Abstract
Aim:
Factors such as the low pH of acidic foods and beverages can affect long-term clinical success of restorative materials in the oral cavity. The aim of this study was to compare the solubility of Bulk-fill and Conventional Nano-Hybrid Resin Based Composites (RBCs) in different solutions.
Material and Methods:
A total of 60 cylindrical specimens were prepared from two RBCs (Tetric N-Ceram, Tetric N-Ceram Bulk-fill) and their constant weight was measured. The specimens were divided into six groups (N = 10) in accordance with RBC type and pH solution (solution 1, 2, 3 with pH = 2.5, 5, 7, respectively), and were immersed in desired solutions for 30 days. Then they were weighed again and solubility levels of the specimens were calculated according to ISO 4049. Data were analysed by two-way ANOVA. The test power was considered at 80%.
Results:
The type of RBC had no significant effect on solubility; while the effect of PH was significant. Solution 1 caused significantly the highest solubility. No statistically significant differences were found among other groups.
Conclusions:
Considering the limitations of the laboratory investigations, it was concluded that solubility rate of Bulk-fill and Conventional RBCs was higher in solutions with lower pH. There was no difference among the RBCs. However, the solubility rates of all groups are within the clinically normal range.
Introduction
Resin Based Composites (RBCs) are widely used in dentistry 1 and considered as a suitable restorative material due to good intrinsic properties, adhesion to the tooth structure and medium price. 2 Nevertheless, polymerization shrinkage and depth of cure are still considered clinical challenges. In addition to being time consuming, incremental layering technique may cause air entrapment, blood and saliva contamination and bond failure between layers. Bulk-fill RBCs were evolved to overcome these problems. 3 Unlike conventional RBCs, which are placed in layers of up to 2 mm, bulk-fills are designed to be placed in more than 4 mm thickness. 1 Since the introduction of bulk-fill RBCs, many efforts have been made to compare the differences between conventional and bulk-fill RBCs and contradictory outcomes have been reported.4,5
The clinical success of restorative materials is not only influenced by their chemical structure but also by the oral environment. Recent studies have shown that acidic conditions in the oral cavity, such as acidic beverages, stomach acid, bacterial activity, or taking certain medications tend to degrade RBCs6,7 which can lead to the collapse of RBC polymer matrix, filler-polymer debonding and release of unpolymerized monomers. This process can affect mechanical and physical properties of restorative materials. 6
Water sorption and solubility may influence aesthetic, integrity, and surface properties of restorative materials. 8 Solubility may affect RBCs by two mechanisms: (a) Uptaking of water leading to increased weight. (b) Dissolution of fillers or monomers in water causes weight loss. 9 On the other hand, some substances released from restorative materials during dissolution, can be potentially irritating and cause delayed allergic reactions. 10
Earlier studies showed that the type of filler and resin matrix has a significant effect on the solubility of RBCs.11–13 Since chemical structure of bulk-fill is different from conventional RBCs, their solubility in acidic environments may also be different. However, studies on the solubility of bulk-fill RBCs are limited. Misilli T evaluated water sorption and solubility of bulk-fills after curing with different modes of a polywave light cure and found that bulk-fills exhibited significantly lower water sorption but the highest solubility values were obtained for a bulk-fill. 14 Another research showed that Acidic drinks may negatively affected on physical and mechanical properties of bulk-fill RBCs. 15
Therefore, this study was conducted to compare the effect of pH on the solubility of bulk-fill and conventional RBCs. The null hypotheses were that (a) there would be no significant difference in the solubility of three different solutions, and (b) there would be no significant differences between solubility of conventional and bulk-fill RBCs.
Methods
Specimen Preparation
This study has been approved by the Research Ethics Committee of Shahid Sadougi University of Medical Sciences (No: IR.SSU.REC.1399.150).
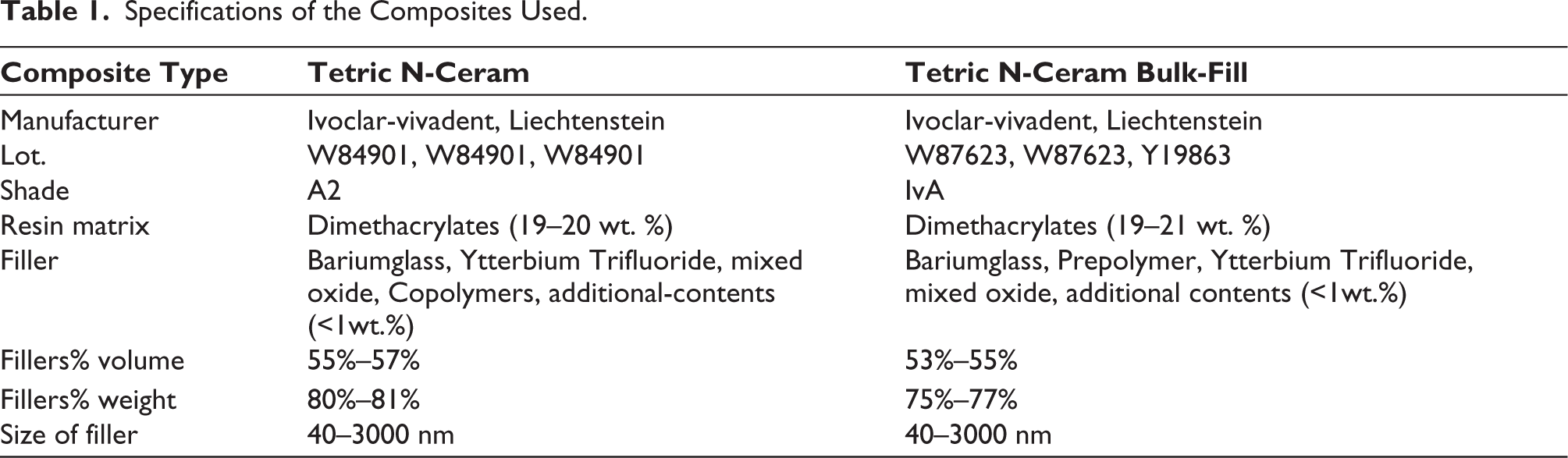
In this in-vitro study, for each nano-hybrid RBC, Tetric N-Ceram (TNC) and Tetric N-Ceram Bulk-fill (TNCBF), 30 specimens were prepared. The specifications of the RBCs used are given in Table 1.
Specifications of the Composites Used.
To prepare specimens, RBCs were manipulated in a Teflon cylindrical mold (4 mm height and 6 mm diameter) and cured using an LED light-curing device (Litex695, Dentamerica_Taiwan) with 1200 mW/cm2 light intensity. Bulk-fill and conventional composites were applied in one step and 2 mm-thick layers, respectively. The specimens were initially polymerized for 40s followed by irradiation of four overlapping peripheral sections for 20s each. The specimen surfaces were wet-grinding using a finishing disc (Tor-VM, Russia). Finally, the specimens were rinsed with water and kept in distilled water at 37°C for 24 hours completing the polymerization process.
Solubility Test
Solubility tests were performed in line with ISO4049. Specimens were inserted in a vacuum oven (Napco, USA) at 37°C and vacuum pressure of 21 mm-Hg for 22 hours. Then they were placed in a desiccator containing silica-gel at 23°C for 2 hours. Specimens were then weighed using a digital balance (NanoPajouhan Raga, Iran) accurate to 0.0001 g. This 24-hour cycle (22 hours at 37°C, 2 hours at 23°C, weight measurement) was repeated continuously (about 14 times) until a constant weight of each specimen was obtained, that is, the weight difference between the two cycles was not more than 0.0001g. This weight was considered as m1 (μg). Also, the volume of each specimen was calculated in mm3 using m1 and density of the RBCs (2.064 g/cm3 for TNCBF & 2.16 g/cm3 for TNC).
The specimens of each RBC were then randomly divided into three groups according to immersion solution. Therefore, in this study there were a total of the following six groups (n = 10):
Group 1: TNC disc immersed in distilled water (pH = 7) Group 2: TNC disc immersed in solution at pH = 5 Group 3: TNC disc immersed in solution at pH = 2.5 Group 4: TNCBF disc immersed in distilled water Group 5: TNCBF disc immersed in solution at pH = 5 Group 6: TNCBF disc immersed in solution at pH = 2.5
The composition of acidic solution is given in Table 2. Each specimen was kept in numbered tubes containing 10 ml of solution in an incubator at 37°C for 30 days of challenge (three periods of 15 min/d). After 15 minutes of storage in each acidic solution, the excess was rinsed with distilled-water. Due to the buffer nature of the prepared solutions, their pH remains almost constant and does not change over time. 16 However, the pH solutions were tested daily using a pH-meter (86502 AZ, Taiwan), and solution be refreshed if pH was changed.
After 30 days, the specimens were cleaned and constant weight was measured under the conditions mentioned earlier. This weight was considered as m2. The solubility rate of each specimen was obtained with the following formula:
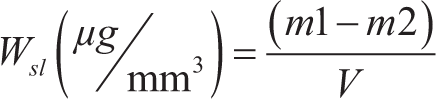
In which, v is the specimen volume in cubic millimetre (mm3). m1 and m2 are the constant weights before and after immersion of the specimen in micrograms (μg), respectively.
Statistical Analysis
SPSS23 software was used to analyse the data. The normality of the solubility values was checked by Kolmogorov–Smirnov test. Two-way ANOVA statistical analysis was used to investigate the effect of two variables on the solubility rate of RBCs. Comparisons between different pHs were performed by Tukey analysis. The statistical significance was considered at p<.05.
Results
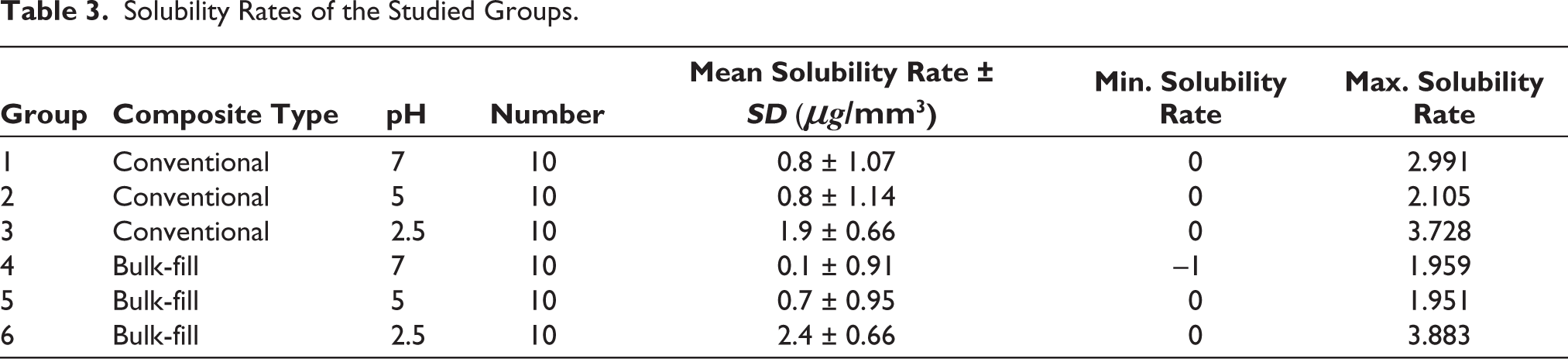
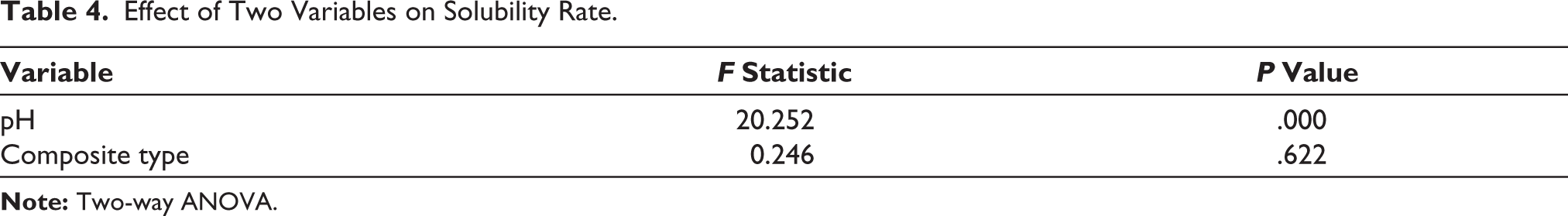
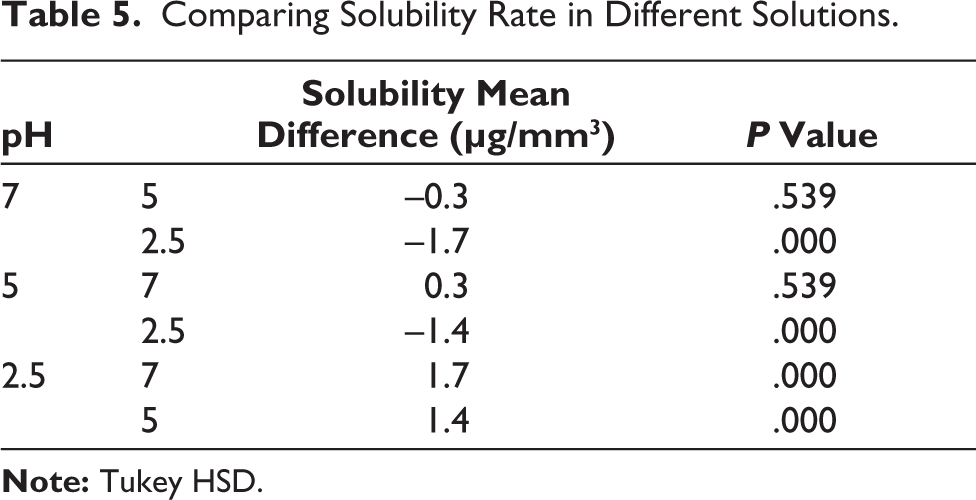
Solubility rates of the studied groups were summarized in Table 3. The maximum and minimum solubility rate was related to group 6 and group 4, respectively. The Kolmogorov–Smirnov test showed normal distribution of data (p value = .065). Based on the results of Two-way ANOVA (Table 4), the type of RBC had no significant effect on solubility (p value = .622); While the effect of pH was significant (p value<.001). Tukey analysis showed that the solubility at pH = 2.5 was significantly higher than two others (p value<.001), but there was no significant difference between pHs 5 and 7 (p value = .539) (Table 5).
Composition of Solutions Used in Study.

Solubility Rates of the Studied Groups.
Effect of Two Variables on Solubility Rate.
Comparing Solubility Rate in Different Solutions.
Discussion
As mentioned by the results, the first hypothesis was rejected because the solubility at pH = 2.5 was significantly different from pHs 5 and 7. The second null hypothesis was confirmed because there were no significant differences between solubility of conventional and bulk-fill RBCs.
In oral environment, composite restorations are subjected to continuous or intermittent acidic attacks. pH is the most important factor in erosive potential of acidic beverages and foods. It has been shown that the pH of beverages determines their erosive potential in the first minutes of exposure. The pH of the drinks tested in one study ranged from 2.43 for Coca-Cola to 3.75 for fruit juices.16–19 Also, the pH of stomach acid is about 1–1.5. 20 Furthermore, critical PH for fluorapatite and hydroxyapatite is 4.5 and 5.5, respectively. Therefore, according to similar previous studies21,22 and to simulate acidic conditions of oral environment, in the present study, pH = 2.5 and 5 were used.
Effect of Composite Type on Solubility Rate
The chemical degradation of RBCs is affected by their monomers and degree of crosslinking. Also, the type and volume of filler affect water sorption and solubility. 23 The presence of OH groups in the Bis-GMA molecule are responsible for the water absorption and solubility properties of RBCs. 24 Many researchers have found that low-adsorption materials also have low solubility. 25 When water infiltrates in organic matrix, expands the spaces between polymer crosslinks and changes polymer dimensions. The unreacted monomers are dissolved in the solvent and may be removed from the polymer mass. Therefore, expansion and leaching processes occur through the diffusion of water into the polymer. Monomers trapped in micropores have been reported to be more prone to leaching than those in microgels; So, leaching of monomers may be affected not only by the amount of unreacted monomers but also by the distribution of the monomers within a polymer. 26
The results of the present study showed that there is no significant difference in the solubility of conventional and bulk-fill RBCs. Resin matrix of the two RBCs was similar. Although previous studies have shown that at 4 mm thicknesses, the degree of curing of TNCBF is higher than TNC. 27 But in this study, since curing process is done from four directions after removing specimens from the mold, depth factor can be ignored and so the polymerization of both RBCs was probably similar. Furthermore, bulk-fill RBC contains less filler than conventional nano-hybrid RBC. 28 Therefore, lower filler percentage may be the reason for the higher solubility of TNCBF at pH = 2.5 than TNC, whereas the difference was not statistically significant.
Comparison of different studies is difficult as a result of differences in type of RBCs, storage solution, storage time and specimen size. 29 Mansouri et al., also showed that water solubility of bulk-fill RBC is slightly lower than conventional, however, this difference was not significant. 26 But the results of the study by Amer. Tiba et al. showed that water solubility of bulk-fill RBCs is significantly lower than conventional. 30 Also, N. Gonulol et al. found that the solubility of bulk-fill RBCs is less than conventional. They attributed the lower water sorption of bulk-fill RBCs to their higher filler-loading, which increases the filler weight ratio, and in turn requires a smaller polymer matrix. 11 Melo RA et al. found that bulk-fill RBC performed better in terms of solubility than conventional. Although the reason for this was not evaluated, the author speculates that the bulk-fill RBC has a higher degree of conversion than the conventional. 28
In the present study, negative solubility was obtained in only one group (TNCBF in solution with pH = 7). negative solubility values can be interpreted as the solubility of materials occurring at low levels, or not occurring at all. 31 Lopes LG et al., reported that negative solubility may occur when RBC is more sensitive to water absorption, which increases the weight and may cover the actual solubility. 32
Effect of Solution pH on Solubility Rate
The current study concluded that pH has a significant effect on solubility; whereas the solubility rate at pH 2.5 is significantly higher than pH 5 and 7, solubility rate at pH 5 and 7 is not statistically different.
Kedici Alp showed that food-simulating liquids such as citric acid and ethanol were effective on surface features of single-shade universal composites. 33 In another study, universal composites were exposed to simulated gastric acid, which resulted in increased surface roughness and decreased microhardness. 34
Lower pHs have been shown to have a negative effect on the wear resistance of RBCs. However, the effect of pH on water sorption and solubility of RBCs has not been sufficiently studied. 23 Eriwati et al. have reported that the amounts of water uptake and solubility of zinc phosphate cement, conventional GIC, RMGIC and RBCs increased after immersion in artificial saliva at pH = 5. 17 The results of the study by Soliman TL et al. showed that the Z100 hybrid restorative material has an increased solubility rate in solutions with pH 5 and 2.5 compared to control group (distilled water). They also found that there was no significant difference between pH 5 and distilled water in the amount of solubility of silorane-based restorative materials. 21 Ortengren et al. investigated the effect of pH and storage time on the sorption and solubility of three RBCs (Spectrum, Z100, ArtGlass). From the results of their study, it seems that pH affects the sorption and solubility behaviour of RBCs, which may be related to the hydrophilicity of the matrix and the chemical composition of the filler. 23
Acidic conditions can increase the plasticity of resin components and weaken the bond between polymer chains in dimethacrylate matrix, which may cause monomer debonding and increase the solubility of restorative materials. 17 It has been reported that the erosive potential of a solution not only depends on pH but also on titratable acidity, exposure time and temperature, type of acid and mineral content. 35 Therefore, previous studies have suggested that after composite restoration, dentist ask the patient to maintain oral hygiene and have a low-sugar diet. Otherwise, long and frequent periods of low pH may result in the solubility of resin composite. 36
According to ISO4049, which is one of the most reliable standards, 31 RBCs as restorative materials must have a maximum value of 7.5 μg/mm3 for water solubility during a week of storage in water. 9 In the present study, solubility values of all groups are in the standard range; Therefore, although there are statistical differences between the study groups, their clinical performance may not be different.
In this study, it was not possible to evaluate the buffering effect of saliva. Also, in laboratory study, all surfaces of a specimen are exposed to solutions which are greater than exposed surfaces of a restoration in the oral environment. Therefore, the results of the laboratory study cannot exactly simulate the clinical condition, so clinical studies are needed to obtain more reliable results. 28
Conclusion
Considering the limitations of the laboratory research, it was concluded that the solubility rate is not affected by the type of composite (Tetric N-Ceram and Tetric-N Ceram Bulk-fill) but the pH has a significant effect on the solubility rate. However, the solubility rates of all groups are within the clinical normal range.
Footnotes
Acknowledgements
We would like to thank the Shahid Sadoughi University of Medical Sciences for Financial support. Also, the authors would like to thank Mr Alireza Chapzadeh for sharing their pearls of wisdom with us during this research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
The study has been approved by the Research Ethics Committee of Shahid Sadoughi University of Medical Sciences (No: IR.SSU.REC.1399.150).
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed Consent
The study was done in an experimental environment and samples were dental resin composite, so patient constant was not required.
