Abstract
Objective
To evaluate the effect of teriparatide on life quality in patients with postmenopausal osteoporosis.
Methods
Patients treated from January 2014 to December 2016 were retrospectively included. Patients were divided into two groups according the treatment received. Those in the teriparatide treatment group were followed up for 24 months, and patients in the control group received calcium supplements and vitamin D. Scores for back pain using a visual analog scale (VAS) and score of the Oswestry Disability Index (ODI) and 36-item Short Form Health Survey of life quality (SF-36) were evaluated at 3, 6, 12, and 24 months and compared between the groups.
Results
In total, 126 patients were included in the teriparatide treatment group and 127 in the control group. There were no significant differences between the groups concerning body mass index, bone density, VAS back pain score, ODI, and SF-36 life quality scores at baseline. At 3, 6, 12, and 24 months’ follow-up, VAS scores were significantly lower in the treatment group than in controls; ODI and SF-36 scores were significantly higher in the treatment group than in the control group.
Conclusion
Teriparatide can significantly decrease pain and increase mobility and life quality in patients with postmenopausal osteoporosis.
Introduction
With increasing older populations, the prevalence of osteoporosis, especially postmenopausal osteoporosis, is continually on the rise. There are more than 54 million people with osteoporosis among people older than age 50 years in the United States and this number is expected to rise to 71.2 by the year 2030. 1 China may have up to 100 million patients with osteoporosis by 2020, 2 and an estimated 9 million new bone fractures occur owing to osteoporosis, 3 with this number increasing in many countries. Osteoporosis causes musculoskeletal pain, reduced mobility, and fracture risk upon falling and lifting heavy items, which can have fatal consequences owing to being bedridden for a long period and an accompanying decline in overall health status and life quality of the patient.
Teriparatide, an artificially synthetized human parathyroid hormone 1-34, has been proven to increase bone density in women with postmenopausal osteoporosis by stimulating osteoblasts and promoting bone formation.4,5 However, there are few reports regarding the effect of teriparatide on life quality among Chinese patients. 6 In the current study, we used different assessment methods to determine the efficacy of teriparatide in promoting quality of life among patients with postmenopausal osteoporosis.
Methods
The present research was approved by the ethics committee of the authors’ affiliated institutions (approval number: HZYLLBA201910). Patients participating in the study provided verbal informed consent. All patients agreed to publication of their clinical data, provided that their personal identity was not revealed. All procedures were conducted in accordance with the Declaration of Helsinki. 7 All included patients had a confirmed diagnosis of postmenopausal osteoporosis and were treated at the authors’ institutions between January 2014 and December 2016. Patients were diagnosed with postmenopausal osteoporosis if they had a T-score less than or equal to −2.5 at the spine or hip or if the patient had experienced fragility fracture. 8
Patient inclusion
Inclusion criteria were as follows: 1) patients with a confirmed diagnosis of osteoporosis; 2) those with no severe cardiovascular or gastrointestinal disorders; 3) those who received teriparatide treatment, calcium or vitamin D supplements but no antiresorptive drugs during the study period; 4) those who were followed up for at least 2 years; and 5) patients over age 60 years as we normally recommend teriparatide treatment to patients with severe osteoporosis in this age group.
Exclusion criteria were: 1) patients with bone sarcomas; 2) those with long-term use of steroids; 3) those undergoing dialysis for chronic renal failure; 4) those with severe cardiovascular and gastrointestinal diseases; and 5) patients who used antiresorptive drugs during the observation period.
Upon analyzing the data of patients treated during the study period (from January 2014 to December 2016), we enrolled patients who received teriparatide treatment for more than 3 months and met the inclusion criteria; these patients comprised the teriparatide treatment group. Among patients receiving only calcium and vitamin D supplements, those who began follow-up after patients in the teriparatide treatment group were retrospectively included in the control group.
Treatment
Patients in the teriparatide treatment group received 20 µg of teriparatide per day, via abdominal subcutaneous injection, for more than 3 months. Patients in the control group received calcium and vitamin D supplements and did not receive any antiresorptive drugs.
Outcome assessment
Demographic characteristics of the included patients, such as age and body mass index (BMI), back pain score assessed using a visual analog scale (VAS), 9 the Oswestry Disability Index (ODI) score 10 , and 36-item Short Form Health (SF-36) life quality score 11 were evaluated every 3 months and compared between the groups.
Statistics
All data were analyzed using IBM SPSS 22.0 statistical software (IBM Corp., Armonk, NY, USA). Quantitative measures were analyzed using independent sample t-tests. Differences were considered significant with P < 0.05. Data are presented as mean ± SD.
Results
A total of 126 patients were included in the teriparatide treatment group, and 127 were included in the control group. Among patients included in the final analysis, there were no significant differences between the two groups concerning BMI, bone density, VAS back pain scores, ODI, and SF-36 life quality scores (Table 1).
Demographic characteristics of patients before treatment. There were no significant differences between the two groups concerning back pain (VAS), ODI, or SF-36 life quality scores at the beginning of the study.

Abbreviations: BMI, body mass index; QCT, quantitative computed tomography; SF-36, 36-item Short Form Health Survey; ODI, Oswestry Disability Index; VAS, visual analog scale.
Data presented as mean ± SD.
Patients in the treatment group reported side effects of teriparatide such as headache (n = 3), vertigo (n = 2), nausea or abdominal discomfort (n = 8), edema (n = 1) of the lower extremities, and pain in the extremities (n = 2). Patients in the control group did not report any effects of vitamin D and calcium supplementation. During follow-up assessment after treatment, there were significant differences between the groups concerning VAS back pain scores, ODI, and SF-36 life quality scores beginning at 3 months’ follow up (all P < 0.01) (Table 2, Figures 1–3).
Back pain scores (VAS), ODI, and SF-36 life quality scores at different time points during follow up.

Abbreviations: SF-36, 36-item Short Form Health Survey; ODI, Oswestry Disability Index; VAS, visual analog scale.
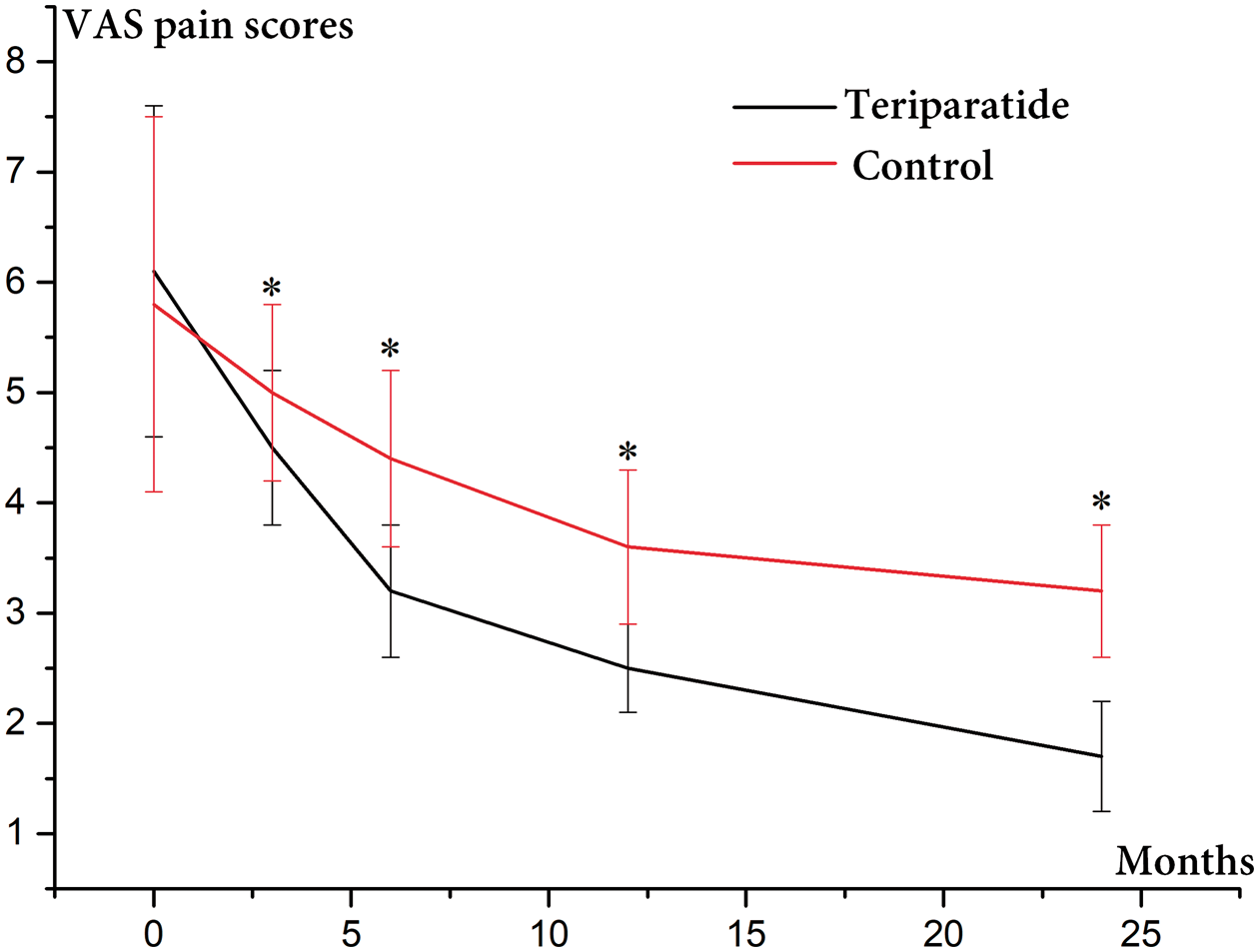
Back pain scores were significantly lower in the teriparatide treatment group than in the control group at 3, 6, 12, and 24 months after treatment .*P < 0.01.
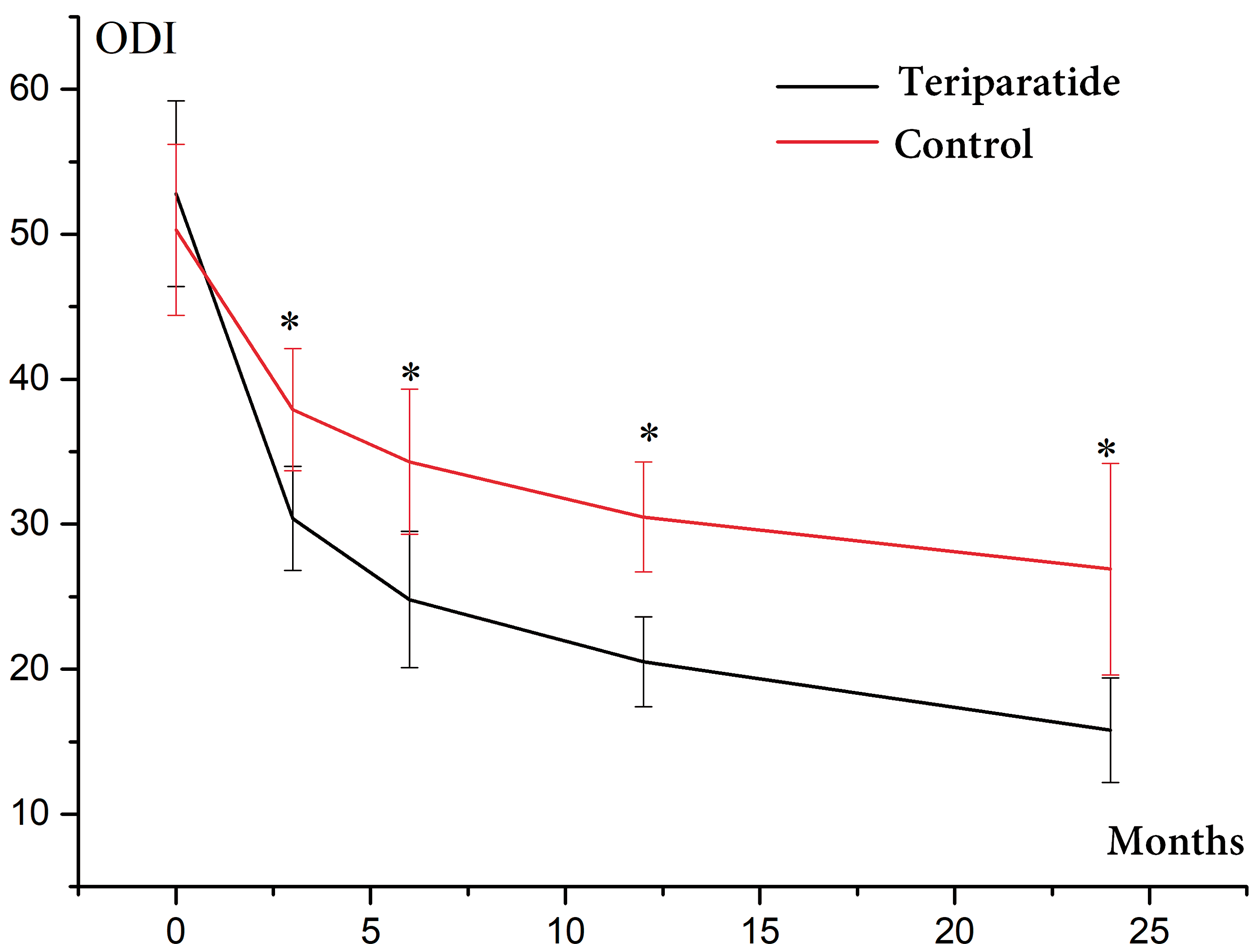
Oswestry Disability Index scores were significantly lower in the teriparatide treatment group than in the control group at 3, 6, 12, and 24 months after treatment. *P < 0.01.

Scores of 36-item Short Form Health Survey, life quality were significantly higher in the teriparatide treatment group than in the control group at 3, 6, 12, and 24 months after treatment. *P < 0.01.
Discussion
Unlike most reports on the effect of anti-osteoporosis drugs, outcome assessment in the current research was focused not only on bone mineral density but also on the subjective assessment of life quality. By analyzing the data of 253 Chinese patients with postmenopausal osteoporosis, we found that use of teriparatide can significantly alleviate back pain, improve patient quality of life, and increase patient mobility.
Anti-osteoporosis drugs include those that prevent bone resorption by inhibiting the activity of osteoclasts, as well as those that promote new bone formation by activating osteoblasts. Bisphosphonates and denosumab have been reported to increase bone mineral density and decrease the risk of fragility fractures.12,13 Teriparatide is the first synthetic human parathyroid hormone that can promote bone formation by stimulating osteoblasts. 14
Over the previous two decades, numerous studies have reported the effects of teriparatide on increased bone mineral density, reduced pain, and improved mobility. A series of meta-analyses of randomized controlled studies of teriparatide revealed that teriparatide positively affects bone mineral density, decreases fracture risk, and reduces VAS back pain scores.15–17
However, there is little evidence in the existing literature on the effect of teriparatide on patients’ life quality, and most studies have been carried out among white populations. These reports have therefore included few data on the effect of teriparatide among patients of Chinese origin. In an 18-month follow-up study by Ying et al. 6 that included 453 postmenopausal Chinese women, there were no significant differences between patients receiving teriparatide 20 µg (200 U) daily for 18 months (n = 341) and those receiving elcatonin 20 U weekly for 12 months (n = 112) with respect to back pain evaluated using a VAS. However, there was no control group that received antiresorptive drugs in that study. In the current study among patients in China, we retrospectively investigated the effects of teriparatide on the life quality of patients with postmenopausal osteoporosis. We found that teriparatide has positive effects on VAS back pain scores, ODI, and SF-36 life quality scores from 3 months after treatment. However, side effects such as headache, vertigo, nausea, abdominal discomfort, edema of the lower extremities, and pain in the extremities were reported by some patients in the teriparatide treatment group. The current literature includes reports of side effects owing to teriparatide such as headache, vertigo, nausea, abdominal discomfort, and pain in the extremities, which are in accordance with the results in our patient series.
To our best knowledge, the current study is one of the few reports on the effect of teriparatide on the life quality of Chinese patients with postmenopausal osteoporosis. Among our participants, those who discontinued the trial after 1 month have reported a significant reduction of pain and increased mobility, indicating that even short-term use of teriparatide can be beneficial to quality of life. Patient compliance is an important factor in achieving satisfactory recovery from chronic diseases such as osteoporosis. 18 It is clear from this study that, in Chinese patients, teriparatide treatment is adequate for increasing patients’ quality of life, which has strong positive effects on patient compliance. Results of this study can be used as a guide for the use of teriparatide in Chinese patients with postmenopausal osteoporosis.
The main limitation of the current study is its retrospective nature. Patients were not randomly assigned to different treatment groups. In addition, we did not investigate patient income; however, as teriparatide is not covered by medical insurance, patients who receive teriparatide treatment are normally wealthier than those who do not. In our study, this might cause patient selection bias. However, we used age over 60 years as a criterion for inclusion of patients in the teriparatide treatment group. To avoid bias arising from the different age groups, patients in the control group were also age 60 years or older. As a result, the baseline characteristics of patients in the two study groups were similar at baseline, and differences were found only after patients had received different therapeutic interventions. Additional randomized controlled trials with larger sample sizes and longer follow-up periods are necessary to further evaluate the effect of teriparatide on the life quality of patients with postmenopausal osteoporosis.
Conclusion
Teriparatide can significantly decrease lower back pain, increase mobility, and improve life quality in patients with postmenopausal osteoporosis.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The research reported in this publication was supported by the Natural Science Foundation of China (No: 81704170, 81873378, 81503669), Young Scholars Cultivation Project of Shenzhen People’s Hospital (No: SYKYPY201925), Project of Traditional Chinese Medicine of Heilongjiang Province (No: ZHY16-003), and Science Fund of Heilongjiang University of Traditional Chinese Medicine (Nos: 2014bs01, 2018pt03).
