Abstract
Introduction:
The number of patients who suffered from osteoporotic vertebral fractures is increasing. Osteoporosis has been reported to affect the healing process using long bone models. However, few studies have reported using vertebrae. In this study, we created a bone defect in the anterior part of vertebral body in ovariectomized rat and evaluated the healing process.
Methods:
Fifty-six 12-week old Sprague Dawley rats were divided into ovariectomy (OVX) and sham operation groups. A bone defect was created in the vertebral body 8 weeks after the first surgery. In both groups, the vertebral bodies were harvested immediately or at 4, 8, or 12 weeks after the second surgery (n = 7 at each time point). Bone volume (BV, mm3), bone volume fraction (BV/TV, %), trabecular thickness (Tb.Th, mm), trabecular number (Tb.N, 1/mm), and trabecular separation (Tb.Sp, µm) were evaluated by micro-computed tomography to assess the new bone formation. Histological analysis was also performed.
Results:
The BV and the BV/TV were significantly lower at 4 and 12 weeks in the OVX group compared with those in the sham group. The Tb.Th was significantly lower at 8 and 12 weeks in the OVX group. Histologically, at 12 weeks, in the OVX group, the bone had a thinner, layered structure on the surface of the defect, and the trabecular structure was less dense.
Conclusion:
This study demonstrated that bone mass formation was suppressed and the quality of repaired bone was poor in the healing process of vertebral body defect under osteoporotic conditions. These findings could be the key to understand the pathology of osteoporotic vertebral fracture and to develop its therapies.
Introduction
Osteoporosis is characterized by reduced bone mass and abnormal bone microstructure, resulting in an increase of bone fragility and the risk of fractures. 1 Over 200 million people are affected worldwide, with a majority of patients being postmenopausal old women, in whom ovarian functions are diminished. 2 The vertebrae are one of the major fracture regions caused by osteoporosis, and the characteristics of osteoporotic vertebral fractures are different from long bone fractures. Since osteoporotic vertebral fractures can occur in the absence of a strong external force, it is estimated that less than one-third of all vertebral fractures are clinically diagnosed. 3 However, all vertebral fractures, whether symptomatic or radiologically identified, are associated with mortality and morbidity, including back pain and decreased physical health status. Osteoporotic vertebral fractures are related to increased risk of further vertebral fractures, with resulting progressive deformity of spine, as well as increased risk of nonvertebral fractures, 4 but the pathological influence of osteoporotic vertebral fractures remains to be fully elucidated.
While most of the current osteoporosis research focused on fracture prevention using a variety of pharmacological agents, the influence of osteoporosis on fracture healing has not been as well studied. Osteoporosis has been postulated to impair the healing process in animal experiments as judged by the formation of callus, mineralization, and mechanical properties. 5 –7 In these studies, long bone fracture models such as the femur and tibia were used, but few studies have reported using the vertebrae. 8,9 The vertebrae change less with growth and contain more trabecular bone compared with long bones. Trabecular bone plays a major role in fractures because it represents over half of the bone mass in lumbar vertebrae. Since the characteristics of vertebrae and long bones differ, an understanding of the pathology and healing process of osteoporotic vertebral fractures requires a study using an animal model of osteoporosis.
Objectives of the current study are to investigate the new bone formation in a defect in osteoporotic vertebral bone. In this study, a bone defect in the anterior part of vertebral body was created in both normal and osteoporotic rats induced by ovariectomy (OVX), and sequential bone healing was evaluated by micro-computed tomography (µCT) imaging and histological analysis.
Materials and methods
Experimental animals
The Committee for Animal Research of Kyoto Prefectural University of Medicine authorized all animal experimental procedures, and the study conformed to international guidelines on the ethical use of animals. Fifty-six 12-week old Sprague Dawley rats were divided into OVX and sham operation groups (n = 28 in each group). We performed surgery to create a bone defect in the third lumbar vertebral body 8 weeks after the first surgery. In both groups, rats were euthanized immediately or at 4, 8, or 12 weeks after surgery (n = 7 at each time point) to collect the second and third lumbar vertebral bodies.
Surgical procedures
OVX and sham operation
All operations in this study were performed under sterile conditions. Rats were anesthetized with 1.5% isoflurane. In both the OVX and sham groups, a 2-cm abdominal incision was made 1 cm from the rostral end of external urethral meatus. The subcutaneous tissue and rectus abdominis muscle were incised. After accessing the abdominal cavity, the ovary surrounded by fat was identified at the lateral end of the uterus. Bilateral ovaries were removed in the OVX subjects. In the sham subjects, the ovaries were left intact.
Bone defect
Eight weeks after OVX or sham operation, a bone defect was created in both groups. All rats were anesthetized with 1.5% isoflurane. The abdomen was shaved and sterilized with povidone-iodine. A 5-cm abdominal skin incision was made over the lumbar region (L2–L4). The subcutaneous tissue and rectus abdominis muscle were incised. After the intestine was covered with saline-moistened cheesecloth for protection, the retroperitoneum was incised and the iliopsoas muscles on both sides were separated to expose the anterior part of the L3 vertebral body. A hemispheric 4-mm bone defect was created in the center of the anterior vertebral body using a 4-mm high-speed drill (Figure 1). After the defect site was sufficiently washed, tramadol (10 mg/kg) was intraperitoneally administered for pain relief. The abdominal muscle tissue and skin were closed with absorbable 4-0 suture. Neomycin (2 mg/mL) was added to drinking water for 7 days to prevent from bacterial infection.

Hemispheric bone defect in lumbar vertebral body. In the center of anterior L3 vertebral body, a hemispheric 4-mm bone defect was generated at 8 weeks after OVX or sham operation. A 4-mm high-speed drill was used to create the defect. OVX: ovariectomy.
Evaluation of osteoporosis
We assessed the bone mineral density (BMD) at 8 weeks after OVX, using the second lumbar vertebra, in which a bone defect was not created. The vertebrae were scanned using µCT (micro focus 2D/3D, ScanXmate-E090S40; Comscan Tecno Co., Ltd., Yokohama, Japan) at 60 kV and 90 μA. Three-dimensional image reconstruction was performed using a software program (FanCT version 1.3; Comscan Tecno Co., Ltd., Yokohama, Japan). The volumetric bone mineral density (vBMD) was measured in three dimensions using an image analysis software (TRI/3D-BON; RATOC System Engineering, Tokyo, Japan). We confirmed shrinkage of the uterine tissue in all rats after euthanization.
Evaluation of bone healing
We used the third lumbar vertebrae to assess the repair process of bone defects over time, using imaging and histology, and performed comparative analysis of the OVX and sham groups.
Bone morphometric analysis
The collected third lumbar vertebrae were scanned with the μCT system. Three-dimensional image reconstruction was performed using a software program. A region of interest was set to a semicylindrical area with a 4-mm diameter and 4-mm height, around the created bone defect site. Bone volume (BV), bone volume fraction (BV/TV), trabecular thickness (Tb.Th, mm), trabecular number (Tb.N, 1/mm), and trabecular separation (Tb.Sp, µm) in the regenerated bone were calculated using the image analysis software (TRI/3D-BON; RATOC System Engineering).
Histological analysis
The collected third lumbar vertebrae were fixed with 70% ethanol and delipidated with 80% ethanol for 3 days. The samples were decalcified with 10% formic acid for 6 days. Slices in the mid-sagittal plane were prepared in a cryostat, and the bone formation at the site of the defect was histologically assessed by hematoxylin–eosin (HE) staining and safranin O staining.
Statistical analyses
Data were shown as mean ± standard error (SE). We used the Mann–Whitney U test in the statistical analysis. p Values less than 0.05 were regarded statistically significant.
Results
Confirmation of OVX-induced osteoporosis
The vBMD values at 8 weeks after OVX were 231.0 ± 15.2 mg/cm3 in the OVX group and 312.6 ± 16.7 mg/cm3 in the sham group (Figure 2(a)). The vBMD was significantly lower in the OVX group. The body weight was 363.6 ± 17.0 g in the OVX group and 288.6 ± 24.2 g in the sham group (Figure 2(b)). The body weight was significantly greater in the OVX group. Marked atrophy of uterine tissue was observed in the OVX group compared with the sham group. These findings indicate that OVX-induced osteoporosis was successfully introduced.

Bone mineral density and body weight after OVX. After 8 weeks from OVX or sham operation, the vBMD and body weight were measured to confirm successful OVX. (a) The vBMD of L2 vertebral body, in which a bone defect was not created, was significantly lower in the OVX group than the sham group. (b) The body weights of OVX rats were shown to be greater than sham rats. Data are shown as the mean ± SE (n =7), * p < 0.01. OVX: ovariectomy; vBMD: volumetric bone mineral density; SE: standard error.
Morphometric changes of bone defect
Changes in BV and BV/TV over time after the creation of a bone defect in the vertebral body were examined (Figure 3). The μCT images exhibited that bone repair progressed over time in both groups, but the bone defect was not completely repaired at 12 weeks in either group (Figure 4). The bone mass of the regenerated bone was lower in the OVX group (Figure 3(a)). The BV values were significantly lower at 4 and 12 weeks after the bone defect creation in the OVX group compared with those in the sham group (4 weeks: 3.8 mm3 in the OVX group vs. 5.4 mm3 in the sham group; 8 weeks: 6.3 mm3 vs. 6.4 mm3; 12 weeks: 7.4 mm3 vs. 10.9 mm3). The BV/TV values were 14.2% (OVX group) and 17.0% (sham group) at 4 weeks, 20.7% (OVX group) and 25.8% (sham group) at 8 weeks, and 22.1% (OVX group) and 31.7% (sham group) at 12 weeks after the bone defect creation. The BV/TV values were significantly lower at 4 and 12 weeks in the OVX group (Figure 3(b)). The Tb.Th was significantly lower at 8 and 12 weeks after the bone defect creation in the OVX group compared with those in the sham group (4 weeks: 173.4 µm in the OVX group vs. 192.4 µm in the sham group; 8 weeks: 208.1 µm vs. 260.1 µm; 12 weeks: 210.7 µm vs. 294.8 µm; Table 1).
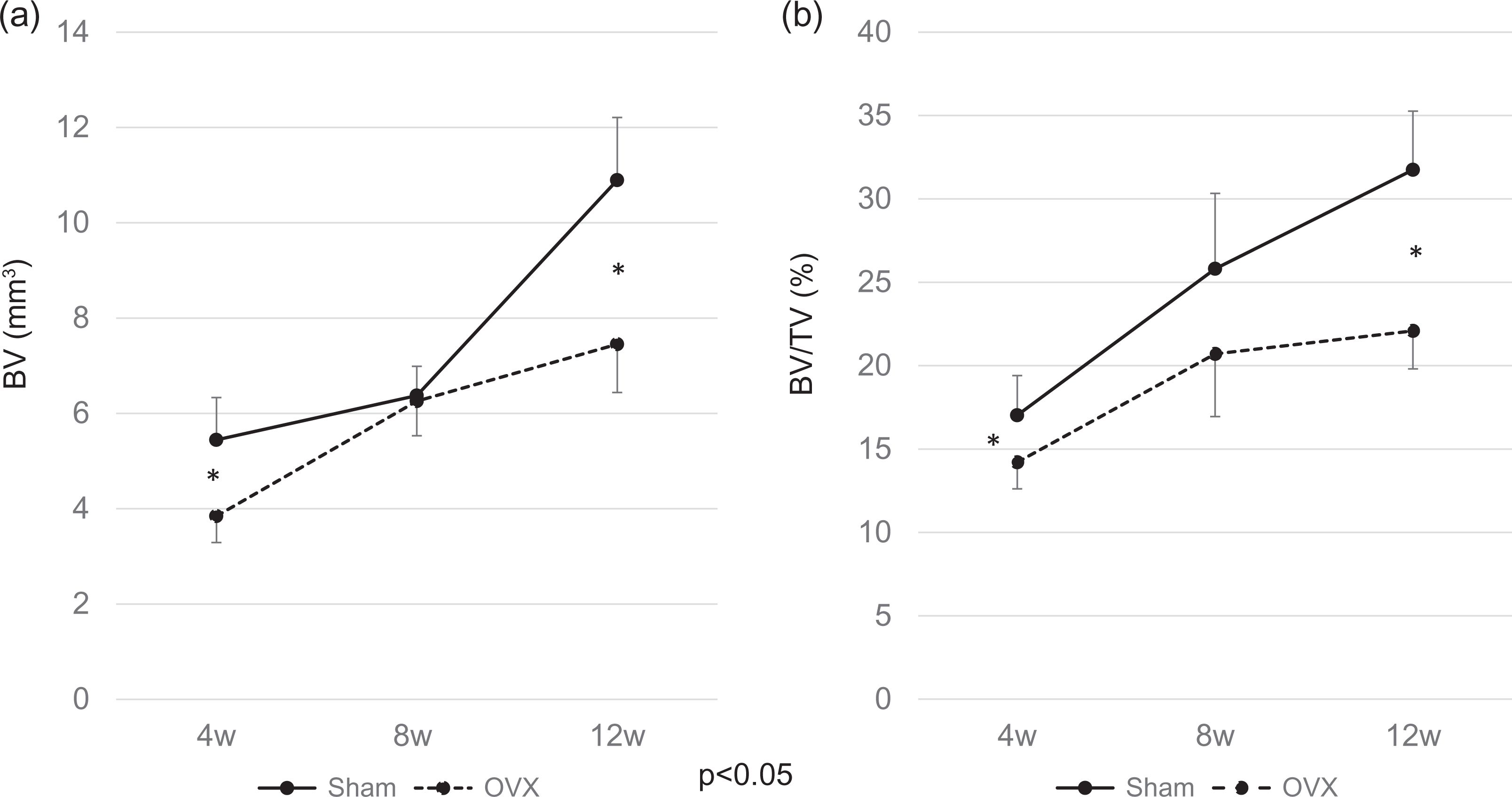
Morphometric changes of lumbar vertebral body defects. The extracted third lumbar vertebrae were scanned with µCT system. The BV and BV/TV in the regenerated bone were calculated using image analysis software. (a) The BV value was significantly lower at 4 and 12 weeks after the bone defect was created in the OVX group compared with those in the sham group. (b) The BV/TV value, an index of bone quality, was also significantly lower at 4 and 12 weeks in the OVX group. Data are shown as the mean ± SE (n =7 at each time point), * p < 0.05. µCT: micro-computed tomography; BV: bone volume; BV/TV: bone volume fraction; OVX: ovariectomy; SE: standard error.

µCT images of bone repair process in lumbar vertebral body defects. Sagittal images of lumbar bodies were shown. In both groups, bone healing was progressing over time. The bone defect was not completely repaired at 12 weeks in either group. µCT: micro-computed tomography.
Tb.Th, Tb.N, and Tb.Sp of lumbar vertebral body defects.a

Tb.Th: trabecular thickness; Tb.N: trabecular number; Tb.Sp: trabecular separation; SE: standard error.
aData are shown as the mean ± SE (n =7 at each time point).
b p < 0.05.
Histological findings during bone healing
The HE-stained specimens of vertebral bodies with a bone defect are shown sequentially in Figure 5. The growth of fibroblast-like cells at the periphery of the bone defect was observed at 4 weeks after the bone defect was created in both groups. Significant outgrowth of fibroblasts entering the spaces of the trabecular structure was observed in the sham group, but it was not apparent in the OVX group at 4 weeks. In the sham group, mature fibroblasts started to be replaced with new bone tissue at 8 weeks, and the bone tissue formed a fine trabecular structure at 12 weeks. In contrast, in the OVX group, the medullary cavity was replaced with non-dense adipose tissue over time. Finally, in the OVX group, the bone had a thinner, layered structure on the surface of the defect, and the trabecular structure was less dense compared with the sham group. In both groups, Safranin O-positive cartilaginous tissue was not observed clearly at any time point (data not shown).

Histological findings during bone healing. The bone repair process at 0, 4, 8, and 12 weeks after creating bone defects was histologically assessed by HE staining. The growth of fibroblasts at the periphery of the bone defect was observed at 4 weeks in both groups. In the sham group, remarkable outgrowth of fibroblasts was observed (arrow); whereas, this outgrowth was suppressed in the OVX group (arrow). At 12 weeks, the bone tissue formed a fine trabecular structure in the sham group. In the OVX group, the trabecular structure was less dense compared with the sham group. The scale bars show (a) 1 mm and (b) 0.2 mm. µCT: micro-computed tomography; OVX: ovariectomy. HE: hematoxylin–eosin.
Discussion
The present study is the first report to reveal the influence of osteoporosis on bone repair process in vertebral body defect. Image assessment showed that a smaller volume of bone mass was generated at the bone defect site and that the bone quality was lower in the OVX group. Histological analysis qualitatively supported these results. Our study demonstrated that osteoporosis negatively affects the healing process of a vertebral body defect.
In this study, the OVX rats were used as an animal model of osteoporosis. This model has been validated as a clinically relevant model of human postmenopausal bone loss. 10 An OVX was performed in rats at the age of 12 weeks, and a bone defect was created 8 weeks later. We used sexually mature rats at this age in order to purely assess osteoporosis caused by OVX, unaffected by age-related changes. The assessment at 8 weeks after OVX showed marked osteoporotic changes including body weight gain, atrophy of uterine tissue, and decreased BMD in the OVX rats compared with that in the normal rats, consistent with previously reported results. 11 –13
Several studies have reported that osteoporosis affects the healing process of bone fractures and defects. 5 –7 It has been demonstrated that osteoporosis impaired the bone healing in terms of the formation of callus, mineralization, and mechanical properties using long bone models. Namkung-Matthai et al. showed that osteoporosis affected the early phase of fracture healing, 5 whereas Kubo et al. 6 and Hao et al. 7 reported that osteoporosis influenced the late phase of healing. In contrast, Cao et al. reported a faster fracture healing process in the osteoporotic rats due to osteoporosis-related acceleration in its callus remodeling. 14 Therefore, the pathophysiology of osteoporotic bone healing was still in debate. In the current study, we examined the repair process over time of a defect in a vertebra, not a long bone. This study provided evidence that bone healing in osteoporotic vertebrae was delayed and newly formed bone quality was decreased in both early and late phases.
There are several steps in bone healing process. In order for bone to regenerate, mesenchymal stem cells (MSCs) would migrate into injured region, proliferate, and differentiate into osteogenic cells. The cartilaginous callus formed from these osteogenic cells undergoes mineralization and resorption, and is then replaced with lamellar bone. The present study showed that fibroblast-like cells proliferated until 4 weeks after the bone defect creation were replaced over time to form high-quality bone in normal group, whereas in the osteoporosis group, fibroblast-like cell induction was not obvious. This lower population of fibroblast-like cells was probably related to the MSCs recruitment and may be a cause of delayed bone repair. Although the cartilaginous tissue was not histologically confirmed, a new bone formation in the defect was observed from 8 weeks to 12 weeks in the normal group. While the bone had a thinner, layered structure on the surface of the defect, the trabecular structure was less dense in the osteoporosis group. This trabecular bone formation process at the vertebral body defect was different from long bone models. The expression of various growth factors and inflammatory cytokines is reportedly regulated in osteoporosis. 15,16 The alteration of molecular cascades controlling bone formation under osteoporotic condition may also affect the decreased quality of regenerated bone. The molecular mechanisms at the cellular level need to be studied in detail.
In bone defect models, long bones of the extremities and cranial bones are frequently used, 17 –19 but these differ in structure from the vertebral body in several aspects. Although analysis using a model in which a burr hole was created in the center of the vertebral body has been reported, 20 the anterior portion is damaged in most vertebral body fractures. In order to mimic the mechanical stress that the vertebral body undergoes in human, as well as an environment similar to that present in actual osteoporotic vertebral fractures, we created a bone defect in the anterior vertebral body. An extensive review of the literature only found a study in normal rats by Liang et al., 21 in which a model using a bone defect was created in the anterior vertebral body. They first described induction of a critical size bone defect in the vertebral body. Our results demonstrated that the bone defect in the anterior vertebral body does not recover completely by self-repair alone in osteoporotic condition at 12 weeks after the defect creation; this is a basic condition for the analysis of treatment of bone defects using this model. This model may be useful for the assessment of the effects of systemic administration of drugs and topical application of artificial materials and growth factors on the healing process at bone defect sites. Thus, we conclude that our animal model possesses characteristics of osteoporotic vertebral fractures encountered in clinical practice.
There are some limitations that should be considered in this study. First, this animal model created vertebral bone defect is different from vertebral fracture. However, bone defect in vertebral body occurs when normal vertebral fracture healing is disrupted in some ways (i.e. delayed union, pseudoarthrosis, etc.). It may be possible to use this animal model for developing the treatment of vertebral fracture in such an adverse condition. Second, the biomechanical environment of the spine is different between quadruped and biped. Further studies are needed to discuss the matter of human vertebrae. Finally, the pathology of osteoporotic vertebral fracture is not fully elucidated in this study. Additional bone histomorphometrical analysis will be helpful.
In summary, this study demonstrated that osteoporosis influences negatively on bone repair in lumbar vertebral body defects from early phase. Consequently, this caused a reduction in bone regeneration and abnormal structure of the regenerated bone. These factors make treatment of osteoporotic vertebral fractures difficult. We believe that this rat model reflects the pathophysiology of bone defects caused by osteoporotic vertebral fracture. Research using the present animal model will lead to an understanding of the pathology of osteoporosis in detail and enable the development of therapies.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a Grant-in-Aid for Scientific Research (No. 16K10832) from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
