Abstract
Purpose:
There is a pressing need for safe venous thromboembolism (VTE) prophylaxis in orthopedic patients with the highest risks of both venous thrombosis and bleeding. Portable intermittent pneumatic compression device (IPCD) has proven to be effective and safe in patients with a high risk of venous thrombosis and low bleeding risk. Therefore, this study examined the effectiveness, safety, and wearing compliance of portable IPCD for postoperative VTE prophylaxis in patients with the highest risks of both venous thrombosis and bleeding.
Methods:
The cases consisted of 38 patients who had used a portable IPCD and had the highest risks of both venous thrombosis and bleeding. We examined the incidence of VTE to assess the effectiveness of the portable IPCD, the presence of hemorrhagic adverse events to assess safety, and the wearing rate to assess wearing compliance.
Results:
The incidences of asymptomatic and symptomatic deep vein thrombosis were 5.3% and 2.6%, respectively. The incidence of hemorrhagic adverse events was 21.1% in patients who received anticoagulants and wore an IPCD simultaneously and 0% in patients who wore an IPCD but did not receive anticoagulants. The wearing rate (i.e. ≥18 h/day) was 100%.
Conclusion:
Portable IPCD has the potential for safe VTE prophylaxis in patients at high risks for both venous thrombosis and bleeding. Therefore, we suggest that such patients use a portable IPCD for VTE prophylaxis.
Keywords
Introduction
Venous thromboembolism (VTE) is a serious complication leading to death after orthopedic surgery. Patients with total hip arthroplasty (THA) have a higher short-term mortality rate within 90 days compared to the general population, and the cause is reported to be VTE. 1 Without VTE prophylaxis, the incidence of VTE after orthopedic surgery is reported to be 22.8–60.0%. 2 –4 Therefore, VTE prophylaxis after orthopedic surgery should be made priority.
The method of VTE prophylaxis should be chosen based on the patient’s risk. 5 Therefore, for the individual assessment of VTE, our institution developed and introduced the computerized clinical decision support system for VTE and bleeding risk assessment (CLI-DES) to all departments of our hospital (details are described in “Material and Methods” section). 6 On the other hand, for patients at a high risk for VTE, a combination of physical and pharmacological prophylactic methods is recommended by the 9th American College of Chest Physicians Guidelines (ACCP) 7 and the Japanese guidelines. However, while anticoagulants are effective against VTE, they are avoided or cannot be used by patients at a high bleeding risk because of concerns of hemorrhagic adverse events. 8 –11
Physical prophylaxis results in fewer hemorrhagic adverse events than pharmacological prophylaxis. 7 Physical prophylaxis includes active ankle exercise, graduated compression stockings, and intermittent pneumatic compression device (IPCD). Graduated compression stockings are reported not to have a high preventive effect on their own in patients with a high risk of venous thrombosis. 12 The ACCP guidelines for VTE physical prophylaxis recommend only using a portable IPCD. 7 A randomized controlled study of patients with total joint arthroplasty found that using a portable IPCD reduced the incidence rate of VTE. 13,14 However, combining IPCD with pharmacological prophylaxis does not further reduce the risk of deep vein thrombosis (DVT). 15 Thus, compared to pharmacological prophylaxis, continuously wearing a portable IPCD for at least 18 h a day is an equally effective and safer alternative.
Despite the pressing need for safe VTE prophylaxis in patients with the highest risks of both venous thrombosis and bleeding, no effective and safe VTE prophylaxis has been reported for such patients. We thought that the portable IPCD would be useful for preventing VTE in patients with the highest risks of both venous thrombosis and bleeding, but the effectiveness, safety, and wearing compliance of the portable IPCD in these patients remain unknown. Therefore, we evaluated VTE prophylaxis with a portable IPCD in patients who have undergone lower extremity orthopedic surgery and have the highest risks of both venous thrombosis and bleeding. Specifically, we evaluated the effectiveness, safety, and wearing compliance associated with the use of a portable IPCD in these patients.
Material and methods
We conducted this case series trial at our hospital in Japan. All 1259 patients who underwent lower extremity orthopedic surgery between 2015 and 2017 were potentially eligible. Among them, we enrolled 38 patients, who had the highest estimated risks of venous thrombosis and bleeding based on CLI-DES and used a portable IPCD for VTE prophylaxis. 6 CLI-DES is a decision-making system that functions in three stages. First, the VTE risk level is categorized in each department of surgery. Second, the system automatically sums the points for additional risk factors (e.g. background factors and patient history) to determine the VTE risk level. Finally, the VTE risk level and recommended VTE prophylactic methods are displayed. This study was approved by the ethics committee of our institutions (no. B17-169) and was conducted in accordance with the principles of the Declaration of Helsinki and the good clinical practice guidelines. Given the retrospective study design, we adopted an opt-out form for informed consent.
All data in this study were collected from electric medical records. The portable IPCD used in this study was ActiveCare + S.F.T. (Medical Compression System Inc., Or Akiva, Israel; Figure 1). The primary effectiveness outcome of the portable IPCD was the incidence of asymptomatic or symptomatic VTE, which was defined as DVT or pulmonary thromboembolism (PTE) confirmed while the patient was wearing a portable IPCD. We confirmed the incidence of asymptomatic or symptomatic VTE by ultrasonography 6–7 days postoperatively. The primary safety outcome for the portable IPCD was the incidence rate of hemorrhagic adverse events, which were defined according to randomized evaluation of long-term anticoagulant therapy (RE-LY) bleeding score. 16 The primary wearing compliance outcome for the portable IPCD was the wearing rate of the portable IPCD during hospitalization. In general, patients continuously wore the portable IPCD on their calves for two postoperative weeks. The attending surgeon changed the wearing period for each patient as necessary. The reason for changing the wearing period was the absence of DVT in two patients, start of gait in rehabilitation in two patients, incidence of DVT in one patient, start of ambulation in one patient, and fear of symptoms of peroneal nerve paralysis in one patient. Patients were instructed to remove the portable IPCD only when necessary (e.g. when going to the bathroom, showering, or undergoing medical examination). The patients also wore the portable IPCD during rehabilitation, although it was temporarily be removed for safety if it was difficult to assist a patient. We defined the wearing compliance of the portable IPCD in two ways: (1) wearing the portable IPCD for at least 18 h/day as recommended by the ACCP guidelines 7 and (2) continuously wearing the portable IPCD except when it was necessary to remove it. If a patient removed the portable IPCD, the time and reason for removal were investigated on the basis of the patient’s electronic medical records.

Portable intermittent pneumatic compression device worn by a patient.
Statistical analysis
The data were summarized using descriptive statistics. The incidence rates of symptomatic and asymptomatic VTE were calculated by dividing the number of occurrences by the number of patients. In addition, the site of occurrence of the thrombus was compiled from the ultrasonography results. On the basis of the RE-LY bleeding score, hemorrhagic adverse events were classified as major bleeding, minor bleeding, or no bleeding; the proportions of each are presented as ratios.
Results
Thirty-eight patients who met the eligibility criteria consented to participate (Table 1). Sixteen patients (42%) underwent hemodialysis. Ten patients (26%) were administered with anticoagulants. The reason for anticoagulant administration was DVT prophylaxis after major orthopedic surgery, such as revision THA, with guidelines 7 recommending prophylactic administration in six patients, a history of cerebral infarction in two patients, history of DVT in two patients, and history of PTE in one patient. From postoperative day 1, all patients began rehabilitation.
Patient characteristics (N = 38).a

DVT: deep vein thrombosis; PTE: pulmonary thromboembolism.
a Age, height, weight, body mass index, and creatinine clearance values are presented as mean ± standard deviation.
Asymptomatic and symptomatic DVT occurred in two patients (5.3%) and one patient (2.6%), respectively. All thrombi occurred in the soleus muscle vein. There were no cases of PTE (Table 2). In addition, the median D-dimer values the patient who developed asymptomatic and symptomatic DVT were 16.8, 7.5, and 10.8 μg/mL on postoperative days 1, 3, and 7, respectively, demonstrating bimodality.
Incidence of asymptomatic and symptomatic VTE.
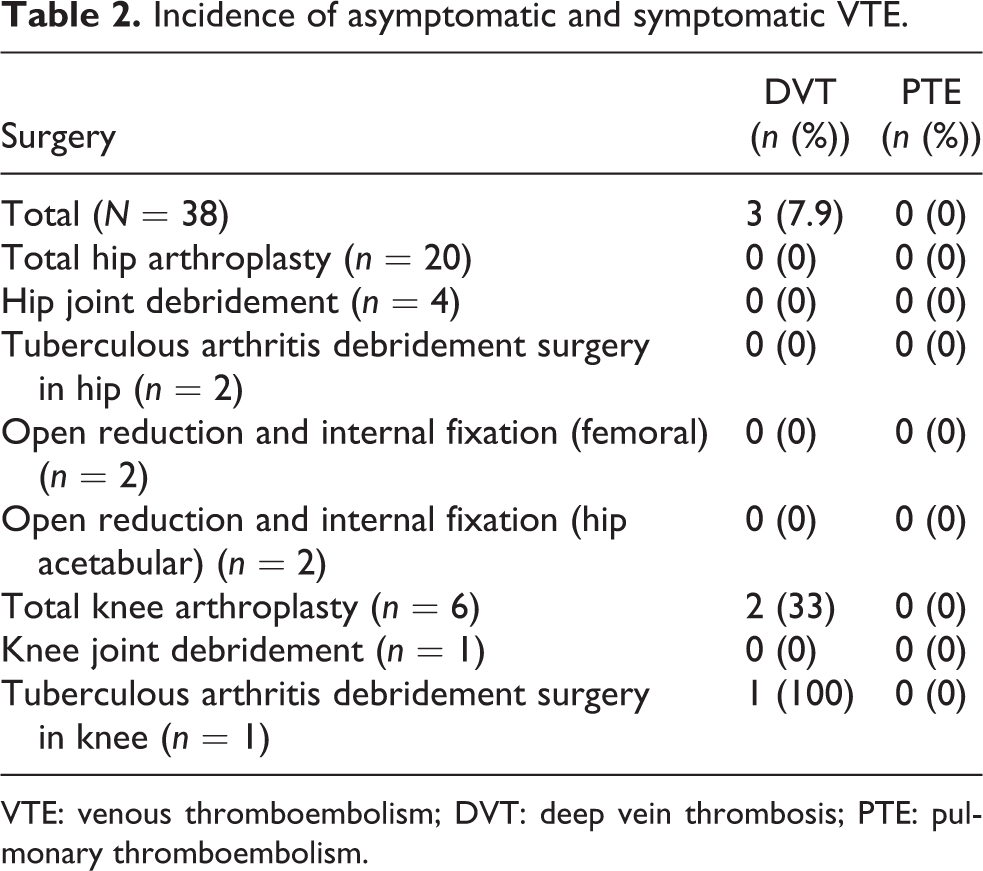
VTE: venous thromboembolism; DVT: deep vein thrombosis; PTE: pulmonary thromboembolism.
Major bleeding occurred in six patients (15.8%), minor bleeding occurred in two (5.3%), and 30 (79.0%) had no bleeding (Table 3). In particular, major bleeding occurred in four patients (10.5%) with a hemoglobin drop ≥2.0 g/dL, one (2.6%) received transfusion of 2 units, and one (2.6%) experienced gastrointestinal bleeding. Among the eight patients who experienced hemorrhagic adverse events, three were on hemodialysis and five were using anticoagulants in combination with a portable IPCD; among these five patients, the reason for anticoagulant administration was the history of cerebral infarction in three patients, and no reason was recorded for the other two.
Incidence of hemorrhage adverse events.
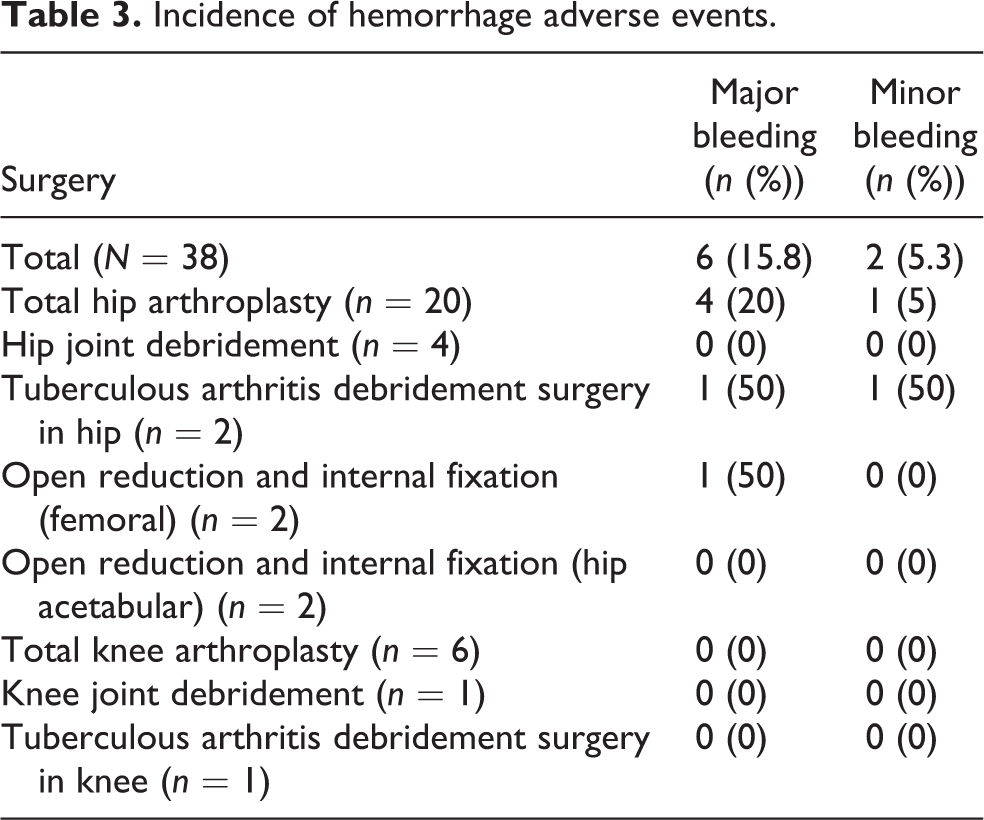
All 38 patients (100%) were able to wear the portable IPCD for at least 18 h a day, whereas 34 patients (89.5%) continuously wore the portable IPCD except when it was necessary to remove it. The average wearing period was 13.1 ± 5.0 days (range 2–32 days). The average portable IPCD wearing time for all patients was 22 h, 57 min per day. The reasons why patients did not wear the portable IPCD continuously included pain in the front of the right lower leg owing to a malfunction of the portable IPCD, tightness in the lower limb, delirium at night, or a conscious decision not to.
Discussion
The incidences of asymptomatic and symptomatic DVT in the present study were 5.2% and 2.6%, respectively. The incidence of DVT with conventional IPCD is reported to be 9.5–15.0%, 2,17,18 whereas that without VTE prophylaxis is reported to be 22.8–60.0%. 2 –4 Thus, the incidences of asymptomatic and symptomatic DVT in the present study are lower than those in a previous study using a conventional IPCD. Early ambulation postoperatively is not only a useful method for VTE prophylaxis but is also useful for postoperative physical function recovery. When a conventional IPCD is used for VTE prophylaxis, the patient is tethered to the bed and cannot move until they are released from the device. Therefore, the use of conventional IPCD prevents early ambulation, meaning that patients may suffer the disadvantages of bed rest. By contrast, because patients can use a portable IPCD when ambulation, such as they go to rehabilitation or to the toilet, the portable IPCD may give patients the advantage of early ambulation. Thus, in patients with the highest risks of both venous thrombosis and bleeding, a portable IPCD might be more effective for VTE prophylaxis than a conventional IPCD alone or no prophylaxis.
In the present study, hemorrhagic adverse events occurred eight patients (21.1%). Given that physical prophylaxis results in fewer hemorrhagic adverse events than pharmacological prophylaxis, it is often adopted for patients with high bleeding risk. 7 In a previous study, among patients who wore a portable IPCD, there were no major bleeding events among those with low bleeding risk, those without a history of VTE, or those without blood coagulation abnormalities. Meanwhile, in that study, the administration of low-molecular-weight heparin resulted in a major bleeding event rate of 6%. 12 It should be noted that because the present study included patients with high bleeding risk, the total hemorrhagic adverse event rate was higher than in previous studies. However, because there were no hemorrhagic adverse events when a portable IPCD was used alone in this study, this suggests that the portable IPCD itself is safe in patients with a high risk of bleeding. In addition, among the eight patients who experienced hemorrhagic adverse events, the three who took clopidogrel were older (age 70–84 years) and had low kidney function (creatinine clearance value: 5.49–32.58). Patients with impaired renal function have been reported to be at increased risk of hemorrhagic adverse events after total joint arthroplasty. 19 In addition, clopidogrel increases the risk of hemorrhagic adverse events in older patients with declining kidney function. Although physical prophylaxis seldom has hemorrhagic adverse events, when using a portable IPCD in such patients, we should carefully monitor hemorrhagic adverse events. Therefore, our results demonstrate that the use of a portable IPCD itself is safe for hemorrhagic adverse events in patients with high bleeding risk, but we are required to observe carefully in some conditions.
With regard to wearing compliance, all 38 patients (100%) wore the portable IPCD for at least 18 h/day, and 34 patients (89.5%) continuously wore the portable IPCD except when it was necessary to remove it. Improving wearing compliance when using a mechanical compression device remains an issue for VTE prophylaxis. Several studies report that the portable IPCD has better compliance than conventional IPCD (77.7–83.0% vs. 49.0–58.9%, respectively). 20,21 Edwards et al. report that 85% of patients who underwent THA or total knee arthroplasty (TKA) wore their portable IPCD for 20.4 h during a 24-h period and that patients who underwent TKA and developed DVT had shorter wearing times. 22 In addition, it has been reported that patients who developed DVT had poorer compliance with a portable IPCD than patients who did not develop DVT (13.4 h/day vs. 19.2 h/day). 23 However, because only four patients removed the portable IPCD for a short time relative to the entire wearing period in the present study, it is unlikely to have influenced the occurrence of DVT. Therefore, the overall wearing compliance of the portable IPCD is excellent, and the portable IPCD which allows patients to ambulate freely can enhance VTE prophylaxis.
Among the four reasons why patients temporarily removed the portable IPCD in the present study, we will focus on the pain on the front of the right lower thigh and the decision not to wear the device. The cause of the pain on the front of the right lower thigh was a malfunction of the calf pump as the portable IPCD was being pressurized. As a countermeasure, it is important to inspect the portable IPCD before use and prepare an environment, where immediate action can be taken if a malfunction occurs. Meanwhile, after guidance from a ward nurse, the patient who decided to remove the portable IPCD did not remove the portable IPCD until the end of the wearing period. Some patients might not have understood the rationale for wearing the portable IPCD. In one report, the compliance rate of patients with guidance from nurses was 85%, while the compliance rate of patients without guidance was 55%. 24 Therefore, all medical staff need to fully understand why a patient needs to wear a portable IPCD for VTE prophylaxis. By doing so, all medical staff can provide proper guidance to patients regarding the wearing method of the portable IPCD, which might consequently improve compliance with VTE prophylaxis and thus reduce the incidence of VTE.
This study has some limitations. First, the sample size was small, because only patients with the highest estimated risks for venous thrombosis and bleeding were included. Second, given the retrospective study design, the data might be incomplete. In a future study, to verify that the portable IPCD is superior to other physical prophylactic methods for patients with the highest risks of both venous thrombosis and bleeding, it will be necessary to set a control group of appropriate sample size and perform a prospective randomized controlled trial. Nevertheless, to our knowledge, this study is the first to examine the effectiveness, safety, and wearing compliance of a portable IPCD for VTE prophylaxis in patients who have undergone lower extremity orthopedic surgery and have the highest risks of both venous thrombosis and bleeding. Thus, the results of this study may help prevent VTE in patients with high risks of both venous thrombosis and bleeding.
Conclusion
Even if no prophylaxis is performed for patients at risks for thrombosis and/or bleeding, suitable VTE prophylaxis should be performed whenever possible. In the present study, the incidences of DVT in asymptomatic and symptomatic patients at high risks for both venous thrombosis and bleeding who wore a portable IPCD were 5.3% and 2.6%, respectively. The incidence of hemorrhagic adverse events was 0% in patients who did not take anticoagulants, and the wearing rate (i.e. ≥ 18 h/day) was 100%. Therefore, we propose the use of a portable IPCD for VTE prophylaxis in such patients.
Footnotes
Acknowledgments
We greatly appreciate everyone involved in our study for their advice on experimental design and assistance in interpreting the results of this study. We also greatly appreciate the contributions of our patients.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Japan Society for the Promotion of Science (JSPS) (KAKENHI grant number: JP17K10940).
