Abstract

Introduction
Duchenne muscular dystrophy (DMD) is an
However, regular corticosteroid use causes osteoporosis, which results in an increased risk of bone loss and osteoporotic fractures [13]. Corticosteroid-induced osteoporosis results from the inhibition of osteoblast formation, decreasing calcium absorption in the gastrointestinal tract, and increasing bone resorption and renal calcium excretion [13]. Progressive development of osteoporosis causes vertebral compression fractures (VCFs). In the pediatric population receiving corticosteroid therapy, the prevalence of VCFs is reported to be between 7% and 34% [11,14,15]. Of pediatric patients with VCF, 44% to 81% are asymptomatic [9,11] and the most symptomatic children have mild and transient symptoms [9]. A small number of patients have severe symptoms and that require treatment. Calcium, vitamin D, and bisphosphonates are used in symptomatic patients [7].
When medication fails to address symptoms, percutaneous vertebroplasty has the potential to enhance patients’ quality of life and mobility. However, the literature regarding this procedure in pediatric osteoporotic fractures is limited; it includes just a few publications [3,5,7] and only one that addresses patients with DMD [3]. As a result, there is a lack of information on indications, outcomes, and patient management, which makes it challenging to plan this complex treatment.
We present a case study involving the successful use of vertebroplasty in the treatment of a patient with DMD whose symptoms, caused by osteoporotic VCFs, could not be alleviated by medication. Management challenges, dilemmas, and implications for the treatment of this compromised patient population are discussed.
Case Description
A 17-year-old male patient with DMD treated with corticosteroid (deflazacort) presented to a tertiary pediatric orthopedic spine clinic with severe low back pain that began while transferring from his wheelchair. Lumbar spine radiography and subsequent magnetic resonance imaging (MRI) demonstrated VCF of L1 vertebra with height loss of 50%, VCF of L3 vertebra with height loss of 40%, VCF of L4 vertebra with height loss of 40%, and minimal VCF of T12 vertebra with height loss of 5% (Fig. 1). Bone marrow edema was seen in the affected vertebrae on short-TI inversion recovery (STIR) MRI images, most extensively in L4. Treatment with calcium, vitamin D3, and appropriate opioid analgesics was initiated. The patient continued to have severe low back pain in the absence of radiculopathy. Treatment was augmented with bisphosphonate (pamidronate). With this treatment protocol, the patient regained his preinjury activity 6 months postinjury and was pain free. Bone mineral density (BMD) testing demonstrated decreased bone mass below the expected range for his age, with a Z score of −3.6 in the lumbar spine.
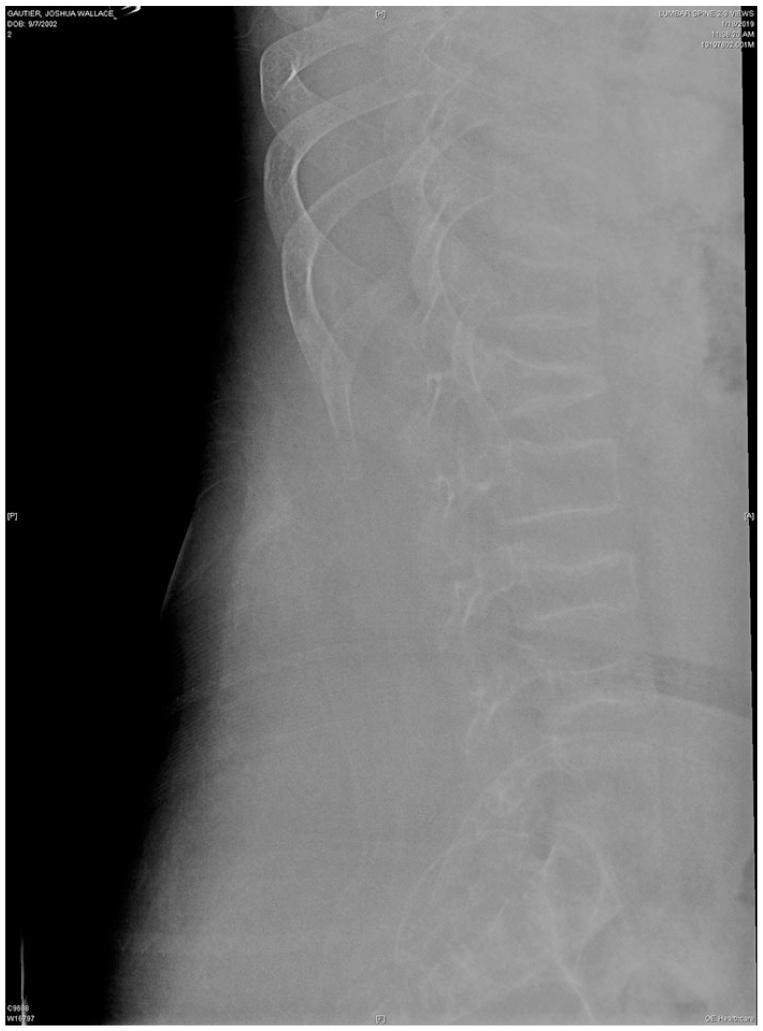
Radiograph showing compression fractures of L1, L3, L4 and mild compression fracture of T12.
The patient then sustained another fall injury that resulted in an acute exacerbation of his symptoms and necessitated hospital admission for management, with a combination of medications prescribed including morphine 7 mg daily for breakthrough pain. He required bed immobilization and could not tolerate transfer to his wheelchair. Imaging studies found compression fractures of T12, L1, L3, L4, and new VCF of L5 (Fig. 2) with height loss of 35% and extensive bone marrow edema in L5 on STIR images.
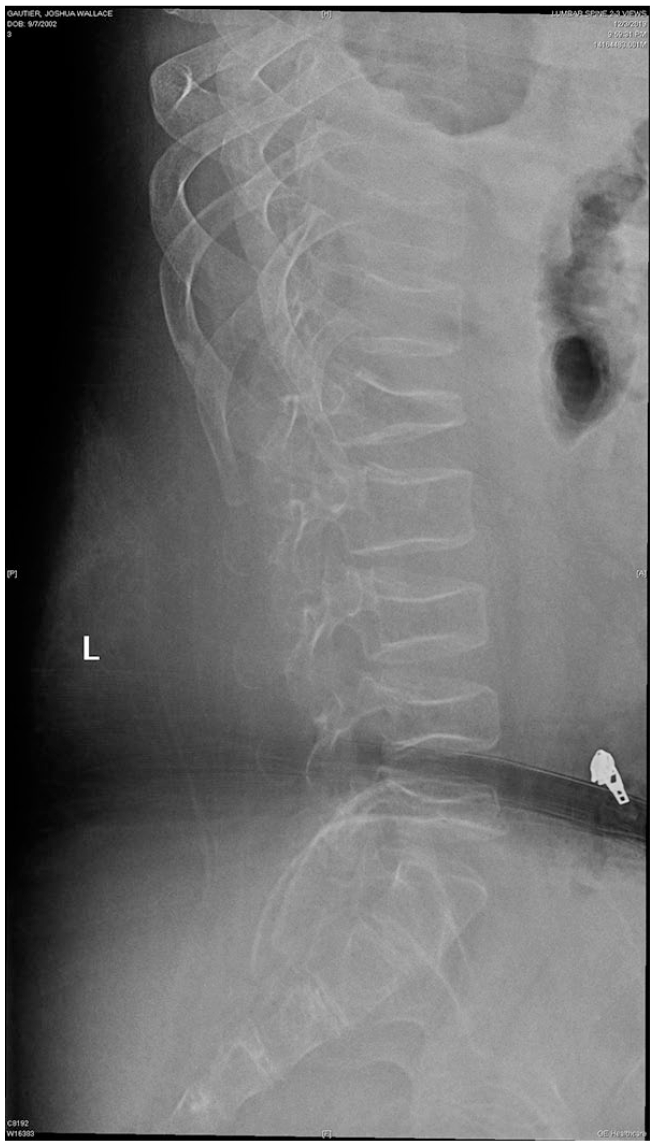
Radiograph showing new compression fracture of L5.
Considering the medications’ failure to alleviate symptoms and prevent osteoporosis progression, treatment with percutaneous vertebroplasty was proposed to ease his pain and improve mobilization.
The patient’s comorbidities were taken into account. In addition to DMD, his medical history included obesity and obstructive sleep apnea that required a continuous positive airway pressure machine. Pulmonary function examination demonstrated significant compromise (35% of predicted). For management planning, we sought input from anesthesiology and pediatric intensive care teams. There was documented concern that general anesthesia and intubation would cause the patient to become ventilator dependent. The patient was assigned American Society of Anesthesiologists (ASA) class IV (severe systemic disease that is a constant threat to life). Admission to the pediatric intensive care unit (PICU) was preplanned.
Vertebroplasty of T12-L5 vertebrae using a bipedicular approach was performed without complication (Fig. 3). The patient tolerated the procedure well and was successfully extubated. There were no respiratory or cardiac events reported.
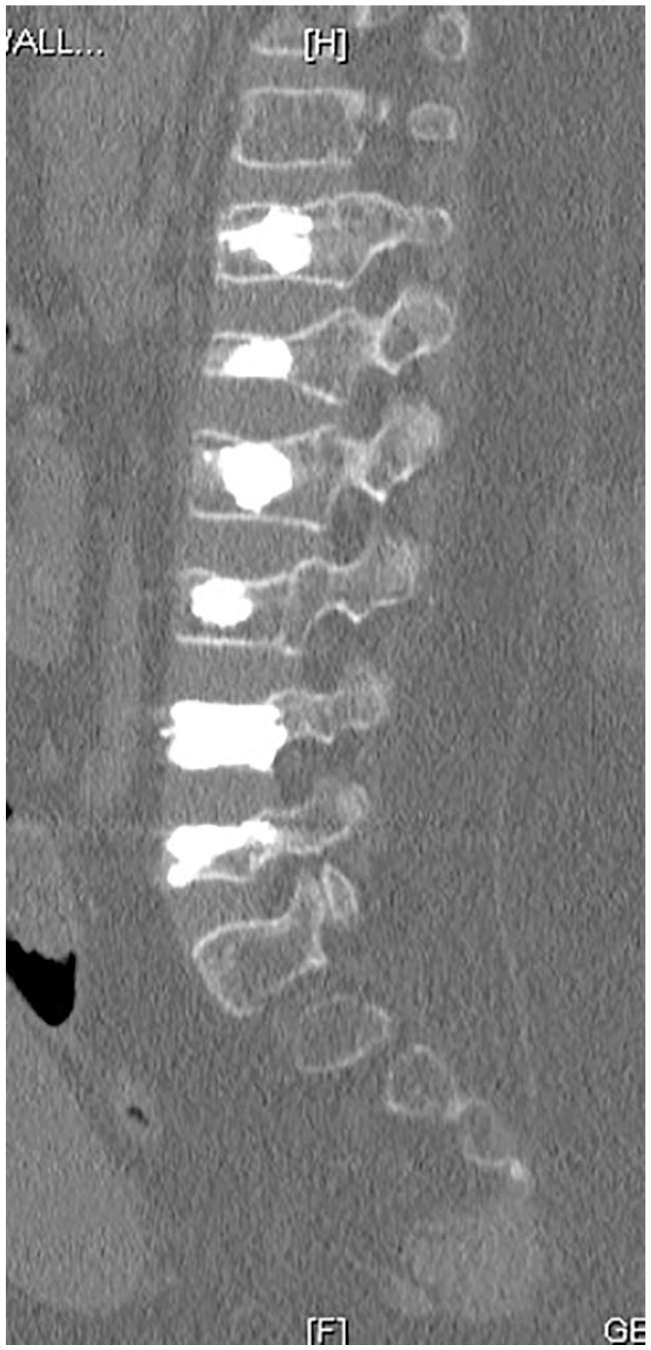
Postprocedure computed tomography.
The day after the procedure, while in the PICU, the patient was found to be tachycardic with oxygen saturation in the low 90% range with supplemental oxygen at 4 L/min. A computed tomographic pulmonary angiogram revealed bilateral atelectasis and consolidations in the lungs and a few tiny cement foci in pulmonary arteries. With oxygen supplementation at 5 L/min, the patient’s oxygen saturation increased to 98% to ~100%. Five days postprocedure oxygen supplementation was no longer required. At discharge, the patient was able to mobilize to his wheelchair and his analgesic needs decreased to hydromorphone 4 mg with a weaning schedule. Shortly after discharge, the patient and his family reported significant improvement in his symptoms and that he was being weaned from opioids.
Discussion
In the elderly population, VCFs are common, with a prevalence of more than 30% in people above the age of 80 years [20]. Linked directly to osteoporosis, VCFs represent the leading cause of disability and morbidity, contributing to debilitating pain and worsening quality of life [18]. Treatment in the elderly population is well studied and established [2]. Strong evidence supports the use of percutaneous vertebroplasty and kyphoplasty in this population [2].
However, while vertebroplasty may improve quality of life and enhance mobility in pediatric patients with VCF, less research has been done on its application in this age group. Only 3 cases describing the treatment of pediatric osteoporotic fractures with percutaneous vertebroplasty/kyphoplasty have been published describing treatment of patients with DMD [3], systemic lupus erythematosus [7], and IPEX-like syndrome [5]. Reports of vertebroplasty or kyphoplasty in pediatric patients with other pathologies are also scarce: 3 patients with traumatic fractures [16] and 6 patients with neoplastic vertebral lesions or other conditions [1,5,6,12,17]. This can probably be explained by the fact that most symptomatic pediatric patients with VCFs have transient symptoms that are treated medically [9]. The paucity of the literature for this cohort makes decision-making challenging in cases of severely symptomatic patients with debilitating pain that is not responding to medical therapy.
When considering treatment of pediatric patients with osteoporotic VCF, the concern was raised about the potential effect of heat produced by cement polymerization on disruption of vertebral growth and remodelization. To prevent this, the use of lower exothermic cements has been suggested [7]. The limited evidence of vertebroplasty/kyphoplasty in the pediatric population does not allow a definitive conclusion on that matter to be drawn and to support this recommendation. The evidence of vertebral growth following cement injection [3] suggests that there might not necessarily be a clinically significant negative effect of the cement injection on vertebral growth.
Another challenge is the decision regarding treatment of the intervening (“sandwich”) vertebral body (L2 in our case) that did not sustain VCF. The literature is inconclusive on the development of adjacent-level vertebral fracture after initial cement augmentation [4,19]. The following considerations are to be taken into account: the necessity of continuing corticosteroid therapy, the degree of osteoporosis, and the risk of the future VCF in intervening vertebra (regardless of conjunction with planned vertebroplasty in adjacent vertebrae). The BMD testing can be used to evaluate the risk of future osteoporotic fracture [14]. In one study, 67% of pediatric patients had a Z score of less than −2.0 (Z score in our patient was −3.6) [15].
Another important consideration is the risk of general anesthesia, which is high in patients with DMD due to reduced pulmonary function, among other factors. Risk increases with the slow and steady progression of DMD. This influenced the decision in our case to perform vertebroplasty of intervening vertebra.
In patients with DMD, decreased pulmonary function and gastric hypomotility may increase risk of complications. This highlights the importance of perioperative planning, which is best accomplished by a multidisciplinary team. Risk factors should be disclosed to the patient and the decision-makers when discussing perioperative morbidity.
In conclusion, vertebroplasty for the treatment of osteoporotic VCF in severely symptomatic pediatric patients who have debilitating pain that is not responding to medication and is extremely limiting mobility is a palliative procedure. It has the potential to relieve pain and improve quality of life. Comorbidities, continuous use of corticosteroids (that cannot be withdrawn), bone growth, anesthesia risks, and perioperative care are among the factors that should be considered in planning and decision-making. This is best carried out by a multidisciplinary team that can determine which supportive measures need to be put in place perioperatively for a successful outcome.
Supplemental Material
sj-zip-1-hss-10.1177_1556331621990648 – Supplemental material for Vertebroplasty for Treatment of Steroid-Induced Osteoporotic Vertebral Compression Fractures in an Adolescent With Duchenne Muscular Dystrophy
Supplemental material, sj-zip-1-hss-10.1177_1556331621990648 for Vertebroplasty for Treatment of Steroid-Induced Osteoporotic Vertebral Compression Fractures in an Adolescent With Duchenne Muscular Dystrophy by Paul C. Missiuna, Mohamed Sarraj, Batool Bosakhar, Patrick Thornley, John Donnellan, Waleed Kishta, Peter Darby and Zeev V. Maizlin in HSS Journal®: The Musculoskeletal Journal of Hospital for Special Surgery
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Human/Animal Rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2013.
Informed Consent
Informed consent was waived from the patient included in this study.
Level of Evidence
Level V (Case Report).
Required Author Forms
Disclosure forms provided by the authors are available with the online version of this article as supplemental material.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
