Abstract
Background:
Bilateral total knee arthroplasty is associated with significant blood loss. Postoperative anaemia retards patients recovery, and blood transfusion increases the risk for major complications including infection. While strategies like hypotensive anaesthesia, tranexamic acid, bone wax and so on are useful to conserve blood, navigation has also shown to decrease blood loss and transfusion in unilateral knee arthroplasty. The aim of this study was to compare the blood loss and transfusion in sequential bilateral knee arthroplasty performed with or without navigation.
Materials and methods:
A retrospective case control cohort study of sequential bilateral knee arthroplasties was performed between 2 groups of 77 patients each, group N operated with navigation and group M operated without navigation. Both groups were matched for age, sex and body mass index. All patients were operated with similar surgical technique. The two cohorts were compared for blood loss calculated by three reliable methods.
Results:
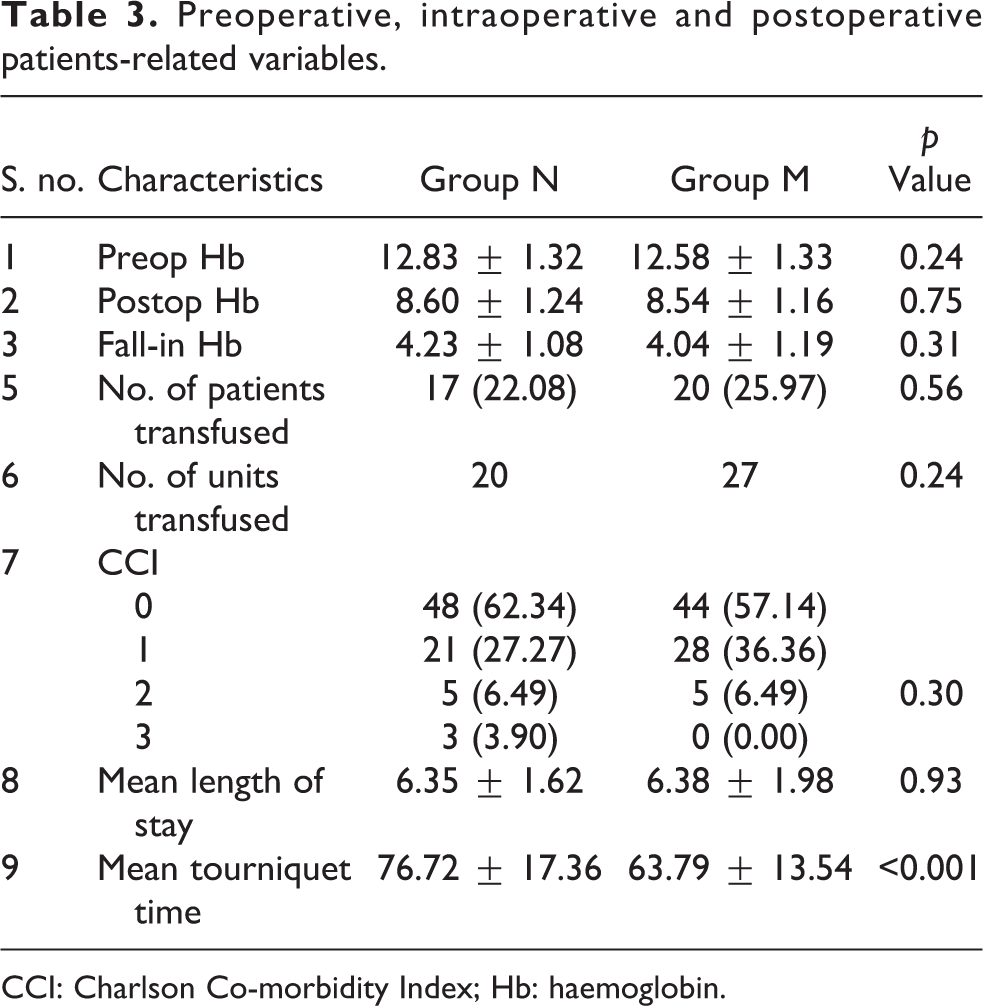
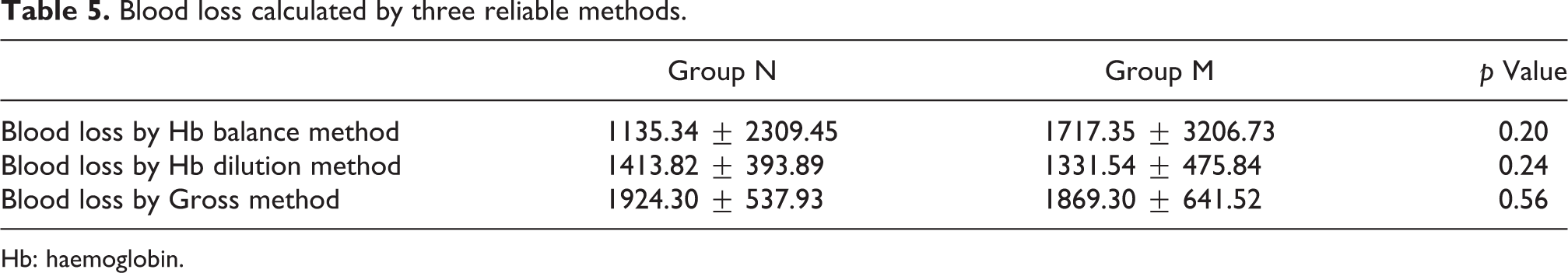
The preoperative haemoglobin (Hb) in group N was 12.83 ± 1.32 and group M was 12.58 ± 1.33 (p = 0.24). The postoperative Hb taken day 4 before any blood transfusions was 8.60 ± 1.24 in group N and 8.54 ± 1.16 in group M (p = 0.75). The average fall in Hb was 4.23 ± 1.08 in group N and 4.04 ± 1.19 in group M (p = 0.31). There was no difference in blood loss between group N and M in all three methods; Gross equation (p = 0.56), Hb dilution (p = 0.24) and Hb balance (p = 0.20). Need for blood transfusion was similar in group N and M (p = 0.56).
Conclusion:
Blood loss and transfusion are equivalent in sequential bilateral total knee arthroplasty perform with or without navigation.
Introduction
Total knee arthroplasty (TKA) is associated with significant blood loss and reducing it is an important goal of this procedure. While the average blood loss in unilateral knee arthroplasty is 800–900 ml, the blood loss in bilateral knee arthroplasty can be in the range of 1800–1900 ml, resulting in loss of one-third of patient’s blood volume. 1,2 Blood loss can lead to postoperative anaemia which is a risk factor for major systemic complications, and the resulting blood transfusion can lead to transfusion reactions, periprosthetic infections and increased hospital stay. 3,4 There have been major advances in reducing blood loss in knee arthroplasty in the last decade which include hypotensive epidural anaesthesia, 5 use of tranexamic acid, 2,6 wound sealants, 7 keeping hip and knee flexed, 8 no drain usage 9 and use of bone wax 10 on bleeding surfaces. Computer navigation has shown to reduce blood loss by eliminating the need for intramedullary hole in the femur. While some studies show the advantages of using computer navigation for reducing blood loss, 11 –13 others have countered that claim. 14 –16 If computer navigation was to decrease blood loss in knee arthroplasty, the advantage could be twofold for cases undergoing sequential bilateral total knee arthroplasty (SBTKA). Most published studies have analysed the effect of navigation in blood loss for unilateral knee arthroplasty but there is paucity of literature for its effect in sequential bilateral knee arthroplasty. The aim of this retrospective case control cohort study was to compare the blood loss and transfusion between sequential bilateral knee arthroplasty performed with or without navigation. We test the hypothesis that navigation will reduce the blood loss and ensuing blood transfusion in SBTKA using three reliable methods of blood loss estimation.
Material and methods
Institutional review board approval was obtained for this retrospective case-controlled study that prospectively collected data from April 2015 and April 2017. We have an ongoing database since 2006 in which we meticulously record all patients’ variables, blood loss, transfusions, tourniquet time and outcome measures. Our inclusion criteria for SBTKA were American Society of Anesthesiologists (ASA) grade 1 or 2 patients of less than 70 years who presented with painful deformity in both knees and received fitness from institution’s empanelled cardiologist, anaesthetist and pulmonologist. We included patients with less than 20° deformity in any plane and more than 90° preoperative arc of movement. Exclusion criteria included age more than 70 years, ASA grade 3 or 4, history of cardiac procedure or poorly controlled diabetes. Patients with severe deformity more than 20° and arc of movement less than 90° were excluded. Patients on anticoagulants, post-traumatic arthritis, inflammatory arthritis, history of deep vein thrombosis, clotting disorders, bleeding diathesis, infection, morbid obesity, revision arthroplasty and unilateral knee arthroplasty were also excluded. All patients with compromised distal vascularity in whom tourniquet couldn’t be applied were also excluded.
We excluded patients on clopidegril or warfarin which have effect on the clotting cascade but included patients on aspirin or NSAID’s that were stopped 48 h prior to surgery. Most patients who are on clopidegrol and warfarin had an angioplasty, cardiac bypass or a stroke and all of these conditions were also among our exclusion criteria for SBTKA. Ten (13%) patients in group M and 12 (15%) patients in group N were on 75 mg of aspirin preoperatively.
Based on the power calculation, we needed 70 patients in each group and hence identified two groups of bilateral patients, 77 operated with computer assisted surgery (CAS) (group N) and a matched group of patients operated without navigation (group M). The matching indices were age, sex and body mass index (BMI). All patients were operated by first 2 senior authors who have been using computer navigation for last 5 years. All patients were operated under spinal epidural anaesthesia with tourniquet applied till after cementation and tourniquet pressure maintained at 225 mm of Hg in all cases to provide blood less field. Meticulous haemostasis was achieved after release of the tourniquet. One gram of intravenous tranexamic acid was given 30 min prior to incision and a second dose repeated 4 h after the completion of second surgery. No femoral entry drill hole was made in the navigation group, but four 3.5 mm unicortical drill holes were made for navigation pins, 2 each for femur and tibia. Small conical bone plugs taken from medial sclerotic part of tibial cut were punched in these four pin holes at the completion of surgery, to reduce stress risers and bleeding (Figures 1 and 2). Bone cement was applied on the box area of the femoral cut and on the femoral bleeding surface of the medial exposed area of the femoral condyle. In the non-navigated group, a drill hole was made in the femur, for intramedullary femur instrumentation and plugged by bone from chamfer cuts to completely seal the bleeding from that hole. A thin layer of cement was further applied over the raw area of box at the time of cementing. All the knees in both groups were posterior stabilized but of three different makes (Smith & Nephew, Zimmer Biomet and Maxx). Patella was resurfaced in all patients. A perioperative anaesthetic cocktail was injected at various points in capsule, tendon, periosteum and subcutaneous tissue. Layer wise closure was done and a compression dressing was applied postoperatively in all knees. The second surgery was started after complete closure of the first knee with fresh instrument set and the team changing into fresh gowns. Drains of both knees and compression dressing were removed after 24 h. Application of ice and mobilization was encouraged by the rehabilitation team. One hundred and fifty milligrams of aspirin were given to all patients for 6 weeks starting from the night of surgery. 17 Venous stockings, active mobilisation and ankle toe pumps were encouraged to reduce venous thrombosis. We evaluated haemoglobin (Hb) and haematocrit on the forth postoperative day 18,19 and transfused blood if the Hb was less than 8 g/dl. 20 None of the patients in both groups were readmitted for anaemia or transfusion hence the transfusion done on last day was the last transfusion of all patients who received packed red blood cell in both groups. The Hb and haematocrit done before operation and a day before last were used to calculate the blood loss by all three methods. The numbers of packed cells transfused were also used in calculating total blood loss by Hb balance method.

Conical bone fragment for pin hole to prevent stress riser.
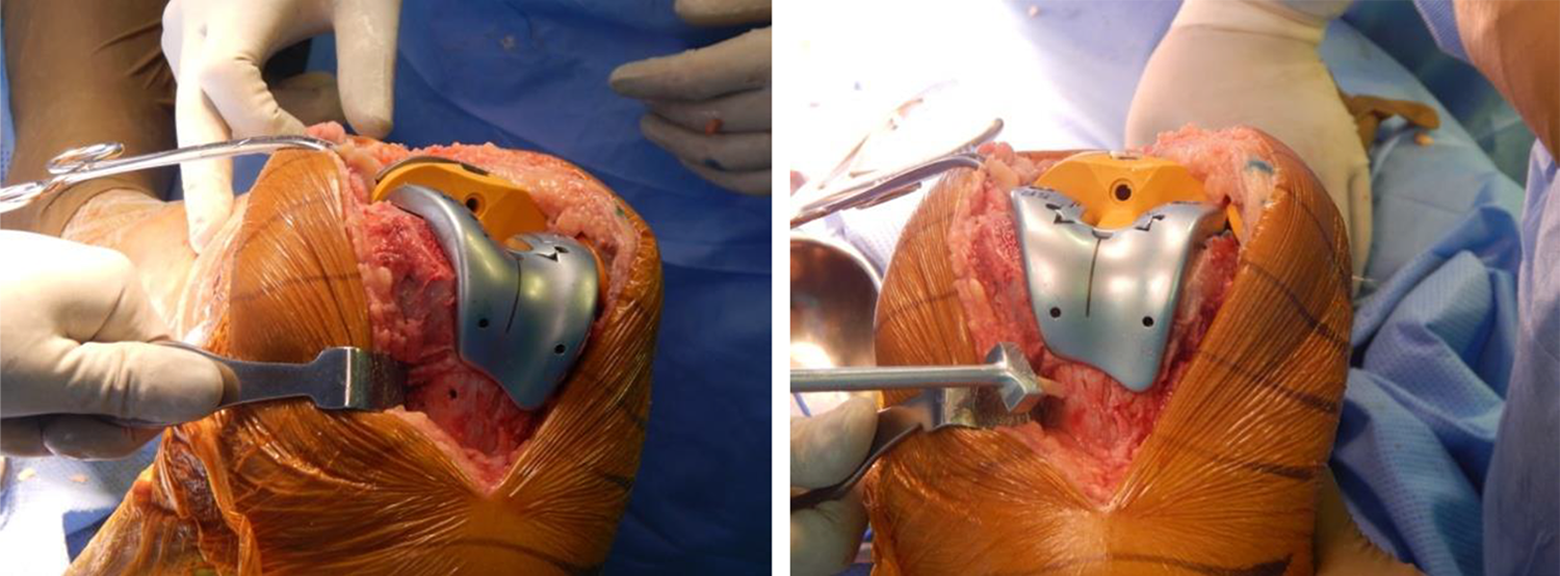
Punching of conical bone fragment in femoral pin hole.
Statistical analysis
The sample size was calculated on the basis of blood loss. The mean difference in blood loss between navigated and non-navigated group was 23 ml with a pooled standard deviation of 84.45. 21 At 5% alpha error and 80% power, the required sample size was 70 patients per group.
All the statistical calculations were done using statistical analysis software STATA 12. Continuous variables were presented as mean and standard deviation, whereas categorical variables were presented as number and percentages. Independent t test and χ 2 test were applied for quantitative and qualitative variables. Pearson correlation coefficient was calculated to determine correlation between three different methods of calculating blood loss. p Value <0.05 was considered statistically significant.
Blood loss calculation
There are number of methods to calculate blood loss post TKA. We used the following three in this study (1) Gross equation, (2) Hb balance and (3) Hb dilution (Table 1).
Methods to calculate blood loss.

Hb: haemoglobin.
Results
The average age in navigated group was 64.53 ± 7.39 years and that of non-navigated group was 64.58 ± 7.49 years which was not statistically different (p = 0.90). The average BMI in navigated group was 29.89 ± 4.57 and that of non-navigated group was 29.82 ± 4.43 which also did not have statistically significant difference (p = 0.92). The ASA grading and the Charlson Co-morbidity Index (CCI) was also similar in both the groups and hence did not account for confounding variable (p > 0.5; Table 2). The mean tourniquet time in navigated group was 76.72 ± 17.36 and in non-navigated group was 63.79 ± 13.54, which was statistically significant (<0.001). The mean length of stay, systemic and local complication did not have any statistical difference (p > 0.05; Tables 3 and 4).
Demographics of both study groups.

BMI: body mass index.
Preoperative, intraoperative and postoperative patients-related variables.
CCI: Charlson Co-morbidity Index; Hb: haemoglobin.
Systemic and local complications in the two study groups post surgery.

The preoperative Hb in navigated group was 12.83 ± 1.32 and in non-navigated group was 12.58 ± 1.33 (p = 0.24). The postoperative Hb taken day 4 before any blood transfusions was 8.60 ± 1.24 in navigated group and 8.54 ± 1.16 in non-navigated group (p = 0.75) (Figure 3). The average fall in Hb was 4.23 ± 1.08 in navigated group and 4.04 ± 1.19 in non-navigated group which was statistically not significant (p = 0.31). Packed red blood cells were transfused in those patient requiring blood transfusion. Seventeen patients in group N were transfused 20 units of blood as compared to 20 patients in non-navigated group who were transfused 27 units of blood. Both the number of patient receiving blood transfusion and number of units transfused were not statistically significant (Table 3).

Comparison of pre and postoperative Hb in between two groups.
There was no difference in blood loss between group N and M in all three methods; Gross equation (p = 0.56), Hb dilution (p = 0.24) and Hb balance (p = 0.20; Table 5). We also calculated the Pearson correlation coefficient of pairwise analysis of blood loss among these formulae, and it showed that the calculation by all three methods was in close approximation with no major difference between the three methods (Table 6).
Blood loss calculated by three reliable methods.
Hb: haemoglobin.
The r-matrix of Pearson correlation coefficient of the pairwise blood loss among three methods.

–: not applicable; Hb: haemoglobin.
Discussion
Blood loss after TKA is a complex and variable phenomenon dependent on patient’s demographics, 22 tourniquet pressure, 23 pre and postoperative medication, 3 coagulation profile 24 and soft tissue dissection. 25 Our matching and exclusion criteria ensured that we have a matched pair of patients especially with respect to factors which could influence blood loss. Apart from BMI, age and sex which were matched, tourniquet pressure and DVT prophylaxis were also the same in all patients of both groups.
To minimize the variability and the inaccuracy in blood loss estimation methods post TKA, we have used three reliable formulae of blood loss estimation post major orthopaedic surgery. We did not find any statistically significant difference in blood loss calculated by Gross equation, Hb dilution or Hb balance method in navigated versus non-navigated SBTKA.
Gross equation 26 is the most popular method to estimate blood loss after TKA and is based on the concept of shifting of fluid from intra compartment to extra compartment to maintain circulation and closely predicts blood loss. It is not valid for cases with large blood loss and does not take into consideration the preoperative volume changes due to anaemia. Because individual factors such as gender, height, weight and volume of red blood cell loss are taken into consideration, the Gross equation reflects actual blood loss to maximum accuracy. However, Gross formula does not account for haemoglobin-related factors which may be a limitation of this method.
Hb balance method 12,26 is the most accurate and scientific method for blood loss calculation after TKA surgery. Many studies prefer it upon other methods for calculation. It takes total blood volume transfusion into consideration and therefore predicts blood loss more precisely.
The Hb balance method is based on the balance of Hb during the perioperative period of primary TKA surgery. Many authors chose this method to investigate the composition and mechanism of blood loss after TKA surgery and this corroborates its clinical utility.
We also calculated blood loss between group N and M by third popular method of Hb dilution. 26 It is a non-invasive method and takes sex, weight and height into account for blood loss calculation. Since it considers subject as normovolemic, it is not recommended for acute blood loss estimation. Also, this method underestimates true blood loss by more than 30% after moderate blood loss of approximately 10% of the total blood volume. 27
We did not calculate the blood loss in the drains as that has been shown to estimate only the apparent blood loss on the first postoperative day and can be subjective and inaccurate. 28 All the studies comparing blood loss in TKA with or without navigation have been on unilateral knee arthroplasty and have used only one of the above mentioned methods for blood loss calculation. Ours is the first study to analyse the blood loss in SBTKA performed with or without navigation. We have used three methods for blood loss calculation and also studied their correlation which is the main strength of this study.
Making a 9-mm intramedullary hole has been cited as the main cause of increased blood loss in the studies done without navigation, 29 but it can be offset by four 3.2 mm holes done for placement of Schanz screws in navigated cases. One of the reasons for the equivalent blood loss in both groups could be related to our surgical technique, wherein we block the intramedullary hole in the non-navigated case with a solid block of bone and then seal it with some cement. We use the same method to block four pin holes in the navigated cases. The reason for equivalent blood loss in both groups can also be correlated with increased tourniquet time in the navigated group which is associated with thrombolytic activity and higher activation of anti-thrombin III leading to increased bleeding. 4
Our study is not without its limitations. The first limitation of this study is that we had one set of navigation instruments and the second case of the day was performed without navigation which could have induced a selection bias. We attempted to mitigate this by rigorous matching criterion and stringent exclusion criteria. Second limitation is the relatively small number of patients in both the groups, as the number of bilateral cases is relatively less in our practise, though 77 cases in each group give sufficient power to this study. Third limitation is the retrospective nature of this study with its inherent biases. Our data from a prospective database and standardized surgical protocols minimize this limitation. Fourth limitation is use of three different implant types in this study. It is well known that implants vary in the medio-lateral dimension and may leave some femoral bone exposed on the medial side that could influence the blood loss from exposed cancellous bone. 30 However, we believe that this would not have affected the blood loss from the bone surfaces as we apply a thin layer of cement on all exposed raw bone surfaces.
It is evident that computer navigation provides no additional benefit in reducing blood loss especially in current practice with the use of tranexamic acid, epidural anaesthesia and use of bone wax or cement on bleeding bone surfaces. Future research should be guided on other modalities to reduce blood loss especially in SBTKA where patient could lose one-third of blood volume.
Conclusion
Blood loss and transfusion is equivalent in SBTKA perform with or without navigation.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
