Abstract
Purpose:
Inflammation plays a major role in tumour development, progression and metastasis. Multiple inflammatory markers such as absolute lymphocyte count, neutrophil–lymphocyte ratio (NLR), lymphocyte–monocyte ratio (LMR) and platelet–lymphocyte ratio have been discovered as prognostic markers for various malignancies. We investigate preoperative lymphocyte count and other cell count ratios and their relation to survival and prognosis of sarcoma patients after surgery.
Methods:
A total of 142 patients from the Orthopaedics Oncology Database were included into this retrospective study. Kaplan–Meier curve and multivariate Cox proportional models were used to calculate the overall survival of patients with sarcoma who underwent radical excision surgery.
Results:
High preoperative LMR is significantly associated with better overall survival and prognosis in sarcoma patients, whereas high preoperative NLR is significantly associated with shorter overall survival and poorer prognosis. Multivariate analysis shows that LMR and NLR are good predictors for overall survival at 3 and 5 years after surgery, respectively. Patients with high preoperative lymphocytes count are associated with longer overall survival, but this association is not statistically significant. Our findings suggest that preoperative NLR and LMR are good predictive markers for survival of sarcoma patients.
Conclusion:
LMR and NLR can be used to identify patients at risk for poor clinical outcome, so that a more aggressive course of treatment can be applied to improve outcome. These are cost-effective prognostic tools as they are calculated from routine preoperative peripheral blood counts. In conclusion, preoperative NLR and LMR are good prognostic markers for predicting the clinical outcome of patients with sarcoma.
Introduction
Sarcomas are tumours of putative mesenchymal origin. They constitute about 20% of all paediatric age group malignant solid cancers and about 1% of all malignant solid cancers of adults. 1 It is difficult to detect early-stage sarcomas as there is lack of symptoms. Majority of the patients present at later stages and approximately 50% succumb to the disease due to metastasis and local recurrence. 2 The survival of metastatic sarcoma patients for the past two decades has stagnated at 20–45%, despite the advancement in chemotherapy, radiotherapy and surgical interventions. 3
Currently, there are about 50 subtypes of sarcoma described in the World health Organization classification and many of them can occur at any age group and at different locations of the body. 4 Further classification according to molecular subtypes is now made possible due to the availability of molecular classification studies literature. The exact overall incidence of sarcoma is not known as most studies analysed different subtypes, adult or children, or soft tissue and bony separately.
Extensive studies are being conducted in search of prognostic markers for sarcoma, so that treatment and follow-up schedules can be specified for each individual sarcoma patient to improve outcome. Current cancer research is aimed at identifying novel biomarkers that are easily available, highly reproducible, cheap and able to identify propensity for recurrence and death. Conventional prognostic factors such as age at diagnosis, tumour size, location of tumour, histological grade, histological subtype and tumour depth are useful as general risk assessment tool to determine the prognosis, surgical strategy and adjuvant management for patients diagnosed with sarcoma. 5
Inflammation is a major component in tumour genesis. 6,7 The microenvironment of tumour is highly influenced by inflammatory cells. Clinical evidence suggest that high neutrophil count, as a surrogate marker of inflammation, has an immunosuppressive effect on the host and translates to poor prognosis as neutrophils suppress the cytotoxicity of immune effecter cells. 8 Meanwhile, lymphocyte count acts as a surrogate marker of host immunity as patients with higher pretreatment lymphocyte count showed significantly better survival rates. 9 The accumulation of inflammatory cells around neoplasm plays an important role in tumour progression and prognosis. Patients with tumour stroma containing higher density of infiltrating lymphocytes are associated with better outcome compared to those with a high density of infiltrating neutrophil. 10 High neutrophil and low lymphocyte counts in patients with various solid tumours have been widely studied and concluded that host immunity was suppressed, translating to poor clinical outcome. 11,12 High tumour infiltrate with lymphocyte subsets, CD4+ and CD20+ lymphocytes, showed to be a significant independent positive prognostic factor for improved disease-specific survival of patients who had undergone surgical resection with wide resection margin. 2
Various biomarkers such as neutrophil–lymphocyte ratio (NLR), C-reactive protein (CRP), platelet–lymphocyte ratio (PLR) and lymphocyte–monocyte ratio (LMR) have been generally accepted as prognostic markers for various types of cancer. 13
In this study, we investigated the significance and relationship of preoperative lymphocyte count and other white cell count ratios (NLR, LMR and PLR) with the overall survival of sarcoma patients after surgical resection.
Methodology
This is a retrospective study involving patients diagnosed with soft tissue and bony sarcoma of the limb treated at the Orthopaedic Oncology Unit. Medical records for patients treated from year 2004 until June 2014 were reviewed. This study was approved by Medical Ethics Committee of University Malaya Medical Centre on 13 October 2016.
The inclusion criteria were patients diagnosed with soft tissue and bony sarcoma of various subtypes treated with surgical resection of primary tumour, with available preoperative full blood count (FBC) and white blood cell differential count results. Patients diagnosed with soft tissue sarcoma and bony sarcoma but treated non-surgically are excluded. Patients with no white blood cell differential count in their preoperative FBC were also excluded.
Preoperative FBC, subtype of sarcoma, and demographic data of all included patients were traced via laboratory database and electronic medical records. NLR, LMR and PLR for each patient were then calculated and recorded. Duration of survival for each patient up to 5 years post-surgical excision of primary tumour was traced via Oncology Unit follow up notes and by cross referencing patients’ identification number with the National Registration Department of Malaysia to identify mortality. All data collected were recorded in a Microsoft excel sheet.
All statistical analysis was performed using the Statistical Package for Social Science software version 20. The primary end point of this study was the duration of overall survival post-surgical excision of primary tumour, which was calculated from the day of operation to the day of death or survival up to 5 years. Cut-off values for preoperative absolute lymphocyte count (ALC), NLR, preoperative LMR and PLR were determined based on figures obtained from existing literature based on the Asian population. Correlation analysis among the dependant and independent variables were performed to determine the significance of the analysis.
Sample size estimation for this study was done with the requirement for significance level at 0.05, power of study of 80%, using power and sample size calculation software. The hazard ratio of 2.97 was obtained from the study by Liu et al. 13 The median survival time of control for the patients was at 53 months and the ratio of control to experimental group, as obtained from literature was at 1.1. Recruited patients were followed up for at least 60 months. 14 An estimated 10% of missing data/lost to follow-up was added to the final figure. A minimum of 43 patients per group were needed for this study.
A total of 142 patients were finally included into this study from the Orthopaedic Oncology Database.
Results
A total of 1072 patients were registered into the Orthopaedic Oncology Database from 1 January 2004 up to 31 December 2014. A total of 589 patients were selected for screening to be included into this study based on diagnosis. A total of 151 patients met the inclusion criteria, while 9 of them were excluded due to inability to determine their mortality status as they were foreigners. A total of 142 patients were finally included into this study.
The mean age of patients with confirmed histological diagnosis of sarcoma was 40.88 years (SD ±21.10 years), with 82 being male (57.7%) and 60 female (42.3%). As for ethnicity, 65 patients were Malay (45.8%), 56 were Chinese (39.4%), 20 were Indian (14.1%) and 1 patient was a native ‘Orang Asli’ (0.7%). Based on sarcoma subtypes, 47 patients were diagnosed with bony sarcoma (33.1%), while 95 patients were diagnosed with soft tissue sarcoma. For the location of the sarcoma, it was located in the trunk for 18 patients (12.7%), while 123 patients had it located at the periphery (86.6%) and 1 patient at an unknown location (0.7%). The median and range for each prognostic ratio were derived from the collected blood parameters as follows: 2.65 (range 0.35–87.75) for NLR, 2.80 (range 0–13.82) for LMR and 174.87 (range 41.62–1800.00) for PLR. The median for ALC was at 1.81 × 109 L−1 (range 0.08–6.44 × 109 L−1). The premorbids of the patients included in the study are as listed in Table 1. Table 2 shows that there is no significant difference in NLR and LMR between group of patients with or without premorbids.
Premorbid conditions in the study group.

DM: diabetes mellitus; HTN: hypertension; ITP: immune thrombocytopenic purpura.
Association between chronic medical premorbid and NLR and LMR.
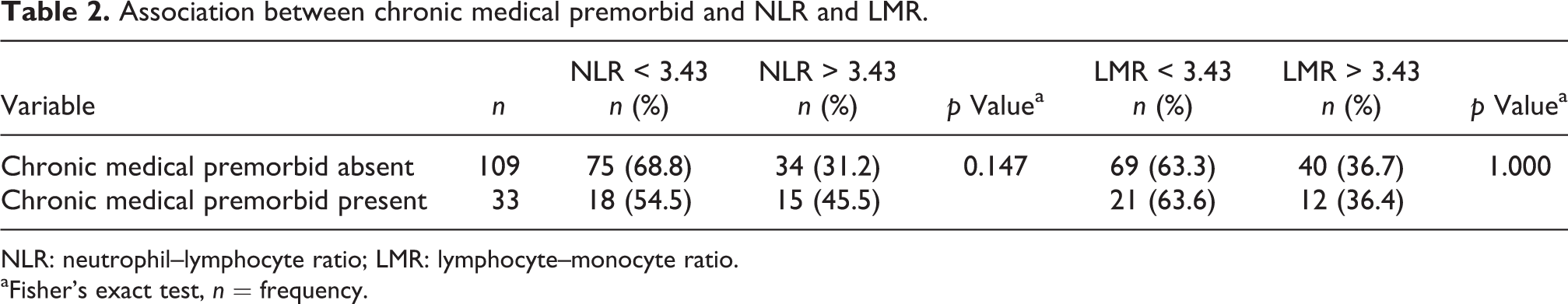
NLR: neutrophil–lymphocyte ratio; LMR: lymphocyte–monocyte ratio.
aFisher’s exact test, n = frequency.
In univariate analysis, patients with low NLR and high LMR had a significantly longer overall survival compared to patients with high NLR and low LMR at 3- and 5-years post-surgery (Tables 3 and 4; Figures 1 and 2). Patients with low PLR showed a significantly longer overall survival compared to high PLR at 3 years post-surgery as shown in Table 4. There was no significant difference in the survival time for patients with ALC of more than 1 compared to patients with ALC of <1 at 3- and 5-years post-surgery.
Mean of survival time among patients with different NLR and LMR ratio at 5 years post-surgery.
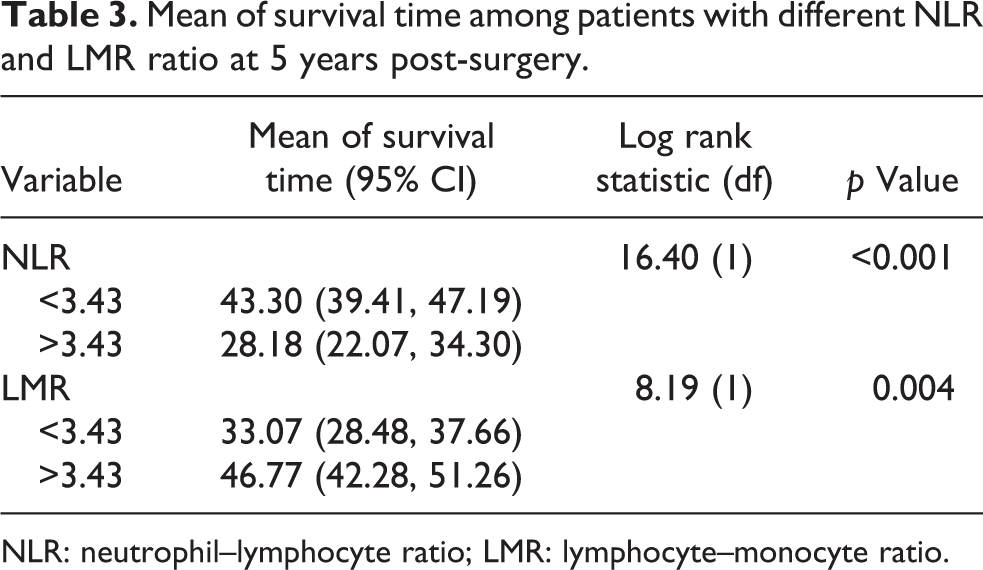
NLR: neutrophil–lymphocyte ratio; LMR: lymphocyte–monocyte ratio.
Mean of survival time among patients with different NLR, LMR and PLR at 3 years post-surgery.

NLR: neutrophil–lymphocyte ratio; LMR: lymphocyte–monocyte ratio; PLR: platelet–lymphocyte ratio.
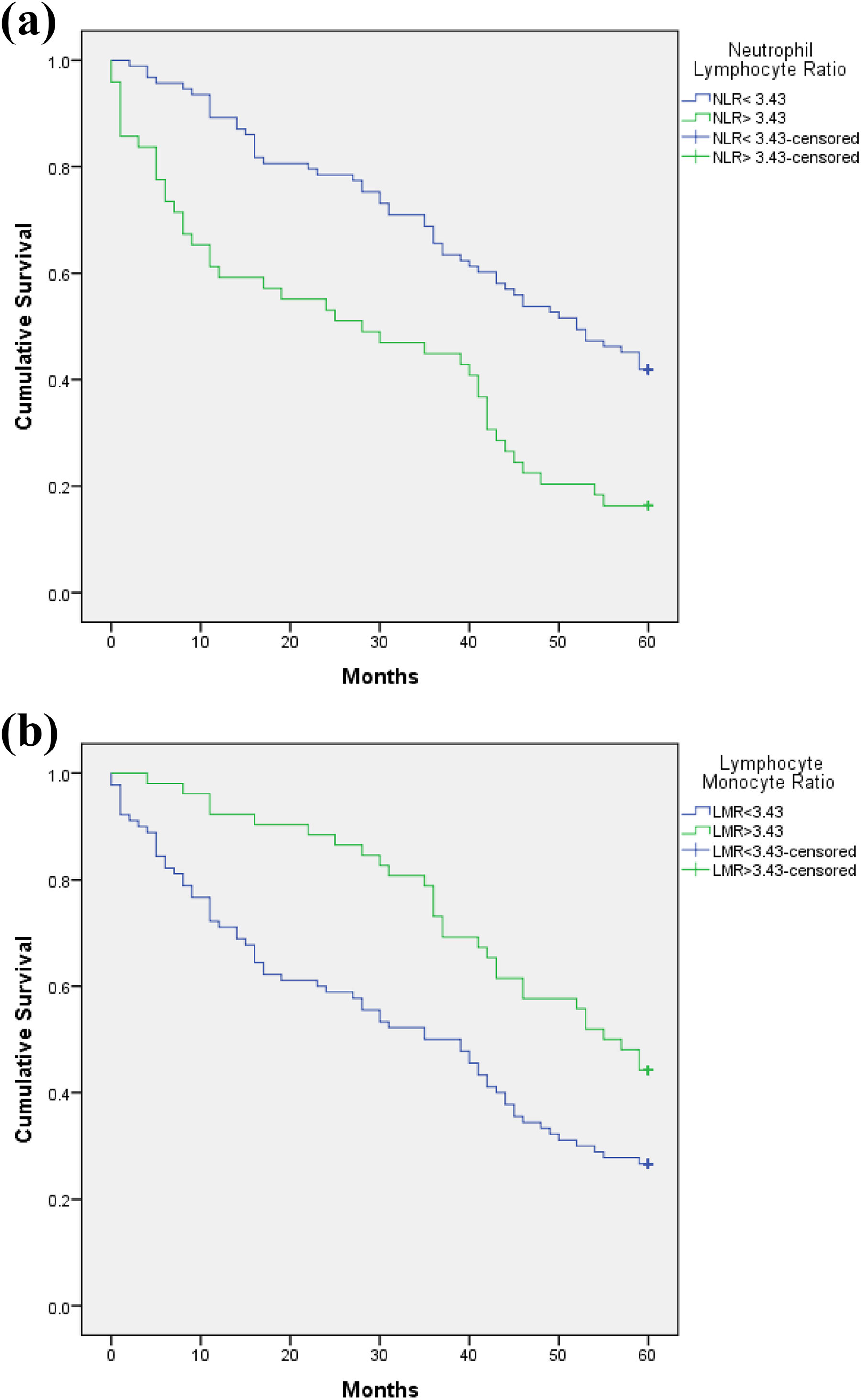
Kaplan–Meier survival curve among patients with different (a) NLR and (b) LMR at 5 years post-surgery. NLR: neutrophil–lymphocyte ratio; LMR: lymphocyte–monocyte ratio.
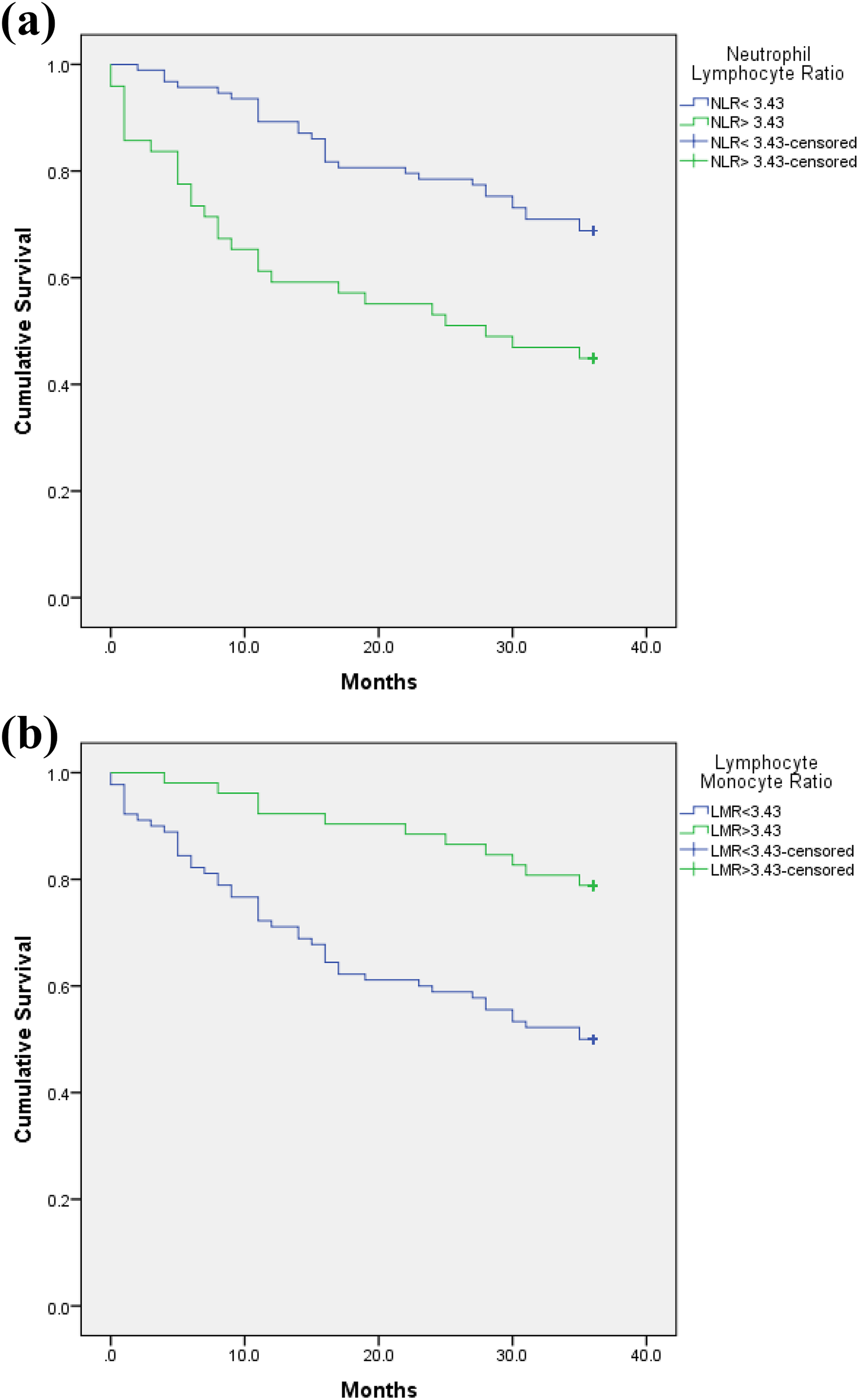
Kaplan–Meier survival curve among patients with different (a) NLR and (b) LMR at 3 years post-surgery. NLR: neutrophil–lymphocyte ratio; LMR: lymphocyte–monocyte ratio.
Multivariate Cox regression analysis applied to all the parameters after adjusted with other confounding factors found that preoperative NLR and LMR were significant parameters affecting the overall survival time at 5 and 3 years post-surgery. Patients with NLR of >3.43 was at 1.87 times higher risk (hazards ratio (HR) 1.87; 95% CI 1.17, 2.99) of dying from sarcoma compared to patients with NLR of less than 3.43 at 5 years post-surgery. Patients with LMR of >3.43 was at 58% lower risk (HR 0.42; 95% CI 0.20, 0.87) of dying from sarcoma compared to the patients with LMR of <3.43 at 3 years post-surgery.
Discussion
Inflammation and neoplasm have been recently proved to be highly correlated with each other, with inflammatory cells playing a major role in pathogenesis and progression of neoplasm. 15 Virchow’s hypothesis in year 1863, which stated that neoplasm arises from sites of chronic inflammation, was later proved by the discovery of leucocytes in neoplastic tissues. 16 Since then, numerous research studies have been conducted and showed the importance of inflammation and its influence in the microenvironment of tumour. Tumour cells induce inflammation by secreting an array of cytokines due to tumour necrosis, which in turn triggers host inflammatory response and attracts inflammatory cells with the capability of secreting factors to promote survival of tumour cells through inhibition of apoptosis, evasion of innate host adaptive immunity, promotion of angiogenesis and cause DNA damage. 10 Host inflammatory response triggered by the tumour causes changes to the composition of cells in the systemic blood circulation. The most common alterations seen are increased neutrophil count, reduced lymphocyte count and increased monocytes count. 17 Other than changes in the number of white blood cells subsets, inflammation caused by tumour is also associated with increased platelet count and anaemia. The above-mentioned changes in the haematological parameters contribute to the survival and progression of tumour and subsequently metastasis. Since systemic inflammation is highly associated with progression of neoplasm, extensive research has been done to look for the association between the systemic blood cell composition changes and the progression of disease, and the possible mechanism of these changes which promotes the growth and metastasis of neoplasm.
In the recent decade, preoperative blood counts in various type of solid neoplasm including sarcoma have been shown to provide a relatively reliable and cost-effective way to predict the outcome of patients who are planned for curative intent surgery. The most commonly studied preoperative blood parameters are the ratios between the cell counts which show the balance between factors that promote the progression of malignancy and anti-tumour efficacy of host immunity. Table 3 provide a list of published literature on the relationship between preoperative blood counts and the outcome of sarcoma patients.
Lymphocyte count is known as the surrogate of host cell-mediated immunity which contributes to the host defence against malignancy. Tumour infiltrating lymphocytes are an indication of host immune reaction towards tumour antigen. Increase in numbers of CD4+ and CD20+ lymphocytes in tumour is associated significantly with improved disease-specific survival in patients who had undergone wide resection margin. 2 Lymphocyte count in a cancer patient can be reduced by lympholytic cytokines secreted by the tumour, where lymphocytes undergo activation-induced apoptosis. This was shown in vivo, where ligands such as FasL or tumour necrosis factor β (TNF-β) produced by patients with cancer-induced lymphocyte apoptosis. Other cytokines such as interleukin 6 (IL-6), prostaglandin E2, IL-10 and TGF-β produced by cancer cells in the tumour environment are released into systemic circulation, which can impair the differentiation of normal functioning dendritic cells needed to maintain the physiological lymphocyte homeostasis and lymphocyte subset pools. 9
Reduced preoperative lymphocyte counts are associated with a higher stage of malignancy and poorer survival outcome. Previous studies have reported a significantly positive correlation between cancer curability and blood lymphocyte count in various cancers with a threshold of 1000 μL−1. 9 Patients with preoperative lymphocyte count of below the 1000 μL−1 threshold have a higher risk of recurrence compared to those with normal or higher lymphocyte count. Preoperative lymphocyte count also has a high predictive value for haematological toxicity and early death in addition to its prognostic values for overall survival in patients with various types of cancers.
Monocytes in the circulation, which is another subset of leucocytes, also play an important role in the progression of tumour. Existing literature shows that monocytes play a controversial role where they inhibit and also enhance the growth of human cancer cells at the same time. 12 Heuff et al. 18 reported that Kupffer cells in the liver eliminate circulating cancer cells which demonstrate their ability to kill tumour cells. 12 Recent studies show that tumour-associated macrophage (TAM), which is derived from circulating monocytes, play a role in promoting tumour growth. These macrophages are selectively recruited into the tumour microenvironment via locally produced chemotactic factors. They are highly correlated with an increased angiogenesis, tumour invasion and poor prognosis in various tumour types. Soluble factors released by these macrophages enhance neo-angiogenesis, which in turn stimulate growth of tumour cells and release of enzymes that regulate digestion of extracellular matrix promoting tumour cell migration and metastasis. Increased monocytes count in the circulation indirectly reflects the increased production of TAM which acts as a surrogate marker for a high-tumour burden. 5
Neutrophil is the most abundant subset of white blood cells in the systemic circulation, which are responsible for inflammation. Tumour growth induces tissue inflammation, which in turn causes the neutrophil count to increase in circulation. High neutrophil count has been widely accepted as the surrogate marker of inflammation, and many have observed its association with patients’ clinical outcome in various types of malignancies in the past decade. Many authors have confirmed that high neutrophil count is highly associated with poor clinical outcome in various types of cancer. 7,19 This association was later supported by in vitro studies which showed that high neutrophil counts suppressed cytotoxic effecter cells in patients which translated into poor clinical outcome. 11
Neutrophil is also known to stimulate and promote the progression of cancer cells via various mechanisms. Various cytokines are released by tumour cells to activate and recruit neutrophils from the peripheral blood into the tumour microenvironment. These neutrophils produce mediators such as vascular endothelial growth factor and matrix metalloproteinase 9, which stimulate angiogenesis in the tumour stroma. Neutrophils also generate reactive oxygen species, nitric oxide and arginase, which suppresses T lymphocytes functions and cause genomic instability, which ultimately produce an optimal environment for tumour cells to thrive in and spread. 10
Platelets in the circulation are the key element that links haemostasis, inflammation and tissue repair. Platelet count in peripheral blood reflects the degree of systemic inflammation, as pro-inflammatory mediators such as IL-1, IL-3 and IL-6 stimulate the proliferation of megakaryocyte which is known to be responsible for platelet production. In recent studies, many published data show that platelets have a major role in the growth and progression of various types of malignancy. They secrete pro-angiogenic factors as well as various growth factors into the tumour microenvironment which stimulate neo-angiogenesis and growth of tumour cells. In vitro experiments have shown that platelets can inhibit tumour cells apoptosis and reverse the cell cycle arrest induced by chemotherapeutic agents such as 5-flurouracil. 20 Platelets have also been found to shield tumour cells from host cell-mediated immune response by attenuating the natural killer cells ability and promote metastasis by inducing epithelial mesenchymal transition as well as microvascular permeability, which promotes extravasations of platelet–tumour cell complex to distant sites. 21
In this study, we have investigated the influence of four blood parameter ratios that are reflective of the patients’ systemic inflammatory response towards sarcoma. The preoperative blood parameters, namely, ALC, LMR, NLR and PLR, reflect the balance between factors which promote the progression of sarcoma and anti-tumour efficacy of host immunity. These preoperative blood ratios, with the understanding of the role played by each type of inflammatory cells at various steps of tumour progression, have shown to have high prognostic value on the outcome of sarcoma patients by many studies and are highly associated with the overall survival of patients with solid tumours.
Preoperative LMR and NLR in this study are found to be significantly associated with the overall survival of patients with sarcoma at 3 and 5 years post-curative intent surgery in univariate analysis. High preoperative LMR is significantly associated with better overall survival and prognosis in sarcoma patients, whereas high preoperative NLR is significantly associated with shorter overall survival and poorer prognosis. These results are similar to many published literature as listed in Table 6 on the association of preoperative peripheral blood LMR and NLR levels with overall survival and prognosis of patients in different types of malignancies. We have also found the association of tumor grade with LMR where higher tumor grade is associated with lower LMR and vice versa as shown in Table 5. However, in multivariate analysis, LMR is only found to be significantly associated with overall survival of patients at 3 years post-surgery and NLR at 5 years post-surgery. This shows that NLR is a good overall survival prognostic marker for a long-term period post-surgery, while LMR is a good prognostic marker for overall survival in the short-term period after taking into account other confounding factors.
Comparing median LMR among tumour grade.

LMR: lymphocyte–monocyte ratio; IQR: interquartile range.
aKruskal–Wallis test.
List of published literature on peripheral blood counts and outcome of sarcoma patients.

NLR: neutrophil–lymphocyte ratio; CRP: C-reactive protein; LMR: lymphocyte–monocyte ratio; PLR: platelet–lymphocyte ratio; STS: soft tissue sarcoma; ESR: erythrocyte sedimentation rate.
The cut-off value for each parameter is different as most authors determine the cut-off value based on the blood test results of the population at the respective region in which the study was carried out. Ethnic variation of patients in different regions also affect the cut-off values for each parameter and also the final results. For example, cut-off values for parameters in European countries are not suitable to be applied to the Asian population. This is because Caucasians have higher peripheral blood neutrophil count and lower lymphocyte count compared to Asian population. 20 In this study, we used the cut-off values suited to the Asian population. The limitations in our study include the small sample size and the retrospective study design done at a single centre with a mixture of various types of bony and soft tissue sarcomas.
Conclusion
In this study, preoperative lymphocyte count correlates with the survival of sarcoma patients post-excision surgery, but not as an independent prognostic factor. Preoperative NLR and LMR are good prognostic predictors of survival in sarcoma patients. The routine combined use of these preoperative markers to identify sarcoma patients with higher risk of mortality is recommended. Future prospective study with larger sample size is recommended to substantiate and validate our results.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
