Abstract
Purpose:
Bone cement is commonly used as a void filler for bone defects. Antibiotics can be added to bone cement to increase local drug delivery in eradicating infection. After antibiotic elution, nonbiodegradable material becomes an undesirable agent. The purpose of this study was to evaluate effects of addition of vancomycin on the compressive strength of injectable synthetic bone substitute, JectOS®. JectOS, a partially biodegradable cement that over time dissolves and is replaced by bone, could be potentially used as a biodegradable antibiotic carrier.
Methods:
Vancomycin at various concentrations was added to JectOS and polymethyl methacrylate (PMMA). Then, the cement was molded into standardized dimensions for in vitro testing. Cylindrical vancomycin–JectOS samples were subjected to compressive strength. The results obtained were compared to PMMA-vancomycin compressive strength data attained from historical controls. The zone of inhibition was carried out using vancomycin-JectOS and vancomycin-PMMA disk on methicillin-resistant strain culture agar.
Results:
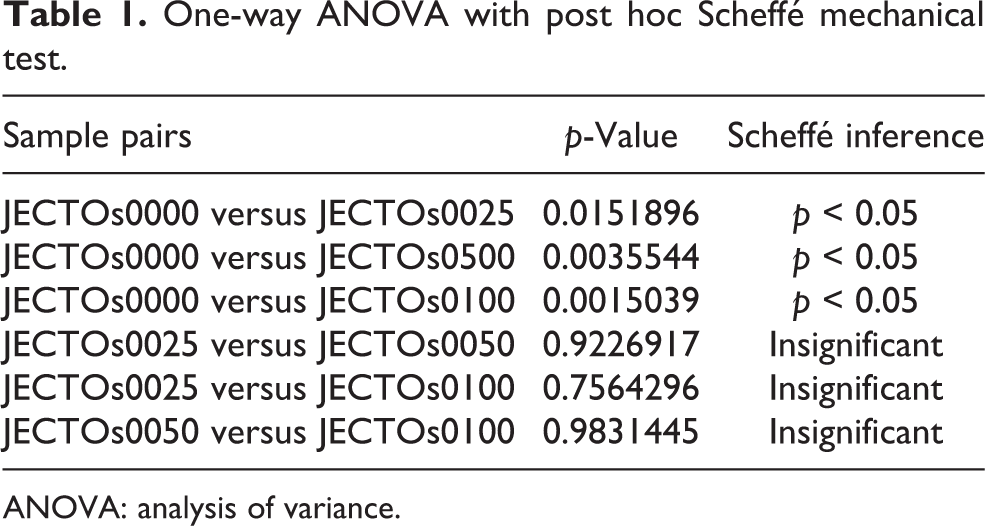
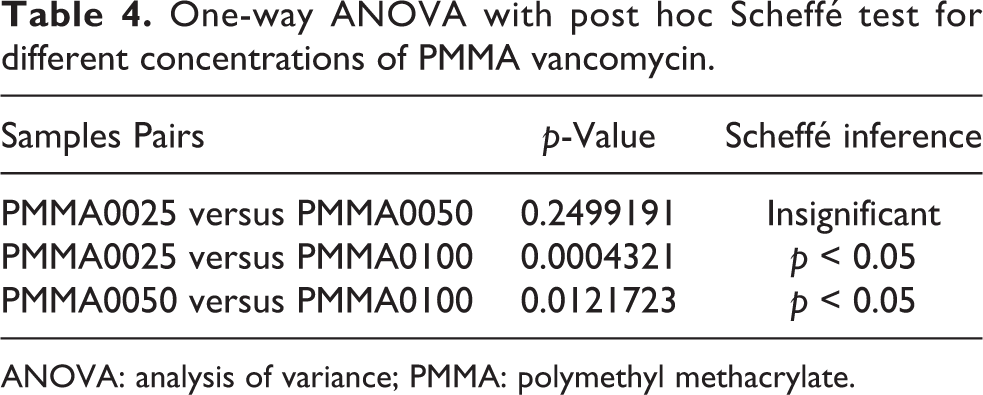
With the addition of 2.5%, 5%, and 10% vancomycin, the average compressive strengths reduced to 8.01 ± 0.95 MPa (24.6%), 7.52 ± 0.71 MPa (29.2%), and 7.23 ± 1.34 MPa (31.9%). Addition of vancomycin significantly weakened biomechanical properties of JectOS, but there was no significant difference in the compressive strength at increasing concentrations. The average diameters of zone of inhibition for JectOS-vancomycin were 24.7 ± 1.44 (2.5%) mm, 25.9 ± 0.85 mm (5%), and 26.8 ± 1.81 mm (10%), which outperformed PMMA.
Conclusion:
JectOS has poor mechanical performance but superior elution property. JectOS-vancomycin cement is suitable as a void filler delivering high local concentration of vancomycin. We recommended using it for contained bone defects that do not require mechanical strength.
Introduction
Bacterial infection in orthopaedics is often devastating and is associated with poor functional outcomes and significant morbidity. 1 Apart from operative options (debridement of devascularized tissues, restoration of soft tissue coverage, stabilization of bone, and obliteration of dead space), antibiotic is a crucial element in management of these cases. 2 The usage of long-term antibiotic treatment can result in systemic side effects and toxicity. Therefore, research on the local delivery of antibiotics to bone has gained considerable interest over the past decade.
The concept of antibiotic cement was proposed in the 1970s. Wahlig and Buchholz used gentamicin to prevent deep surgical site infection following prosthetic hip implantation. 3 Antibiotic-impregnated bone cement remains the current gold standard for local antibiotics delivery. 4 The conventional antibiotic-impregnated cement is polymethyl methacrylate (PMMA). 5 As PMMA carrier is clinically inert, it does not trigger the host immune response and is able to release antibiotic steadily. However, it is not biodegradable, and the porous surface can harbor biofilm-forming bacteria. 6 Therefore, if infection is not completely eradicated, the PMMA can act as a potential nidus for bacteria proliferation and chronic infection. A second surgery is often required to remove the residual cement after completion of antibiotic elution.
The commonest pathogen implicated in musculoskeletal infections is Staphylococcus aureus, 7 the most notorious being methicillin-resistant strains (MRSA). Not all antibiotics can be mixed in cement. It should be heat stable, water soluble to enable diffusion, active against common pathogens, and elute above minimal inhibitory concentration (MIC) and act locally without entering systemic circulation. It should also be hypoallergenic with low rate of primary resistance and available in powder form. 8,9 Based on the aforementioned characteristics, vancomycin (glycopepetide effective against gram-positive bacteria and MRSA) was chosen in this study.
Biodegradable materials have been developed to avoid secondary surgical procedures. 10 They can be implanted in vivo as the void filler but exhibit osteoconductive properties. Secondary release of antibiotic may also occur during the degradation process, which could further increase the antibiotic efficacy compared to nonbiodegradable carriers. 11
JectOS® (Kasios®, France) is an injectable synthetic bone substitute that is partially biodegradable. Compared to cortical bone and PMMA, the mechanical properties of biodegrable cements are inferior. 12 Based on the literature search, there are no mechanical studies published which demonstrate how biodegradable cement behaved after the addition of antibiotics (vancomycin). This experiment was carried out to determine the feasibility of adding vancomycin to JectOS and the optimum concentration as well as the effect on its biomechanical and microbiological properties.
Materials and methods
JectOS comprises 55% dicalcium phosphate dihydrate and 45% tricalcium phosphate (TCP). The PMMA used in this study was Simplex P®, and the antibiotic used was vancomycin (Dhacocin, Agila Specialties, Bangalore, Karnataka, India).
Test samples were prepared by hand-mixing vancomycin powder in concentrations of 2.5%, 5%, and 10% to JectOS. Then, the cement was poured into stainless steel molds to produce standardized dimensions for testing.
Preparation of test samples and mechanical testing
For compressive strength, cylindrical (6 mm diameter and 12 mm height)-shaped antibiotic cement test samples were made (Figure 1). They were subjected to International Organization for Standardization (ISO)-5833 testing protocol using the Instron® 3365 machine, Biomechanics Lab (NOCERAL) University Malaya; (Figure 2). These results were compared with the PMMA-vancomycin compressive strength data attained from previous publication, 13 and no actual mechanical testing was conducted using PMMA-vancomycin in this study due to limitation of budget.

Cement cylinder test samples of 6 × 12 mm2 for biomechanical testing.

Instron® machine model 3365.
Four test groups of six samples each were prepared: plain JectOS, 2.5%, 5%, and 10% vancomycin–JectOS. The vancomycin powder, cement powder, and liquid monomer were measured on an electronic precision scale. The precise amount of vancomycin powder was mixed with the correct amount of cement powder. Both materials were dry mixed together. The correct amount of liquid monomer was then added to the powder and hand-mixed together until a homogenous liquid cement mixture was formed. The cement paste was pressed into the aluminum mold and kept at the room temperature to harden for 24 h. For removal of the samples, the base plate of the mold was detached, and the samples were pushed out gently. The samples were then stored in a plastic container with its composition labeled clearly.
Instron® microtester Model 3365 Materials Testing System was used to study mechanical properties. The setup of the machine involved in compressive strength testing included 1-kN loads cells and double-plate jig to hold the test sample in a vertical position in between the plates. The test sample specifications and protocols were set according to the ISO-5833 protocols. According to the ISO requirement, compressive strength test is the only one important gold standard test for cement mechanical strength test. Other tests such as bending, shearing, tensile, and torsional strength tests are not mandatory. Other paper published also adapted compressive strength test only. 14 Handling of JectOS was difficult as it is very brittle and the addition of antibiotic made it worse; therefore, only one biomechanical test was chosen according to the ISO gold standard. This experiment was limited by financial constrains too. The test samples were designed in the shape of a cylinder of 6 mm diameter and 12 mm height.
After unmolding, all samples were checked with calipers to ensure the specified dimension. Those samples with visible cracks, holes, or flaws were discarded. The test samples were loaded in between the two plates in the upright position. A constantly increasing load with a constant crosshead speed of 20 mm min−1 (strain rate = 2.0 × 10−3 s−1) was applied; the load versus deformation was recorded. The average compressive strength was calculated for at least six samples. The end point of the test was load to failure or upper yield point, whichever occurs first, as required by the standard.
The relevant formula for calculating the mechanical strength was as follows:
Antimicrobial properties testing
The Kirby-Bauer zone of inhibition test was carried out. The samples were prepared as done under the mechanical testing; the dimensions of the disk were 6 mm in diameter and 3 mm in thickness (Figure 3). Six groups of samples were prepared: three groups of vancomycin-JectOS disk (2.5%, 5%, and 10%) and three groups of vancomycin-PMMA disk (2.5%, 5%, and 10%).

Cement disk test samples 6 × 3 mm2 for antimicrobial testing.
Bacteria strain selection and inoculum broth preparation
Three MRSA strains (ST 1409-3, ST 1410-5, and ST 11105-12) were used in this study and were obtained from the infected tissues and bones from the orthopedic patients found in our hospital. All the strains were sensitive to vancomycin.
Inoculation and antibiotic disk placement
MRSA was swabbed uniformly over the entire surface of blood culture agar. Antibiotic disks were placed manually using sterile forceps on the surface of the agar with adequate spaces in between the disk to allow the zone of inhibition reading. The plates were then incubated at 35°C for 24 h with the pH controlled between 7.2 and 7.4. Each group of antibiotics disks was tested against one strain of MRSA for at least twice.
The measurement of the size of zone of inhibition was carried out using Scan®500. The diameter of the zone of inhibition was measured from margin to margin of the clear zone surrounding the antibiotic disk. The accuracy of the machine was 0.2 mm.
Results
Biomechanical testing
The one-way analysis of variance (ANOVA) was the statistical method used to compare and analyze the differences among the groups of JectOS samples. The p-value corresponding to the F-statistic of the one-way ANOVA was lower than 0.05, suggesting that the one or more groups were significantly different. The post hoc Scheffé test showed that significant differences (p < 0.005) were found comparing plain JectOS with all other groups. The plain JectOS certainly had higher compressive strength. The addition of vancomycin of any concentration above 2.5% significantly reduced its compressive strength (Table 1). The results of the biomechanical testing are shown in Table 2 and Figure 4.
One-way ANOVA with post hoc Scheffé mechanical test.
ANOVA: analysis of variance.
Compressive strength of JectOS® with various concentrations of vancomycin.

Note. *JECTOs0000–plain JectOS®; JECTOs0025–JectOS® with 2.5% vancomycin; JECTOs0050–JectOS® with 5% vancomycin; JECTOs0100–JectOS® with 10% vancomycin; SD: standard deviation.

Average and standard deviation of the compressive strength of JectOS® samples.
Microbiological testing
The ANOVA was used to compare the differences within the groups of JectOS and PMMA samples. For comparison between JectOS and PMMA samples of the same vancomycin concentration, independent t-test was performed. The p-value corresponding to the F-statistic of the one-way ANOVA was higher than 0.05, suggesting that the sample groups were not significantly different from each other. Post hoc Scheffé test did not show any significant difference among the sample groups. There was no statistical difference in the antimicrobial effect of vancomycin added to JectOS in the concentration ranging from 2.5% to 10% (Table 3). There was no statistical difference in the antimicrobial effect of 2.5% vancomycin added to PMMA (Table 4). The results of both vancomycin-laden JectOS and PMMA are shown in Table 5 and Figures 5 and 6.
One-way ANOVA with post hoc Scheffé test for different concentrations of JectOS®-vancomycin.

ANOVA: analysis of variance.
One-way ANOVA with post hoc Scheffé test for different concentrations of PMMA vancomycin.
ANOVA: analysis of variance; PMMA: polymethyl methacrylate.
Diameter of the zone of inhibition of JectOS® and PMMA cements with various concentrations of vancomycin.

Note. *JECTOs0025–JectOS® with 2.5% vancomycin; JECTOs0050–JectOS® with 5% vancomycin; JECTOs0100–JectOS® with 10% vancomycin; PMMA0025–PMMA cement with 2.5% vancomycin; PMMA0050–PMMA cement with 5% vancomycin; PMMA0100–PMMA cement with 10% vancomycin.
PMMA: polymethyl methacrylate; SD: standard deviation.
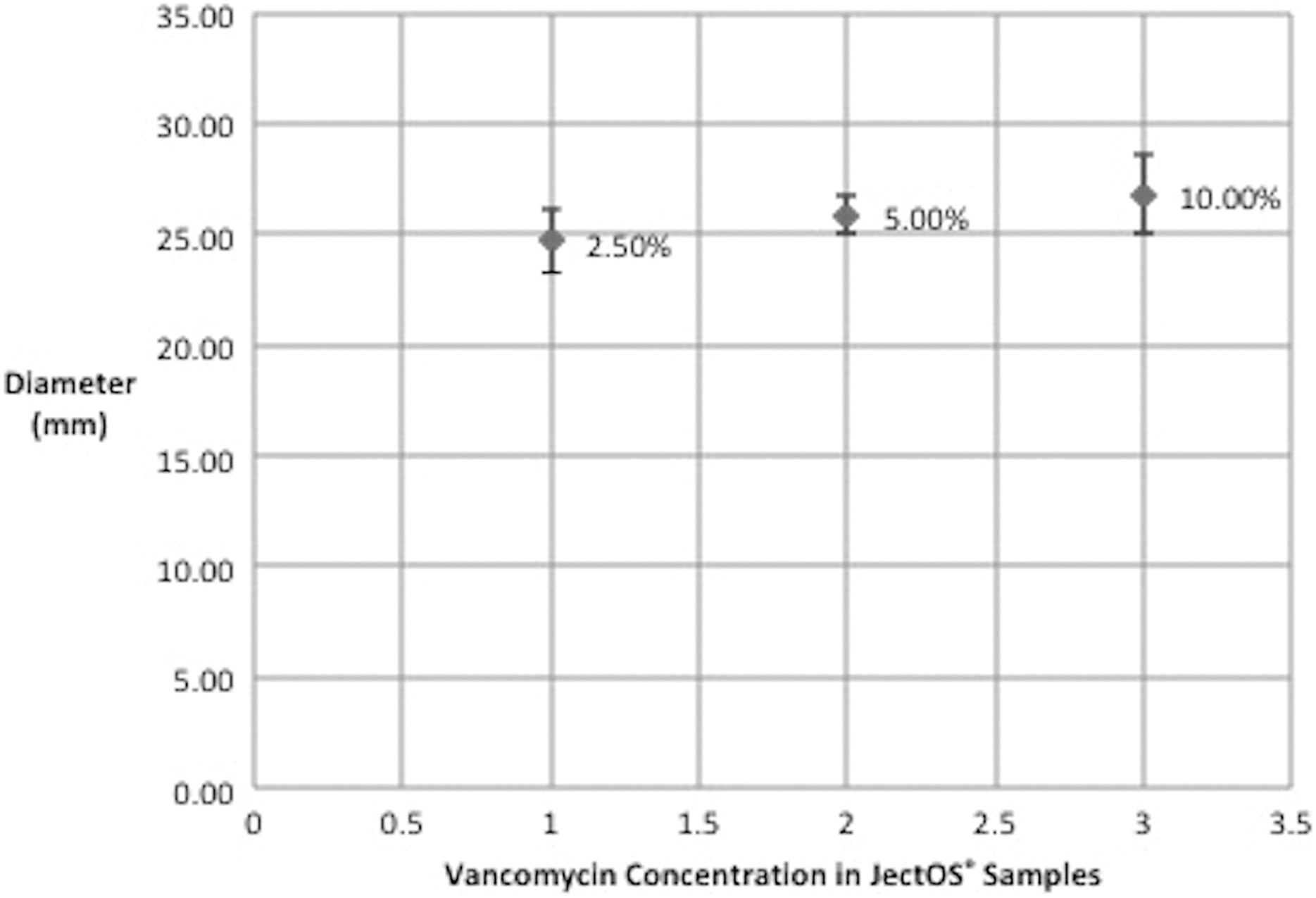
Average and standard deviation of the diameter of the zone of inhibition of JectOS® samples.
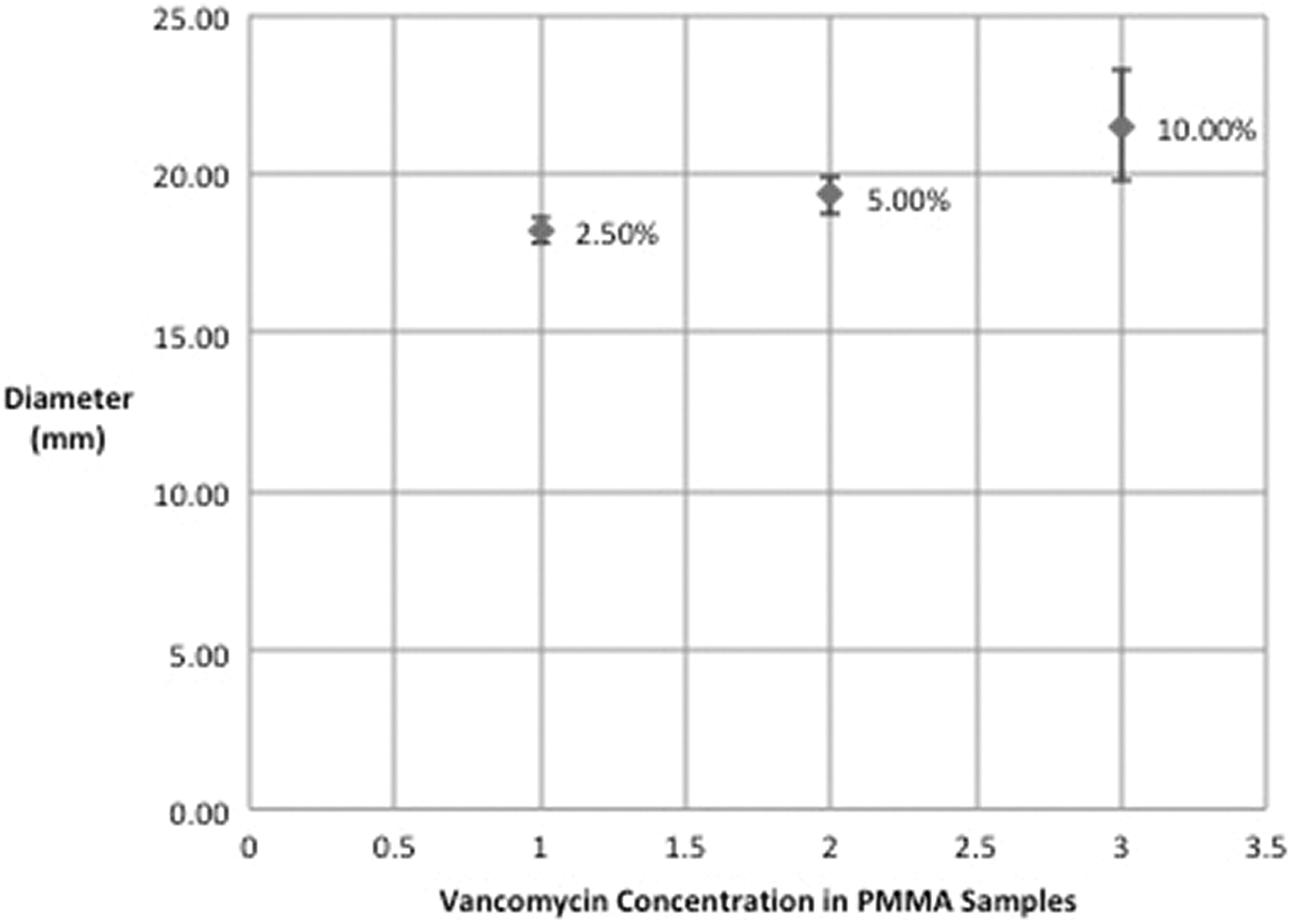
Average and standard deviation of the diameter of the zone of inhibition of PMMA samples. PMMA: polymethyl methacrylate.
Discussion
Lilikakis and Sutcliffe studied the addition of vancomycin to Palamed G (bone cement) and found that the average compressive strength of plain PMMA (PMMA0000) was 91 MPa. With the addition of 2.5% (PMMA0025), 5% (PMMA0050), and 10% (PMMA0100) vancomycin, the average compression strengths were 79.8, 82.3, and 74.6 MPa, respectively, with a reduction of 12.4%, 9.6%, and 18.1%. 13 It is advised that the addition of up to approximately 10% of antibiotic is safe. 15 However, the highest safe combination in the study tested was 5%. They did not test the 10% ratio of antibiotic–cement and made their recommendation based on extrapolating the graph. Another group of researchers found that a concentration of up to 7.5% vancomycin to plain cements had no significant effect on the compression strength. 16,17
Biodegradable cement is generally brittle in nature, and its compressive strength ranges from 1 to 40 MPa, depending on the composition of the cement and the setting condition. 18 In our study, the average compressive strength of plain JectOS (partially biodegradable) was 10.62 MPa, which is about one-ninth that of PMMA strength (91.09 MPa). With the addition of vancomycin 2.5% (TCP0025), 5% (TCP0050), and 10% (TCP0100), the average compression strengths were 8.01, 7.52, and 7.23 MPa, respectively, with a reduction of 24.6%, 29.2%, and 31%. The low compressive strength of JectOS was largely due to higher porosity and low-density microstructure. The porosity was in turn largely dependent on the setting process of the cement. Many processing parameters, such as cement particle size, composition of cement reactants, and additives, could be adjusted to control the setting process and influence its mechanical performance. 18 The statistical analysis showed that the addition of vancomycin significantly weakened JectOS. However, there was no significant difference in the mechanical strength at increasing concentrations of vancomycin. Thus, a concentration of as high as 10% vancomycin did not further weaken the cement compared with vancomycin at 2.5%.
The advantage of using JectOS is the ability to elute antibiotic more efficiently due to its porosity. Apart from the initial release of antibiotics similar to other materials, it has a characteristic secondary release from the degradation of cement. 11
Biodegradable cements are a good platform to deliver local antibiotics, but at the expense of losing mechanical strength. The Kirby-Bauer test in our study clearly demonstrated that the zone of inhibition of JectOS was significantly larger than that of PMMA. Based on the results, it could be concluded that JectOS was able to diffuse antibiotic more readily even at concentration as low as 2.5%. One major concern was regarding the duration of sustained release of vancomycin above MIC. Further studies would be required to study the elution characteristics of JectOS. Another interesting aspect to note is that TCP-based cement set at body temperature, which opens the possibility of using both heat stable or heat sensitive antibiotics.
Conclusion
In general, JectOS has a poor to moderate mechanical performance, compared with its polymeric counterparts. The intrinsic weakness is due to its highly porous morphology. In view of the mechanical strength that is comparable to cancellous bone, it can only be used as a bone filler, in non–weight-bearing bones. It is not mechanically suitable for immediate structural support. The mixing of JectOS with vancomycin can be carried out with a simple hand-mixing technique under normal operating theatre condition. Concentration ranging from 2.5% to 5% of vancomycin is effective without changing its mechanical performance significantly, but 10% vancomycin may result in higher viscosity, lower injectability, and shorter handling time. In short, it is feasible to use vancomycin-JectOS as a partially biodegradable antibiotic cement.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

