Abstract
Background:
Ceftaroline is a cephalosporin that is effective against methicillin-resistant Staphylococcus aureus (MRSA) infections. The objective of this study was to determine the feasibility of using ceftaroline-loaded Polymethyl methacrylate (PMMA) as antibiotic cement against MRSA versus vancomycin-loaded PMMA in an in vitro setting.
Methods:
PMMA pellets were prepared with three separate concentrations of each of the two antibiotics tested. They were tested to determine the effect of increasing concentration of antibiotics on the biomechanical properties of PMMA and antibiotic activity by measuring the zone of inhibition and broth elution assay.
Results:
Ceftaroline PMMA at 3 wt%, three-point bending was 37.17 ± 0.51 N (p < 0.001) and axial loading was 41.95 N ± 0.51 (p < 0.001). At 5-wt% vancomycin-PMMA, three-point bending was 41.65 ± 0.79 N (p = 0.02) and axial loading was 49.49 ± 2.21 N (p = 0.01). Stiffness of ceftroline-loaded PMMA in low and medium concentration was significantly higher than the vancomycin. The zone of inhibition for ceftaroline was higher than vancomycin. Ceftaroline at 3 wt% eluted up to 6 weeks (0.3 ± 0.1 μg/ml) above the minimum inhibitory concentration (MIC) and vancomycin at 2.5 wt% eluted up to 3 weeks, same as MIC, that is, 0.5 ± 0.0 μg/ml.
Conclusions:
Ceftaroline, loaded at similar concentrations as vancomycin into PMMA, is a more potent alternative based on its more favourable bioactivity and elution properties, while having a lesser effect on the mechanical properties of the cement. The use of 3-wt% ceftaroline as antibiotic laden PMMA against MRSA is recommended. It should be noted that this was an in vitro study and to determine the clinical efficacy would need prospective, controlled and randomized studies.
Introduction
Background
Ceftaroline Fosamil-TAK-599 or PPI-0903 (Zinforo™) is a new broad-spectrum cephalosporin that targets a wide group of microorganisms such as gram-positive pathogens, including methicillin-resistant Staphylococcus aureus (MRSA), multidrug-resistant Streptococcus pneumoniae and common gram-negative organisms. 1,2 In vitro studies has shown that ceftaroline is broad spectrum with specific affinity toward MRSA and S. pneumoniae. 1,3,4 It can be administered for a shorter duration, and it is suggested that this drug may be as efficacious as the customary treatment using vancomycin or azteonam. 5 Ceftaroline binds to penicillin binding protein 2a (PBP 2a), which is an MRSA specific PBP that has a lower affinity for most other β-lactam antibacterials. As such, ceftaroline has been deemed as a specific drug to be used to combat MRSA bone and soft tissue infections.
Implant-related bone and soft tissue infections are a disaster in the field of orthopaedic surgery often requiring antibiotic-laden cement spacers for staged revision arthroplasty and infected implants (plates or intramedullary devices that have led to osteomyelitis). Cephalosporins are broad-spectrum antibiotics with proven efficacy and favourable safety profile. Thus, ceftaroline would be a more acceptable drug for the treatment of MRSA. 3,6,7 A combination of ceftaroline and PMMA as an antibiotic cement spacer for treating MRSA-related bone and soft tissue infections have not been reported. Therefore, a study to determine the prospect of using ceftaroline as an antibiotic-laden cement for local treatment of implant-related MRSA infection and compare its efficacy to the standard practice of vancomycin-laden antibiotic cement would be beneficial.
The objective of this study was to determine the feasibility of using ceftaroline-loaded PMMA as antibiotic cement against MRSA in an in vitro setting and to compare this to the addition of vancomycin to PMMA cement.
Materials and methods
Isolation and refinement of bacteria strains
Four strains of MRSA were used (ST0702, ST0408, ST1409-17 and ATCC29213) and isolated from tissue or blood from our orthopaedic ward. These strains were isolated and refined prior to streaking on blood agar for purity check before commencement. Test organism was propagated and handled in accordance with American Type Culture Collection (ATCC) recommendations for broth media and incubation specifications.
Fabrication of PMMA test samples
All samples were prepared in the same operation theatre with temperature maintained from 16°C to 18°C at atmospheric pressure.
Designated amounts of ceftaroline manufactured by Forest Laboratories, Inc. (New York, NY, USA) and vancomycin manufactured by LF Asia (Malaysia) were poured into a vacuum mixer. Subsequently, 40-g SmartSet® HV bone cement powder (DePuy Synthes, Raynham, MA, USA) was poured and stirred uniformly under vacuum. Twenty millilitres (18.88 g) of Liquid component was finally added to the mixture and uniformly blended. The resultant cement was poured into a 60-cm3 medical syringe and extruded into aluminium moulds that yielded cylinders measuring 10 × 30 mm2, 6 × 12 mm2 and pellets measuring 3 × 6 mm2, this technique provided pressurization to prevent defects in the samples. To ensure uniformity, all the pellets and cylinders prepared were weighed with a digital scale and those with a weight difference of more than 5% were discarded. Any nonuniform, broken, defective or cylinder with bubble was discarded. Eight sets of samples for each concentration were chosen, and dimensions were measured using a digital vernier caliper and labelled accordingly. Control samples were prepared in a similar fashion. The concentrations of antibiotics were quantified as wt% per packet of PMMA. A fixed concentration as per vial of antibiotic was used in order to mimic the actual operative setting where weighing the powder is not feasible. One vial of ceftaroline is 0.6 g, and 1 vial of vancomycin is 1 g. Ceftaroline 0.6 g (1.5 wt%), 1.2 g (3 wt%) and 1.8 g (4.5 wt%) were used. Vancomycin 1 g (2.5 wt%), 2 g (5 wt%) and 3 g (7.5 wt%) were used. They were further categorized into three groups for comparison (Table 1).
Showing the three major categories of the antibiotics used that were clustered into groups.

Bioassay of antibiotic activity
The bacterial suspension was swabbed on to agar plates. Antibiotic bone cement pellets (3 × 6 mm2) were seeded on to each plate. After the stipulated incubation period, that is, 18 h, the plates were examined for zone of inhibition.
Minimum inhibitory concentration analysis
Minimum inhibitory concentration (MIC) analysis was performed according to the Clinical and Laboratory Standards Institute guidelines. In brief, 0.1 ml of different dilutions of ceftaroline (0.06–512 mg/l) was aliquoted into 3 wells of a 96-well plate to have triplicates for each sample. Microbial suspension was prepared by suspending the colonies (selected from 18 h to 24 h growth) in 0.85% sodium chloride to a final concentration of 0.5 McFarland (equivalent to 108 colony forming unit/ml). Then, 0.01 ml of the suspension was inoculated into each well. The inoculated microtitre plate was incubated at 35 ± 2°C for 20 h in an ambient air incubator. Control wells (without inoculum) and ATCC 29213 were used for quality control in this study according to the guidelines. The growth of the strains in the wells was recorded. On the other hand, mass spectrometric analysis of the samples was performed on a 1260 Infinity Quaternary LC System equipped with a 6540 quadrupole time-of-flight mass spectrometer attached to a Dual Agilent Jet Stream Electrospray Ionization (Dual AJS ESI) ionization source (Agilent Technologies, Santa Clara, CA, USA). The samples were separated and analysed in the positive mode. Water with 1% formic acid (A) and acetonitrile with 0.1% formic acid (B) were used as the mobile phase. A linear gradient was set to run from 2% to 98% B over 25 min, at 0.5 ml/min with 3 µl of injections (3×) for each sample. The ESI condition was set at voltage 3.0 kV, gas temperature 300°C, drying gas 8 l/min, nebulizer 35 psig, VCap 3500 V, fragmentor 175 V and skimmer 65 V. The instrument was set to acquire over the m/z range of 100–1700 with an acquisition rate of 1 spectra/s, and the concentration of eluded ceftaroline was measured by monitoring the MS/MS transition m/z 605 → 208. The actual concentration was calculated based on a calibration curve (external standard; linearity 0.25–512 mg/ml).
Antibiotic broth elution assay
Each cement pellet was immersed in a polypropylene tube with 5-ml phosphate buffered saline (PBS; pH 7.3) and stored in a shaker incubator at 35 ± 2°C. PBS was chosen because it was cost-effective, and an experimental and clinical study done on antibiotic cement found no difference in the elusion into phosphate buffer or pooled human serum. 8 Every time a sample was collected, the pellet would be rinsed with distilled water and immersed in a new batch of PBS solution (5 ml).
Elution samples of 2 ml of PBS were collected daily for 1 week. Of the 2 ml, 1 ml was used for each high-performance liquid chromatography (HPLC) run. After 1 week, the solution was changed according to the time point, that is, every week for 7 weeks. Making it a total duration of 8 weeks. The concentrations of antibiotics were determined using HPLC. This method of HPLC analysis was based on previous published methods. 9 –11 Concisely, the elution samples were diluted with mobile phase and injected into the HPLC system. Eight samples for each concentration were tested and the resulting chromatogram was recorded.
Mechanical testing of antibiotic-loaded PMMA
Cylinders measuring 10 × 30 mm2 for three-point bending and 6 ×12 mm2 for axial loading were used. The cylinders were then loaded on to a jig designed to fit the Instron® (Norwood, MA) microtester Model 3365 Material Testing System.
Based on extensive literature review, the mechanical property of PMMA bone cement is measured using several methods. 12 –14 The forces tested were three-point bending and axial loading (compression) only, due to the limitation of resources and financial constrain. Stiffness (brittle) was calculated from the stress–strain curve. Testing to failure was interpreted as fracture of the samples to mimic real-life scenarios.
For three-point bending a preload of 1 N was applied at the beginning of each cycle to ensure that the PMMA cylinder remained in place throughout the test. A sine wave shaped load was applied, and the test was done under load control using a 2-kN load cell. The test was conducted at a rate of 0.5 Hz and was performed until the fixation failed with a sudden drop in the resisted load. Data were captured at 0.1 kHz. 13 For axial loading 2-N preload and displacement value of 0.5–5 mm/min were set. The test was done under load control using 500-N load cell. 12 Eight specimens for each concentration were tested.
Test of normality, Shapiro–Wilk test was used to determine the nature of distribution of the variables since the number of samples was less than 50. It was seen that the data were parametric; therefore, independent T-test was conducted for the significant difference between means of two groups. Analysis of variance test was used to determine significance within an individual group comparing means of more than two groups. A p value of less than 0.05 was adapted to have a statistically significant comparative result.
Results
Minimum inhibitory concentration
MIC for ceftaroline was achieved via sequential dilution test. This was performed as a quality control. The MIC for MRSA (ST0408) is 0.25 μg/ml and MRSA (ST0702, ST1409-17) is 0.19 μg/ml. An overall range for MIC is 0.19–0.25 μg/ml (Table 2).
Sequential dilution test results.a

MIC: minimum inhibitory concentration; ATCC: American Type Culture Collection.
aN = no growth; Y = growth.
Broth elution assay
Ceftaroline at 1.5 wt% eludes up to 3 weeks (0.3 ± 0.0 μg/ml) above MIC and fourth (0.2 ± 0.1 μg/ml) and fifth (0.2 ± 0.1 μg/ml) weeks just below MIC. Three wt% eluted up to sixth week (0.3 ± 0.1 μg/ml) above the MIC and 7.5 wt% eluded up to seventh week (0.3 ± 0.0 μg/ml) above MIC (Figure 1).

Broth elution assay for ceftaroline over 8 weeks for the respective concentrations with the MIC. 1.5-wt% ceftaroline eludes up to 3 weeks (0.3 ± 0.0 μg/ml) above MIC and fourth (0.2 ± 0.1 μg/ml) and fifth (0.2 ± 0.1 μg/ml) weeks just below MIC. 3 wt% eluted up to sixth week (0.3 ± 0.1 μg/ml) above the MIC and 7.5 wt% eluded up to seventh week (0.3 ± 0.0 μg/ml) above MIC. MIC: minimum inhibitory concentration.
Vancomycin-PMMA at 2.5 wt% elutes up to 3 weeks with the same concentration as MIC, that is, 0.5 ± 0.0 μg/ml, whereas both 5 wt% (0.5 ± 0.1 μg/ml) and 7.5 wt% (0.7 ± 0.1 μg/ml) of vancomycin PMMA elute at or above MIC till 5 weeks, respectively. However, 7.5 wt% has a higher concentration eluted at fifth week compared to 5 wt% (Figure 2).

Broth elution assay for vancomycin over 8 weeks for the respective concentrations with the MIC. 2.5-wt% vancomycin PMMA elutes up to 3 weeks with the same concentration as MIC, that is, 0.5 ± 0.0 μg/ml, whereas both 5 wt% (0.5 ± 0.1 μg/ml) and 7.5 wt% (0.7 ± 0.1 μg/ml) of vancomycin PMMA elute at or above MIC till 5 weeks, respectively. MIC: minimum inhibitory concentration; PMMA: Polymethyl methacrylate.
Zone of inhibition
The greater the wt% the greater the zone of inhibition. Ceftaroline-PMMA pellet at 4.5 wt% showed the largest zones of inhibition (Table 3). Ceftaroline was more potent against ST0408 and there was a significant increase in zone of inhibition from 1.5 wt% to 3 wt% (p < 0.001) and 3 wt% to 4.5 wt% (p < 0.001) for ST1410-5. For ST1410-5 strain, there was a significant increase in zone of inhibition comparing 1.5 wt% to 3 wt% (p < 0.001) and 3 wt% to 4.5 wt% (p < 0.001). Ceftaroline 4.5 wt% produced a significantly higher zone of inhibition for both the tested strains of MRSA when compared to 3 wt%.
Comparing the zone of inhibition of ceftaroline with vancomycin (values are in mm).
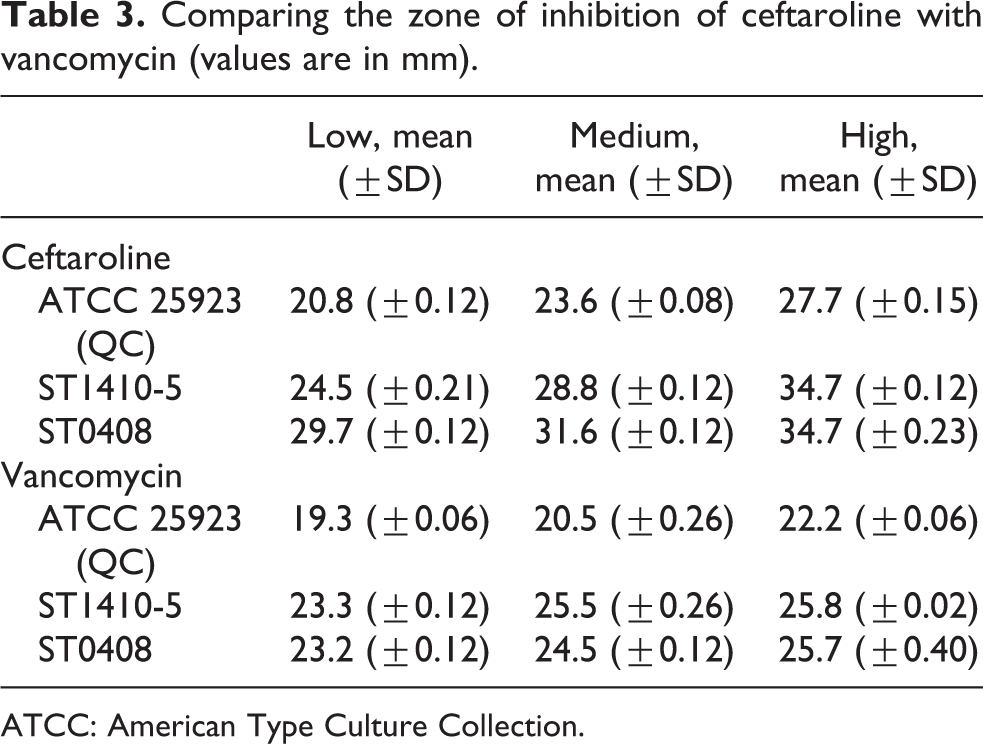
ATCC: American Type Culture Collection.
Vancomycin-PMMA pellet at 7.5 wt% showed the highest zone of inhibition (Table 3) though not significantly higher than 5 wt% (p = 0.22). However, there was a significant increase in zone of inhibition from 2.5 wt% to 5 wt% for ST1410-5 and ST0408 (p < 0.001). For ST0408, there was a significant increase in zone of inhibition from 5 wt% to 7.5 wt% (p = 0.03). It can be summed that vancomycin is slightly more potent against strain ST1410-5 of MRSA compared to ST0408.
Biomechanical testing
The control group had no antibiotics. Three concentrations were used for each respective antibiotic and categorized into low, medium, and high concentration for comparative study, as concentrations of antibiotics used were not equal (Table 1).
It can also be noticed that PMMA gets weaker with increasing concentration of antibiotic.
Load to failure of ceftaroline comparing vancomycin
Three-point bending
At 1.5-wt% (42.45 N ± 1.12, p = 0.098) and 3-wt% (42.10 N ± 1.27, p = 0.065) ceftaroline, there was no significant drop in strength. At 4.5-wt% (37.17 N ± 0.51, p < 0.001) ceftaroline, there was a significant drop (Figure 3).
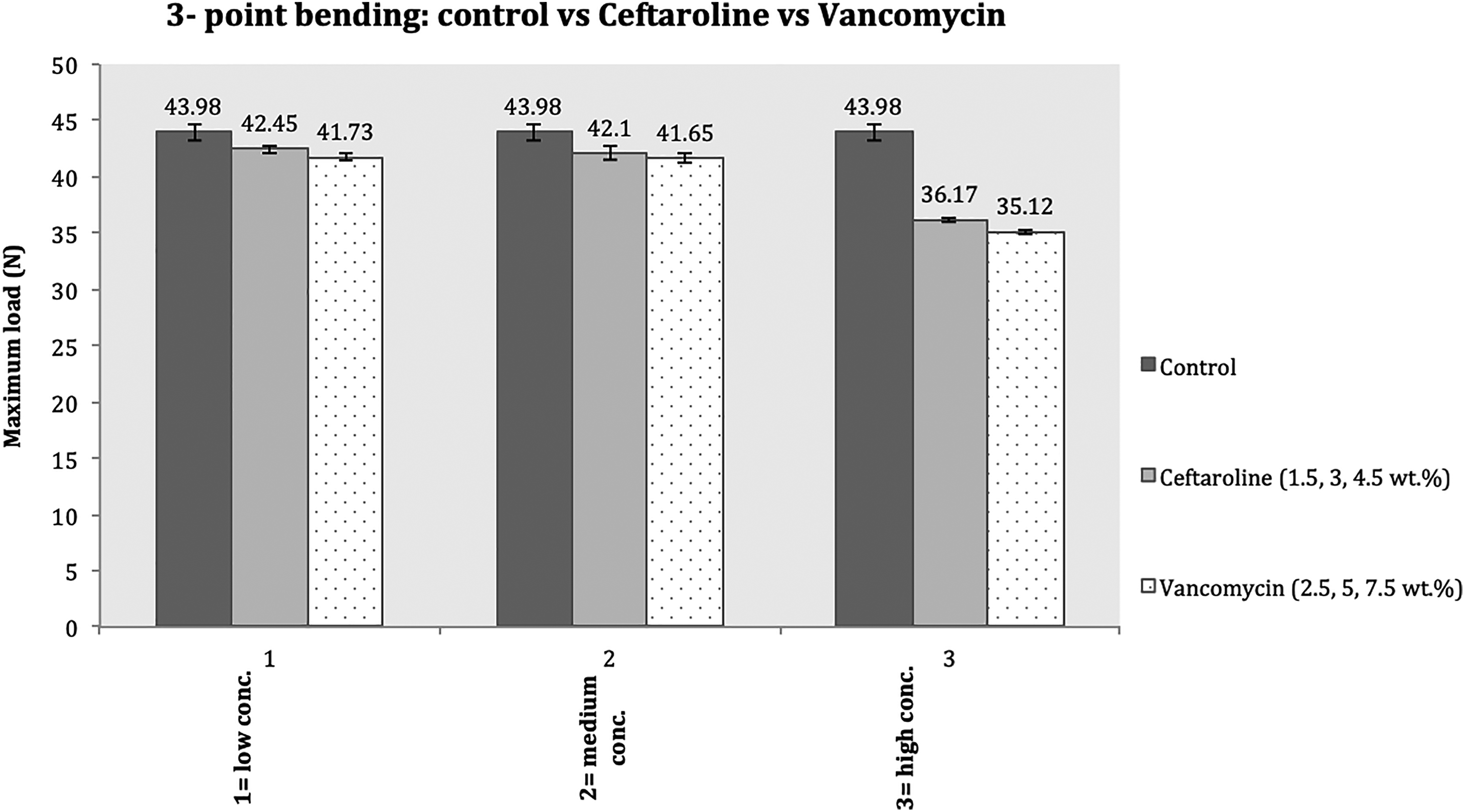
Tested three-point bending comparing control with ceftaroline and vancomycin. Comparing the means between the low (42.45 N ceftaroline vs. 41.7 N vancomycin groups; p = 0.57) and medium (42.10 N ceftaroline vs. 41.65 N vancomycin groups; p = 0.40) high-concentration groups (37.17 N ceftaroline vs. 35.12 N vancomycin groups; p = 0.002).
At 2.5-wt% (41.7 N ± 3.28, p = 0.13) vancomycin, there was no significant drop in the strength. There was significant drop in strength at 5 wt% (41.65 N ± 0.79, p = 0.02) and 7.5 wt% (35.12 N ± 0.59, p < 0.001) of vancomycin (Figure 3).
There was no significant difference between the low (42.45 N ceftaroline vs. 41.7 N vancomycin groups; p = 0.57) and medium (42.10 N ceftaroline vs. 41.65 N vancomycin groups; p = 0.40) concentration subgroups. There was a significant difference in the high-concentration group (37.17 N ceftaroline vs. 35.12 N vancomycin groups; p = 0.002; Figure 3).
Axial loading (compression)
At 1.5-wt% (51.96 N ± 0.27, p = 0.47)] and 3-wt% (51.04 N ± 1.27, p = 0.055) ceftaroline, there was no significant decrease in the strength. At 4.5-wt% (41.95 N ± 1.49, p < 0.001) ceftaroline, there was a significant drop in the strength of PMMA (Figure 4).

Tested axial load comparing control, ceftaroline and vancomycin. Comparing the means between the vancomycin and ceftaroline subgroups; low concentration (51.96 N ceftaroline vs. 51.91 N vancomycin groups; p = 0.88), medium concentration (51.04 N ceftaroline vs. 49.57 N vancomycin groups; p = 0.13) and high-concentration groups (41.95 N ceftaroline vs. 40.07 N vancomycin groups; p = 0.065).
At 2.5-wt% (51.91 N ± 0.63, p = 0.39) and 5-wt% (49.59 N ± 2.21, p = 0.01) vancomycin, there is no significant drop in strength. There was a significant drop in the strength when 5-wt% vancomycin was added, and 7.5-wt% vancomycin was added, 40.07 N ± 2.17 (p < 0.001; Figure 4).
Figure 4, comparing the means between vancomycin and ceftaroline subgroups, revealed no significant difference; low-concentration (51.96 N ceftaroline vs. 51.91 N vancomycin groups; p = 0.88), medium-concentration (51.04 N ceftaroline vs. 49.57 N vancomycin groups; p = 0.13) and high-concentration groups (41.95 N ceftaroline vs. 40.07 N vancomycin groups; p = 0.065).
Stiffness
There was a significant drop in stiffness upon addition of 1.5-wt% ceftaroline, that is, 21.81 ± 0.70 (p = 0.01), 3-wt% ceftaroline, that is, 19.73 ± 0.88 (p < 0.001) and 4.5-wt% ceftaroline, that is, 18.6 ± 1.66 (p < 0.001; Figure 5).
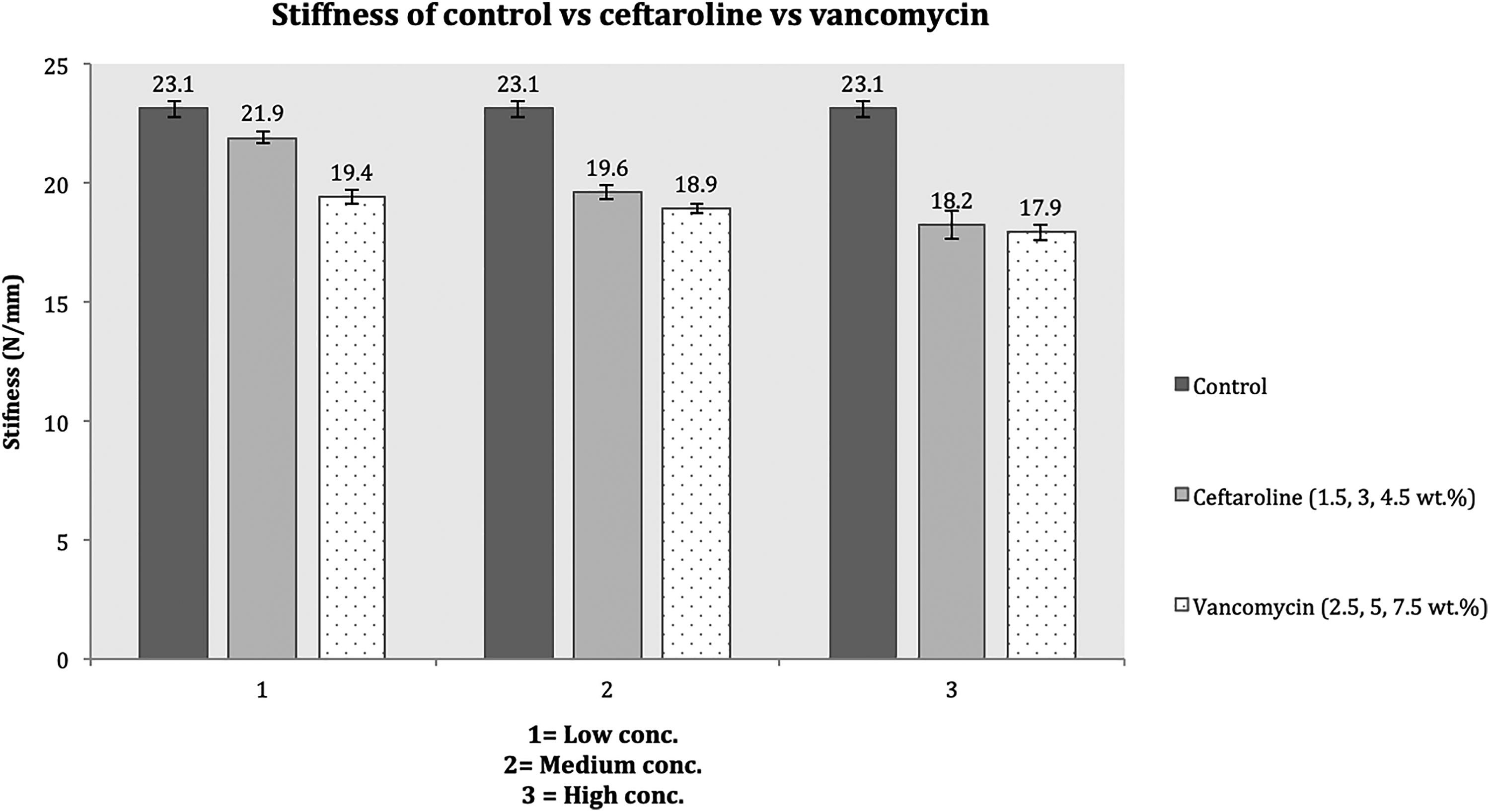
Comparing the stiffness of ceftaroline with vancomycin and control PMMA with their varying concentrations, divided into three groups. In low-concentration group, the mean of 1.5-wt% ceftaroline was significantly higher than 2.5% vancomycin, p < 0.001. In the medium-concentration group, the mean of 3-wt% ceftaroline with 5-wt% vancomycin was also significantly higher, p = 0.039. There was no significant difference between the high-concentration groups, p = 0.31. PMMA: Polymethyl methacrylate.
There was a significant drop in stiffness upon addition of 2.5-wt% vancomycin, that is, 19.4 ± 0.92 (p < 0.001), 5-wt% vancomycin, that is, 18.89 ± 0.51 (p < 0.001) and 7.5-wt% vancomycin, that is, 17.9 ± 0.87 (p < 0.001; Figure 5).
As shown in Figure 5, in the low-concentration group, the mean of 1.5-wt% ceftaroline was significantly higher than 2.5% vancomycin (p < 0.001). In the medium-concentration group, the mean of 3-wt% ceftaroline with 5-wt% vancomycin was also significantly higher (p = 0.039). There was no significant difference between the high-concentration groups (p = 0.31).
Discussion
MRSA infection involving bone and soft tissue has been a challenge to treat, often requiring the administration of prolonged intravenous antibiotics. 15 Since the early 1950s, vancomycin has been the choice for treating MRSA infection. However, a major problem has developed in most countries since the emergence of vancomycin-resistant S. aureus (VRSA). 16
Preparing PMMA with antibiotics produces a spacer with both antibacterial and mechanical properties. 17,18 Vancomycin is the antibiotic of choice that is added into PMMA bone cement to eradicate MRSA. 19 With the introduction of ceftaroline, which is a new generation of cephalosporin, we now have an alternative. Ceftaroline has superior activity against MRSA and demonstrates a better safety profile. 20 It also has better activity against isolates positive for the Panton–Valentine leukocidin (pvl) gene, heterogeneous vancomycin-intermediate S. aureus (hVISA), vancomycin-intermediate S. aureus (VISA), vancomycin-resistant S. aureus (VRSA) and quinupristin/dalfopristin-non-susceptible, tetracycline-resistant, mupirocin-resistant, linezolid-resistant, daptomycin non-susceptible and fluoroquinolone-resistant isolates. 2,4
It is worth noting that during the process of blending antibiotics into bone cement, polymerization of PMMA occurs, which results in a rise in temperatures of between 70°C and 120°C. 21 Not tested directly, but indirectly showing that ceftaroline was able to elute out and inhibit bacterial growth, that is, it was able to stand the increase in temperature and did not denature.
MIC was performed as a quality control to check both purity of MRSA isolated and antibiotic used. The MIC achieved in this study ranged from 0.19 μg/ml to 0.25 μg/ml for the three MRSA strains (ST0702, ST0408 and ST1409-17) tested, which was similar to the MIC published in other articles. Reported MIC for MRSA in vitro study (105 samples) as 0.5–2 μg/ml and 0.25–2 μg/ml for clinical isolates (1237 samples). 1 Kaushik et al. reported an MIC range of 0.25–2 μg/ml. 4 The slight variation in the values of MIC when comparing different studies could be due to the different strain of MRSA and purity of antibiotic used.
Barber et al. compared ceftaroline to vancomycin, daptomycin and teicoplanin also found ceftaroline to have a lower MIC than vancomycin. In their study, the ceftaroline MIC range was 0.125–1.5 μg/ml and vancomycin was 0.19–8 μg/ml. This study also compared parent strains and mutant strains. Ceftaroline proved to be active against both parent and mutant strains with a lower MIC compared to vancomycin. 20 In our study, MIC range for vancomycin was not determined, as the standard curve for vancomycin was already available in the HPLC database. The MIC range for vancomycin found in literature is 0.5–2 μg/ml. 22
The elution rate over time for ceftaroline and vancomycin showed that both antibiotics did elute some amount of antibiotic up to 8 weeks, but the concentration dropped over time eventually dropping below MIC. For ceftaroline, it was observed that the amount eluted drastically dropped within 1 week. The concentration of ceftaroline eluted after 24 h was extremely high and the amount eluted depended on the initial concentration added to PMMA. We also found that, adding higher concentration of antibiotic to PMMA eluted over a longer period above the MIC. On the other hand, vancomycin maintained a constant concentration up to 1–2 weeks. Similar to ceftaroline, the amount of vancomycin eluted depended on the initial concentration of vancomycin added to PMMA. Penner et al. stated that a higher concentration of antibiotic can be considered if the concentration and duration of elution needs to be longer. 23
Ceftaroline demonstrates a more rapid elution but higher diameter zone of inhibiton at a lower concentration than vancomycin. This finding could be attributed by vancomycin having different chemical property and higher molecular size. Our study did not venture into specific strains such as VRSA. But other studies as aforementioned concluded that ceftaroline is effective even against vancomycin-resistant strains. 2,4
An ideal spacer would be one that does not significantly weaken PMMA. We found that, 3-wt% ceftaroline can be added to PMMA without significantly compromising its biomechanical properties. The safest concentration of vancomycin that can be added to PMMA without compromising its biomechanical properties is 2.5 wt%.
Penner et al. predicted that 5% concentration of vancomycin negatively affected the fatigue strength of cement with a poor elution rate even with high doses. 23 The findings of that study are in line with our study.
It is recommended that antibiotics should be prescribed at low concentrations, that is, between 5 wt% and 10 wt%, the biomechanical properties are not adversely reduced. 24 This general rule cannot be applied to all antibiotics. It depends on the type of antibiotic used and the conditions in which the cement was prepared. 25 A similar conclusion can be made based on our findings, that is, ceftaroline altered the biomechanical property of PMMA differently than vancomycin.
Generally, high-dose antibiotic cement spacers are clinically safer than the systemic effects brought about by parenteral antibiotics. 26 In order to be safe when considering higher doses in high-risk patients, ceftaroline will be a safe choice as some articles have reported systemic toxicity within 24 h and local cellular (skeletal cell) toxicity with the use of vancomycin-loaded PMMA. 26 –28 Ceftaroline has a superior safety profile compared to vancomycin in renal impairment patients as evident by Monte Carlo simulations; no dosage adjustment is needed for patients with mild renal impairment. 29
The only drawback of ceftaroline in developing countries is the cost. Where 1 vial of ceftaroline (0.6 g) is roughly 10 times higher than vancomycin (0.5 g). However, for renal failure patients or other patients where systemic side effects produced by vancomycin are undesired, ceftaroline should be considered.
The limitations of this study were not all mechanical properties were sought into, for example, creep, fatigue, yield strength and tensile properties. High-viscosity PMMA was utilized due to availability and cost-effectiveness. It would be interesting to look into other different viscosity and compare. The specimens were weighed, dimensions measured, the outliers and ones with obvious defects or bubbles were discarded. A precise method to scan for defects would have been a computed tomography scan but due to lack of funds it was not performed.
Conclusions
Ceftaroline maintains its antimicrobial activities even after being incorporated into PMMA. Ceftaroline was able to elute from PMMA and inhibit the growth of MRSA in vitro. Ceftaroline PMMA has higher inhibitory properties, lower MIC against MRSA and elutes longer than vancomycin PMMA.
The use of 3 wt% for ceftaroline and 2.5 wt% for vancomycin as antibiotic laden PMMA against MRSA would be ideal if weight bearing is desired. However, higher concentrations can be added to PMMA if the duration of treatment is deemed longer than 3–6 weeks with protected weight bearing. Ceftaroline at 1.5 wt% can be used as a prophylactic grout for primary arthroplasty/tumour reconstruction surgery or even for the second stage of two-staged revision surgery because at this concentration it does not significantly affect the biomechanical property of PMMA. It should be noted that this was an in vitro study and to determine the clinical efficacy would need prospective, controlled and randomized studies.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
