Abstract
Prosthesis with antibiotic-loaded acrylic cement was designed as a temporary articulating cement spacer in a two-stage procedure before definitive reimplantation for the treatment of periprosthetic joint infections. It is designed to remain in situ for about 6–12 weeks, until evidence of infection is controlled before reimplantation of a definitive total hip replacement. This study presents a case of a patient with prosthetic articulating spacer retention for 6 years, previously performed for an infected unipolar hemiarthroplasty for which he refused second-stage reimplantation. He remains relatively asymptomatic with no evidence of infection, implant loosening, or fracture. The patient is able to walk with a frame with minimal hip pain.
Introduction
Infection after total hip arthroplasty is a dreaded complication feared by patients and surgeons alike. The two-stage approach is commonly employed in periprosthetic joint infection with success rate of over 90% in the eradication of infection. 1 The first stage involves removal of the prosthesis and debridement of infected tissues. The patient undergoes antibiotic treatment during this interim period for about 6–12 weeks, until evidence of infection control before the second-stage reimplantation of a definitive total hip replacement.
Traditionally, a Girdlestone procedure was performed in the intervening period, which made reimplantation in the second stage extremely difficult due to debris occupying the potential space and soft tissue contractures after removal of the components. The patient often could not bear weight and had limited hip function.
Prosthesis with antibiotic-loaded acrylic cement was designed as a temporary articulating cement spacer in the intervening period before reimplantation of a permanent arthroplasty. Whereas earlier methods greatly limited patient mobility, articulating spacers can allow weight bearing and functional hip movement with early mobility and improved range of movement during this time. The spacer can eliminate the joint dead space, decreasing the amount of soft tissue contracture and restoring limb length, to facilitate reimplantation and soft tissue approximation. 2
This study shows a case of a patient who underwent implantation of an antibiotic-loaded cement spacer, then refused second-stage revision as he remained relatively asymptomatic. He has had antibiotic-loaded cement spacer retained for 6 years in situ without significant complications.
Case study
A 75-year-old man had bilateral unipolar arthroplasty performed for fracture neck of femur in 2003 (Figure 1). The left hip arthroplasty was later complicated with dislocation and methillin-resistant Staphylococcus aureus (MRSA) infection. He presented with hip pain and raised inflammatory markers. Hip joint aspiration showed MRSA. Implant removal with extended trochanteric osteotomy and antibiotic-loaded cement spacer was performed in 2010, complicated with a fracture at the greater trochanter which was stabilized with cerclage wires (Figure 2).
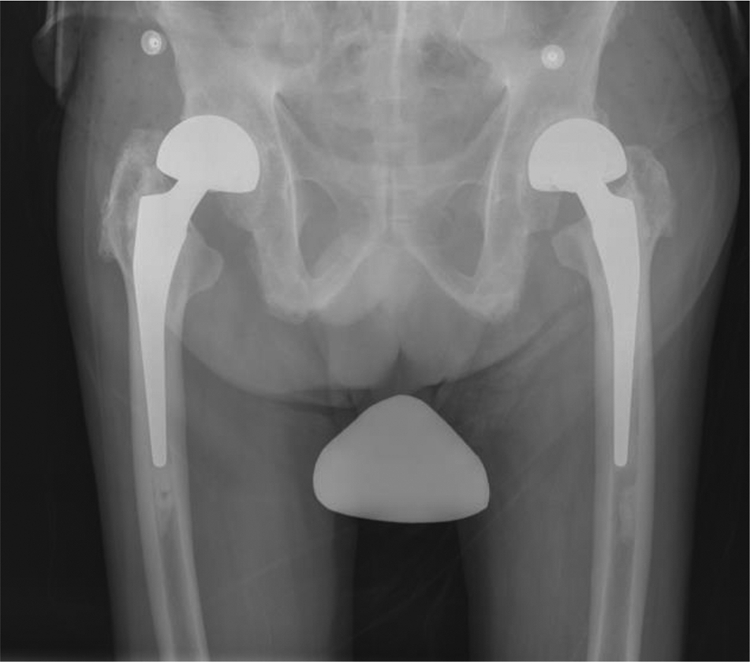
Initial X-ray of the pelvis with bilateral hip unipolar arthroplasty for bilateral fracture neck of femur.
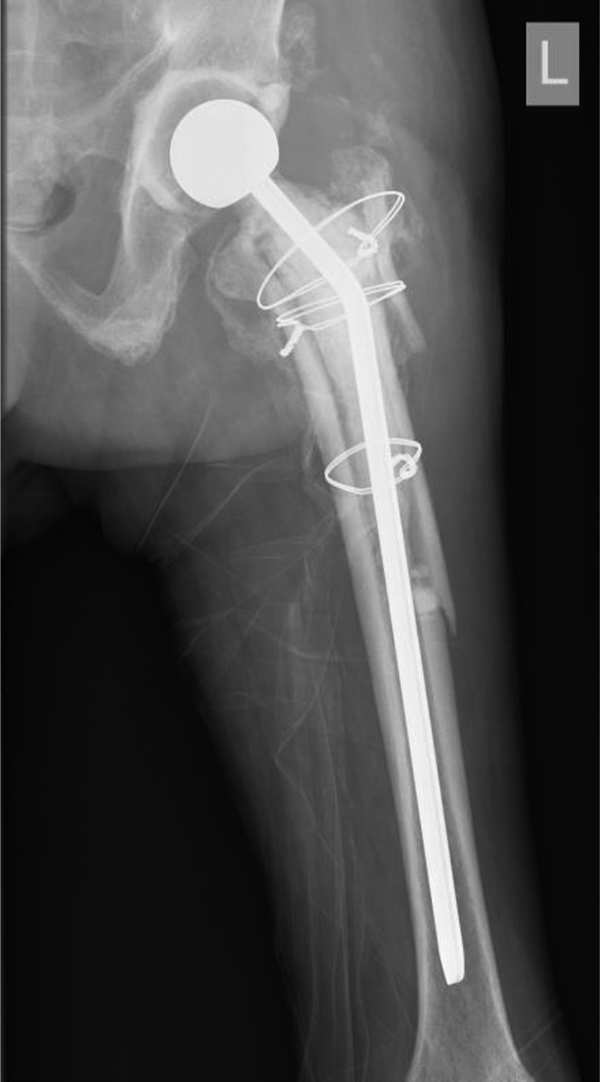
Early postoperative X-ray of an antibiotic-loaded spacer implantation after removal of unipolar implant with extended trochanteric osteotomy. Iatrogenic fracture at the greater trochanter was reduced with cerclage wires.
Repeat hip aspiration at about 12 weeks later showed persistent infection and revision was performed with repeat antibiotic-loaded cement spacer implantation and debridement. He was given a prolonged course of antibiotics for about 7 months until inflammatory markers normalized. He refused further surgery due to minimal hip pain and he was satisfied with the current hip function. He is currently 6 years with an antibiotic-loaded cement spacer in situ without long-term antibiotics. There is no clinical or laboratory evidence of recurrent infection. C-reactive protein and erythrocyte sedimentation rate levels are in normal range.
He had a history of ankylosing spondylitis, T11/T12 pseudoarthosis with laminectomy and T9-L3 posterior spinal fusion performed in 2009, complicated by wound infection, non-union, and implant loosening, for which the patient refused further spinal surgery.
He has minimal hip pain and is able to walk with a frame for about 1 hour. His ambulation is mainly limited by his spinal problem with persistent bilateral lower limb numbness. The range of movement of the left hip is well preserved. There is a left lower limb shortening of 1.5 cm. Functionally, the patient has moderate limitation in activities involving the hip. He experiences difficulty with wearing socks and shoes. Overall, the patient has no hip pain and is satisfied with his current function. Over the course of 6 years since antibiotic-loaded cement spacer implant, there is no radiological loosening, and previous fractures have healed (Figure 3).
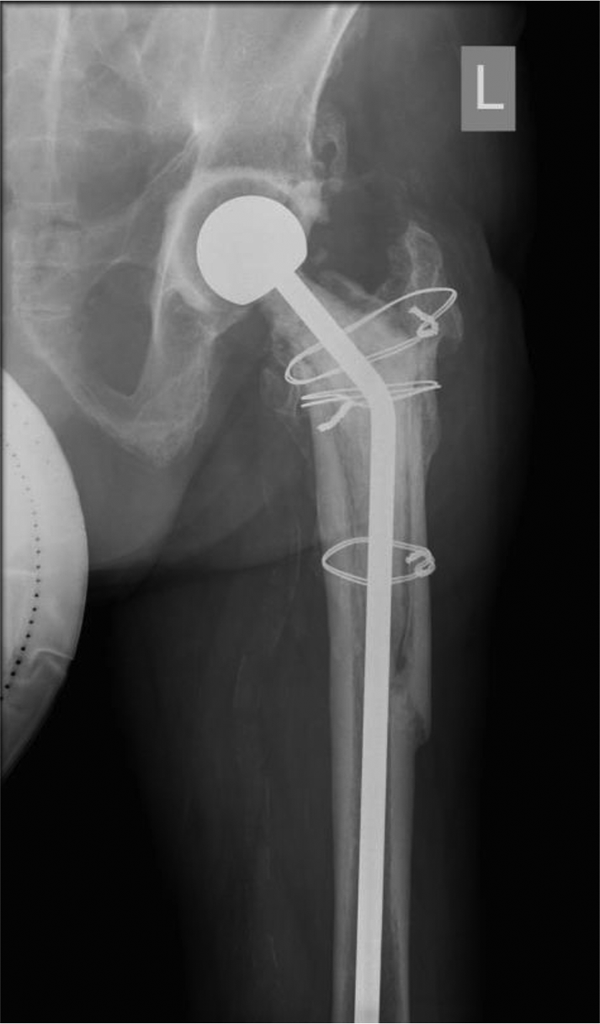
Prosthetic spacer retention at 6 years follow-up without evidence of loosening or breakage.
Discussion
Antibiotic-loaded cement spacers are designed to be a temporary prosthesis, hence its retention as a permanent/semi-permanent prosthesis has been rarely studied. However, it may not be as uncommon as previously thought. A study by Scharfenberger et al. 3 has shown that up to 65% of their study patients did not go on to complete second-stage surgery, with the majority of patients being high-risk surgical candidates, or patients were satisfied with the current function and declined second-stage surgery.
The treatment goals are to eliminate infection, improve function, and range of movement during rehabilitation. Antibiotic-loaded cement spacers have shown to be effective in eradication of infection cure rate of 90–95% 4 and has been shown to elute antibiotics for at least 118 days after initial implantation. 5
Scharfenberger et al. 6 reported lower pain scores and better function with antibiotic-loaded cement spacer compared to scores for patients with osteoarthritis awaiting primary total joint replacement, but worse compared to those 6 months post-primary total hip replacement.
Articulating spacers have shown to be promising as a permanent spacer in non-axial joints in the upper limb, but its long-term outcome in the weight bearing lower limb joints has yet to be elucidated. The obvious advantage of spacer retention is to avoid a second surgery. On the other hand, there are concerns about biomechanical complications, such as fracture, loosening, and dislocation. Theoretically, articulating cement spacers are weaker due to the increased cement porosity and a weaker metal endoskeleton when compared to a standard total hip replacement. Vacuum mixing of cement is avoided to increase porosity for maximal antibiotic elution, at the sacrifice of mechanical strength. 7 The increased ratio of antibiotic to cement may also increase rate of fracture. 8 There is also proximal bone loss in the femur producing a cantilever effect which may increase the rate of spacer fracture. This may be remedied by adequate cementation proximally and post-operative protected weight bearing.
Choi et al. 9 studied the long-term outcome of unplanned retention of prosthetic articulating temporary spacers and found that in a group of 18 patients, 15 (83.3%) maintained well-functioning articulating spacers up to 6 years without further surgical intervention. Spacers were revised in three patients (16.7%) for recurrent infection at 24 months and mechanical loosening at 74 and 50 months. The variable cementation technique may also be a confounding factor in the mechanical strength of the spacers, as most were coated with cement and inserted without pressurization, in contrast to a one-stage revision with high-grade cementing techniques.
Summary
The retention of articulating spacers may serve as an alternative as a semipermanent spacer in a selected group of patients up to 6 years. The patient in this case had a low functional demand. At 6 years spacer retention, he had satisfactory hip function and without major biomechanical complications. He will require regular follow-up to observe the long-term outcome.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
