Abstract
Background:
Few studies have focused on the correlation between the clinical variables and the survival in Epithelioid Sarcoma (ES). The aim of this study was to investigate the relevant clinical variables influencing the survival of ES patients.
Methods:
From March 2000 to April 2018, 36 patients (median age, 38 years, range 22–61 years) with ES were evaluated, treated, and followed up.
Results:
All 36 patients underwent resection in our hospital. Among them, the 2 and 5 years local recurrence rates were 32.0% and 45.1%, respectively, with a better prognosis in patients with R0 resection margin. Distant metastasis rates for the 33 patients with M0 after 2 and 5 years were 51.5% and 70.8%, respectively. Overall survival rates at 2 and 5 years for 36 patients were 74.8% and 43.3%, respectively. Tumor size (>5 cm) and M1 were significantly associated with a poor overall survival. But the R0 resection margin was the only prognostic factor for influencing the LRFS and DMFS.
Conclusions:
The R0 resection margin and small tumor size were critical for a better prognosis.
Introduction
Epithelioid sarcoma (ES) is rare, high-grade, heterogeneous group of malignancy derived from mesenchymal tissues that constitutes <1% of all soft tissue sarcomas (STS). 1 It was first described in 1961 by Laskowski, 2 and further perfected by Enzinger. 3 ES originates from the mesenchymal tissue with a complex histogenesis morphology, which makes it extremely easy to be misdiagnosed. It can either be a slow-growing solitary painless firm nodule or it can take the form of multiple, sometimes with overlying skin ulceration, which occurs mainly in young adult males, predominantly affecting fasciae, subcutaneous tissues, or tendon sheaths of the extremities, with a predilection for forearms and hands. 4 ES has a propensity to spread via hematogenous and lymphatic system, and shows tenacious distant and local progression, 5 and the reported overall 5-year overall survival rate is about 32–78%. 6 –10 Surgical resection en bloc with or without radiotherapy/chemotherapy is the principle of treatment for primary-localized sarcoma. 2 Even with wide resection, there remains a high risk of local recurrence and metastatic spread. 1,11,12 The therapy for epithelioid sarcoma is unclear especially for advanced stage tumors.
The aim of this study was to investigate the impact of the relevant clinical variables on the survival of ES patients. We sought to investigate the oncological outcomes of ES patients post-surgical treatment, identify the independent predictors of survival among clinical variables and demonstrate what can be done to benefit the patient.
Materials and methods
Between March 2000 and April 2018, 36 patients with ES were evaluated, treated, and followed up at our hospital. The immunohistochemical examinations of all pathologic specimens were reviewed by two seasoned pathologists. All operations were performed by the same senior surgeon in our hospital. The types of all patients specimens were confirmed according to the 2013 World Health Organization STS classification. 13 Either patients or their guardians, where applicable, provided written informed consent according to the rules approved by the respective institutional review board. All data were compiled by a review of medical records, and the study was approved by the Clinical Ethics Committee.
The inclusion criteria for patients enrolled in this study were: (1) surgery, including the first surgery or the immediate second surgery post-unplanned surgery, must be performed at our hospital, (2) patients who just received radiotherapy and/or chemotherapy were excluded, (3) all patients for whom the final diagnosis, as well as the surgical margins, were determined by our pathology department, and (4) follow-up data were recorded completely. The patient’s age was determined by the time of the first surgery in our hospital. Primary and recurrent patients were defined as per whether the surgery was done for a primary tumor or recurrent tumor. The proximal subtype was first described as a distinct subtype of ES in 1997. 14 Histologically, the proximal subtype differs from the classic type by its larger epithelioid cells, more proximal distribution (mainly, but not exclusively, involving the pelvic, perineal, and genital areas), more deep-seated location, and more aggressive clinical behavior from the outset. 15
Surgery is the mainstay of any treatment approach for a localized primary or recurrent ES. If the main nerve and blood vessels were involved, amputation came to be accepted as the standard for surgical therapy; otherwise, local tumor resection or wide resection would be performed. Surgical margin assessment was performed according to the resection quality, which were classified into R0 and R1/R2. R0, referred to microscopic tumor-negative surgical margins; R1, referred to microscopic tumor-positive surgical margins; and R2, referred to macroscopic tumor-positive surgical margins.
Follow-up
Follow-up data were obtained from medical records, local tumor extent was assessed using magnetic resonance imaging (MRI). Lymph node and/or distant metastasis post-treatment was assessed using enhanced CT and/or Whole Body Positron Emission Tomography. The median follow-up period was 40.0 (range 7–178) months overall and the follow-up deadline was September 30, 2020. Local recurrence-free survival (LRFS), distant metastasis-free survival (DMFS), and overall survival (OS) were dated from the time of definitive surgery performed until the most recent follow-up at our institution for M0 patients. Patients were followed up for a minimum of 24 months unless they died earlier of disease.
Statistical analysis
The statistical softwares used in this article were SPSS 22.0 and GraphPad Prism 6, and Kaplan-Meier method and Cox regression model were used for univariate and multivariate analysis, respectively. Prognostic factors with statistical significance (p < 0.10) in the univariate analysis were included in the multivariate analysis of Cox’s proportional hazard method. P-values less than 0.05 were considered statistically significant. Survival time was calculated from the time of first surgery in our hospital to the time of death or the last documented time. Patients who died of non-tumor related factors were considered to have been censored.
Results
Clinical features
A total of 36 patients (25 males, 69.4%) were studied with a ratio of male to female being 2.27:1. The median age at diagnosis was 38 years, with a mean age of 38 years (range, 22–61 years) and the median follow-up duration was 36 months (range, 7–178 months). Herein, 18 (50.0%) patients presented with primary tumors, whereas 18 (50.0%) patients presented with recurrent tumors. 3 (8.3%) patients showed with distant metastasis and 6 (16.7%) patients showed with lymph nodes metastasis when they first visited our hospital. Table 1 shows patient characteristics according to tumor at the time of presentation.
Clinicopathological characteristics of 36 patients with epithelioid sarcoma.

All 36 patients underwent surgery in our hospital (11 amputations, and 6 regional lymphadenectomies). 31 patients of them has also received adjuvant therapy, including 17 patients who only underwent radiotherapy, 11 patients who only underwent chemotherapy, and 3 patients who underwent radiotherapy plus chemotherapy. The radiation dose was 56–66 Gy, with an average dose of 60 Gy. Chemotherapy was administered for 2–9 cycles (doxorubicin, ifosfamide, pirarubicin, vincristine, etc.) (Table 1).
Local recurrence and risk factors
Local recurrence occurred in 14 patients (38.9%) among them with a median time being 12 months (range 2–45 months). Local recurrence rates at 2 and 5 years were 32.0% and 45.1%, respectively (Figure 1(C)). Local recurrence arose in 9 of 31 patients (29.0%) treated in R0 resection quality, and in comparison, all 5 patients with R1/R2 resection quality developed local recurrence.
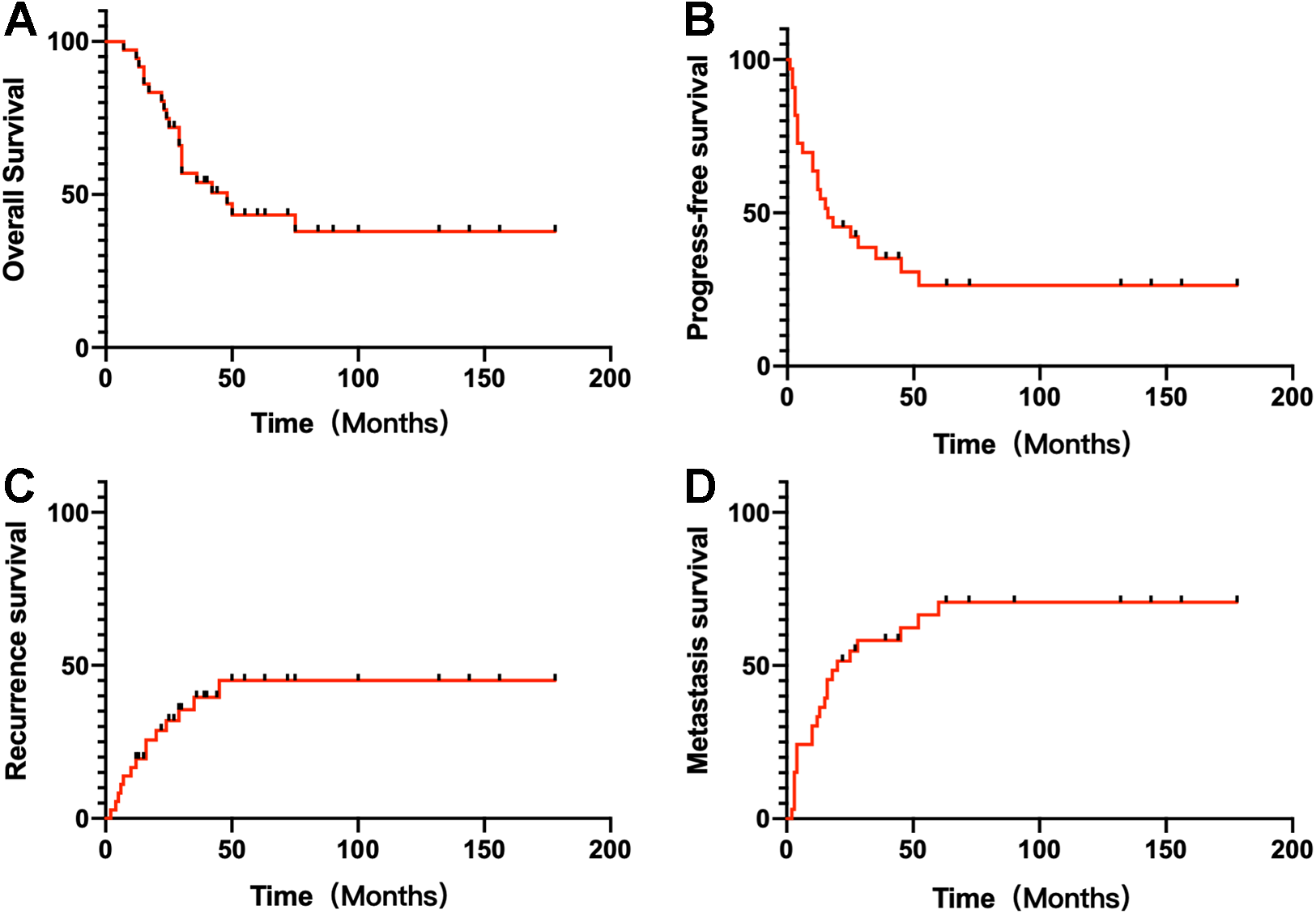
Kaplan–Meier analysis of survival in Epithelioid sarcoma patients after surgery. (A) Overall survival rates at 2 and 5 years for 36 patients were 74.8% and 43.3 %, respectively. (B) Progress-free survival rates for the 33 patients with M0 were 45.0% and 26.4%. (C) Local recurrence rates for the 36 patients were 32.0% and 45.1% at 2 and 5 years, respectively. (D) Distant metastasis rates for the 33 patients with M0 after 2 and 5 years were 51.5% and 70.8%, respectively.
To identify the possible factors impacting the postoperative local recurrence, further analysis was conducted in these 36 patients. Univariate analysis showed that tumor size (p = 0.029), surgical margin (p < 0.001) independently affected local recurrence (Table 2; Figure 2(A)). However, on multivariate analysis, only R0 surgical margin was identified as factors associated with local recurrence (p < 0.001, Table 2).
Univariate and multivariate analysis of factors influencing local recurrence-free survival in 36 patients.
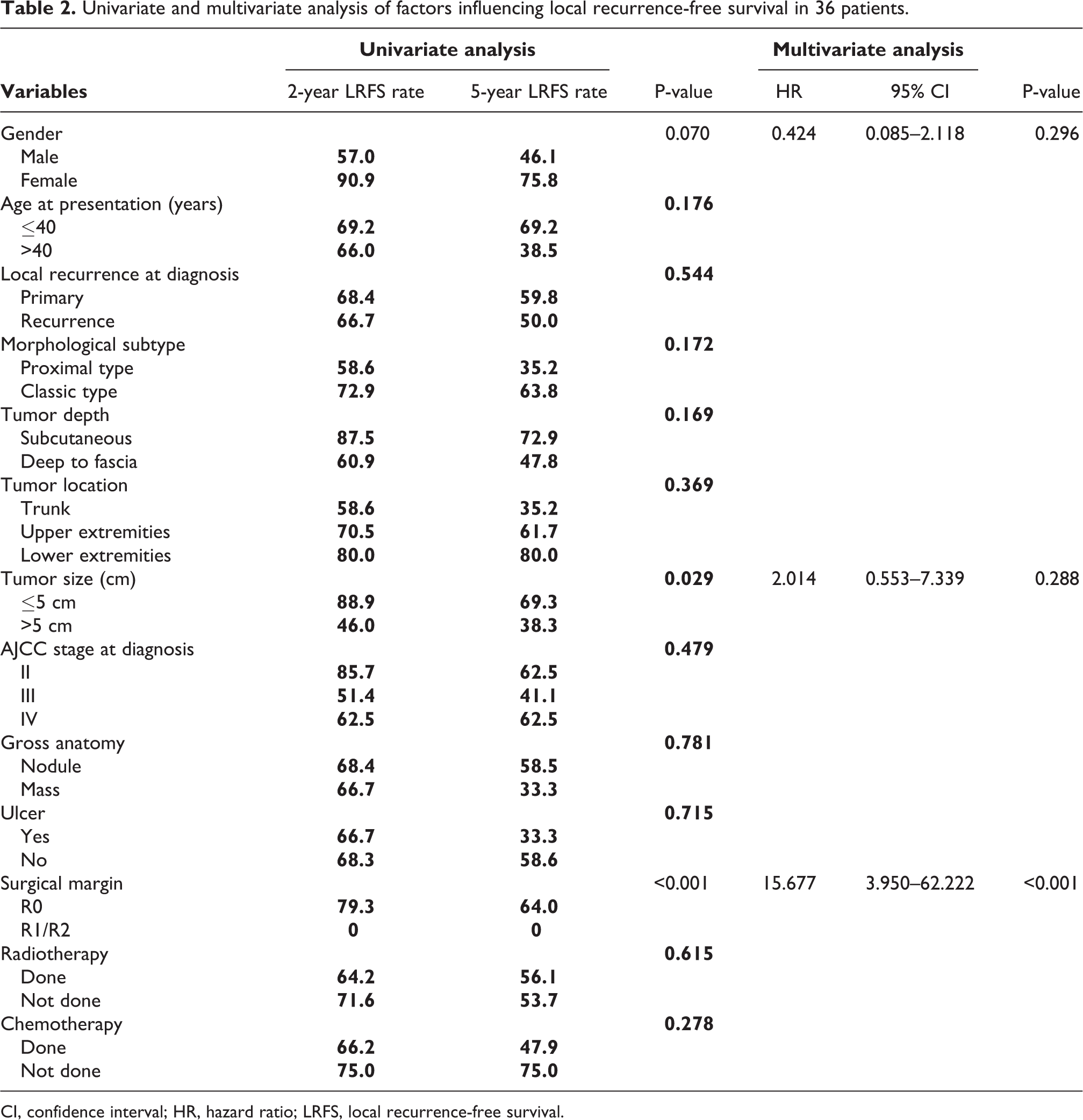
CI, confidence interval; HR, hazard ratio; LRFS, local recurrence-free survival.

Kaplan–Meier analysis of survival in Epithelioid sarcoma patients after surgery. (A) Kaplan-Meier local recurrence-free survival curve according to the surgical margin in 36 ES patients. (B) Kaplan-Meier distant metastasis-free survival curve according to surgical margin in 33 ES patients with MO. (C) Kaplan-Meier progress-free survival curve according to gender (male vs female) in 33 ES patients with MO. (D) Kaplan-Meier distant metastasis-free survival according to the tumor sizes in 33 ES patients with MO. (E) Kaplan-Meier overall survival curve according to tumor size (≤5 cm vs >5 cm) in 36 patients with epithelioid sarcoma. (F) Kaplan-Meier overall survival curve according to M stage (M0 vs >M1) in 36 patients with epithelioid sarcoma.
Distant metastasis and risk factors
Three of the 36 patients presented with metastases before resection in our hospital; 1 of these 3 patients had lymph node metastases, and 2 patients had metastases in the lungs. Of the 33 patients without distant metastases at diagnosis, 22 subsequently developed metastases at a median follow-up of 12 months (range 2–60 months). The 2 and 5 years distance metastasis rates for these 33 patients were 51.5% and 70.8%, respectively (Figure 1(D)). Univariate analysis found that gender (p = 0.019), tumor size (p = 0.011) and surgical margin (p = 0.045) were risk factors for tumor metastasis post-resection in M0 patients, but tumor size (p = 0.041), surgical margin (p = 0.006) and chemotherapy (p = 0.044) remained independent prognostic factors for DMFS (Table 3; Figure 2(B) and (D)).
Univariate and multivariate analysis of factors influencing distant metastasis in 33 patients with M0 tumors.

CI, confidence interval; DMFS, distant metastasis and risk factors; HR, hazard ratio.
Progress-free survival and risk factors
At a median follow-up of 40 months, 10 patients had survived without disease progress among the 33 patients with M0. For our series of 33 patients, the 2 and 5 year progress-free survival was 45.0% and 26.4%, respectively. For patients who presented with localized disease with R0 margins post-surgery, the 2 and 5-year survival rates were 53.6% and 31.1%, respectively. The univariate and multivariate analysis of risk factors associated with 2 and 5 year progress-free survival for patients with ES in the 33 patients is shown in Table 4. On multivariate analysis using the Cox model, surgery margin (p = 0.033) was found to be the only significant factor affecting progress-free survival.
Univariate and multivariate analysis of factors influencing PFS in 33 patients with M0 tumors.
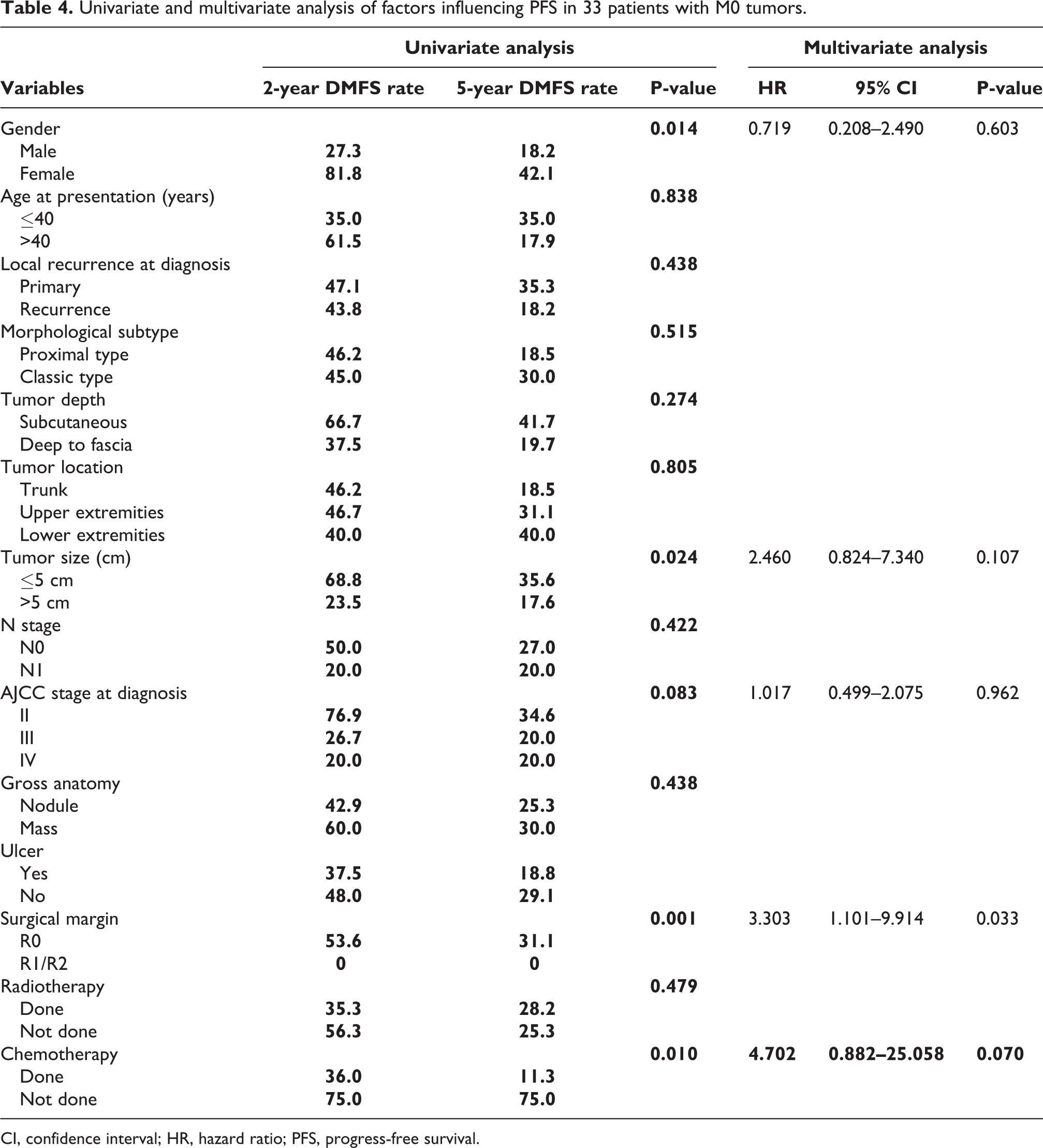
CI, confidence interval; HR, hazard ratio; PFS, progress-free survival.
Overall survival and risk factors
By the end of final follow-up, 17 patients had died due to the metastases among the 36 patients with a median interval being 29 (range 7–75) months. The OS rates at 2 and 5 years for ES were 74.8% and 43.3%, respectively (Figure 1(A)). OS was found to significantly differ between patients with metastasis at diagnosis and those with localized disease (0 vs 47.4% at 5 years, respectively, P < 0.05). Univariate analysis showed that gender (p = 0.001), tumor size (p = 0.003), M stage (p = 0.013) and local recurrence post-treatment (p = 0.036) were significant prognostic factors for OS. On multivariate analysis using the Cox model, tumor size (p = 0.008) and M stage (p = 0.010) were found to be the significant factors affecting OS (Table 5; Figures 2(E) and (F)).
Univariate and multivariate analysis of factors influencing overall survival of all 36 patients with epithelioid sarcoma.
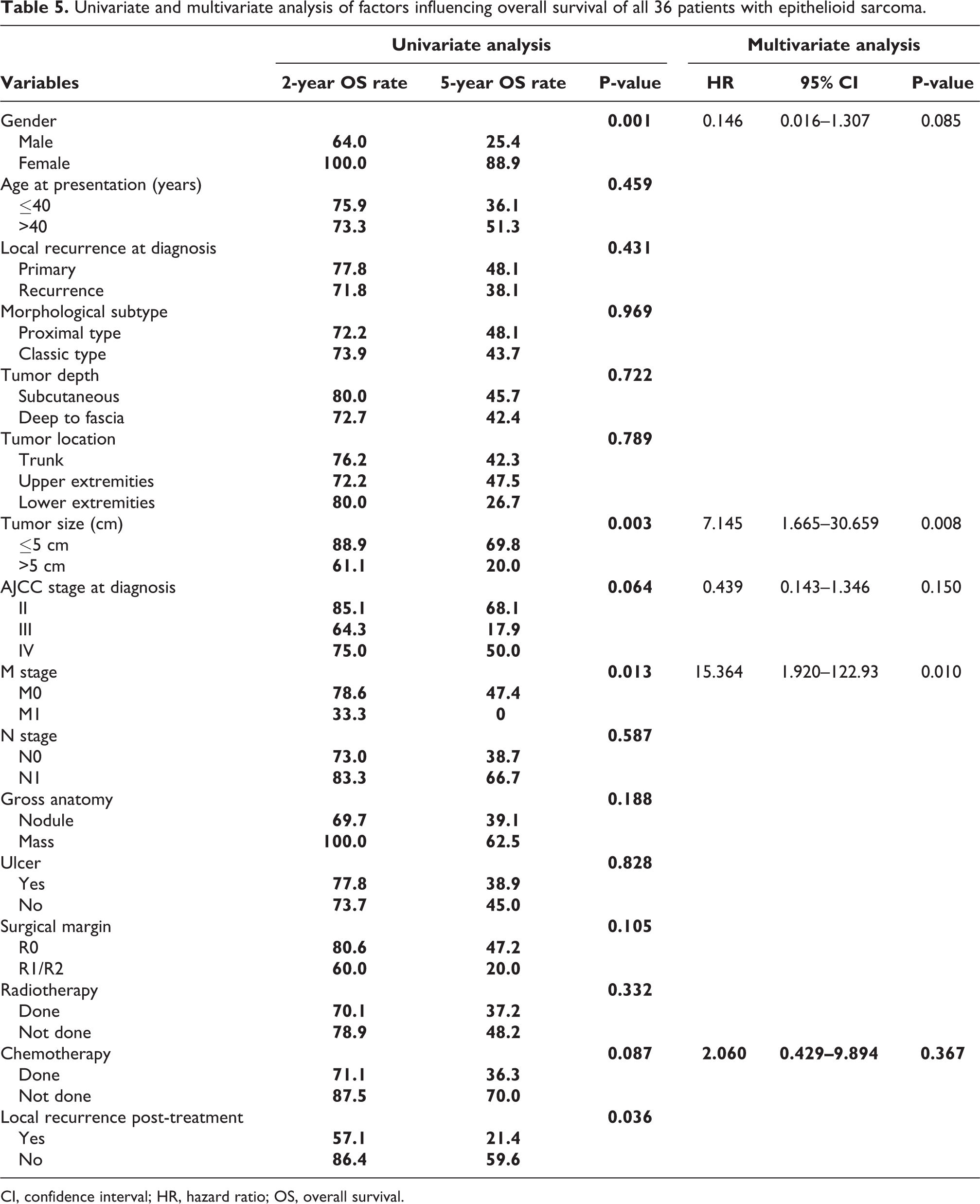
CI, confidence interval; HR, hazard ratio; OS, overall survival.
Discussion
ES is a rare mesenchymal tumor with severe consequences even in multiple treatment modes. Compared with other soft tissue sarcomas, ES is prone to early local lymph node and lung metastasis and is less responsive to radiotherapy and chemotherapy. 2 Approximately 77.7% of the lesions were located in the extremities, and the upper and lower limbs amounted for 44.4% and 33.3% of the lesions, respectively. The others were mainly located in the trunk (22.2%). ES has no evident specificity in clinical manifestation, and mainly presents as a local mass and some patients may have ulcers (about 19.4% of patients in this study developed ulcer symptoms). Most patients had undergone unplanned surgery before attending our hospital. Due to the lack of vigilance, postoperative tumor residuals are often caused, which increases the possibility of postoperative recurrence and the difficulty of secondary surgery.
In our series, the OS rates for ES at 2 and 5 years were 74.8% and 43.3%, respectively. Compared with OS rates reported 32–78%, 6 –10 the OS rate at 5 years (43.3%) was lower but remained within the previously described range. This can be explained by the early metastasis in patients in this group [16.7%, (8/36)]. We found that gender (p = 0.001), tumor size (p = 0.003), and M stage (p = 0.013) were significant prognostic factors affecting OS. Some related studies also report that tumor size was a key factor for predicting prognosis survival. 1,16 The 5-year survival rate of patients with a tumor diameter less than 5 cm (69.8%) was 3.49 times that of those with a tumor size greater than 5 cm (20.0%). The faster the tumor grows, the larger the tumor volume. The more intuitive the characteristics of malignant biological behavior, the more frequent mitosis, and the greater the risk of secondary metastasis, which indirectly affects the survival rate of patients. In our study, 3 patients with lung metastasis received palliative surgery, and both of them died within 3 years of the operation.
The principle of therapy for ES is also wide resection with negative margins (R0). Many researches have confirmed that R0 resection margins could reduce the recurrence rate. Sambri et al. 4 reported that among 47 patients presenting with primary tumors, local recurrence-free rate was 72.9% at 5 years, and 61.9% at 10 years, and a better local control achieved in patients with radical margins (P = 0.026). However, Asano et al. 10 revealed that 10 patients (30%) among the 33 ES patients with M0 tumors who underwent curative surgery showed local recurrences. Further, multivariate analysis showed that lymph node metastasis (p = 0.013) and tumor depth (p = 0.040) remained independent prognostic factors for LRFS. In our series, local recurrence occurred in 14 (38.9%)patients among the surgically treated patients, which is in agreement with previous results. In addition, significant prognostic factor with LR was surgical margins (P < 0.001). Grossly positive tumor margin is the factor responsible for local recurrence of soft tissue sarcoma. 17 Just like de Visscher et al. 1 reported that epithelioid sarcoma was still an only surgically curable disease.
A high rate of distant metastasis (40–73%) in ES was reported in previous reports. 1,6,10,16 The metastasis rates for these 33 patients were 51.5% and 70.8% after 2 and 5 years, respectively. However, the tumor size were identified as risk factors affecting the development of distant metastases (P = 0.041, Table 3) and the OS (p = 0.008, Table 5). Among the ES patients in our data, the R0 surgical margins was the independent prognostic factors for local recurrence, distance metastasis and progress-free survival in multivariate analysis. However, R0 surgical margins are not considered to be an important factor affecting overall survival. In this study, local recurrence post-treatment in our hospital did significantly affect the analysis of the prognostic factors of OS (P = 0.036) (Table 5). So decreasing the LR is critical for improving the OS. Sambri et al. 4 reported that OS of ES patients referred to their centers with primary ES and radical margins could be improved, where patients presenting with local recurrence did not have a better prognosis despite the radical surgical margins post-operation again. The following factors may explain this: ES is prone to metastasis early, and even there is not recur, it still dies due to rapid progression of metastases.
It is generally believed that chemotherapy has positive implications for tumor metastasis and provides better survival for highly malignant sarcomas. 18 –20 Multivariate analysis of DMFS showed that chemotherapy was also an independent prognostic factor (P = 0.044). But after analyzing the data, it was found that the DM rate was higher for those receiving chemotherapy than for those not receiving chemotherapy. It may be explained by the fact that patients who received chemotherapy had larger tumors and achievement of R0 resection margin was difficult in these patients with ES. This could also lead to statistical selection bias. A theory that can be inferred from this perspective is that chemotherapy could not have any beneficial role in the therapy of ES, which can be supported by some studies. 6,21 Asano et al. 10 showed that radiotherapy and chemotherapy did not significantly improve the prognosis, which is in line with the results of the current study.
With the successful impression of immune checkpoint inhibitors just like anti-programmed death-1 (PD-1)/programmed death ligand-1 (PD-L1) in melanoma, NSCLC and other solid tumors, 22 –24 much more research was performed to investigate the clinical significance of PD-1/PD-L1 axis in sarcoma. Recent study revealed that 42.7% of the sarcoma patients had positive expression of PD-L1, which varied according to histologic subtypes, but ES had a significant positive expression rate of 100%. 25 So PD-L1 blockade could be a promising therapeutic strategy in ES. Touati et al. 2 also reported that pazopanib administration achieved 1 partial response (11.1%), 4 stable disease (44.4%), and 4 progressive disease (44.4%) in advanced ES patients. Targeted treatment of the majority of soft tissue sarcomas is still in the exploration phase.
As a retrospective analysis, due to the extremely low incidence of the disease, the number of cases was still limited. Moreover, more cases should be accumulated and analyzed to reflect more actual characteristics of this disease objectively by multiinstitutional collaborative studies.
Conclusions
Characteristics of ES are high-grade malignancy, tenacious local and distant progression, and poor survival. Therefore, the current status of ES treatment is still very challenging. In this study, we found that tumor size (>5 cm) and M1 were significantly associated with a poor overall survival. But the R0 resection margin was the only statistically significant prognostic factor influencing the LRFS and DMFS. Epithelioid sarcoma is still an only surgically curable disease, and the R0 resection margin was critical for a better prognosis. Immune checkpoint inhibitors and targeted therapy could be promising therapeutic strategies in advanced ES patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: (1) CAMS Innovation Fund for Medical Sciences (CIFMS) (no. 2017-I2M-1-005); (2) Capital characterized clinical application research Fund of Beijing Municipal Science &Technology Commission of China (no. Z171100001017210); (3) Beijing Hope Run Special Fund of Cancer Foundation of China (no. LC2016L01); and (4) Special Fund for Clinical Research of Wu Jieping Medical Foundation (no. 320.6750.14298).
