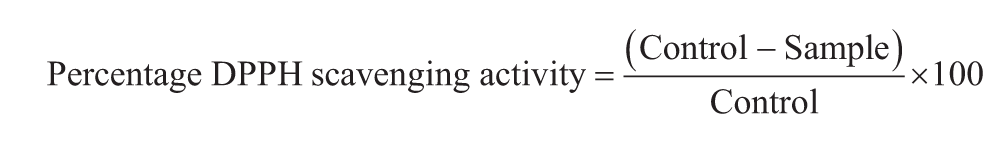Abstract
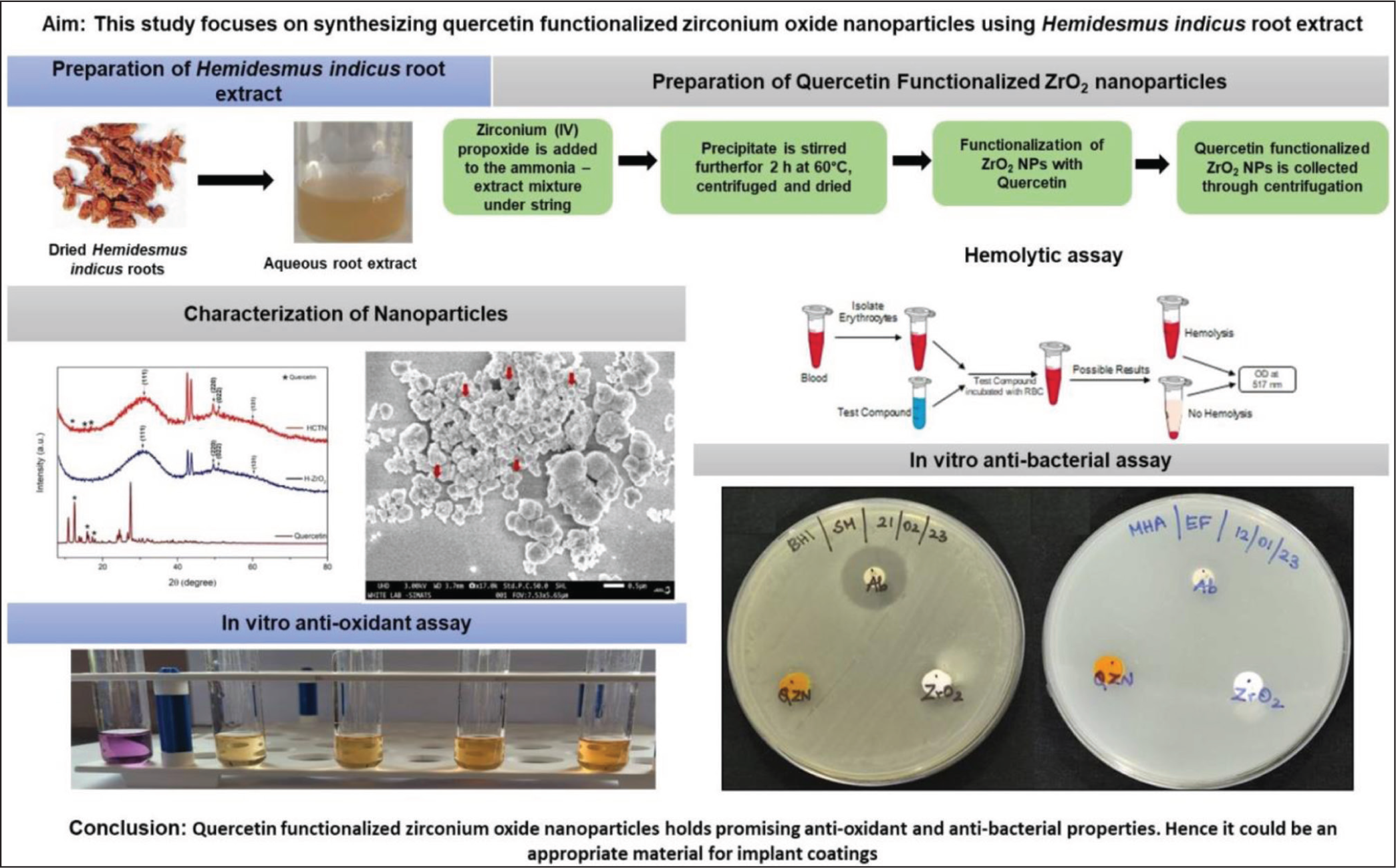
Aim:
Zirconium oxide nanoparticles have unique properties that could be useful in various applications, including implants, biosensors, anticancer, and antimicrobial agents. To enhance their suitability as coatings for prosthetic implants, quercetin, a flavanol abundant in plants, serves as an effective functionalization agent, enhancing nanoparticle solubility, stability, and bioavailability. This study focuses on synthesizing zirconium oxide nanoparticles using Hemidesmus indicus (H. indicus) root extract and functionalizing them with quercetin. Further, their biocompatibility, antioxidant and antimicrobial activity are evaluated to target their application as coatings for prosthetic implants.
Materials and Methods:
Zirconium oxide nanoparticles were prepared using H. indicus root extract (H-ZrO2NPs) and decorated with quercetin followed by pegylation. The nanoparticles were characterized using various characterization techniques including UV–vis spectroscopy (UV–Vis), Fourier-transform infrared spectroscopy (FTIR), X-ray diffraction (XRD), scanning electron microscopy, and energy-dispersive X-ray analysis. The biological activity of the nanoparticles was evaluated using biocompatibility, antioxidant, and antibacterial assays. Biocompatibility was tested by investigating the interaction between the quercetin-functionalized zirconium oxide nanoparticles (HQZN) and erythrocytes. The antibacterial and antioxidant efficacy of H-ZrO2NPs and HQZN was tested and compared with the standard drug. The results were statistically analyzed using one-way ANOVA followed by Dunnett’s multiple comparison tests.
Results:
H-ZrO2NPs were successfully synthesized using H. root extract and functionalized with quercetin. The characterization of nanoparticles using various techniques confirmed their morphology, elemental composition, functionalization, crystallinity, size, and stability. The nanoparticles were appearing spherical in shape with a size range of 20–120 nm. The crystallinity of H-ZrO2NPs is found to be enhanced with quercetin functionalization. In vitro, bioactivity studies revealed the antioxidant and antibacterial efficacy of HQZN. The hemocompatibility of HQZN was proved by investigating its interaction with erythrocytes.
Conclusion:
The study findings revealed that the biocompatible HQZN holds notable antioxidant and antibacterial properties. On the other hand, further comprehensive investigation using an in vivo model is vital to appreciate their efficacy and toxicity profile.
Keywords
List of Abbreviations
HQZN: quercetin-functionalized zirconium oxide nanoparticles
DMSO: dimethyl sulphoxide
DPPH: 2,2-diphenyl-1-picrylhydrazyl
E. faecalis: Enterococcus faecalis
EDX: Energy-dispersive X-ray analysis
FTIR: Fourier-transform infrared spectroscopy
H. indicus: Hemidesmus indicus
H-ZrO2NPs: H. indicus root extract-based ZrO2NPs
MHA: Mueller–Hinton agar
PBS: phosphate-buffered saline
S. mutans: Streptococcus mutans
SEM: scanning electron microscopy
SPR: surface plasmon resonance
UV–Vis: UV–vis spectroscopy
XRD: X-ray diffraction
Introduction
Coatings for prosthetic implants play a pivotal role in enhancing their biocompatibility and performance. In the realm of prosthetic implants, such as dental and orthopedic applications, advanced coatings have become a focal point of research. 1 These coatings aim to improve implant-tissue interactions, reduce inflammation, and prevent microbial colonization, ultimately leading to improved implant integration and long-term success. 2
Nanomaterials have gained huge attention in medicine, biomedical research, and environmental biotechnology. The nanoparticle size ranges from 1 to 100 nm and exhibits excellent magnetic, electrical, optical, mechanical, and catalytic properties. 3 The nanoparticles can be prepared by chemical method (chemicals used as reducing agents), physical method (microemulsion, coprecipitation, etc.), and green synthesis method (using plants or microorganisms as reducing or capping agents). 4 The green synthesis method eliminates energy consumption, cost, and toxic wastes and also increases the functionality of the nanoparticles. 5 In a previous study, a plant source was used for the synthesis of nanoparticles where phytocompounds in plants acted as a reducing and stabilizing agent. 6
Zirconium is a transition metal renowned for its remarkable corrosion resistance and exceptional mechanical, thermal, and catalytic properties. 7 Consequently, it has gained widespread usage in the development of implant biomaterials and dental crowns. 8 Zirconium oxide has also exhibited significant potential in various areas, including adsorption, photo-degradation, antimicrobial activity, and structural reinforcement.9,10 A multitude of studies has highlighted the versatility of zirconium oxide nanoparticles (ZrO2NPs), which have been employed as biosensors, anticancer, antimicrobial, and antioxidant agents, as well as in implant applications.11,12 The unique physicochemical properties of ZrO2NPs make them particularly apt for biomedical applications. 13 Moreover, plant pigments that contribute to the color of fruits have been shown to possess antioxidant potential, which can bolster the immune system and mitigate the risk of illnesses such as cataracts, cancer, and macular degeneration. 14
Quercetin is a powerful antioxidant and is also known to play a role in cell-cycle progression and cellular signal transduction pathway regulation. 15 However, quercetin has poor solubility, low bioavailability, poor permeability, and instability. 16 This can be overcome by coating quercetin onto nanoparticles which can improve the solubility, stability, and bioavailability. The ability of microorganisms to develop drug resistance has led to a rapid decrease in the effectiveness of the existing implants. However, nanoparticles offer a promising solution for combating microbial resistance. 17 Nanoparticles with different compositions, such as those containing metals, employ several mechanisms simultaneously to combat microbial infections. This multifaceted approach makes it difficult for microbes to develop resistance to nanoparticles because they would need to acquire multiple gene mutations to overcome these mechanisms. 5 Therefore, nanoparticles have the potential to be effective against drug-resistant microbes. Several studies have demonstrated the antioxidant activity of ZrO2NPs in both in vitro and in vivo studies. 18 In animal models, it is found that the free radical scavenging activity is done by reducing lipid peroxidation known to cause oxidative stress. 19
Therefore, in this study, we mainly focused on synthesizing quercetin-decorated Hemidesmus indicus (H. indicus) root extract-based ZrO2NPs (H-ZrO2NPs) and characterizing the nanoparticles using various techniques. Further, this study aimed to investigate the biological activities of nanoparticles.
Materials and Methods
Collection of Sample
The H. indicus roots used for extract preparation were collected from a village in Kanyakumari, Tamil Nadu, India, and authenticated by Dr R. Siva, the botanist from Shrimathi Devkunar Nanalal Bhatt Vaishnav College for Women, Chennai.
Chemicals and Reagents
Zirconium isopropoxide (99%) is procured from Sigma-Aldrich India. 2,2-Diphenyl-1-picrylhydrazyl (DPPH), quercetin, and standard antibiotic disks were obtained from HiMedia. Analytical grade reagents were used in this current preparation. MilliQ water was used throughout the study.
Preparation of H. indicus Root Extract
H. indicus roots were cut into pieces, and the residual impurities were removed by repetitive washing under distilled water and dried at room temperature. The dried roots were pulverized into a coarse powder. About 2 g of coarse H. indicus root powder was heated with 50 mL of double distilled water for 30 min at 80°C. The extract was then filtered and used for H-ZrO2NP synthesis.
Preparation of H-ZrO2 NPs
H-ZrO2 NPs were prepared by adding 3 mL of zirconium isopropoxide into 30 mL of ammonia–root extract mixture (1:3) slowly with stirring. The precipitate formed followed by 2 h of stirring at 60°C was separated using centrifugation at 8,000 rpm for 10 min. The pellet was purified with acetone and dried overnight at 70°C.
Functionalization of H-ZrO2 NPs with Quercetin
H-ZrO2 NPs were pegylated prior to quercetin functionali-zation. About 200 mg of H-ZrO2 NPs was added into the aqueous PEG 4000 (0.05%) solution and stirred for another 1 h at room temperature. The quercetin solution in DMSO (20 mg/mL) was added dropwise to pegylated H-ZrO2 NPs under stirring at 1,200 rpm. Then, the mixture was stirred further for 30 min and centrifuged at 8,000 rpm for 10 min. The pellet collected from the centrifuged sample was freeze-dried under vacuum and used for further studies.
Characterization of Nanoparticles
H-ZrO2NPs and HQZN were subjected to various characterization techniques. The UV–visible spectroscopy (UV–Vis) spectra of the HQZN and H-ZrO2NPs were recorded using a UV–visible spectrophotometer (Jasco, Japan). The Fourier-transform infrared spectroscopy (FTIR) spectra of HQZN and H-ZrO2NPs were obtained in the region of 4,000–500 cm−1 using Fourier-transform infrared spectrometer (Bruker IR, Germany) in ATR mode. The X-ray diffraction (XRD) pattern of NPs was recorded in the range of 20°–80° at 0.05°/s scan rate using an X-ray diffractometer of typical Cu–Kα radiation (λ = 1.5406 Å). The surface morphology was explored with scanning electron microscopy (SEM) (JEOL JSM–IT800 SEM, Tokyo). The particle size distribution was determined from the SEM image using ImageJ software. An energy-dispersive X-ray (EDX) spectrum was accomplished using a JSM–IT800 SEM instrument with a silicon drift detector (Oxford X-MaxN 50 mm2, Oxford Instrument, UK).
Antimicrobial Assay
Antibacterial activity of H-ZrO2 NPs and HQZN was evaluated against Streptococcus mutans (S. mutans) and Enterococcus faecalis (E. faecalis) using agar well-diffusion technique as per the report published earlier. 20 The Mueller–Hinton agar (MHA) was prepared (pH 7.0) and sterilized for 15 min at 121⁰C in an autoclave. The sterile MHA was then poured into the petri plate and solidified at room temperature inside the laminar flow. The MHA plate was swapped with the suspension of the freshly prepared culture containing 10 6 cfu/mL of the bacterial strain. The three wells (9 mm in diameter) were made into the MHA plate and filled with standard antibiotic disk along with different concentrations (100 and 25 μg/mL) of H-ZrO2 NPs and HQZN. The plate was kept at room temperature for 4 h to allow the compounds to diffuse into the medium and then incubated at 37°C for 24 h. After incubation, the diameter (mm) of the zone of inhibition was measured from each well and compared with that of the standard.
In Vitro Antioxidant Activity
The antioxidant activity of the H-ZrO2 NPs and HQZN was assessed by quantifying the DPPH absorbance at 517 nm.21,22 About l.5 mL of 0.1 mM methanolic DPPH solution was mixed with different concentrations of H-ZrO2 NPs and HQZN. The mixture was shaken vigorously and kept intact for 20 min at 27°C. Then, the absorbance was quantified at 517 nm. The antioxidant activity was determined using the formula as follows:
Hemolytic Assay
The hemocompatibility of H-ZrO2NPs and HQZN was determined by calculating the hemoglobin concentration released from the erythrocytes treated with H-ZrO2NPs and HQZN. The hemolytic assay was performed as per previously reported studies.
23
The erythrocytes (RBCs) were separated from the whole human blood centrifuged at 1,500 × g for 5 min and washed thrice with phosphate-buffered saline (PBS) (pH 7.4) to remove the impurities. RBCs were diluted to 10% of their initial concentration after washing with PBS. To the different nanoparticle concentrations (12.5, 25, 50, 100, and 200 µg/mL), 200 µL of erythrocyte suspension was added, and the final volume of the suspension was made up to 1 mL to the using PBS. The suspension was incubated for 1 h at 37°C and centrifuged for 5 min at 1,500 × g. The supernatant of the centrifuged mixture was collected, and their absorbance was measured at 540 nm. PBS-treated cells were considered as a negative control, and cells treated with distilled water were considered as a positive control. The experiment was done thrice, and the hemolysis percentage was calculated using the following equation:
24
Statistical Analysis
The experiments were carried out in triplicates and the data expressed as mean ± SD (n = 3) were subjected to statistical analysis using GraphPad Prism 6.0 software. Data from various time points were interpreted using one-way ANOVA followed by Dunnett’s multiple comparison tests. The statistical significance standard was fixed at P < .05.
Results
In the current study, we have synthesized H-ZrO2 NPs and functionalized the nanoparticles using quercetin. The nanoparticles were validated using different characterization techniques. The biocompatibility of the nanoparticles was tested by performing the hemolytic assay. The antioxidant and antimicrobial potential of the nanoparticles was determined using in vitro studies.
Characterization of HQZN
Figure 1 represents UV–visible spectra of H-ZrO2NPs and HQZN that displayed a characteristic surface plasmon resonance peak at 285 and 278 nm, respectively. In addition, the UV–visible spectrum of HQZN displayed a typical peak with maximum absorbance at 390 nm, evidencing the quercetin capping on H-ZrO2NPs.
UV Spectra of H. indicus extract (HI), Quercetin, H-ZrO2 NPs and HQZN.
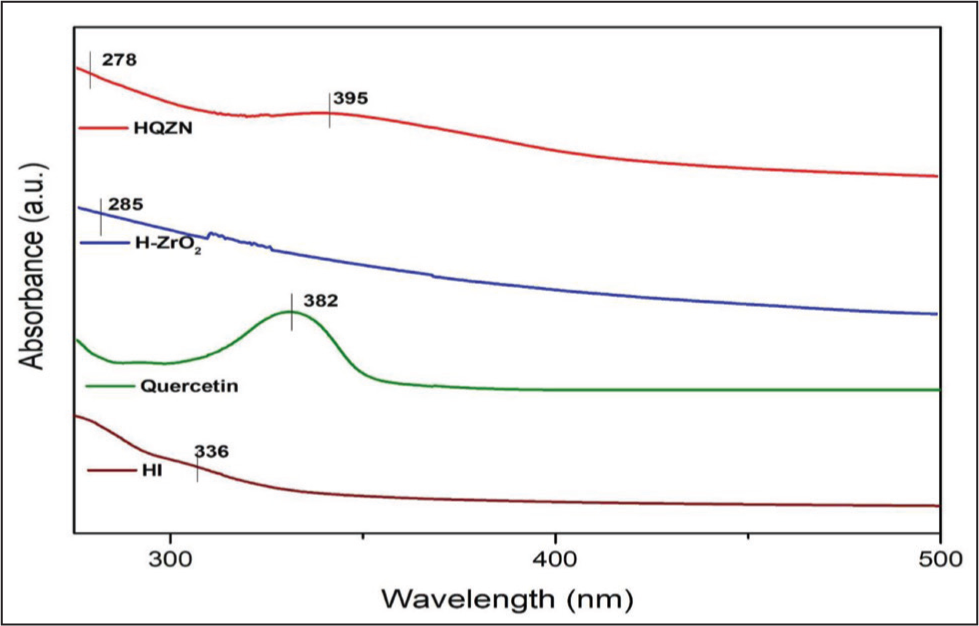
Figure 2 represents the FTIR results of the H-ZrO2NPs and HQZN recorded between 4,000 and 500 cm−1. The FTIR spectrum of H-ZrO2 NPs presented peaks at 3,394, 2,321, 1,633, 1,558, 1,341, 1,046, and 846 cm−1. The specific peak stretching at 3,394 cm−1 confirms the OH group present in the extract used for H-ZrO2 NP formation. In the same way, the FTIR spectrum of HQZN showed strong bands at 3,390, 2,320, 1,621, 1,493, 1,198, 1,097, 1,000, and 820 cm−1. Characteristic stretching at 3,390 cm−1 represents the OH group present in the extract and quercetin used for HQZN formation. The additional peaks observed at 1,493, 1,276, 1,170, and 840 cm−1 in the FTIR spectrum of HQZN, evidencing the presence of quercetin as it coincides with the spectrum of quercetin.
FTIR Spectrum of Quercetin, H-ZrO2 NPs and HQZN.
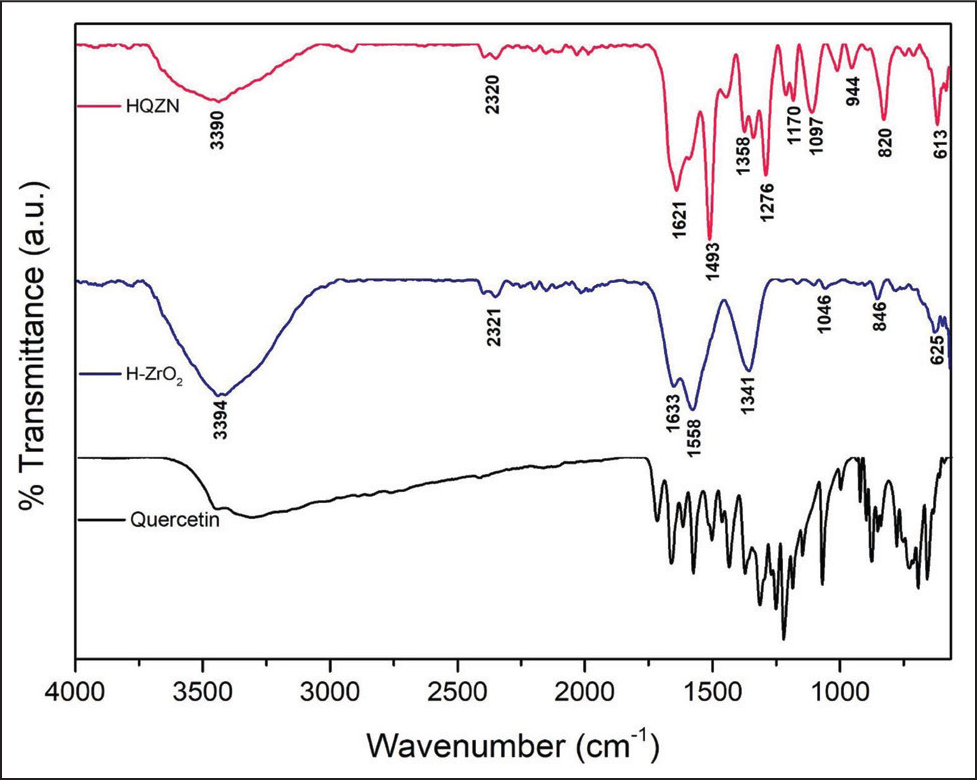
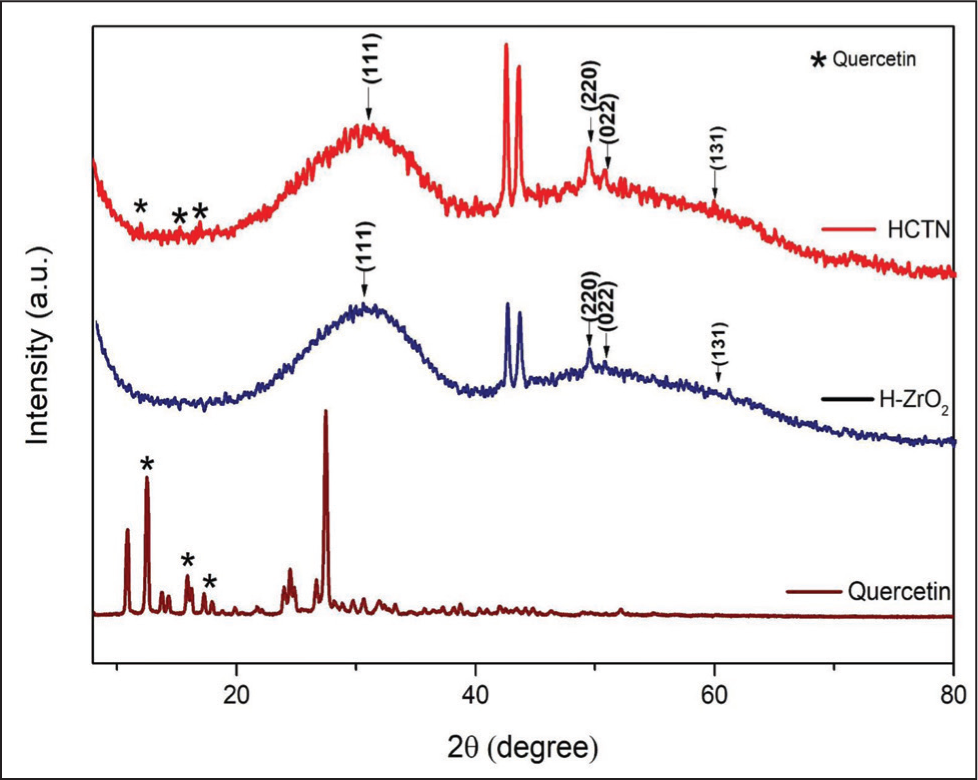
Figure 3 represents the XRD pattern of quercetin, H-ZrO2 NPs, and HQZN. The pattern displayed the amorphous nature of the NPs. Peaks were observed at 2 theta values of 30.86°, 49.3°, 50.7°, and 60.2°, in both H-ZrO2 NPs and HQZN. The positions of the peaks were in line with the standard pattern reported in the JCPDS 37-1484, which revealed that the sample exhibits a monoclinic crystal structure, consistent with zirconium oxide. In addition to the zirconium oxide peaks, HQZN showed peaks below 20° corresponding to the quercetin, indicating the successful coating of quercetin over H-ZrO2 NPs.
XRD Pattern of Quercetin, H-ZrO2, and HQZN.
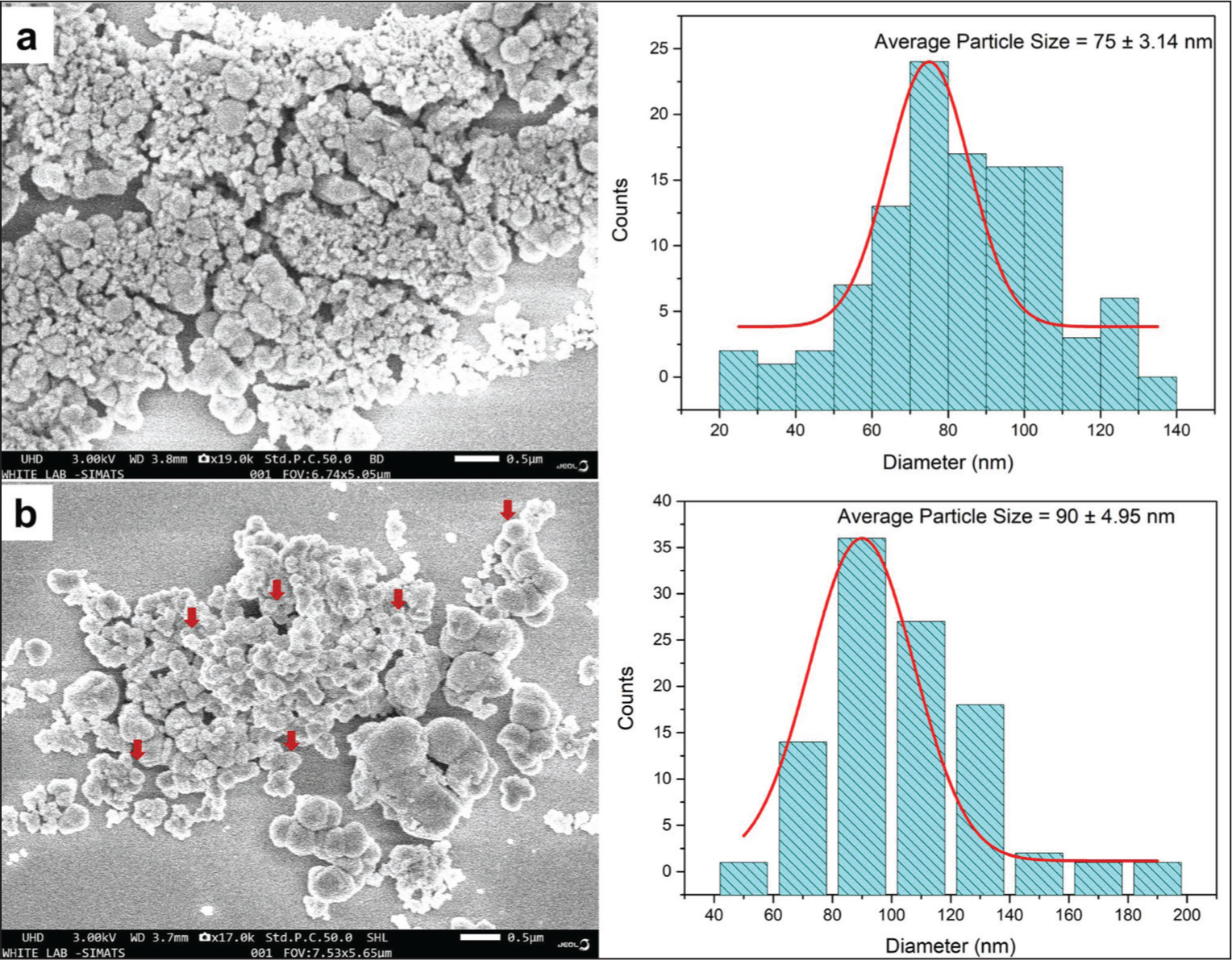
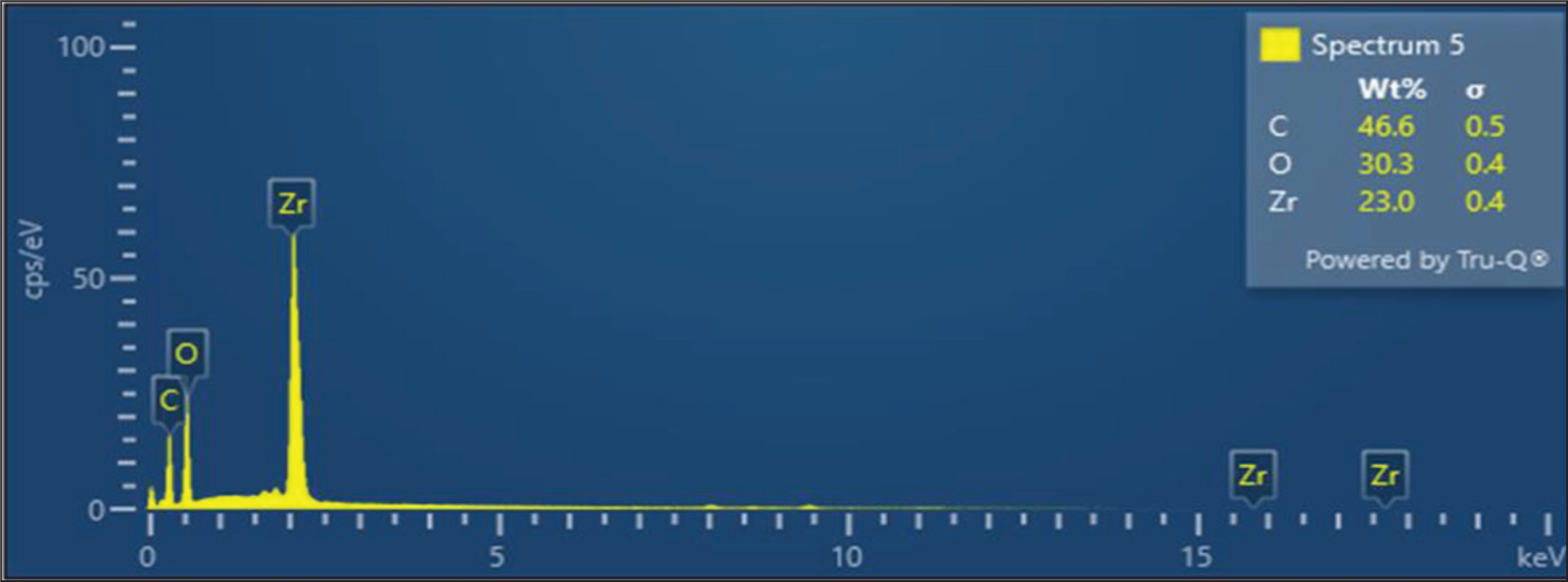
The morphology of the H-ZrO2 NPs and HQZN was determined by SEM. H-ZrO2 NPs exhibit aggregated spherical shapes with a size range of 20–120 nm. However, the quercetin coating increases the particle size to a range of 60–140 nm. Figure 4 represents the morphology and particle size distribution of H-ZrO2 NPs and HQZN. The average particle size of H-ZrO2 NPs and HQZN was found to be 75 ± 3.14 and 90 ± 4.95 nm, respectively. Figure 5 displays the EDAX spectrum of synthesized HQZN. EDAX of green synthesized HQZN showed 23 wt% of Zr, 30.3 wt% of O, and 46.6 wt% of C. The presence of higher carbon content confirms the coating of quercetin on H-ZrO2 NPs.
SEM Micrograph and Particle Size Distribution of (a) H-ZrO2 NPs and (b) HQZN. Arrows Indicating the Spherical HQZN Particles.
EDAX Spectrum of HQZN.
In Vitro Antimicrobial Assay
The antimicrobial activity testing of H-ZrO2 NPs against S. mutans and E. faecalis did not show any significant change in the zone of inhibition. The HQZN exhibits significant antimicrobial effects against S. mutans and minimal effects against E. faecalis under the current experimental conditions. Hence, further research is needed to explore potential antimicrobial properties using alternative approaches. The antimicrobial activity of H-ZrO2 NPs and HQZN against S. mutans and E. faecalis is shown in Figure 6.
Antimicrobial Studies of H-ZrO2 NPs and HQZN Against (a) S. mutans (b) E. faecalis.

In Vitro Antioxidant Activity
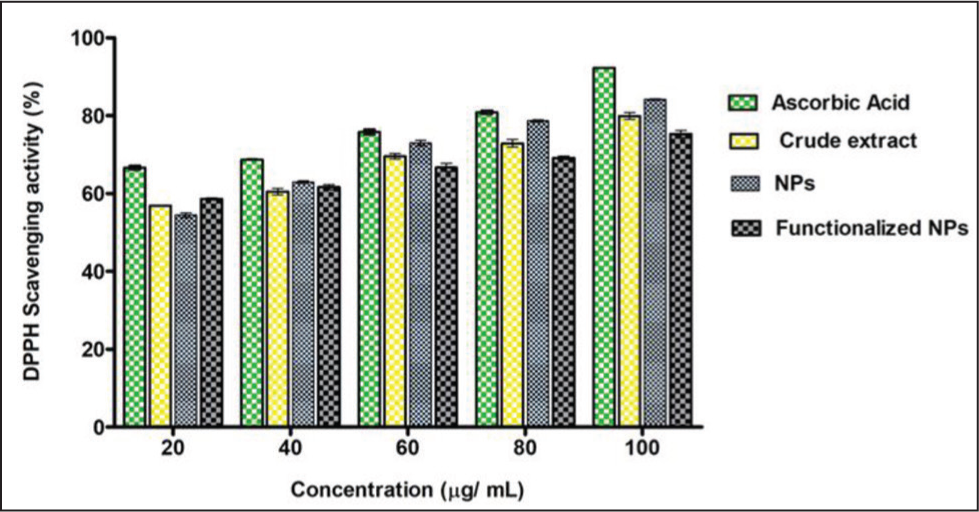
The DPPH assay was used to assess the in vitro antioxidant activity of H. indicus crude extract, H-ZrO2 NPs, and HQZN. The sample’s ability to scavenge free radicals is directly correlated with the decline in DPPH radical absorbance. A dose-dependent rise in the DPPH radical’s scavenging activity percentage was observed and at the maximum concentration tested (100 g/mL) was found to be 79.8 (1.2%) (Figure 7).
Hemolytic Assay
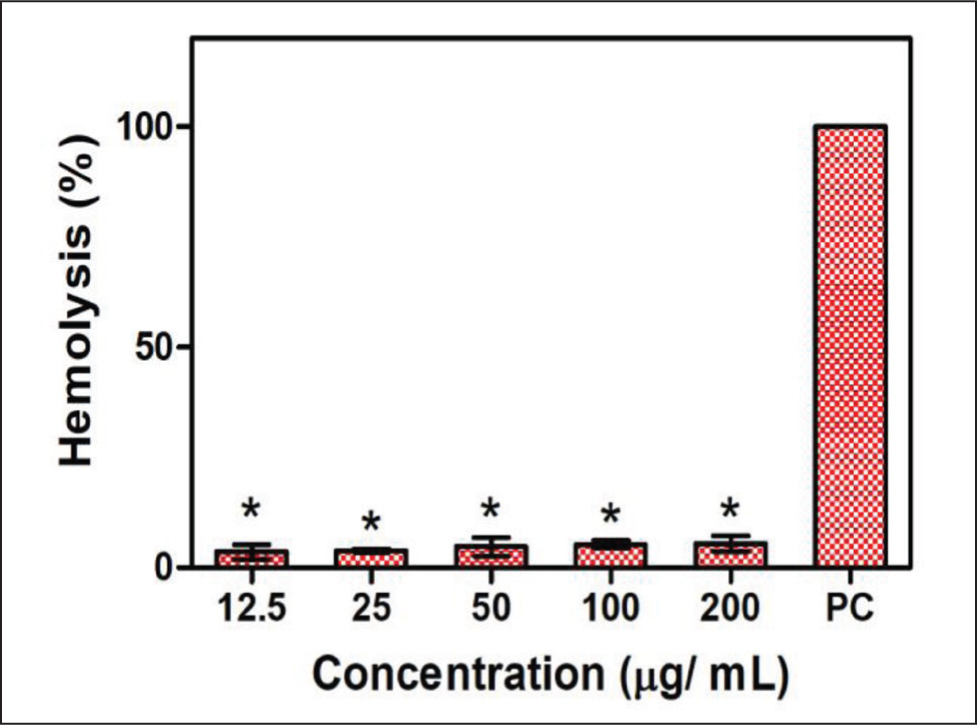
The results of HQZN (12.5, 25, 50, 100, and 200 μg/mL) treated erythrocytes presented less than 5% hemolysis at various concentrations compared to the control. Figure 8 shows the hemolytic results of HQZN toward erythrocytes.
Discussion
The present study aimed to synthesize HQZN by using H. indicus root extract and quercetin as capping agent and investigate their in vitro antioxidant, hemolytic, and antimicrobial activities. UV–Vis spectra of H-ZrO2 NPs and HQZN, had peaks at 285 and 278 nm, respectively. This finding is supported by previous studies on ZrO2 NPs synthesized via hydrothermal method showing maximum absorption peak at 285 nm. 25 The quercetin functionalization on the surface of H-ZrO2 NPs is responsible for the additional peak at 390 nm. Our previous on quercetin-coated silver nanoparticles showed maximum absorbance at 390 nm is in line with our current findings. 26 This indicates that quercetin was successfully functionalized on the surface of H-ZrO2NPs.
The characteristic FTIR peaks of H-ZrO2NPs and HQZN were connected with the study reported by Sagadevan et al. in 2016. 27 The FTIR spectrum of H-ZrO2NPs showed that a typical peak at 3,394 cm−1 corresponds to N–H stretching of aliphatic primary amine. The peak at 2,169 cm−1 corresponds to strong S–CΞN stretching of thiocyanate. The peak at 1,633 cm−1 relates to strong C=C stretching of alkene. The stretching peak at 1,558 cm−1 displays strong N–O stretching of the nitro compound present in the extract. The peak at 1,341 cm−1 resembles the absorbed CO2 molecules. 25 The peak at 613 cm−1 represents the Zr–O stretching. A previous report by Sagadevan et al. in 2016 suggested that 680 cm−1 corresponds to Zr–O vibration of ZrO2 with a tetragonal structure, which is correlated with our results.25,27 Similarly, the FTIR spectrum of HQZN showed a characteristic peak at 3,390 cm−1 that corresponds to medium N–H stretching of aliphatic primary amine. The peak at 1,621 cm−1 corresponds to medium C=C stretching of conjugated alkene. The peak at 1,198 cm−1 represents the medium C–N stretching of amine. The C–O stretching of the secondary alcohol was represented by the peak at 1,097 cm−1. The peak at 1,000 cm−1 corresponds to strong C=C bending of alkene. The peak at 613 cm−1 corresponds to strong Zr–O stretching.25,28 Overall, the FTIR spectra of H-ZrO2NPs and HQZN confirmed the presence of different secondary metabolites of the H. indicus and quercetin, which act as a capping and stabilizing agent.
Peaks at 2 theta values of 30.86°, 49.3°, 50.7°, and 60.2°, in both H-ZrO2 NPs and HQZN, correspond to the (111), (220), (022), and (131) crystallographic planes of monoclinic ZrO2. These findings align with previous research, validating their correlation with earlier studies.29,30 Moreover, the peaks with 2 theta values of 11.9, 14.88, and 16.79° observed at the HQZN pattern evidenced the quercetin coating. The broadening at 2 theta value of 30.86° in the pattern is mainly due to the higher amorphous nature of the particles. 31 The crystallinity of H-ZrO2 NPs and HQZN is found to be 2.4% and 3.1%, respectively. The quercetin coating increased the crystallinity of H-ZrO2.
The H-ZrO2 and HQZN had a uniform spherical morphology with an average particle size range of 20–120 nm as seen in the SEM images, which is in line with earlier research indicating a correlation with previous findings. 32 The nanoparticles were evenly disseminated and clumped together. The creation of well-defined nanoparticles with homogeneous sizes and shapes is confirmed by the SEM analysis. The particle size was further confirmed by calculating the particle size distribution from the SEM micrograph image. Dan et al.’s study of spherical-sized ZrO2 particles with 80 nm using the hydrothermal-assisted sol–gel method support our findings. 33 The existence of Zr, O, and C components was confirmed by EDX analysis of the HQZN, demonstrating quercetin’s successful functionalization of the H-ZrO2 NP surface.
The H-ZrO2 NPs and HQZN were nonhemolytic up to a concentration of 200 g/mL, which correlated according to the results of the hemolytic assay of the previous study by Wang et al. 29 The nanoparticles are, thus, safe suggesting their biocompatibility and safety as coatings for prosthetic dentures. The H-ZrO2 NPs and HQZN showed considerable antioxidant efficacy when compared with that of the ascorbic acid standard which is essential for improving the longevity and biocompatibility of prosthetic dentures. This study’s findings are corroborated by previous research, highlighting their correlation with earlier studies. 34 Despite conducting thorough antimicrobial activity tests on S. mutans and E. faecalis, minimal changes in the zone of inhibition were observed. These negative results suggest that the extract did not exhibit significant antimicrobial effects on the tested microorganisms under the specified experimental conditions. Further research with alternative approaches is warranted to explore the possibility of antimicrobial properties for the extract against these specific microorganisms.
Conclusion
The present study promotes the green synthesis of ZrO2NPs using H. indicus root extract. The characterization of H-ZrO2 NPs and HQZN was done using various techniques. The hemolytic assay revealed the biocompatibility of HQZN underscoring their potential in curbing bacterial colonization on dental prosthetic surfaces. The successful development of HQZN represents a significant advancement in dental implant technology, offering a pathway to enhance both the performance and biocompatibility of dental prosthetic materials. These findings hold great promise for advancing dental implantology, paving the way for safer and more effective dental prosthetic coatings. Continued research and clinical investigations are warranted to fully unlock the practical applications of HQZN in the realm of dental prosthetic implantology.
Footnotes
Acknowledgements
The authors extend their sincere gratitude to the Saveetha Dental College and Hospitals for their constant support and successful completion of this work.
Author Contributions
Conceptualization, A.P.F., R.G.; study design, S.F.P., A.P.F., K.S.; writing–original draft preparation, S.F.P., K.S.; writing–review and editing, G.R., V.V.P., A.P.F.; project administration, A.P.F.
Data Availability Statement
Data are available on request from the authors.
Declaration of Conflicting Interests
The authors declare that there is no conflict of interest.
Ethical Approval
Not applicable
Funding
This work is supported by Shereen Cotton Center, Chennai and Saveetha Dental College and Hospitals, Saveetha Institute of Medical and Technical Sciences, Chennai.
Informed Consent
Not applicable