatitis B virus surface antigen; N/A, not applicable; HBVsAb, hepatitis B virus surface antibody; HBVcAb, hepatitis B virus core antibody; R, reactive; HDV Ag, hepatitis D virus antigen; HDV Ab, Hepatitis D virus antibody; HDV RNA, hepatitis D virus ribonucleic acid; HDV IGM AB EIA, hepatitis D virus immunoglobulin M antibody enzyme immunoassay; AFP, alpha fetoprotein.
Discussion
Immune escape occurs as a result of immunization, therapeutic measures, and/or by spontaneous mutation. Despite availability of vaccines, these variants continue to incite challenges against HBV immunity. Emergence of HBV variants of the preS/S genomic region whether naturally occurring or therapeutically induced, frequently occur in chronic HBV disease yet diagnosis and treatment monitoring remain limited, and associated risks of transmissibility remain unknown.
Hepatitis B virus statistics are notably underestimated. 1 S-variant HBV disease is further grossly unaccounted for, despite common knowledge of its occurrence. 2 PreS/S variants have been associated with fulminant hepatitis, fibrosing cholestatic hepatitis, cirrhosis, and hepatocellular carcinoma (HCC). One meta-analysis of 43 studies evaluated more than 11 000 cases of HBV and revealed that patients with s-variant HBV infection were associated with a 3.77-fold higher risk for HCC. 12 Multiple experimental studies resonate this finding.
S-variant HBV pathogenesis may be attributable to several specific mutations in the preS/S gene, at 1 or more of the 3 coding sites on DNA. 2 These naturally occurring or therapeutically induced s-variant HBV infections may present with mutations that lead to an imbalanced amount of envelope proteins known to accumulate into the endoplasmic reticulum (ER) of the hepatocytes. The ER stress pathways then become activated which have been linked to oxidative radicals. Oxidative stress leads to DNA damage, and genomic instability thus leading to hepatocyte destruction and eventual cirrhosis or HCC.2-4 Ultimately, liver damage may occur due to the accumulation of cytotoxic intracellular enzymes, favoring disease progression to cirrhosis and HCC. 2 Early detection of s-variants may be helpful in identifying patients in need of more aggressive treatment and monitoring strategies. For increased risk of the aforementioned and unknown transmissibility risk regardless of immunoprophylactic status, s-variant HBV poses a major public health concern. 2
Chronic carriers with serologic evidence of HBsAg and HBsAb could be considered potential reservoirs of immune escape variants. S-escape variants are relatively untraceable by current industry standards. Screenings and vaccinations via standard methods are inadequate for diagnosing and preventing s-variant HBV infection. Quantification of surface antigen is rarely obtained. In our patient, qualitative antigen assay was the only available option, which is the acceptable standard.2,4 HBV DNA viral load by polymerase chain reaction was also useful. However, quantitative HBsAg may have unique interpretations with clinical significance. While they are often used in research and clinical trials, use of surface antigen quantitative assay has been trending as a future industry standard for HBV management. The HBsAg and HBV DNA assays are projected as determinants of disease activity and prognosis.2,4 Development of more sensitive, standardized, and validated assays for detection of serum HBsAg, s-variants, HBsAg with immune complexes, and HBsAb are required. 3
Mutations involving amino acids 126, 129, and 141 have been documented responses to immunization but the clinical significance has not been well documented. 13 Most reports found that the immunization-related HBV s-variants were absent in maternal carriers. This may indicate mutation selection by vaccine and/or hepatitis B immunoglobulin. 14
One study in Taiwan found that the prevalence of s-variant HBV in HBsAg positive pediatric population increased with immunizations (7.8% in 1984 to 25% in 1994, and 23% in 1999). 15 Interestingly, carrier rates decreased (9.8% to 0.7%) in the same time frame. Another study in 2009 found no increase in breakthrough s-variant HBV infections over a 25-year course. 15 These data indicated that immunization-induced s-variant HBV infections are rare and have not diminished the efficacy of HBV vaccines.
Familial transmission is of reasonable concern by the G145R mutation, regardless of adequate serologic HBsAbs. 15 Improvement of detection system for the G145R and other s-variants is required for their effective control.
Because not all HBV immunizations result in immune escape, protection against HBV is probable for the majority. 3 Meanwhile, expansion of evidence-based guidelines for improved management of s-variant HBV disease is needed.
Conclusions
Concern for immune escape s-variants of hepatitis B is warranted when serologies of both the surface antigen and antibody coexist. 2 HBsAg is not routinely quantified but may have diagnostic value, although not explicitly endorsed by reference guidelines. 4 Commercial testing limitations exist and do not account for s-variant HBV infections. Immunizations are recommended for at-risk, non-immunized patients, despite potential for immune escape against prophylaxis. The risk of transmission of s-variant HBV, even to individuals with adequate immunity against HBV, remains unknown. 2
Footnotes
Acknowledgements
This case has been presented at the Infectious Disease Association of California’s (IDAC) 34th Annual Fall Symposium, fascinating cases, November 2019, and the American Federation of Medical Research’s (AFMR) Western Medical Research Conference, January 2020.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval to report this case was obtained from the Kern Medical Institutional Review Board; approval ID: 19079.
Informed consent
Informed consent was obtained for patient information to be published in this article.
