Abstract
Previous studies have shown that the recurrence rate of HBV (hepatitis B virus) after liver transplantation (LT) can be as high as 80% without any preventive measures. Therefore, prevention of HBV recurrence after LT is always an essential part of clinical work worldwide. The NAs that have been approved for HBV treatment include lamivudine, adefovir dipivoxil, entecavir (ETV), and telbivudine, tenofovir disoproxil fumarate (TDF), and tenofovir alafenamide (TAF). They are often combined with HBIG to prevent HBV recurrence after LT clinically. However, NAs with a higher genetic barrier, such as ETV, TDF, and TAF, can improve liver function by strongly inhibiting HBV replication and reducing the risks of HBV resistance. Recently, some NAs with a higher genetic barrier, such as ETV, TDF, and TAF, have been adopted as monotherapy for preventing the recurrence of hepatitis B after LT in multiple organ transplant centres and have achieved effective outcomes. This article aims to review the advances for NAs monotherapy in prophylaxis for HBV recurrence after LT.
Introduction
The prevention of hepatitis B virus recurrence after liver transplantation (LT) remains an important consideration worldwide, especially in East Asian countries. The proportion of LT recipients with HBV-related end-stage liver disease is as high as 74.79%, and HBV-related liver disease affects 71.25% of the LT recipients in China. 1 Without introducing preventive measures, the recurrence rate of HBV after LT can be as high as 80%, and one of the critical factors that cause graft loss and declined long-term survival rate in recipients is HBV recurrence. 2
However, the mechanisms of HBV recurrence after LT are still complex and have not been fully elucidated. Several factors are believed currently to increase the risk of HBV recurrence after LT.1,3,4 That includes occult HBV infection (OBI) in the native liver, the persistence of covalently closed circular DNA (cccDNA) detectable in the hepatocytes of the graft, non-clearance of HBV in the host extrahepatic tissues, HBV genotype-specific and genetic variations, infusion of blood products during LT, the application of immunosuppressive agents after LT, nucleos(t)ide analogues (NAs) with weaker genetic barriers, and lower anti-HBs titers after LT.
Existing regimens for prophylaxis against HBV recurrence after LT include combinations of HBV immunoglobulin (HBIG) and nucleos(t)ide analogues, active immunization programs for HBV vaccines, and NA monotherapy prophylaxis. 5 In addition, previous studies have suggested that a single application of HBIG after LT can also reduce the recurrence rate of HBV to 40%. However, the long-term use of HBIG is expected to cause higher recurrence rates, which have led it to be rarely adopted alone to prevent HBV after LT.4–6 Currently, CHB (chronic hepatitis B) is treated with nucleos(t)ide analogues (NUCs) and interferon (IFN). NUCs are viral polymerases and reverse transcriptase inhibitors that effectively inhibit HBV replication, resulting in a rapid reduction of HBV DNA. Since NUCs do not inhibit covalently closed circular DNA (cccDNA) transcription, they have a negligible effect on circulating hepatitis B e antigen (HBeAg) and hepatitis B surface antigen levels (HBsAg). 7 Prolonged therapy with highly active NUCs with the fewest drug resistance profiles can achieve long-term viral suppression and halt disease progression. The advantages of NUC therapy include oral administration, typical side effects, and a high rate of on-treatment virological responses; however, the risk of drug resistance, long-term safety, and indefinite duration of therapy remain the main concerns. 8
The NAs that have been approved for HBV treatment include lamivudine (LAM), adefovir dipivoxil (ADV), telbivudine (LdT), entecavir (ETV), tenofovir disoproxil fumarate (TDF), and tenofovir alafenamide (TAF). The TAF, which was recently approved and had a more favourable renal profile than TDF, will likely be the more appropriate formulation if TNV is used in the post-LT setting. Based on early results, TAF appears safe and effective in LT recipients. TAF is preferred over LAM and ADV because it is more potent and has lower resistance. 9 Some of the earlier NAs (i.e. LAM, ADV, and LdT) have limited barriers against HBV resistance, and they are used alone for prophylaxis against HBV recurrence after LT. The recurrence rates are likely to be higher; therefore, these tend to be used in combination with HBIG.10,11 However, NAs with more substantial genetic barriers, such as ETV, TDF, and TAF, can improve liver function by strongly inhibiting HBV replication and reducing the risks of HBV resistance and have been approved as first-line oral medicines for the treatment of chronic HBV infection.1,5,12
Acute hepatitis D caused by HBV/HDV co-infection occurs simultaneously when an individual susceptible to HBV becomes infected with both HBV and HDV. As with HBV mono-infection, HDV/HBV co-infection is typically transient and self-limiting in adults. The rate of progression to chronicity is the same as that of HBV mono-infection (i.e. less than 5%). 13 Infection with the Hepatitis D virus (HDV) characterizes a subset of hepatitis B surface antigen (HBsAg)-positive patients who are suffering from a particularly aggressive form of chronic liver disease (hepatitis D). For this reason, a productive HDV infection is invariably associated with an HBV infection. This is because the assembly and release of HDV particles are both dependent on the presence of HBV within the same hepatocytes. Co-infection with HBV and HDV and superinfection with HDV in a person who is chronically infected with HBV are the two significant patterns of infection that have been identified. 14
Recently, particular NAs with more substantial genetic barriers, including ETV, TDF, and TAF, have been adopted as monotherapy options to prevent HBV recurrence after LT in multiple organ transplant centres in China and other countries to facilitate effective outcomes. Simultaneously, this monotherapy approach as a prevention strategy has also significantly reduced the heavy medical burden brought to the government, society, and the family by combining NAs and HBIG and shows good potential for widespread clinical application. This article reviews the advances in NAs in prophylaxis for HBV recurrence after LT.
A short course of HBIG combined with long-term NA monotherapy
Short course of HBIG combined with long-term NAs monotherapy.
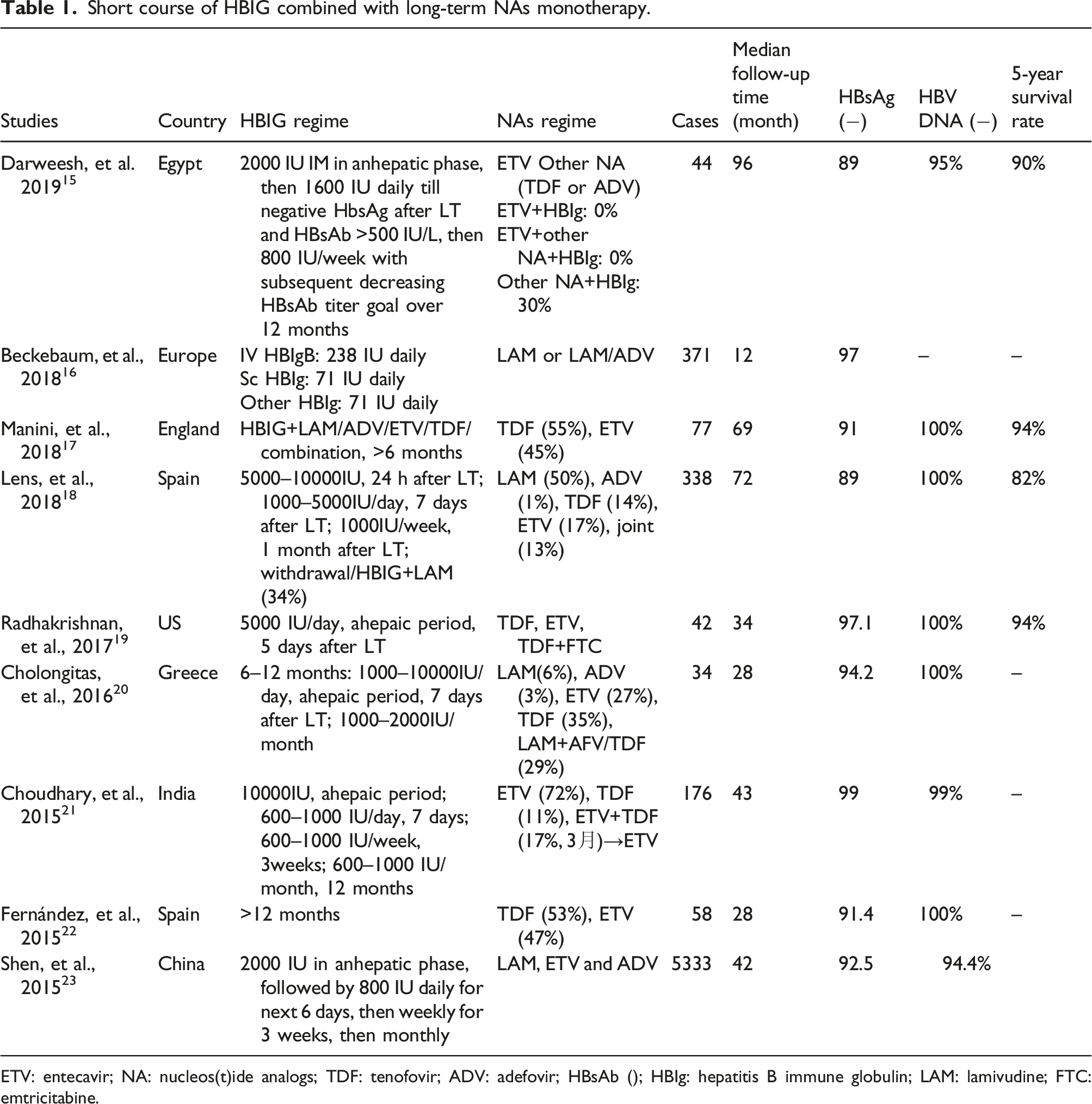
ETV: entecavir; NA: nucleos(t)ide analogs; TDF: tenofovir; ADV: adefovir; HBsAb (); HBIg: hepatitis B immune globulin; LAM: lamivudine; FTC: emtricitabine.
Despite recent advancements in the field, a growing gap exists between the availability of organ donors and the number of people on the waiting list for liver transplantation (LT). One of the current efforts to alleviate the organ shortage is based on grafts from donors with antibodies against the hepatitis B core antigen (anti-HBc). But do not have antibodies against the hepatitis B surface antigen (HBsAg); these donors are referred to as “anti-HBc positive donors.”. 29 These grafts are quite prevalent in countries with a high or even a moderate prevalence of HBV infection, such as Asia and the Mediterranean basin. However, liver donors who are anti-HBc positive frequently have occult HBV infection, defined as persistent HBV DNA in the liver and/or serum without serologic evidence of active HBV infection (negative HBsAg with or without positive anti-HBs). Indeed, several studies in HBsAg-negative subjects have demonstrated that covalently closed circular DNA (cccDNA) and pre-genomic RNA are frequently detected in the liver. 30
Manini et al.
17
investigated 77 cases (55% TDF, 45% ETV). There were 69 HBV mono-infected individuals in Group A and 8 HBV/HDV co-infected patients in Group B. Patients in Groups A and B were followed for 69 and 61 months after HBIG removal, respectively. After 18 months, no Group B patients developed HBsAg or HBV-DNA recurrence, while in Group A, 6 patients developed HBsAg. The 5-year cumulative incidence of HBsAg recurrence was 9%. During a further 30 months of follow-up, all six patients had undetectable HBV-DNA levels and consistent graft function. Seroconversion was temporary in three of the six patients, whereas the remaining three had HBsAg levels of less than 0.13 IU/mL throughout the study. This study also confirmed that the duration of HBIG was not related to the likelihood of HBsAg recurrence (
Lens et al.
18
conducted a retrospective multicenter analysis of the effect of discontinuing HBIG and continuing long-term NA therapy on preventing HBV recurrence following liver transplantation. They included 383 liver transplant recipients and compared the effects of short- and long-term HBIG regimens. During the median 72-month follow-up period, 37 patients (11%) developed HBV recurrence; discontinuation of HBIG did not affect the rate of recurrence or disease progression. At the same time, recurrence of hepatocellular carcinoma and LAM resistance were independent predictors of HBV recurrence following LT (
Another centre conducted a preliminary study about using HBIG only in the high-dose perioperative period followed by long-term NA use post-LT to prevent HBV recurrence. Radhakrishnan et al. 19 enrolled 42 pre-transplant HBV DNA-negative LT recipients with HBIG (5000 IU/day) in the anhepatic phase and daily for 5 days after LT. They underwent a single injection application of TDF, ETV, or TDF in combination with emtricitabine after LT to prevent HBV recurrence. During the 16-month follow-up period, only one patient was re-infected with HBV, and the survival rates post-LT were 98% and 94% at one and 5 years, respectively. These findings proved that the short-term combination of high-dose HBIG and NA monotherapy effectively prevents HBV recurrence after LT and should be the preferred strategy for managing pre-transplant HBV DNA-negative LT recipients.
According to the results outlined in the above studies, the recipients remain safe. They can show good clinical outcomes by adhering to a prevention strategy of short-term HBIG followed by long-term maintenance of NAs with more substantial genetic barriers, such as ETV or TDF after LT.
NA prophylaxis without HBIG in preventing HBV recurrence
NAs prophylaxis without HBIG in preventing HBV recurrence.
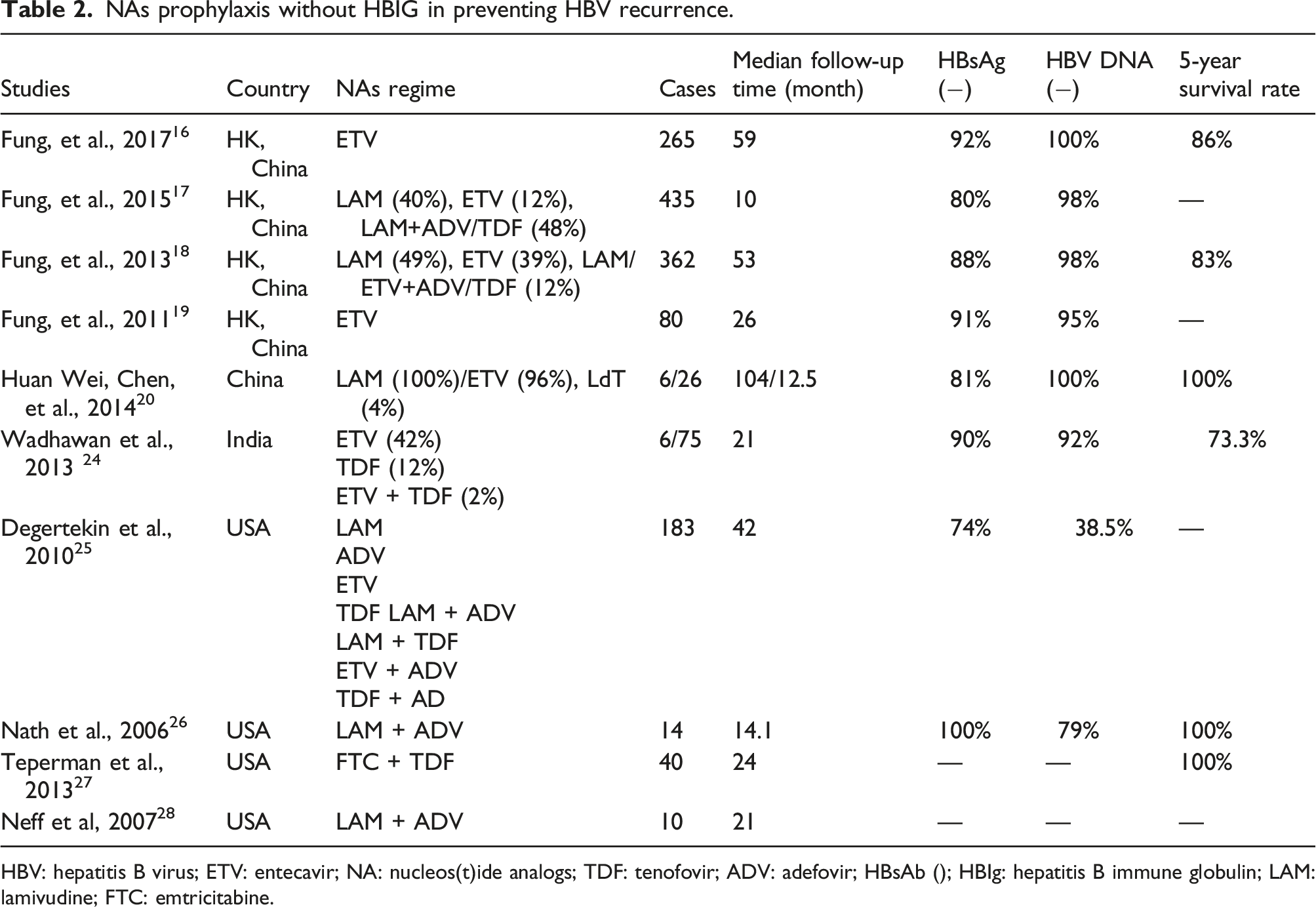
HBV: hepatitis B virus; ETV: entecavir; NA: nucleos(t)ide analogs; TDF: tenofovir; ADV: adefovir; HBsAb (); HBIg: hepatitis B immune globulin; LAM: lamivudine; FTC: emtricitabine.
In another study conducted by the same research team,
33
the clinical effects of different NAs alone in preventing HBV recurrence after LT were compared. During the 53-month follow-up period, 362 recipients given NAs were divided into three groups, including 176 who received LAM, 142 who received ETV, and 44 who received LAM + ADV at the time of LT, respectively. Although 60% of these recipients presented detectable HBV DNA before LT, they retained a higher HBsAg serum clearance rate and achieved viral suppression. The incidence rates of HBV recurrence at 3 years for LAM, ETV, and the combination group were 17%, 0%, and 7% (
The majority of published data reports boast limitations, including small sample size, a single-centre design, patient selection bias (e.g. low-risk subjects), a shorter follow-up period; lack of consensus on the anti-HBs scheme after HBIG discontinuation, or a lack information regarding the virological profile before LT (e.g. genotype, drug-resistance mutation, HBsAg titer). 10 However, the available findings also indicate that the prevention regimens of NAs with more substantial genetic barriers and/or a short course use of HBIG can achieve good clinical outcomes after LT. Their survival rates were not significantly different from the combination prevention regimens. It could be argued that the single use of NAs with more substantial genetic barriers for preventing HBV recurrence after LT is feasible and worthy to study further.
Side effects and limitations of the single administration of NAs
As HBV can only be suppressed but cannot be eradicated, long-term NA monotherapy always pairs with the risk of HBV resistance. Some research indicates the 5-year mutation probabilities of LAM can be as high as 70%,
5
and the 5-year drug-resistance rate of ADV can reach 29% (also including side effects of renal impairment).5,34 The drug-resistance rate of LdT can also get 17% (prone to cause polyneuropathy or myopathy), respectively.5,35 The ARES research was a multicenter, randomized, open-label trial in which 175 HBeAg-positive patients received 24 weeks of entecavir (ETV) lead-in therapy and were then randomly assigned to receive PegIFN-α2a + ETV for 24 weeks (
Therapeutic vaccination
Despite universal vaccination being available for over three decades, an estimated 248 million persons are chronically infected with the hepatitis B virus (HBV) globally, constituting a significant health burden. 44 One factor contributing to the chronicity of HBV infection is persistent immunological dysfunction. They are partly caused by the exhaustion of HBV-specific CD8+ and CD4+ T-cell responses in the presence of chronically high HBV antigen loads. Although LT significantly reduces the antigenic load by removing all infected hepatocytes, this alone appears insufficient to restore the immune deficit. Following transplantation, additional vaccination strategies appear ineffective at stimulating the production of neutralizing antibodies. Perhaps this is unsurprising given that these patients have already been exposed to HBV antigens throughout their lives with a persistent inability to mount an effective immune response. 45
Discussion
According to current clinical studies, most researchers still have advocated for LT recipients to receive lifelong NA therapy to prevent HBV recurrence. However, it may be feasible to discontinue NA therapy after LT altogether in specific populations. Lenci et al. 46 considered the lifelong use of NA prophylaxis after LT to prevent HBV recurrence in a study that included 30 patients with LT who were preoperatively positive for HBsAg but negative for HBeAg and HBV DNA who received HBIG in combination with LAM for at least 3 years. These patients underwent testing for intrahepatic HBV DNA and cccDNA un-detectability at 24 weeks following the discontinuation of HBIG, with LAM withdrawal. The results showed that the 2-year HBsAg-negative rate was negative, 83.3%. Of the 25 recipients who did not relapse, 23 presented negative HBV DNA findings in the liver tissue. In contrast, in the five relapsed recipients, results of HBV DNA were positive in the liver tissue (cccDNA was detected positively in one recipient). Recent studies have suggested that, 47 in the sixth year without any preventive measures, 90% of recipients showed an HBsAg negativity rate of 93.3% and an HBV DNA negativity rate of 100%. In comparison, 60% of LT recipients developed spontaneous seroconversion of anti-HBs. Therefore, it may be feasible to withdraw NAs in a specific post-LT recipient population. However, it is not recommended to discontinue HBV prevention treatment after LT altogether. The effectiveness and safety of this strategy still need to be confirmed by further research efforts. 48
Studies have shown that chronic HBV carriers who acquire HDV infection (HDV superinfection) develop acute hepatitis Delta, resulting in recurrence. It frequently worsens pre-existing liver damage and often results in the development of fulminant hepatitis in 7–15% of patients. Moreover, chronic HDV infection is defined by the persistence of antibodies to HDV and HDV RNA or HD-Ag in serum. At least 6 months after infection, it results in chronic hepatitis Delta (HDV-CH), a more severe disease than HBV-CH, with elevated aminotransferase levels and fibrosis progression rates. 49 Additionally, patients with HDV-CH are twice as likely as those with HBV mono-infection to develop and die of hepatic decompensation or HCC. The chance of survival 20 years after diagnosis with HDV-CH has been estimated to be 86%. HDV replication persistence is the sole component linked with increased mortality risk. 50
In addition, some studies have reported that 53.7% of recipients receiving the HBV vaccine after LT produced an active immune response. A 14.9% of those required periodic vaccination to boost their response, and younger recipients receiving a double-dose HBV vaccine 51 or an adjuvant recombinant HBV vaccine 52 may experience an improved response rate. However, the clinical efficacy of the HBV vaccine after early LT was poor, and it was usually deployed in recipients with longer survival times after LT. Therefore, HBV-associated LT recipients with more extended postoperative periods, stable liver function, HBV DNA negativity, and minor immunosuppressant doses can receive conventional double doses of the recombinant HBV vaccine, 53 which would allow them to produce protective anti-HBs. 54 However, in general, the strategy of HBV vaccination after LT to prevent HBV recurrence is still in the clinical trial stage. There remains a lack of uniform and standardized application programs, such as indications for target vaccination populations, the best vaccination dose, the ideal frequency of vaccination, and whether the vaccine should contain an adjuvant or not. Further, there is quite a difference in the strategy and efficacy of vaccination among major centres, 55 so it is necessary to conduct large-scale, multicenter randomized controlled clinical trials to explore and confirm this prevention program.
Limitations of the present study
The retrospective design of this study is a drawback, even though it included a large patient group and a long follow-up time. Anti-HBs levels were not consistently documented because this was a non-interventional trial. However, a significant drawback is the lack of information on the recurrence of HBV DNA following therapy in the trials included in this meta-analysis. Different HBIG and antiviral procedures utilized in the trials are other sources of bias.
Conclusions
In conclusion, while long-term immunosuppressive therapy is ineffective in preventing HBV recurrence after LT, the HBV viral load in patients after LT has significantly decreased. Fewer side effects and greater anti-HBs efficacy of novel NAs with considerable genetic barriers enable the avoidance or alleviation of several downsides, such as the enormous cost burden associated with long-term HBIG injections or the recipient’s body’s limited pain adherence to treatments. Thus, while the single-use of high-genetic-barrier NAs to prevent HBV recurrence following LT may enhance recipients’ compliance and long-term survival, larger-sample, multicenter controlled studies are required to confirm our findings. Additionally, establishing the clinical effectiveness of HBV prevention programs such as HBV vaccination or complete discontinuation of NAs and HBIG and achieving a higher level of long-term efficacy is still a key area of research in preventing HBV recurrence following LT.
Footnotes
Acknowledgements
We thank Xie Man for editing and retouching the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Clinical Medicine+X” program of Qingdao University (2017223).
Ethics approval
This study was approved by the Ethical Affairs Committee of the Affiliated Hospital of Qingdao University, and the study adhered to the tenets of the Declaration of Helsinki.
