Abstract
Background: This study aimed to determine the prevalence of HCV and occult HBV among newly diagnosed pre-treatment Egyptian lymphoma patients and evaluate patients’ outcomes based on the presence of the viral infections. Methods: The study included 80 therapy-naïve lymphoma patients including 71 non-Hodgkin lymphoma (NHL) and 9 Hodgkin lymphoma disease (HD) in addition to 100 healthy volunteers. HBV screening using HBsAg and anti-HBc IgM and HCV using AB/Ag ELISA and real-time RT-PCR were screened in tested and control groups. The diagnosis was confirmed by histopathology. Overall survival (OS) and progression-free survival (PFS) were conducted to diseased patients. Results: Healthy patients showed 4/100, (4%) active HCV infection and 1/100, (1%) active HBV infection and no occult HBV infection. Among NHL patients, 28 were positive for HBV (6 active and 22 occult HBV infection). Occult HBV was also detected in 5/9 HD patients. HCV was detected in (30/71, 42.3%) of NHL patients and in a single HD patient. Ten occult HBV NHL patients showed a mixed infection with HCV. The incidence of both HCV and HBV are higher in NHL than HL patients. After antitumor treatment, complete remission for lymphoma was achieved in 45% of patients. Both overall survival (OS) and progression-free survival (PFS) were correlated and significantly associated with patients’ LDH levels. Conclusions: Our findings claim the suggestive role of HCV and occult HBV infections in NHL but not HL patients in comparison to healthy control, suggesting pre-screening of related factors including occult HBV in for potential better therapy response.
Introduction
Liver inflammation is caused by multiple viral infections leading to viral hepatitis, which can occasionally result in liver fibrosis, cirrhosis, liver failure, and hepatocellular carcinoma (HCC). Most common hepatitis viruses are (hepatitis A virus, HBV, HCV, HDV, or HEV). HBV and HCV are the most prevalent chronic viral infections affecting people globally due to their ability in causing high rate of morbidity and mortality thus becoming of utmost importance. They can cause acute and chronic infections that can lead to cancer and liver cirrhosis. The estimated level of infection with HCV is More than 180 million people worldwide (3% of the world’s population), with prevalence rates being highest in North Africa, the Middle East, Central Asia, and East Asia ( rate of prevalence up to 15%), if to be compared with Italy and the United States of America (of a prevalence rate of 3%).1,2 70% of HCV patients can progress to a state of chronicity reaching a level of 71 million worldwide according to recent updates. 3 In 2019, the WHO has estimated the number of chronic HBV infections to be around 296 million globally. The highest rate of infection being in Western Pacific and African region. 4 Both viruses can coexistence together due to the similar mode of infection. The incidence of coexistence of HCV and occult HBV (HBV DNA-positive, HBsAg-negative) in Mediterranean population area is estimated to be 16%. 5
Clonal proliferation of B-, T-, or NK-cell subtypes of lymphocytes during their stages of maturation lead to development of hematological malignancies mainly lymphoma. 6 Hodgkin disease (HD) and Non-Hodgkin disease (NHL) are the main subtypes of such progression contributing to (10%), (90%) of lymphomas, respectively. HD is defined by the presence of Hodgkin Reed-Sternberg cells either classical or non-classical types while NHL represents a heterogeneous group of malignancies of different biology and prognosis. 7 The National Comprehensive Cancer Network (NCCN) guidelines for NHL provide general recommendations on classification whether the NHL is indolent or aggressive and whether it is B-cell, T-cell, or NK-cell lymphoma.7,8 Most common is classified into B-cell, T-cell, and NK-cell subtypes.7,8 B-cell type lymphoma contribute to (about 90%) of NHL and were more common western countries, while T-cell lymphoma represent about (10%) and were more frequent in Asian countries, and NK lymphoma were less than 1%. 8
Lymphoma incidence is increased in areas where HCV infection is high in comparison to general population (15%). 9 This theory by the successful regression of lymphoma upon administration of anti-viral drugs. 10 The detection of HBV-DNA is proven now to be a more accurate tool due to the underestimation of HBV infection as a predisposing factor in lymphoma. Upon determining the correlation between HBV and lymphoma, this has proven to be of utmost importance due to the risk of reactivation of occult HBV during chemotherapy which represent a challenge to NHL therapy or the therapy in itself lead to reactivation of a dormant virus (up to 20% of cases) leading to poor prognosis. 11 HBV infection is frequently detected in NHL 12 and it was detected in stage III and IV NHL patients as well as in progressive disease thus further proving the correlation between HBV and NHL. 13
The aim of the study is to estimate the incidence of HCV and occult HBV among newly diagnosed pre-treatment Egyptian lymphoma patients and evaluate patients’ outcomes based on the presence of the viral infections.
Materials and methods
Ethical approval
The study has been reviewed and approved by National Cancer Institute, Cairo University Ethical Committee, Egypt, with approval No.201001415025.2 in accordance with the ethical principles outlined in the declaration of Helsinki. An informed written consent was obtained from the patients who participated in the study.
Study cohort
This study was conducted from Jan 2015 through Dec 2018 at the National Cancer Institute, Cairo University, Egypt. A subset of 80 newly diagnosed lymphoma patients and one hundred control healthy volunteers (blood donors) were included in this study. In all cases, the diagnosis was confirmed by histopathology and classification was done according to the World Health Organization classification. All subjects were therapy-naïve. Age, gender, complete blood panel, liver enzymes, performance status, tumor stage, B symptoms, lactate dehydrogenase count, international prognostic index, bone marrow infiltration, treatment protocols, censored status, and death date were collected. Patients were treated according to the standard institutional protocol and treatment response was defined according to the international guidelines. 14
Selection criteria for patients; LPD confirmed patients, who did not receive any type of therapy, negative for infection with HIV. Exclusion criteria were patients who receive chemo or radiotherapy, positive results for HIV.
Non-Hodgkin lymphoma cases received either anti-CD20 monoclonal antibody (Rituximab)—containing chemotherapy regimen (R-CHOP) rituximab-free schedule CHOP or COP); while HD cases received ABVD (Adriamycin + Bleomycin + Vinblastine + Dacarbazine) for 4–8 cycles as per national guidelines. All positively infected cases were treated as per national guidelines (oral anti-viral or injections) at the Liver Egyptian Institute.
Detection of HBV and HCV
Detection of Hepatitis B surface antigen (HBsAg) in sera was done by ELISA technique using Monolisa HBsAg ULTRA kit according to manufacturer’s instruction. Immunoglobulin M (IgM) to hepatitis B core antigen (anti-HBc IgM) was determined in sera by Monolisa HBc IgM ULTRA kit according to manufacturer’s instruction. Detection of HCV in sera was done using a combined screening kit for anti-HCV antibodies and the viral antigen of the hepatitis C virus using an ELISA technique Monolisa HCV Ag-Ab ULTRA V2 kit according to manufacturer’s instruction. Patients’ samples tested positive for anti-HCV antibodies were tested for HCV-RNA using quantitative real-time PCR. Patients’ samples tested negative for both HBsAg and anti-HBc IgM were tested for the presence of HBV-DNA by nested PCR.
Detection of HCV-RNA by quantitative real-time PCR
Plasma HCV viral load detection was performed with the Artus1HCV-RG RT-PCR Kit (cat no. 4518265, QIAGEN1) according to the manufacturer’s protocol, and amplification was done by the ABI 7500 Fast Real-Time PCR Thermal cycler (Applied Biosystems, Foster City, CA, USA). Negative and positive controls were obtained from healthy volunteers and HCV-infected patients, respectively.
Detection of occult HBV
HBV and HCV biomarkers among lymphoma patients.

aHD: Hodgkin's disease.
bNHL: Non-Hodgkin's lymphoma.
cAmong the 6 patients showed positive HBsAg, there are two patients showed positive PCR for s gene, one patient for core gene, and 3 patients were positive for c, s, and x gene.
Morphological and immunophenotypic analysis
Morphological analyses of blood and bone marrow (BM) smears were carried out using May-Grünwald-Giemsa stain, hematoxylin, and eosin stains (H&E). The bone marrow trephine biopsy panel was performed with every lymphoma patient to assess lymphoma staging according to the international prognostic staging system. 16 Tumor grading were carried out based on morphologic examination of the tumor tissues according to the World Health Organization classification. 17
Data analysis
Overall survival (OS) was defined as the interval between histological diagnosis and death or last follow-up visit, while progression-free survival (PFS) was calculated from the start of first-line chemotherapy treatment until the date of disease progression or death. Survival distribution was estimated by the Kaplan–Meyer method. Significant differences in the probability of surviving between the strata were evaluated by the long-rank test. The Cox multivariate proportional hazard regression model was used to evaluate the effects of the prognostic factors on PFS and OS.
Statistical analysis
Data obtained from the present study were presented as numbers and percentages. Statistical comparisons were achieved using a chi-square test or Fisher exact test as appropriate.
Results
Demographic data
The present study included 80 newly diagnosed lymphoma patients with 71 (88.8%) NHL, 51 were B subtype and 20 were T-NHL and 9 (11.3%) HD. At the time of diagnosis, 20 (25%) patients were in stage I, 22 (27.5%) patients were in stage II, 21 (26.2%) patients were in stage III, and 17 (21.3%) patients were in stage IV. The median age for patients was 42 and 46 years for HD and NHL, respectively, with a gender ratio of 29 (male): 51 (female). All HD patients were females, but 59.2% and 40.8% of NHL were females and males, respectively.
HCV and HBV among HD and NHL patients
Seropositive HCV patients were also found positive for HCV real-time RT-PCR. The percentage of HCV-positive patients was highly significant (
Chemotherapy of the affected patients
Treatment protocols and responses in HD and NHL patients.

CR: Complete remission; PR: Partial remission; SD: Stable disease; RD: Relapsed disease.
Effect of different parameters on the response of lymphomas’ treatment
Response to treatment and variable affecting the remission response among lymphoma.

CR: Complete remission; WBCS: White blood cells; RBCs: Red blood cells; PS: The ECOG scale of performance status; LDH: Lactate dehydrogenase; IPI: international prognostic index.
Multivariate analysis of the type of lymphoma, HBV infection and response of treatment.
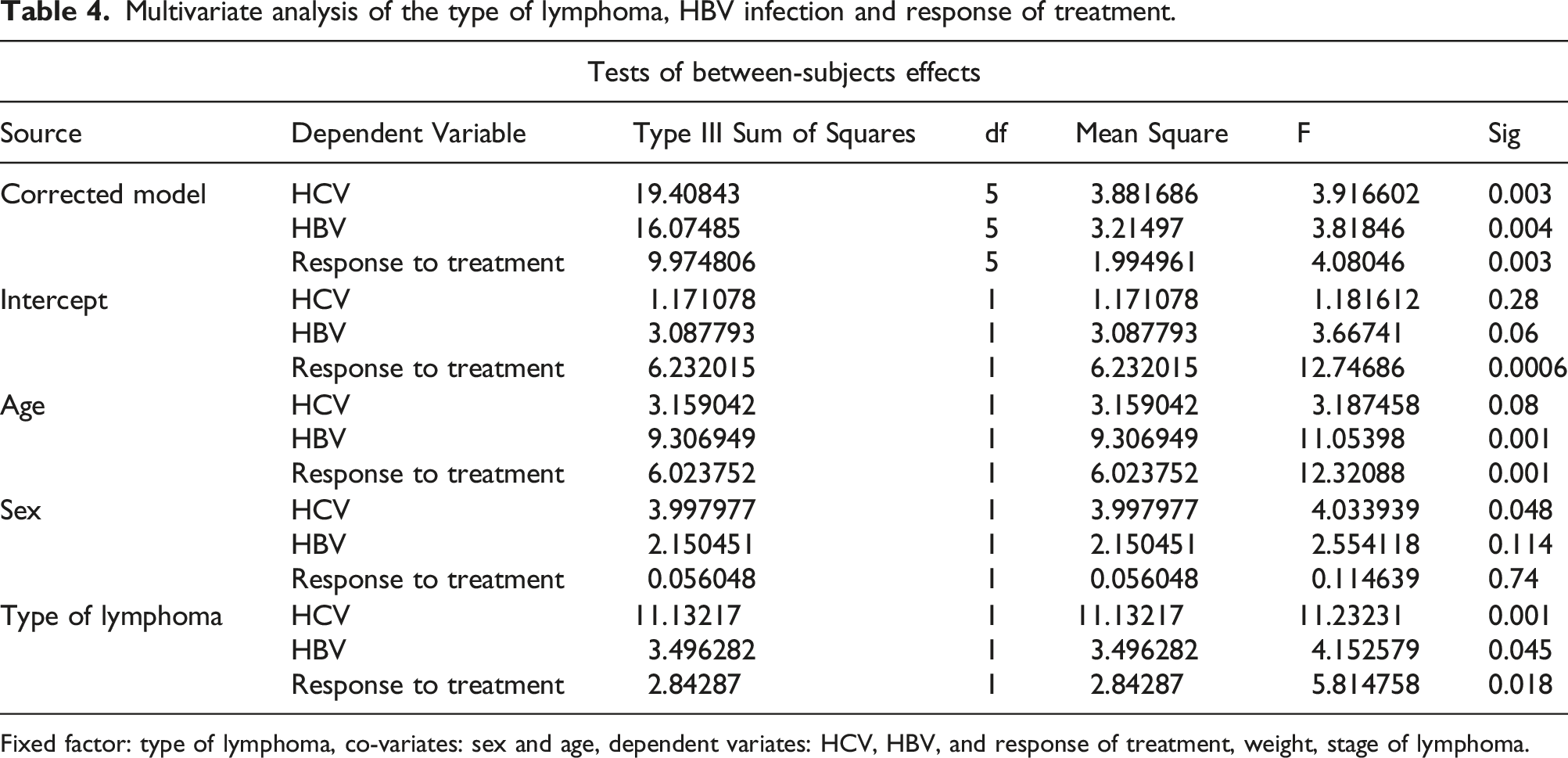
Fixed factor: type of lymphoma, co-variates: sex and age, dependent variates: HCV, HBV, and response of treatment, weight, stage of lymphoma.
Survival curves
The Kaplan-Meyer overall was used to calculate the survival curves according to tumor response and status of being progression-free at 12, 36, and 48 months (Figure 1). The overall survival (OS) and its relation to the prognostic factors showed a significant association with patients’ post-treatment normalized LDH, and CR ( Kaplan–Meyer curves of overall survival according to tumor response in the study group. (a) The median survival time for all lymphoma patients (b)The median survival time for lymphoma patients with normal post-treatment LDH. (c) (b)The median survival time for lymphoma patients who were given therapy. (d) The median survival time for lymphoma patients with CR. Kaplan–Meyer curves of Progression-free survival (PFS) according to tumor response in the study group. (a) The median survival time for all lymphoma patients (b) The median survival time for lymphoma patients with normal post-treatment LDH (c) The median survival time for lymphoma patients who were given therapy. 

Discussion
Lymphoma, a type of blood cancer, arises when lymphocytes grow out of control in the lymph system, particularly in the lymph nodes, or initiate somewhere, metastasize, and travel to the lymph nodes. There are two main types of lymphoma: (i) Hodgkin lymphoma (HD) and (ii) non-Hodgkin lymphoma (NHL). In our study, occult HBV infection was reported in many NHL patients. A total of 28/71 (39.4%) of NHL was found to be infected either persistent active (n:6) or occult HBV infection (n:22), in HD and NHL patients, respectively. This finding may indicate that a screening test target on DNA is required for all patients with B-cell lymphoma.
Many studies have found association between NHL and the presence of HBsAg.18-21 In addition, occult hepatitis B was also found to be associated with increased risk of developing NHL.22-24 Similarly, a Korean study reported 12.4% occult HBV prevalence in lymphoma patients. 25 Several cohort studies reported that HBV reactive persons have 2–3-folds increased risk to develop NHL than general populations,16,26 as demonstrated in the present study group. NHL patients infected with HBV usually present in stage 3 or 4, and a cohort study showed that HBV infection increases the risk of developing CD20+ aggressive lymphoma. 27
Our results showed an elevated ratio of occult HBV surface gene, due to its high sensitivity in serum samples. 28 Several studies reported that occult HBV infection is detected in several conditions including HCV-infected patients.29-31
Our study suggests that HCV is a possible risk factor for the development of NHL which agree with different studies from many countries.32-36 Interestingly, such association was not linked to the certain B-cell NHL subtypes (diffuse large B-cell, follicular, marginal zone, and chronic lymphocytic leukemia/small lymphocytic lymphoma) as confirmed by different studies form different countries.37-39 With an established role of HCV in the association with NHL development in several cohort studies, benefits of prevention and treatment of HCV infection are of the highest interest, as reported that individuals with chronic HCV infection with circulating HCV-RNA are at increased risk of NHL development.9,10 High incidence of HCV, 42.6% of NHL patients were HCV-positive, was reported in the current study. One could say that such finding is expected since there is high rate of widespread of HCV infection among Egyptian population. It is known that areas where HCV more prevalent, there is an increasing in the attribution to NHL that is estimated to be 10% higher than reported in other studies. 40 However, studies from Sweden, with low HCV prevalence reported detect a significant correlation between HCV and NHL. 36
A high incidence of HCV co-infection with occult HBV was reported in several studies, especially in the Mediterranean region, reaching up to 30% of HCV-infected individuals.41,42 Likewise, our study showed co-infection of HCV and occult HBV in a total of 12 lymphoma patients (16.7%). This co-infection was explained in other studies as both HBV and HCV viruses, rather than sharing the same route of transmission, but it was reported that HCV infection most probable can cause HBV replication inhibition with negative serology testing.41,43-45 HCV co-infection and occult HBV was reported in several studies in Egypt ranging to 8.5% a, 44 while others reported a wide range from 1.8% to 38.3%,29,30 with a difference in a ratio reported between northern and southern parts in Egypt.31,46 Another study made on Egyptian patients with hematological malignancy reported a high incidence of HCV co-infection with occult HBV reaching up to 32%, 47 which agrees with our results.
Screening for HBsAg and HBV-DNA, as well as screening for HCV-RNA for positive cases should be done for patients diagnosed with NHL as chronic viral infection may have a role in NHL B-cell development. In areas with a high prevalence of HCV and HBV, prevention and treatment of infection could diminish NHL incidence. Up on Comparison HBV and HCV incidence in HL with NHL patients, there is a significant higher rate of incidence of both HCV-RNA and HBsAg in NHL than in HL patients (
Overall, the high incidence of HBV and HCV in NHL and HL in the current study and in others can be explained by two hypotheses: (i) virus reactivation and, (ii) HBV and/or HCV may be possible risk factor. Both HBV and HCV viruses are reactivated due to the lowered immunity in cancer patients. This hypothesis is supported by that fact that reactivation of both HBV and HCV is not uncommon complication in patients with cancer. 49 Although reactivation of HBV was reported hepatitis B surface antigen (HBsAg) positive, it can occur in previously infected patients who apparently recovered from the HBV viral infection. HBV reactivation is enhanced by immunosuppression as associated with using chemotherapy. It is indicated by increase the rate of viral replication which is followed by an increase in the ALT levels within up to 3 weeks from the beginning of reactivation. 49 Similar to HBV reactivation, HCV reactivation is induced by immunosuppression with subsequent loss of control on the virus replication. 50 Cancer patients including those with NHL possess an elevated risk for HCV reactivation.51,52 The second hypothesis is that HBV and HCV may be considered risk factors for the development of NHL and HL. Incorporation of the HBV genome into the host genome may lead to overexpression and/or down regulation of oncogenes and tumor suppressor genes, respectively. 53 HCV intracellular proteins may also affect B-cells in an indirect manner and can induce oncogenic transformation due to chronic antigen stimulation that leads to E2 antigen-driven polyclonal B-cell proliferation 54 which could play a role in the development of NHL. 55 Meanwhile, the chronic inflammation during HCV and HBV is a strong inducer of cancer. 56 Meta-analyses studies suggested that each of HBV 12 and HCV 57 is considered a risk factor for NHL. However, conflicting results about the risk of the HBV in the induction of NHL since some studies confirmed variable range of risk58-60 while others found no significant link correlation between HBV infection and NHL.35,61,62 A large-population case–control study reported that the incidence of HBV and HCV in the NHL group in higher incidence than control group. 58 It is apt mention that chronic HCV infection resulted in 2-fold increase of lymphoid tumors from 2001 to 2005 including NHL. 63
Treatment for lymphoma mainly depends on the type of lymphoma (HD or NHL) and tumor stage. Chemotherapy, immunotherapy (targeted antibodies, immunomodulators, and adoptive cell therapy), radiotherapy, or combinations are typically used to treat both types of lymphoma. In this study, all HD patients were treated with chemotherapy ABVD regimen for 4–8 cycles. More than 50% of HD patients were chemo-sensitive and showed a complete remission. Given that most lymphomas are developed from B-cells, most of NHL patients (70%) were treated with Rituximab-containing CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisolone) regimen for 4–8 cycles. Rituximab is a monoclonal antibody that targets CD20+ expressed on B-cells, thus disrupting the CD20 pathway. A complete remission was achieved in 21 (20%) NHL patients and 6 (12%) patients were chemo-resistant and showed relapse. The overall survival (OS), the duration of patient survival from the time of histological diagnosis/treatment initiation to death or last follow-up visit, is a universally accepted direct measure of clinical benefits. In the same line, the progression-free survival (PFS), the time from the start of first-line chemotherapy treatment until the date of disease progression or worsening, could be used as a direct measure of clinical benefits. OS is comprised of PFS plus post-progression survival, therefore both OS and PFS would be related to each other. However, the relationship between the two outcomes is not always straightforward and it depends highly on the cancer type. 64 In the recent study, associations between OS and PFS were established. We identified a significant correlation between median OS and PFS to CR and normalized LDH post-treatment. The patient-level analysis showed that responders have better PFS and OS compared with non-responders.
Limitation
The main limitation of this study was we did not conduct any power analysis to calculate the sample size selected for this study. In addition, we did not have data about the history of infection in the affected patients prior to the development of HL or NHL so we could not confirm the hypothesis of virus reactivation.
Conclusion
In comparison to healthy control, both HCV and occult HBV were reported in considerable numbers in Egyptian lymphoma patients. Chemotherapy-treated patients have shown a complete remission in 45% of patients which was significantly associated with many parameters (patients’ age ≤45 years, reactive occult HBV core gene, normal SGPT levels, PS I, stage I disease, and post-treatment normal LDH, and low IPI). Data have demonstrated that both OS and PFS were correlated and significantly associated with patients’ post-treatment normal LDH with a median time of 21.1 and 10.1 months for OS and PFS, respectively.
Supplemental Material
Supplemental Material - The impact of hepatitis B virus and hepatitis C virus infections in patients with Hodgkin's and non-Hodgkin’s lymphoma
Supplemental Material for Hepatitis C and B viruses as potential risk factors in patients with Hodgkin and non-Hodgkin lymphoma by Dalia Y Kadry, Mostafa A Elbahnasawy, Mohamed TM Mansour, Omnia K EL Gebaly, Hala Aziz, Mahmoud M Kamel, Ahmed S Abdel-Moneim and Samah Radwan in International Journal of Immunopathology and Pharmacology
Footnotes
Acknowledgments
The authors would like to acknowledge the Deanship of Scientific Research, Taif University, Taif 21944, Saudi Arabia for funding this work.
Author contributions
D.Y.K., M.A.E., S.M., and O.E.G. designed research; D.Y.K. and S.R. performed research; D.Y.K., S.R., M.T.M.M., M.M. K, A. S. A, and O.E.G. responsible for the data analysis. M.A.E. wrote the manuscript. A.S.A., M. M. K and D.Y.K critically revised the manuscript. All authors reviewed and approved the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
