Abstract
Introduction
Osteoporosis is becoming increasingly prevalent owing to the ageing population worldwide, posing a potentially heavy disease burden to affected individuals and undermining their functionality. 1 It contributes significantly to the frailty of elderly individuals and has become an important concern in the worldwide healthcare system. 2 It is a skeletal disorder characterized by a reduction in bone strength and density, and affected individuals are susceptible to an increased risk of fractures in the hip, spine, and other skeletal sites. 3 Old age and the female sex are the two most important risk factors for osteoporosis.2–4 The prevalence of osteoporosis in postmenopausal women ranges from 29.1% to 50.4% in various studies, with Caucasians generally having a higher prevalence than Asians.5–9
Anti-osteoporosis medications have been widely used to control osteoporosis progression either in patients with dual-energy x-ray absorptiometry (DEXA) proven osteoporosis or those with atypical fractures. Common medications to treat osteoporosis include bisphosphonates, parathyroid hormone analogues, receptor activators of nuclear factor kappa-B ligand inhibitors (RANKL inhibitors), oestrogen agonists/antagonists, and calcitonin. 10 The effects of these agents on bone mineral density (BMD) and fracture risk reduction have been comprehensively discussed in the current literature.11–15 However, the effects of anti-osteoporosis medications on the quality of life (QOL) of patients are often overlooked though it is equally important as clinical parameters such as BMD. Currently, only a limited number of studies have investigated the effects of anti-osteoporosis medications on QOL,16–19 with most of them focusing on one medication only. Our study aimed to directly evaluate and compare the QOL outcomes of three commonly used osteoporosis agents, namely bisphosphonates, RANKL inhibitors and parathyroid hormone analogues. A more extensive set of questionnaires was used in our study for a more comprehensive understanding of QOL outcomes as compared to existing literature.
Methods
Study design
A prospective observational study was conducted at a tertiary referral spine clinic from 25 February 2016 to 31 December 2020. The study was ethically approved by the local institutional review board (UW 15-628). All patients were well informed of the study content and provided written consent for participation. Subjects with parathyroid hormone
The following inclusion criteria must be met for an individual to be included in the study: (1) Chinese women aged ≥ 55 years, (2) diagnosed with osteoporosis by DEXA (T-score SD < 2.5) or prior fragility fracture and (3) able to understand and complete the patient questionnaires. These patients were recruited and assessed at one-year follow-up. Patients with concomitant use of other anti-osteoporosis medications, hormone replacement therapy and corticosteroid, and those who failed to complete the treatments/follow-up questionnaires were excluded. All study subjects were newly initiated with their assigned anti-osteoporosis medications. No specific restriction was applied to the study subjects’ concomitant use of calcium and vitamin D supplements. All study subjects who managed to continue their assigned medications during the 12-month period and completed their follow-up questionnaires were included in the analysis.
Study assessments
Patient baseline characteristics were collected from their medical records, radiographs and questionnaires. These included age, body height/weight, age of menopause, number of births, maternal history of osteoporosis/fragility hip fracture, comorbidities, current drug use (calcium/vitamin D supplement, painkillers, drug-related increased risk of osteoporosis), fracture history, risk factors for fall/fracture and socio-economic status. Drug-related increased risk of osteoporosis included the use of glucocorticoids, aromatase inhibitors, anti-androgen, high-dose thyroxine, thiazolinediones, calcineurin inhibitors, anticonvulsants, oral anticoagulants, heparin, antiretroviral drugs, selective inhibitors of serotonin reuptake, loop diuretics and proton pump inhibitors. 20 Risk factors for fall/fracture and socio-economic status of study subjects were obtained from questionnaires whereas patient demographics, past medical history, family history and drug history were based on medical records. At the 12-month assessment, medical records and radiographs were reviewed for treatment compliance, new fractures, new major diseases, current medications, and new BMD measurements if any. No changes in the analgesic drug regimen were made for the purposes of this study.
QOL was assessed at baseline and 12 months using ambulatory status assessment, back pain visual analogue scale (VAS), 21 the Osteoporosis Assessment Questionnaire-Physical Function (OPAQ-PF) 22 and EuroQol 5-level 5-dimension (EQ-5D-5L) questionnaire. 23 The ambulatory status was graded according to the New York University Classification of Ambulatory Function. The back pain VAS is based on a scale ranging from 0 to 10 and measured the most severe and average pain felt by the study subjects in the past 24 hours. 2.5 was taken as minimally clinically important differences (MCID) in chronic lower back pain (LBP). 24 OPAQ-PF 22 is a validated questionnaire that consists of 15 questions related to the daily activities of living. A total score of 90 is generated and a higher score is associated with better physical function of study subjects (MCID: 4.0). 22 For the EQ-5D-5L questionnaire, two scores are used for analysis: (1) EQ-VAS from 0 to 100 concerning patients’ self-rated health status and (2) an adjusted EQ-5D summary index from −0.864 to 1 concerning patients’ mobility, self-care, usual activities, pain/discomfort and anxiety/depression. 23 High EQ-VAS and EQ-5D summary index are associated with a better patient-reported general health status. 23 From our knowledge there is no research on the MCID of EQ-5D-5L on osteoporosis, but we used the MCID of 0.07 for improvement, and − 0.05 for worsening in EQ-5D summary index, derived from a study investigating the MCID of EQ-5D-5L on hip osteoarthritis. 25 Adherence Evaluation of Osteoporosis treatment (ADEOS-12) is an objective disease-specific measure taken at the 12-month follow-up to assess treatment adherence, which includes questions on ease of use, motivation and any missed episode when taking osteoporotic medications. Our interpretation of the ADEOS-12 results followed the validation study by Breuil et al. 26 An index of ≥ 20 is interpreted as a high probability of persistence, whereas an index ≤ 16 is associated with a high probability of treatment discontinuation.
Statistical analysis
Test of normality was applied to the weighted QOL results using the Shapiro-Wilk test, and this determined the subsequent statistical tests used for comparison. The baseline and one-year values were compared within each group to evaluate the effect of treatments in each group. Wilcoxon signed-rank test was performed to evaluate the difference between baseline and one-year values. For the comparison across the three groups, covariates including respective baseline values for each parameter, BMI, vitamin D/calcium/painkiller intake and history of osteoporotic fracture were controlled. Quade's ANCOVA was utilized to compare the pre- and post-treatment QOL outcomes across the study groups. Denosumab and alendronate groups were combined (n = 12) and compared with the teriparatide group (n = 11) which was an accepted method to balance group sizes. 27
Predictive factors for improvement for average back pain VAS were identified through a general linear regression model with satisfactory Levene's test results after the inverse probability of treatment weighting (IPTW) adjustment with propensity score to minimize potential confounding biases from baseline discrepancy. This was particularly chosen due to the smaller subject sample size. We have considered a range of patient baseline characteristics, baseline age, BMI, use of painkillers and calcium/vitamin D use in the generation of the propensity score. The propensity score weighting method for comparison in a three-treatment group setting had been validated by Yoshida et al. 28 Adjusted marginal means of the change of VAS of average back pain for each study group were also presented. Post-hoc pairwise comparison among three groups was conducted for any intergroup difference.
Statistical testing was carried out using Toolkit for Weighting and Analysis of Non-equivalent Groups (TWANG) R package (RAND Corporation, Santa Monica, California, US) and SPSS version 26 (IBM, Armonk, New York, US). Unless otherwise specified, all continuous variables were reported as mean ± standard deviation, with results considered significant at p < 0.05. Post-hoc power analysis was performed for significant factors.
Results
Patient characteristics
A total of 31 Chinese female patients with osteoporosis were enrolled from 25 February 2016 to 13 December 2019. Six patients in the teriparatide group were excluded as they discontinued treatment before the 12-month follow-up, while two in the denosumab group were excluded as they refused treatment after enrolment. After the exclusion, a total of 23 patients were evaluated in our study (Figure 1). The patients had mean baseline age of 78.6 ± 7.2 years, mean height of 152.1 ± 5.7 cm, mean body weight of 51.5 ± 7.5 kg and mean BMI of 22.3 ± 3.0 kg/m2. 91.3% and 52.2% of the study subjects were taking calcium/vitamin D supplements and painkillers respectively at the beginning of the study. None of the study subjects had a maternal history of osteoporosis and/or fragility hip fracture. The baseline characteristics and baseline QOL outcomes of each treatment group are summarized in Table 1.
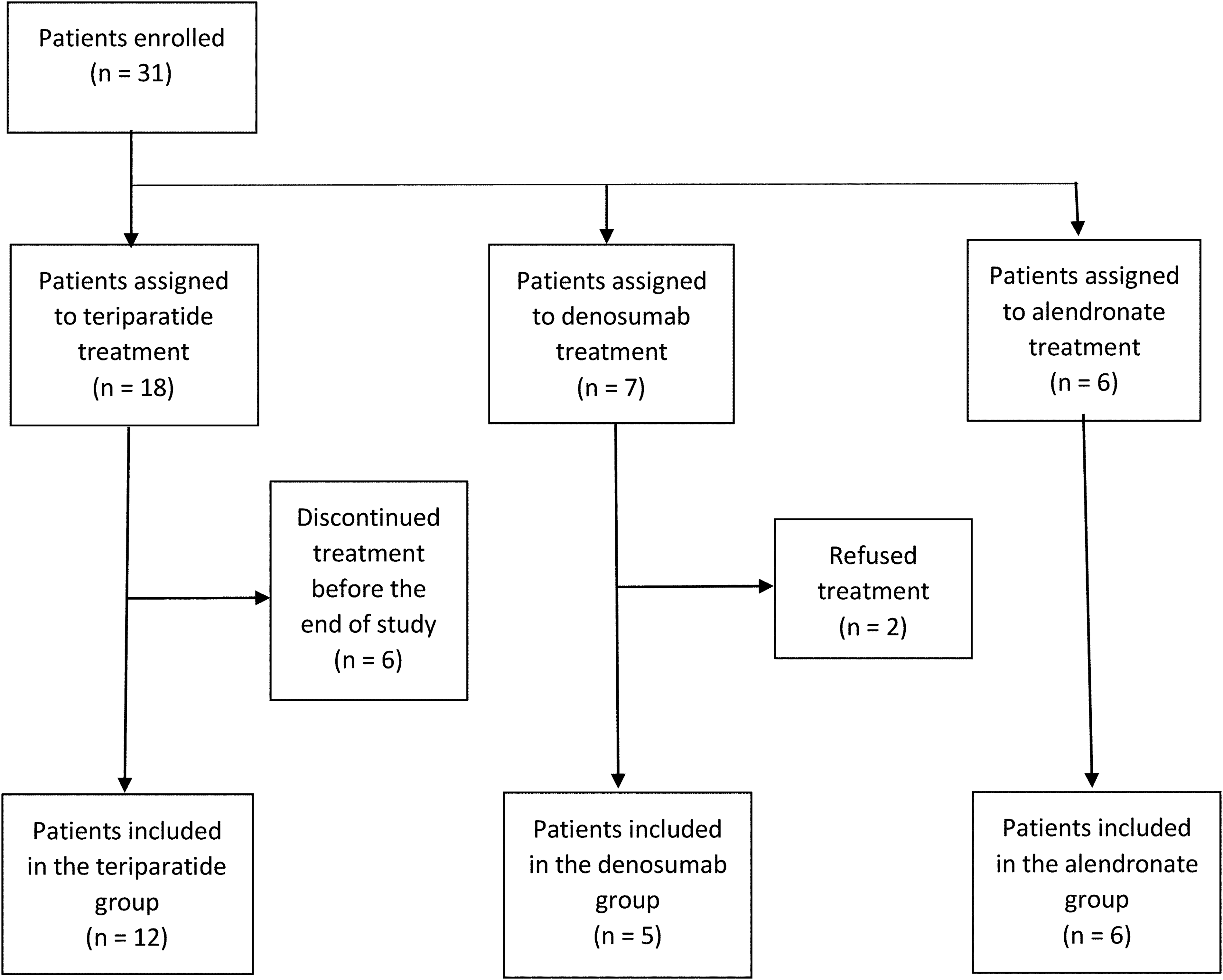
Flowchart on patient recruitment.
Baseline characteristics.
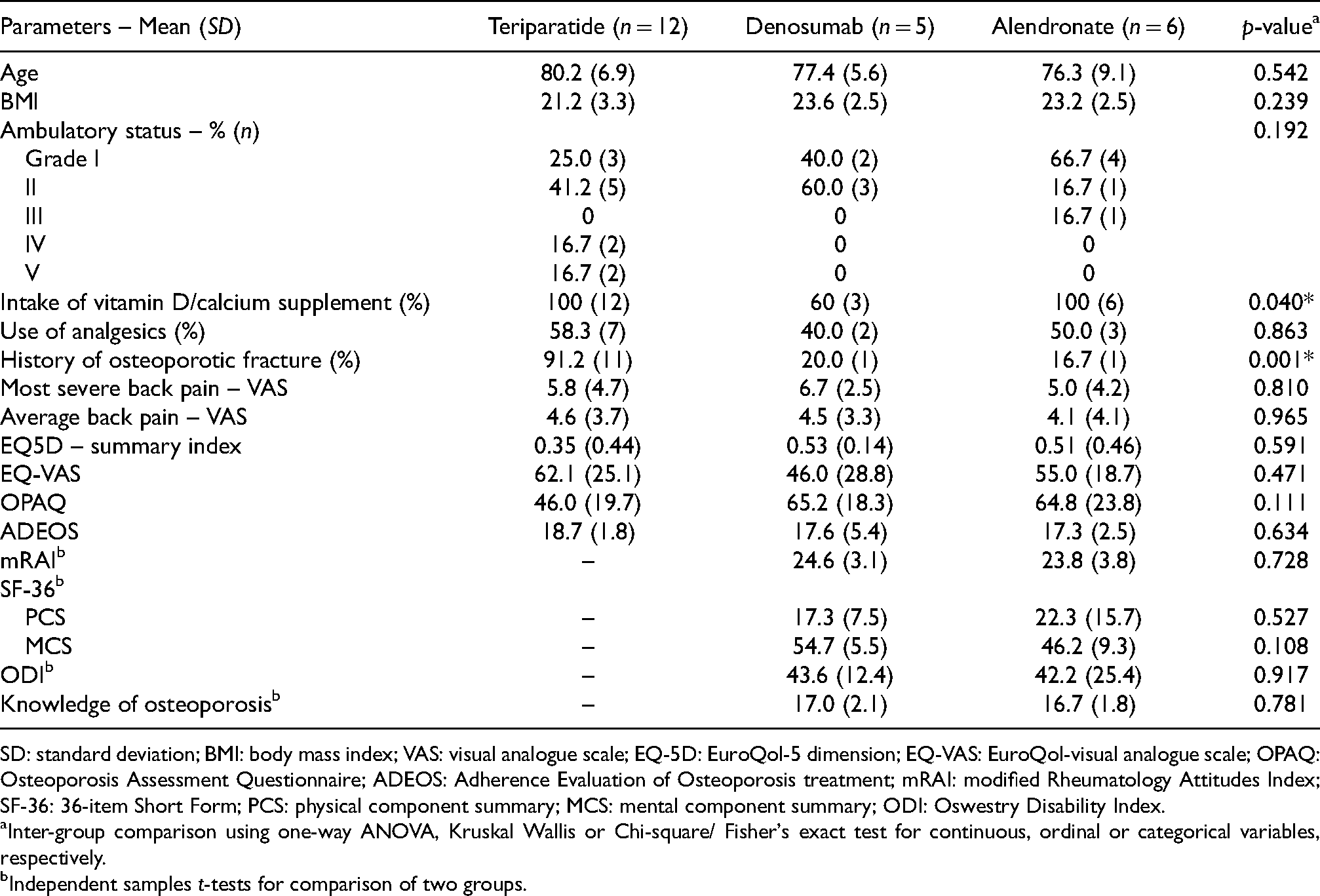
SD: standard deviation; BMI: body mass index; VAS: visual analogue scale; EQ-5D: EuroQol-5 dimension; EQ-VAS: EuroQol-visual analogue scale; OPAQ: Osteoporosis Assessment Questionnaire; ADEOS: Adherence Evaluation of Osteoporosis treatment; mRAI: modified Rheumatology Attitudes Index; SF-36: 36-item Short Form; PCS: physical component summary; MCS: mental component summary; ODI: Oswestry Disability Index.
Inter-group comparison using one-way ANOVA, Kruskal Wallis or Chi-square/ Fisher's exact test for continuous, ordinal or categorical variables, respectively.
Independent samples t-tests for comparison of two groups.
Comparison of pre- and post-treatment QOL outcomes
The teriparatide group demonstrated improvements in five parameters (Table 2). The median values of most severe and average back pain reduced from 8.0 to 0.0 (p = 0.029) and from 6.0 to 0.0 (p = 0.006), respectively, for the teriparatide group. Significant improvement in general health status was demonstrated by the increased EQ-5D summary index (from 0.47 to 0.87, p = 0.001) and EQ VAS (from 65.0 to 80.0, p = 0.008). The OPAQ physical function improved from 40.0 to 79.5 after 1 year of teriparatide treatment (p < 0.001). On the contrary, the denosumab and alendronate group showed worsened VAS back pain but improved function/QOL. The average back pain of the denosumab group increased from 3.4 to 6.0 (p = 0.013), but the EQ-VAS and OPAQ scores improved from 50.0 to 70.0 (p = 0.002) and from 61.0 to 75.0 (p = 0.005), respectively. A similar trend was observed in the alendronate group, with a worsened most severe back pain score (from 4.0 to 6.5, p = 0.042) but an increased OPAQ score (from 70.0 to 80.0, p < 0.001).
Comparison of pre- and post-treatment QOL outcomes for each study group.
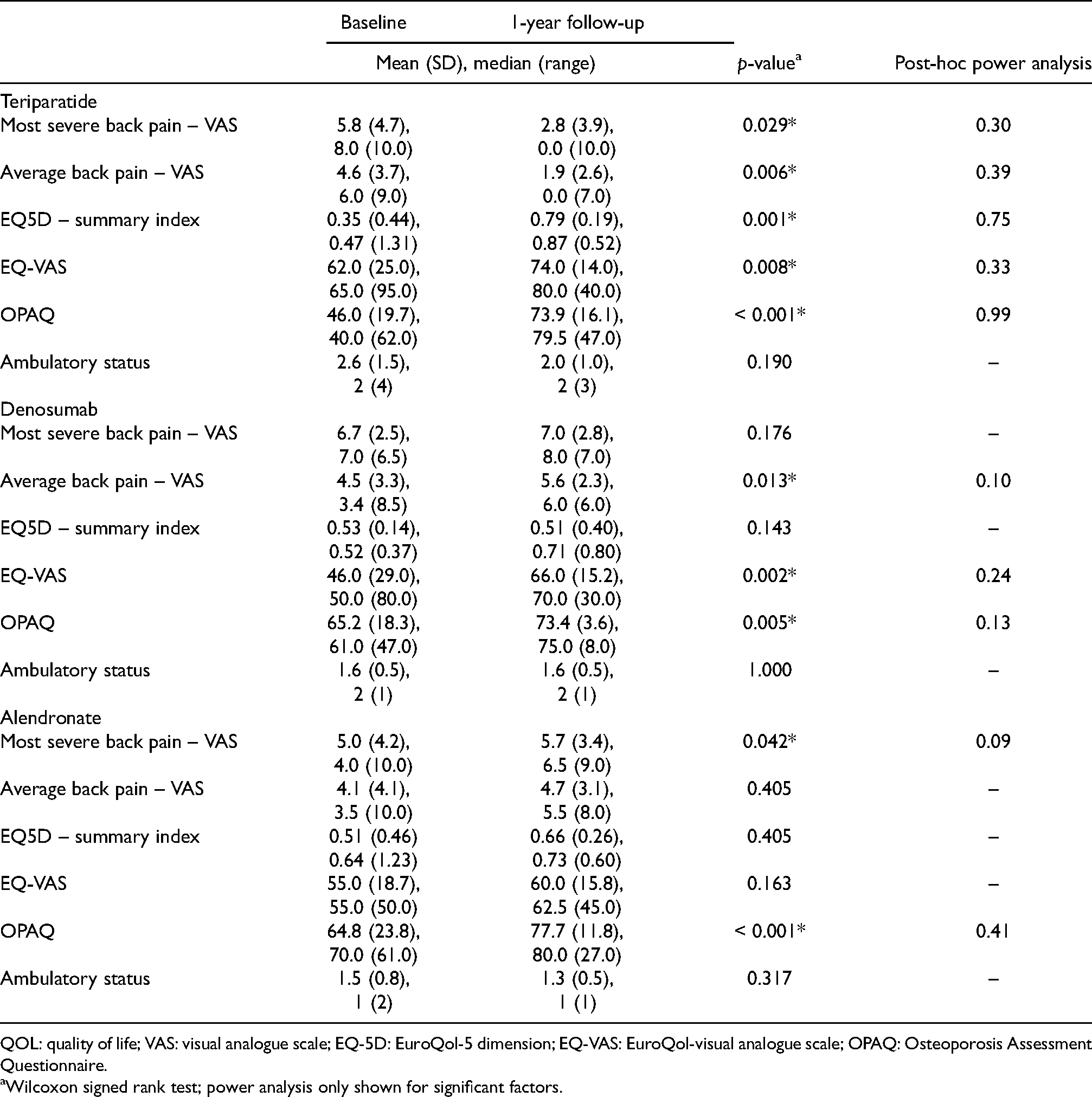
QOL: quality of life; VAS: visual analogue scale; EQ-5D: EuroQol-5 dimension; EQ-VAS: EuroQol-visual analogue scale; OPAQ: Osteoporosis Assessment Questionnaire.
Wilcoxon signed rank test; power analysis only shown for significant factors.
In the comparison across treatment groups (the teriparatide group vs the combined denosumab and alendronate group), no significant difference was demonstrated when comparing the changes in the back pain scores (most severe and average VAS back pain) and QOL outcomes (EQ summary index, EQ-VAS and OPAQ-PF score) after treatment (p > 0.05 in all five parameters).
Predictive factors for the improvement of average VAS back pain
7 (30.4%) patients achieved a reduction of average VAS back pain greater than or equal to 2.5 points. Six out of 12 individuals (50%) in the teriparatide group demonstrated MCID. One patient in the denosumab group achieved MCID, whereas none in the alendronate group had MCID.
Factors that predict the clinically significant improvement in average VAS back pain were investigated in our study. In the model (Table 3) before propensity score weighting (adjusted R2 = 0.636), the baseline average VAS back pain and the treatment group assignment were the predictive factors for back pain improvement. Higher baseline VAS was associated with higher VAS pain at a one-year follow-up (p < 0.001). For the pairwise comparison in the change in VAS back pain (Table 4), significant differences were observed in two pairs, that is, teriparatide versus denosumab (p = 0.007) and teriparatide versus alendronate (p = 0.024). Teriparatide treatment was associated with more minimally clinically significant back pain improvements as compared to the two other groups.
Multiple regression models for change of average back pain at one-year follow-up.
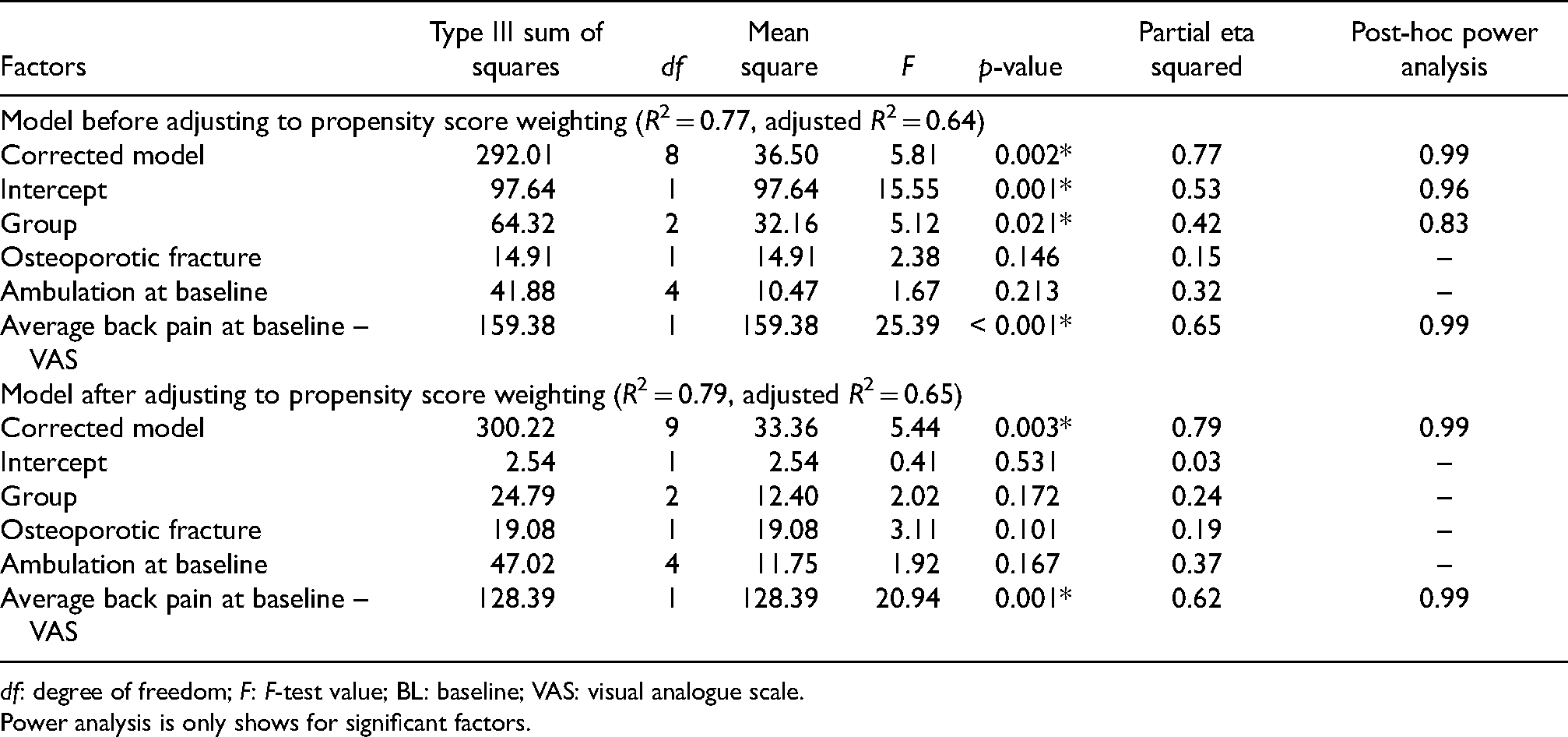
df: degree of freedom; F: F-test value; BL: baseline; VAS: visual analogue scale.
Power analysis is only shows for significant factors.
Post-hoc pairwise comparison of adjusted mean change of average back pain at one year (in VAS).
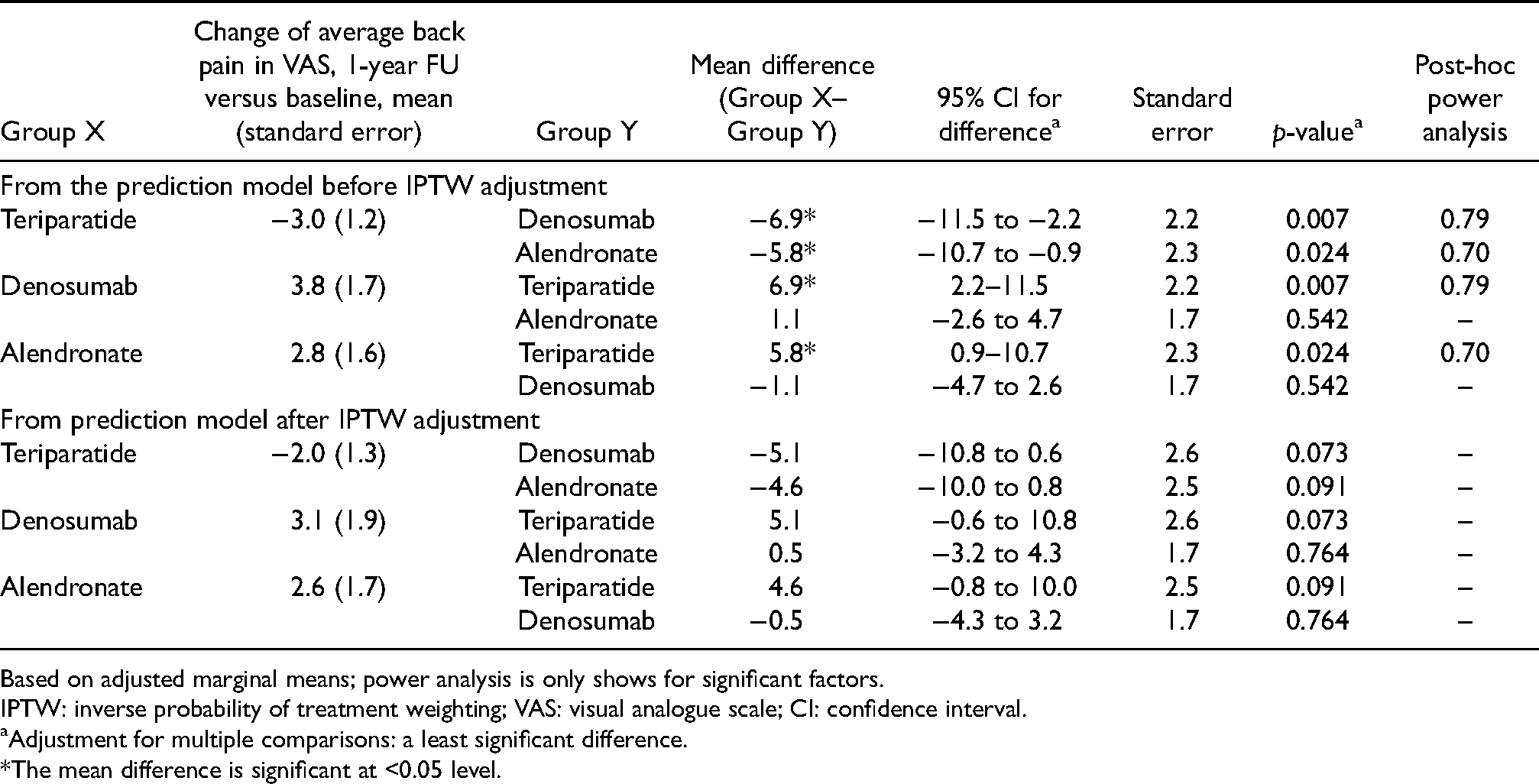
Based on adjusted marginal means; power analysis is only shows for significant factors.
IPTW: inverse probability of treatment weighting; VAS: visual analogue scale; CI: confidence interval.
Adjustment for multiple comparisons: a least significant difference.
*The mean difference is significant at <0.05 level.
Nevertheless, in the propensity score weighted model (adjusted R2 = 0.645), baseline average VAS back pain was the only significant predictive factor (partial eta squared = 0.617, p = 0.001) (Table 3). The predictive effect of the baseline VAS was observed in all three treatment groups in our study. This reached satisfactory power. After IPTW adjustment (Table 4), the teriparatide group demonstrated improvement of more than 2.5 points in average VAS back pain, while deterioration in VAS score was observed in the other two groups (2.8 points in denosumab and 2.3 points in alendronate). The intergroup difference between teriparatide and denosumab was −5.1 (p = 0.073), and that between teriparatide and alendronate was −4.6 (p = 0.091). Since p > 0.05 for both intergroup comparisons, treatment group assignment was no longer a significant factor contributing to the change in VAS back pain.
Adherence to anti-osteoporosis medications
Out of the three medications, alendronate had the lowest ADEOS-12 score (17.33 ± 2.65). Only 16.7% of patients taking alendronate had an ADEOS-12 score ≥ 20 (indicating a high probability of treatment persistence), but a significant portion (33.3%) in the group had scores ≤ 16 (indicating a high probability of treatment discontinuation).
Discussion
The effect of anti-osteoporosis medications on BMD has been well discussed in different literature. However, despite being the single most important factor in guiding treatment, BMD is not the sole factor clinicians should consider. Moreover, the improvement in BMD is often subclinical and may not drive patient-perceived outcomes. Thus, patients are seldom strongly motivated to adhere to their treatments. On the contrary, QOL measures, such as pain assessment and functional status, may be more relevant to the patients and allow patients to perceive the treatment effects more effectively.
Study results suggest that continuous treatment with parathyroid hormone analogues demonstrated significant improvements in multiple QOL parameters over one year, encompassing aspects like back pain, general health status and physical function. All the improvements were greater or equal to the MCID and were considered clinically significant. Similar trends were not observed in the other two treatment groups. In the RANKL inhibitor and bisphosphonate groups, only the OPAQ scores improved but the back pain VAS worsened. The results of our study are different from those of Sambrook et al. 16 and Hagino et al., 19 in which bisphosphate treatment was shown to be associated with significant improvement in specific domains of QOL. One of the reasons to explain the difference is that the mean age of study subjects in our study is about 5 years higher than the mean age of the two studies (around 73 years), and older patients may be more prone to additional back pain. This hypothesis was justified by the subgroup analysis in the study by Hagino et al., 19 in which the older subgroups (age ≥ 75 years) failed to show significant improvement in QOL with once-weekly bisphosphonates. Thus, we should be aware that older patients are generally less likely to benefit from anti-osteoporosis medications in terms of QOL outcomes.
Back pain in osteoporosis is a complicated process with elements of fractures, postural alterations and muscle atrophy. 29 Vertebral compression fracture is a well-recognized cause of back pain in osteoporotic patients. Its pain severity can vary from mild chronic pain to incapacitating acute back pain. 30 However, the incidence of back pain correlates well with the vertebral fracture severity. 31 In osteoporotic patients without vertebral fractures, their back pain mainly originates from muscle weakness and postural alterations. In elderly patients, osteoporosis and sarcopenia often coexist, and muscle weakness plays an important role in inducing kyphosis and chronic back pain. 29 Our study results are consistent with previous studies which showed teriparatide can reduce back pain to a greater extent as compared to alendronate.31,32 In addition to comparison with alendronate, superiority in back pain reduction in teriparatide over denosumab was observed in our study as teriparatide is the only agent showing statistically and clinically significant back pain reduction. The reason for that is largely unknown. It is believed that back pain reduction in teriparatide is mainly mediated by the reduction in the number and severity of vertebral fractures, 33 but this mechanism also applies to most anti-osteoporosis medications. Moreover, unlike alendronate and denosumab which may improve muscle strength and back pain in sarcopenia,34,35 teriparatide is not known to exert any effect on muscle strength. However, a recent study showed that teriparatide can significantly reduce mechanical hyperalgesia and the upregulation of transient receptor potential channel vanilloid 1 (TRPV1) and calcitonin gene-related peptide (CGRP) expression in mice. 36 This may offer new possible explanations for the greater back pain reduction in teriparatide.
Another interesting phenomenon was observed regarding the predictive factors for the progression of back pain in osteoporotic patients. To our knowledge, there is no existing study that particularly investigates the factors that predict the progression of back pain in osteoporotic patients. However, back pain is one of the more commonly encountered symptoms by postmenopausal women with osteoporosis with reported prevalence from 85.1% to 91.4%.29,37,38 78.3% of patients in our study also complained of variable degree of back pain at baseline. Thus, reduction in back pain is of important concern in osteoporotic patients. In our study, a general linear model was constructed to identify the predictive factors that account for MCID in average VAS back pain. Initially, without PS weighting, both baseline pain level and treatment group were significant predictive factors, with parathyroid hormone analogues outperforming the other two. Despite this, the intergroup differences (teriparatide versus denosumab: −5.1; teriparatide versus alendronate: −4.6) were quite remarkable. Our study results raised the hypothesis that teriparatide treatment may predict better back pain improvement when compared to denosumab and alendronate, but larger-scale studies are needed to prove whether the choice of anti-osteoporosis medications can influence back pain progression significantly. Nevertheless, baseline pain level remains the most important factor in predicting future back pain progression in our study, and we should not expect certain anti-osteoporosis medications to significantly improve back pain in patients with poor baseline pain levels.
Poor adherence to bisphosphonates was observed in our study and several other studies.39,40 Patients in the alendronate group particularly performed poorly in the questions concerning forgetting their medications and inconvenient drug intake instruction. Thus, we propose that the once-weekly regimen of bisphosphonate may be difficult for elderly patients to remember when they should take drugs. The requirement of patients to maintain an upright posture for 30–60 min after ingesting bisphosphonates to avoid erosive esophagitis may also cause inconvenience to the patients and account for the lower treatment adherence score. On the other hand, the uses of parathyroid hormone analogues and RANKL inhibitors are more convenient with less restriction on drug intake. However, it is important to note that six patients (33.3%) discontinued teriparatide before the end of the study. It means that the most noncompliant patients have left the study and are not included in the analysis, so the ADEOS-12 score of the teriparatide group may not reflect the true extent of treatment adherence.
The first limitation of our study is the unequal numbers of patients in the three study groups, with significant differences between the teriparatide group and the other two groups. Comparison of QOL outcomes across treatment groups was difficult as the unequal numbers limited the power of the statistical tests. Nevertheless, our study design with propensity score matching should have reduced the effect of these various confounders. In addition, from our post-hoc power analysis, the main outcome of back pain was satisfactorily powered (Table 3). Another limitation is the unequal drug-taking frequency (daily for teriparatide, 6 months for denosumab, weekly for alendronate) which may impose health-related quality of life outcomes and not able to be identified through our questionnaires.
One-year continuous teriparatide treatment was associated with significant improvements in VAS back pain, EQ-5D-5L general health status and OPAQ physical function in Chinese postmenopausal women, whereas denosumab and alendronate only demonstrated improved OPAQ scores but worsened back pain. The highest proportion of patients with MCID in average VAS back pain was also observed in the teriparatide group. In the propensity score-adjusted model, baseline average VAS back pain was the only significant factor predicting one-year back pain progression detected in our study.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the SK Yee Medical Foundation Grant (grant no. 2171223).
