Abstract
Purpose:
The aim of this study was to examine the inhibitory effect of teriparatide (TPTD) on pain and on bone loss in ovariectomized (OVX) mice. The mechanism of osteoporotic pain in OVX mice was evaluated through an examination of pain-related behavior as well as immunohistochemical examinations.
Methods:
Eight-week-old female ddY mice were OVX and assigned to one of three groups: (1) OVX mice treated with vehicle (OVX), (2) OVX mice treated with teriparatide (OVX-TPTD), or (3) SHAM-operated mice treated with vehicle (SHAM). Starting immediately after surgery, vehicle or TPTD was injected subcutaneously. After a 4-week treatment, mechanical sensitivity was tested using von Frey filaments. The proximal tibial metaphyses were analyzed three-dimensionally by microcomputed tomography (μCT). Calcitonin gene-related peptide (CGRP) and transient receptor potential channel vanilloid 1 (TRPV1) expressions in L3–5 dorsal root ganglion (DRG) neurons were examined using immunohistochemistry.
Results:
Ovariectomy induced bone loss and mechanical hyperalgesia in the hind limbs with upregulation of CGRP and TRPV1 expressions in DRG neurons innervating the hind limbs. Bone loss was prevented more effectively in the OVX-TPTD mice than in the OVX mice. Furthermore, mechanical hyperalgesia and upregulation of CGRP and TRPV1 expressions were significantly lower in the OVX-TPTD mice than in the OVX mice.
Conclusion:
TPTD treatment prevented ovariectomy-induced bone loss and ovariectomy-induced mechanical hyperalgesia in hind limbs, and it suppressed CGRP and TRPV1 expressions in DRG neurons. These results suggest that TPTD is useful for the treatment of osteoporotic pain in postmenopausal women.
Keywords
Introduction
Osteoporosis is a chronic skeletal disorder characterized by decreased bone density and changes in bone microarchitecture. These changes lead to increased skeletal fragility and increased risk of low-trauma fractures with associated complications, eventually leading to reduced health-related quality of life (HRQoL). 1 –3 In clinical practice, 80% of osteoporotic patients complain of lower back pain 4 and at least 10% do not demonstrate any signs of fractures or diseases other than bone demineralization. 5
Teriparatide (TPTD) (Forteo®; Eli Lilly, Indianapolis, Indiana, USA), a recombinant 1–34 N-terminal sequence of human parathyroid hormone, is the first anabolic agent approved for the treatment of patients with osteoporosis. 6 Treatment with TPTD is an effective therapeutic option for postmenopausal women with severe osteoporosis. According to the European Forsteo Observational Study, the clinical administration of TPTD in postmenopausal women with severe osteoporosis resulted in a significant reduction in the incidence of fragility fractures and back pain, resulting in an improvement in reported HRQoL in these patients. 2,7
In Japan, a postmarketing surveillance study (24 months of treatment) of 1847 osteoporotic patients showed a significant increase in bone mineral density, a decrease in the incidence of new vertebral and nonvertebral fractures, and a decrease in the severity of back pain, with an improvement in HRQoL, following TPTD treatment. 8 Regardless of whether recent vertebral fractures were present, TPTD treatment was associated with a significant reduction in back pain in the European Study of Forsteo, which included 868 postmenopausal women. 9 In a meta-analysis study, the role of TPTD in preventing back pain in osteoporotic patients was assessed through a meta-analysis of four completed, randomized, double-blinded trials of TPTD versus an alternative treatment. 10
The role of TPTD in reducing back pain has been widely demonstrated; however, its mechanism in reducing back pain has not been well investigated, and the effect of TPTD on bone loss and pain markers can only be investigated to a limited degree in the clinical setting, where the main focus of treatment is on pain reduction. The objective of the current study was to investigate the effect of TPTD on pain-related behavior in ovariectomized (OVX) mice.
Materials and methods
Reagent
The reagent used was TPTD (Forteo®; Eli Lilly, Indianapolis, Indiana, USA).
Animals
This research has been approved by the institutional review board of the authors’ affiliated institution. The animal studies adhered to the Planning Research and Experimental Procedures on Animals: Recommendations for Excellence guidelines. We followed a randomized, prospective, and controlled animal model design. All efforts were made to minimize animal suffering and the number of animals used. Sample size was determined using a power analysis for an α of 0.05 and a power of 0.80 using G*POWER3. 11
Seven-week-old female ddY mice were purchased from Japan SLC (Hamamatsu, Japan) and acclimated for 1 week before the start of the experiment. Experiments were conducted on 8-week-old female ddY mice weighing 34–39 g, as per previous reports. 12,13 The mice were housed in a temperature-controlled room (23 ± 1°C) with a 12-h light/12-h dark cycle (lights on from 7:00 to 19:00) and with free access to food and water.
Experimental protocol
At 8 weeks of age, the mice were randomly assigned based on body weight to one of three groups: (1) sham-operated mice treated with vehicle (SHAM; n = 8), (2) OVX mice treated with vehicle (OVX; n = 8), or (3) OVX mice treated with TPTD (OVX-TPTD; n = 8). The mice were either OVX bilaterally under anesthesia with intraperitoneal administration of pentobarbital sodium (Sankyo, Tokyo, Japan) or sham-operated (ovaries exteriorized but not removed). Immediately after reload, vehicle (physiological saline) or TPTD was injected subcutaneously. TPTD (40 μg/kg) was injected subcutaneously five times a week, as described previously. 14,15
At the end of the 4-week treatment period, the mechanical sensitivity of the hind limbs was examined using von Frey filaments. Following the examination, mice were euthanized using an intraperitoneal injection of pentobarbital sodium (0.5 mg/kg). Bilateral hind limbs were removed to conduct microcomputed tomography (μCT) and the bilateral dorsal root ganglions (DRGs) from L3 to L5 were collected for immunohistochemical analysis.
Analysis of three-dimensional bone structure using μCT
At the end of the 4-week treatment period, isolated tibias were imaged using a μCT scanner (R_mCT; Rigaku Corporation, Tokyo, Japan), as described previously, to determine the three-dimensional bone microstructure. 12,13 Three-dimensional images were reconstructed and analyzed using three-dimensional image analysis software (TRI/3D-BONE, RATOC System Engineering, Tokyo, Japan). Osteoporotic evaluation was performed on the basis of bone volume and tissue volume fraction (BV/VT, %), trabecular number (Tb.N, /mm), trabecular thickness (Tb.Th, μm), and trabecular separation (Tb.Sp, μm).
Histological analysis of the hind limb bone
Isolated hind limb bones and immunostained sections were prepared as described previously. 12,13 Sections were stained with hematoxylin and eosin for histological analysis of the bone structure. To identify osteoclasts in hind limb bone, the tartrate-resistant acid phosphatase (TRAP) method was used. In the proximal tibia, the number of TRAP-positive osteoclasts was determined within an area that was 0.5 mm in length and 2 mm in width, apart from the most distal part of the growth plate. The sections were reviewed independently by three investigators.
Measurement of pain-related behavior with von Frey filaments
The mechanical nociceptive threshold of the hind paw was determined as described previously. In a recent study, measurement of pain-related behavior with von Frey filaments to the hind paw was reported as pain-related behavior related to knee joint inflammation or knee osteoarthritis. 16,17 Thus, mechanical hyperalgesia around knee osteoporosis was assessed using von Frey filaments. The von Frey tests were conducted at 4 weeks after treatment. The von Frey tests were performed as described previously. 12,13 Briefly, the frequency of the withdrawal response, the withdrawal threshold, and the 50% withdrawal threshold were determined.
Immunohistochemical analysis in DRG
Previous studies showed that the L3–5 DRG neurons innervate the hind limb bones. 18,19 Immunohistochemical analysis of calcitonin gene-related peptide (CGRP) expression and transient receptor potential channel vanilloid 1 (TRPV1) expression were completed for the L3, L4, and L5 DRG neurons. Four weeks after treatment and 2 h after the behavioral tests, the mice were euthanized, and their spines and hind limb bones were removed. Tissue preparation and immunostaining of sections were performed as described previously. 12,13 The primary antibodies used included anti-CGRP antibody (rabbit polyclonal; Sigma-Aldrich, St Louis, Missouri, USA) at a 1:1000 dilution or anti-VR1 C-terminus (Guinea Pig polyclonal; Neuromics, Edina, Minnesota, USA) at a 1:1000 dilution overnight at room temperature. There were no observable immunoreactions in controls. The average percentages of immunoreactive L3, L4, and L5 DRG neuron sections were calculated on immunostained section examination. The immunostained sections were reviewed independently by three investigators.
Statistical analysis
All numerical values are expressed as means ± standard deviation. Statistical analyses were performed using one-way analysis of variance, and p < 0.05 was accepted as indicating significance.
Results
Analysis of three-dimensional bone structure by μCT
The three-dimensional images of the proximal tibial metaphysis (Figure 1(a)) showed less cancellous bone in the OVX group than in the SHAM group. Cancellous bone loss was significantly less in the OVX-TPTD group than in the OVX group.

Micro-CT analyses of the proximal tibial metaphysis. (a) The three-dimensional images, (b) BV/TV (%), (c) Tb.N (/mm), (d) Tb.Th (μm), and (e) Tb.Sp (μm). Data are shown as means (±SD). *p < 0.05; **p < 0.01; ***p < 0.005; ****p < 0.001 (n = 8 in each group). CT: computed tomography; BV/TV: bone volume/tissue volume; SD: standard deviation; Tb.N: trabecular number; Tb.Th: trabecular thickness; Tb.Sp: trabecular separation; OVX: ovariectomized; TPTD: teriparatide.
μCT analysis of the proximal tibial metaphysis showed that the BV/TV was significantly lower in the OVX group than in the SHAM group. BV/TV was significantly higher in the OVX-TPTD group than in the OVX group (Figure 1(b)). The Tb.N and the Tb.Th were significantly lower in the OVX group than in the SHAM group. The Tb.N and Tb.Th were significantly higher in the OVX-TPTD group than in the OVX group (Figure 1(c) and (d)). Tb.Sp was significantly higher in the OVX group than in the SHAM group. The Tb.Sp was significantly lower in the OVX-TPTD group than in the OVX group (Figure 1(e)). Marked bone loss occurred in the proximal tibial metaphysis in the OVX group, and the loss at the end of the 4-week treatment period was prevented by treatment with TPTD.
Histological analysis of the hind limb bone
The OVX group had less cancellous bone in the proximal tibial metaphysis than the SHAM group (data not shown). Cancellous bone loss was less in the OVX-TPTD group than in the OVX group (Figure 2(a)). The number of TRAP-positive osteoclasts in the proximal tibial metaphysis was significantly higher in the OVX group than in the SHAM group (Figure 2(b)). Furthermore, the number of TRAP-positive osteoclasts of the proximal tibial metaphysis tended to be lower in the OVX-TPTD group than in the OVX group.

Histological analysis of the hind limb bone. (a) TRAP staining for histological examination of the proximal tibial metaphysis. (b) Histological analysis of the number of TRAP-positive osteoclasts in the proximal tibial metaphysis (**p < 0.01). TRAP: tartrate-resistant acid phosphatase; OVX: ovariectomized; TPTD: teriparatide.
Measurement of pain-related behavior with von Frey filaments
The paw withdrawal threshold and the 50% paw withdrawal threshold were also significantly lower in the OVX group than in the SHAM group and were higher in the OVX-TPTD group than in the OVX group (Figure 3(a) and (b)). The paw withdrawal frequency stimulated by von Frey filaments with strengths of 0.6–2.0 g (0.4 g, 0.6 g, 1.0 g, 1.4 g, and 2.0 g) were significantly higher in the OVX group than in the SHAM group (Figure 3(c)). The paw withdrawal frequency stimulated by von Frey filaments with strengths of 1.0 g and 1.4 g was significantly lower in the OVX-TPTD group than in the OVX group. TPTD improved mechanical hyperalgesia in hind limbs.
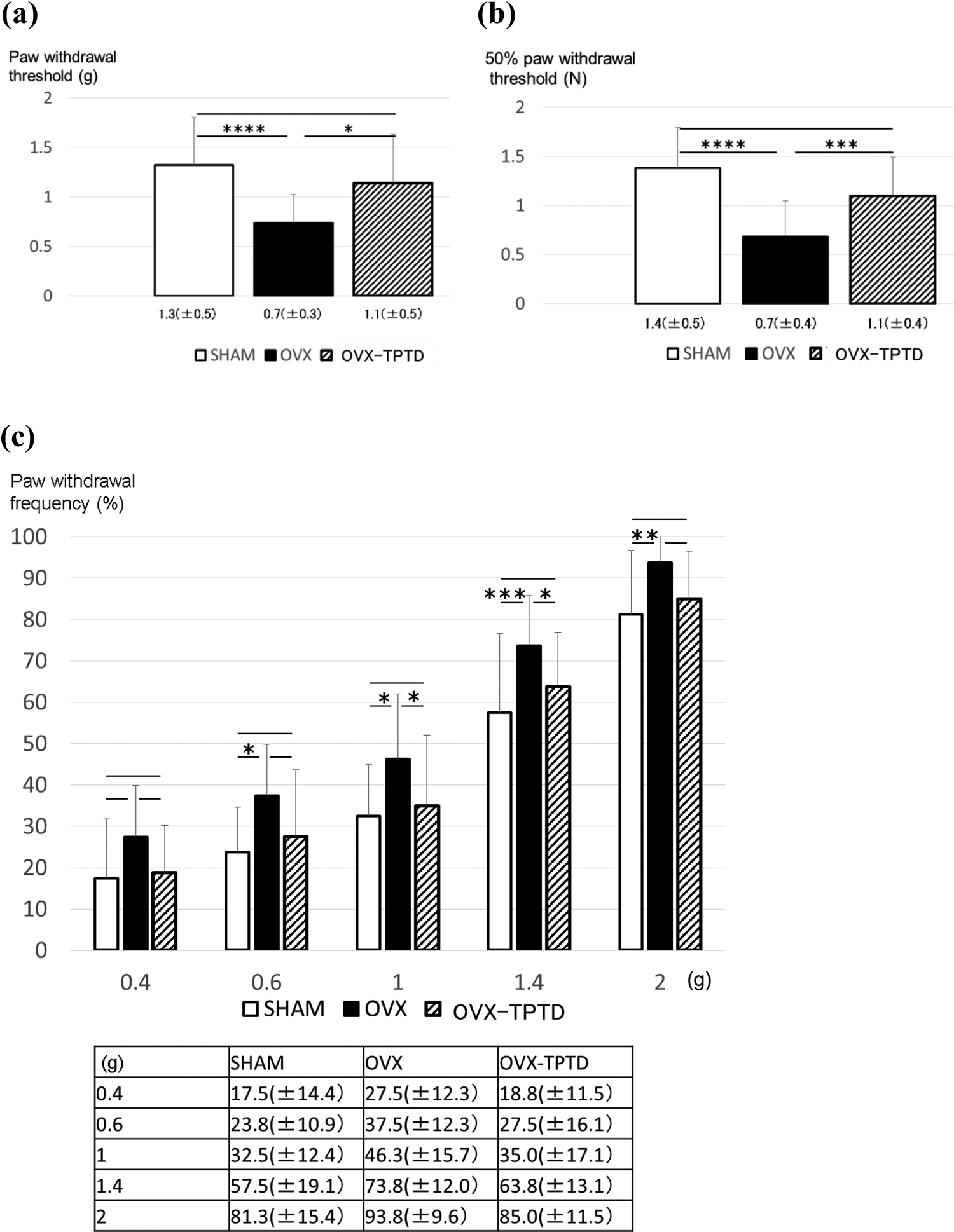
Measurement of pain-related behavior with von Frey filaments. (a) Paw withdrawal threshold, (b) 50% paw withdrawal threshold determined using the up-down method, and (c) paw withdrawal frequency stimulation. Data are shown as means (±SD). *p < 0.05; **p < 0.01; ***p < 0.005; ****p < 0.001. SD: standard deviation; OVX: ovariectomized; TPTD: teriparatide.
Immunohistochemical analysis in the DRG
The immunohistochemical analysis showed that the percentage of CGRP-immunoreactive L3, L4, and L5 DRG neurons was significantly higher in the OVX group than in the SHAM group, whereas it was significantly lower in the OVX-TPTD group than in the OVX group (Figure 4(a) to (d)). The percentage of TRPV1-immunoreactive L3, L4, and L5 DRG neurons was significantly higher in the OVX group than in the SHAM group, whereas it was significantly lower in the OVX-TPTD group than in the OVX group (Figure 4(e) to (h)).
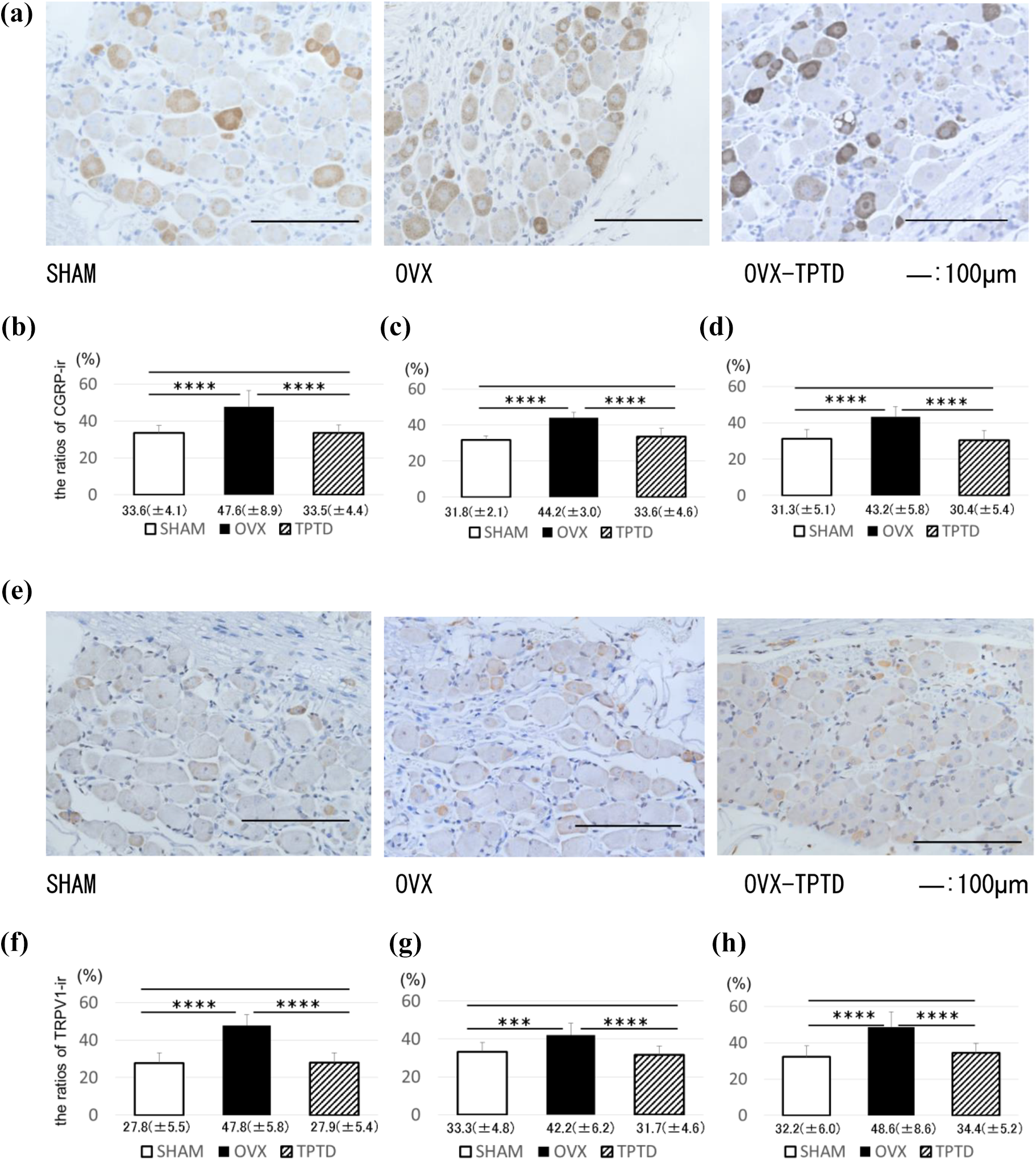
Immunohistochemical analysis of CGRP and TRPV1 expressions in DRG neurons. (a)–(d) CGRP expression and (e)–(h) TRPV1 expression in the DRGs. The ratios of immunoreactive (ir), (b) and (f): L3, (c) and (g): L4, and (d) and (h): L5 DRGs. Data are shown as means (±SD). *p < 0.05; **p < 0.01; ***p < 0.005; ****p < 0.001. CGRP: calcitonin gene-related peptide; DRG: dorsal root ganglion; TRPV1: transient receptor potential channel vanilloid 1; SD: standard deviation; OVX: ovariectomized; TPTD: teriparatide.
Discussion
This study aimed to investigate the effect of TPTD on pain-related behavior in OVX mice. The results of the study found that ovariectomy induced bone loss and mechanical hyperalgesia in the hind limbs of mice, with upregulation of CGRP and TRPV1 expressions in DRG neurons innervating the hind limbs. There was significantly less bone loss in mice treated with TPTD. Furthermore, mechanical hyperalgesia and upregulation of CGRP and TRPV1 expressions were significantly lower in the mice treated with TPTD.
In 2010, 23% of the Japanese population were aged 65 years and over, and this is projected to increase to 32% in 2030 and to 40% by 2050. 20 The prevalence of osteoporosis in Japan is expected to increase due to the rapidly increasing proportion of elderly people.
Even without the occurrence of fractures, osteoporosis has been associated with undesirable consequences such as chronic pain, impaired physical ability, reduced social activity, poor well-being, and depressed mood, which overall leads to a reduced QoL. Therefore, studying the impact of medications on the improvement of back pain and QoL is particularly relevant to the treatment of osteoporosis. 21
Bone pain is one of the most common complications in cancer patients with bone metastases. In a bone cancer model, Yoneda et al. reported that osteoclasts dissolve bone minerals by releasing protons and creating an acidic microenvironment, and the acidic condition causes bone pain through the activation of acid-sensing receptors, such as acid-sensing ion channels and the TRPV subfamily members. 22,23
Suzuki et al. described osteoporotic pain as follows. The establishment of acidic microenvironments in osteoporotic bones, due to osteoclast hyperactivity, might trigger bone pain. These decreases in pH likely stimulate acid-sensing receptors and increase the production and secretion of proinflammatory cytokines in osteoporotic bones. Furthermore, CGRP-immunoreactive pain-related sensory nerve fibers increase in osteoporotic bone marrow. These acidic and innervation changes can affect sensory input, creating a local neurogenic inflammatory state and osteoporotic pain. 24
As previously suggested, sensory innervation of OVX mice showed increased expressions of CGRP, a neuropeptide marker of pain, and TRPV1, an acid-sensitive ion channel, in DRG neurons. 12,25 In addition, bisphosphonate (BP) inhibited bone pain associated with osteoporosis in an OVX model and inhibited osteoclast proliferation, upregulation of CGRP and TRPV1, and bone pain-related behavior.
In this study, the number of osteoclasts tended to be lower in the TPTD group than in the OVX group. TPTD, as well as BP, inhibited bone loss and osteoclasts, caused an increase in the pain threshold value, and prevented CGRP and TRPV1 expressions in DRGs.
Although BP and TPTD are potent anti-osteoporosis agents, they demonstrate contrasting bone remodeling effects in increasing bone mass. BP has an inhibitory effect on osteoclast activation, whereas TPTD is an anabolic therapy for osteoporosis and has a stimulatory effect on osteoblast activation leading to osteoclast activation and a high bone turnover state.
Daily treatment with TPTD accelerates bone turnover, and bone formation markers such as the carboxyterminal propeptide of type 1 procollagen and the aminoterminal propeptide of type 1 procollagen are elevated from the beginning of treatment. 26,27 Bone resorption markers such as type I collagen N-telopeptide and type I collagen C-telopeptide are also elevated several months after the commencement of treatment, and this is termed the “anabolic window” of TPTD. 28,29
TPTD has both anabolic and catabolic effects on osteoblasts in vitro, and the mechanisms underlying its promotion of bone formation are unclear. Isogai et al. reported that immediately after the administration a single dose of TPTD, bone resorption was promoted through transient osteoclast activation and bone formation was suppressed through inhibition of osteoblast function. However, after TPTD was cleared from the blood, the osteoblast number increased, resulting in increased bone formation. 30 Moreover, TPTD decreases bone resorption by decreasing the number and activity of osteoclasts. This report supports the results of our study. Decreasing bone resorption may lead to an improvement in the acidic condition in bone minerals and improve osteoporotic bone pain.
A previous in vivo study reported that treatment with TPTD for 2 weeks (from 6 weeks to 8 weeks after OVX) significantly improved pain-like behavior in the von Frey test in OVX mice compared to that in untreated mice. In addition, the improvement effect on pain-like behavior in OVX mice was associated with the downregulation of inflammatory cytokine expression, including interleukin (IL)-1β, IL-6, and tumor necrosis factor-α. 31 In a recent study, although anti-IL-6 R antibody treatment had no effect on ovariectomy-induced bone loss, the treatment prevented ovariectomy-induced mechanical hyperalgesia in the hind limbs and suppressed CGRP expression in DRG neurons. 13 TPTD may inhibit bone nociceptors sensitized by lowering pH and by inflammatory cytokines.
The present study had several limitations. First, we did not evaluate the expression of bone formation markers in the bone. Second, changes in CGRP and TRPV1 expressions in bone tissue were not directly evaluated. Third, the dose- and time-dependent effects of TPTD were not examined. Fourth, analysis of temporal changes in bone loss, mechanical hyperalgesia, and immunohistochemical analyses in DRG neurons of OVX mice was not performed. Fifth, whether TPTD administration starting at the beginning prevents osteoporosis is yet to be investigated. Future studies should address the timing effects of treatment in OVX -induced bone loss.
Conclusion
In this study, TPTD prevented bone loss, and it improved mechanical hyperalgesia in the hind limbs of OVX mice and downregulated the expression of pain-related marker in the DRG neurons of OVX mice.
Footnotes
Acknowledgements
The authors would like to thank Katsura Chiba for excellent technical assistance.
Author contributions
SK and HW contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
