Abstract
Purpose:
To compare the yearly cost involved per patient in the use of magnetically controlled growing rod (MCGR) and traditional growing rods (TGRs) in the treatment of early onset scoliosis (EOS) and to assess the overall cost burden of MCGR with reference to patient and health-care infrastructure.
Methods:
For a hypothetical case of a 5-year-old girl with a diagnosis of EOS, a decision-tree model using TreeAge Software was developed to simulate annual health state transitions and compare the 8-year accumulative direct, indirect, and total cost among the four groups: (1) dual MCGRs with exchange every 2 years, (2) dual MCGRs with exchange every 3 years, (3) TGR with surgical distraction every year, and (4) TGR with surgical distraction every 6 months. Base-case values and ranges of clinical parameters reflecting complication rate after each type of surgical distraction were determined from a review of literature and expert opinion. Government gazette and expert opinion provided cost estimation of growing rods, surgeries, surgical complications, and routine follow-up. Microsimulation of 1000 individuals was conducted to test the variation in total direct costs (in 2016 Hong Kong dollars (HKD)) between individuals, and estimated the standard deviations of total direct costs for each group.
Results:
Over the projected treatment period, indirect costs incurred by patients and family were higher for the MCGR as compared to the TGR. However, the total costs incurred by MCGR groups (group 1: HKD164k; group 2: HKD138k) were lower than those incurred by TGR groups (group 3: HKD191k; group 4: HKD290k). Although the accumulative costs of three groups (TGR with distraction every year and MCGR replacing every 2 and 3 years) were approaching each other in the first 2 years after initial implantation, at year 3 the accumulative cost of MCGR exchange every 2 years was HKD36k more than the yearly TGR surgery due to the cost of implant exchange. The cost incurred by both the MCGR groups was less than that incurred by the TGR groups from year 4 to skeletal maturity.
Conclusions:
The use of dual MCGRs, regardless of its 2- or 3-year exchange, was only cost saving and less expensive than the dual TGRs for EOS treatment from the fourth year of continuous treatment. Despite higher patient-related costs during MCGR treatment, it is important to consider the reduced risks and mental burden suffered by these children during repeat surgeries. With improved knowledge of the costs associated with long-term MCGR use, better constructed cost-effectiveness studies can be performed in the future.
Introduction
In this modern age, increased health-care costs influence the type of medication or device that is available to health-care providers. A new drug or medical device may be effective clinically in treating a certain condition, but occasionally the cost to the patient and establishment may be too significant and becomes a deterrent to health-care providers to implement. It is necessary for health administrators and government officials to see not only data proving the superiority of a certain treatment method in terms of clinical effectiveness but also cost saving to push forward any new drug or medical device. Hence, health-care economics is an important factor that the industry and clinician must consider prior to testing or initiating any new treatment. Health-care economics is, in particular, a significant factor in guiding decision-making of spine surgery. The field of spine surgery is evolving at a rapid pace with new and possibly improved implant systems introduced into the market. Thus, spine surgeons must be able to balance the necessity of certain devices with their cost.
Early onset scoliosis (EOS) is one spine condition that requires special attention. The associated spinal deformity occurs in young children and if left untreated, the deformity progresses and cause cosmetic disfigurement, poor pulmonary development, and overall poor quality of life. 1 –8 Traditional growing rods (TGRs), which require open surgery under anesthesia every 6–12 months for manual rod distraction, have been the mainstay surgical option available for gradual correction of scoliosis in young children. 9 –11 Implantation of these TGRs allows regular distractions for spine growth and thus avoids spinal fusion. 9,10,12,13 However, if we assume a 5-year-old girl with EOS is implanted with the TGR, she may require up to 16 lengthening procedures if skeletal maturity is at 13 years. This imposes significant psychological and financial burden to the child and family including repeat hospital admissions, visits to clinics and therapists, and increased rate of anesthetic and wound complications with distraction surgery. 1,2,11 The overall wound complication rate from TGR is 16%, which increases by 24% during each additional surgery. 2
The magnetically controlled growing rod (MCGR) is growing in popularity as it avoids repeat manual distractions with the TGR by utilizing an external magnet to drive its internal distraction device. 14 This allows outpatient distractions with the patient awake and under constant neurological monitoring. Thus, the MCGR is a preferred alternative to the TGR. Studies have shown that the MCGR has similar clinical effectiveness in curve correction, 15 –18 better long-term length gains, 19 and can provide safe gradual correction of severe deformities in young patients with EOS. 20 In addition, distractions can be done at more frequent and regular intervals to better mimic normal growth. Although more frequent imaging may be required to monitor these increased distractions, radiation-free techniques like the ultrasound can be used as proxy. 21
Being able to perform more frequent distraction procedures, however, may have potential drawbacks of increased traveling and time off of work for the parents. Furthermore, the cost of the MCGR is significant due to the manufacturing cost of its internal magnet/actuator as compared to more simple or “home-made” constructs used for TGRs. Yet, a possible cost-saving value of the MCGR is to reduce the number of operations. This has the benefit of reduced anesthetic and surgical risks, cost for surgical consumables, and duration of intensive care stay. Several cost analysis studies 22 –24 have attempted to compare the TGR and MCGR in terms of associated costs and have found the MCGR to be cost saving in comparison to the TGR. However, several flaws of these studies include short-term follow-up with limited outlook on the risk of complications with prolonged use of the MCGR and the cost of rod exchanges. Multiple MCGRs are necessary to be implanted over the course of an EOS patient’s treatment since the housing portion of the rod can only accommodate 4.8 cm of distraction, far less than the expected spine growth in a child. 14 Furthermore, implant-related complications such as rod fracture, implant dislodgment, and proximal junction failure have been reported. 14 The authors have the longest experience of using the MCGR with follow-up of subjects until skeletal maturity and rod removal or spinal fusion. 14,17 Hence, it is the opportune moment for us to present a thorough cost analysis comparing TGR and MCGR based on our long-term data. The aim of study as such is to identify the direct and indirect costs associated with TGR and MCGR use with respect to the patient and the health-care infrastructure.
Methods
Model structure and setting
For a hypothetical 5-year-old girl with EOS, a decision tree with individual patient microsimulation model was developed to simulate annual health state transitions and to compare the 8-year accumulative cost among four commonly adopted strategies based on the authors’ experience: (1) dual MCGRs with exchange every 2 years, (2) dual MCGRs with exchange every 3 years, (3) dual TGRs with surgical distraction every year, and (4) dual TGRs with surgical distraction every 6 months. Only dual rods were considered as they have superior outcomes compared to single rods and thus is generally preferred. 9,17 The microsimulation (or first-order Monte Carlo simulation) model started with a decision mode from which one of the four groups (two MCGRs and two TGRs) originated. Within each decision mode, microsimulation generated a hypothetical group of EOS using 1000 patients and tracked them until the end of simulation. Based on the results of these 1000 patients, mean value and standard deviation of accumulative costs per strategy were calculated. A time horizon of 8 years was used (initial surgery at 5 years to projected maturity at 13 years) to cover the whole treatment trajectory between strategies with respect to the types of growing rods and the frequency of distraction surgery. No cross-over between strategies (i.e. individual opt out or transferred from one strategy to another) was allowed in this model. Full compliance was assumed for all surgical and nonsurgical treatments and for routine follow-up assessments. The structure of the costing model is depicted in Figure 1 .

Schematic presentation of the magnetically controlled growing rod and traditional growing rod treatment costing model with four strategies. Microsimulation of 1000 individuals was conducted for each study from implantation till skeletal maturity.
Literature search were performed to identify the types and risks of surgical complications to be included in this model after review of the authors’ own experience. The probabilities of complications after distraction were obtained from a study that specifically compared complication rates between MCGR and TGR for EOS patients. 25 Surgical complications after distraction were single event in nature and were classified into three major subtypes: superficial infection, deep infection, and implant pullout. More than one complication might occur during specific interventions. Since the study 25 did not capture surgical complications other than those three major subtypes, no other surgical complications were assumed to occur after surgery. In particular, rod fracture was not included as it was due to a mechanical weakness in the rod design that has been addressed in the latest generation of MCGRs. 14 No surgical mortality and all-cause mortality were accounted for in this model. Probabilities at base-case scenario are reported in Table 1 .
Clinical parameters associated with magnetically controlled growing rod (MCGR) and traditional growing rod (TGR) in early onset scoliosis patients.

MCGR: magnetically controlled growing rod; TGR: traditional growing rod.
aPatients in TGR group may have at least one surgical complications.
Direct costs
The 8-year cumulative costs per strategy were aggregated based on the health-care provider’s and societal perspectives and expressed as 2016 Hong Kong dollars (HKD) (converting to US dollars (USD) with exchange rate pegged at US$1 = HK$7.8). Medical costs were considered from the health-care provider’s perspective, while both medical and patient time costs were considered from the societal viewpoint. Relevant unit costs of the growing rod, anchor, cross-connector, and cross-link implants were retrieved from the rod manufacturer and cost reports from the Department of Orthopaedics and Traumatology, the University of Hong Kong and its affiliated hospitals: The Duchess of Kent Children’s Hospital and Queen Mary Hospital. As health care in Hong Kong is a government-based public system, all direct costs are charged to the institution while individual patient need to pay only a daily flat rate as cost recovery for health-care system.
In our center, the CD Horizon® Legacy™ system from Medtronic® is the most commonly used instrumentation system for scoliosis surgery. For the purposes of this study, we adopted the costs of using this system for the anchors, cross-connectors, and rods for TGR and the anchors for the MCGR. The routine instrumentation construct used was four anchors at the upper end and four anchors at the lower end. For simplicity of calculation, all anchors analyzed in this study consisted of pedicle screws only. For dual TGR, two rod connectors were used to connect the four rods. However, only the cost of two rods were used for analysis since the length of one manufactured rod was usually adequate to be cut into two rods for creating the TGR construct. In the TGR group, patients were assumed to have one to two surgeries every year depending on the frequency of reoperation and stop surgery at year 7 or the date of projected skeletal maturity. By repeating surgery every 2 years, the patients in the MCGR group were assumed to undergo surgery at initial year, year 2, year 4, and year 6. By repeating surgery every 3 years, the patients in the MCGR group were assumed to undergo surgery at initial year, year 3, and year 6.
Costs associated with each surgery accounted for spinal cord monitoring, duration of operating theater use, manpower (nurse/anesthetist), intraoperative and follow-up X-ray imaging, outpatient visits, and length of hospitalization (general ward) of a total of 3 days inclusive of one night at the intensive care unit (ICU). Costs associated with hospitalizations, outpatient, and physiotherapy visits were based on the latest charges to nonentitled persons for use of health services in the hospital authority/governing health-care body (as cost recovery charges for health services), as published in 2013 government gazette. 26 Based on the authors’ practice, the number of routine follow-up visits given to TGR every year, TGR every 6 months, and MCGR groups was 2, 4, and 12 per year, respectively. This was based on one postoperative visit for wound assessment and one preoperative visit for operation consent and imaging between each TGR distraction, and monthly outpatient distraction visits for the MCGR. Costs accrued for surgical complication were estimated from expert opinion on the average health resources utilized for managing individual complications. For superficial infection, the cost of 2-week oral antibiotics was considered. For deep infection, the costs incurred included one surgical debridement (involving anesthetist, theater cost, and personnel), 1 week hospitalization (one night at ICU), antibiotics (1 week intravenous antibiotics followed by 5-week oral antibiotics), and wound dressing. For implant pullout, the cost incurred included the cost of four screws (for revision of the upper anchors that were the common site of failure), imaging, operating theater, and hospital stays of 3 days (inclusive of one night at ICU). Table 2 outlines the unit cost for each service component associated with the use of growing rods in EOS patients. The consumption of health-care resources in this study was based on the authors’ experience in managing EOS patients. Undoubtedly, the number of items used and the cost per item would be subjected to individual center and region’s variability.
Unit cost ($HKD) for each service component associated with the use of growing rod in early onset scoliosis patients.

P+O: prosthetics and orthotics; MCGR: magnetically controlled growing rod; TGR: traditional growing rod; HKD: Hong Kong Dollar; O&T: orthopedics and traumatology; HKU: The University of Hong Kong.
Indirect costs
Indirect costs were valued based on a human capital approach theory. Lost productivity due to the caring for EOS patients was counted as parental work loss because these patients are by definition less than 18 years old for the entire simulation. Parents were assumed to take either half-day or full-day off from work, equivalent to approximately 4 hours or 8 hours spent accompanying the EOS patient, respectively. Surgical treatment was assumed to take one full day for preoperative assessment and one full-day postoperatively to accompany the patient before discharge, while outpatient visits for routine follow-up and distraction were assumed one half-day off from work. Monetary values were estimated with all working hours lost due to the treatment and their related transportation and gross hourly salary of the working population in Hong Kong. Gross hourly salary in Hong Kong working population aged from 15 to 65 was quoted from Census and Statistics Department of Hong Kong Government 27 and adjusted to year 2016.
Base-case and sensitivity analyses
To take time preferences into account, future direct and indirect costs were annually discounted at a rate of 3%, recommended by established guidelines 28 and frequently adopted in health economic evaluation applied to the setting of Hong Kong. 29 Accumulative costs for each strategy from both patient and institution perspective were calculated. TreeAge software Pro version 2016 (Treeage Software, Inc., Williamstown, MA) was used for development of decision tree and microsimulation analyses. As for base-case scenario, MCGR group was considered as the cost-saving or dominant strategy when compared to TGR group if accumulative costs for both groups 1 and 2 were less than those for groups 3 and 4.
Sensitivity analysis was performed to assess uncertainty and robustness of model parameters such as the discount rate and time horizon. Each model parameter had varied theoretical possible range, whereas the value of other parameters kept constant. Threshold of parameter value at which the accumulative costs of MCGR and TGR groups were the same was captured. Range of parameter values at which either MCGR or TGR group was cost saving was provided.
Results
The medical costs accrued in the initial surgery per patient in MCGR and TGR groups were HKD134652 and HKD27528, respectively. In this 8-year period, the total costs for the MCGR group repeating surgery every 2 years and every 3 years were HKD1635650 and HKD1383033, respectively, while the total costs for TGR group repeating surgery every 1 year and every 6 months were HKD1914117 and HKD2897967, respectively. From the health-care provider’s perspective, both strategies for TGR (groups 3 and 4) cost more than the two strategies for MCGR (groups 1 and 2) despite the incorporation of increased indirect costs with more frequent follow-up visits and time off from work for the MCGR. Among the strategies in the MCGR group, the cost for patients repeating surgery every 2 years is slightly more than those repeating surgery every 3 years, due to the discounting factor having a greater reduction in the costs in the later years which is contributed by the difference in final surgery dates. The MCGR groups with repeat surgery every 2 years and every 3 years underwent the final surgery at year 5 and 7, respectively. Hence, it involved less costs in the former group compared to the latter group. Table 3 summarizes the direct, indirect, and total costs for each strategy considered in this costing analysis. The accumulative costs for each strategy from institution and patient perspectives are listed in Table 4 .
Accumulative direct, indirect, and total costs for each growing rod strategy.

MCGR: magnetically controlled growing rod; TGR: traditional growing rod; HKD: Hong Kong Dollar.
Accumulative institution, patient, and total costs for each growing rod strategy.

MCGR: magnetically controlled growing rod; TGR: traditional growing rod; HKD: Hong Kong Dollar.
Sensitivity analysis was performed by varying the model parameters to verify the uncertainty and robustness of conclusions. However, no change in the conclusion was observed by unit costs of treatment and discount rate. Figure 2 shows the direct costs for each strategy at each year cycle. Although the accumulative costs of three groups (TGR repeating surgery every 1 year and the two MCGR groups) were approaching each other in the first 2 years after initial implantation and distractions, the accumulative cost per patient in the yearly TGR distraction group was higher than that of MCGR groups in year 2 and from year 4 onward. The accumulative cost per patient in MCGR with exchange every 2 years (HKD808120) was only HKD36k more than that of TGR repeating surgery every 1 year (HKD771497) in year 3 when the former group had implant exchange. The threshold for being cost saving for both MCGR groups was 4 years after initial surgery.
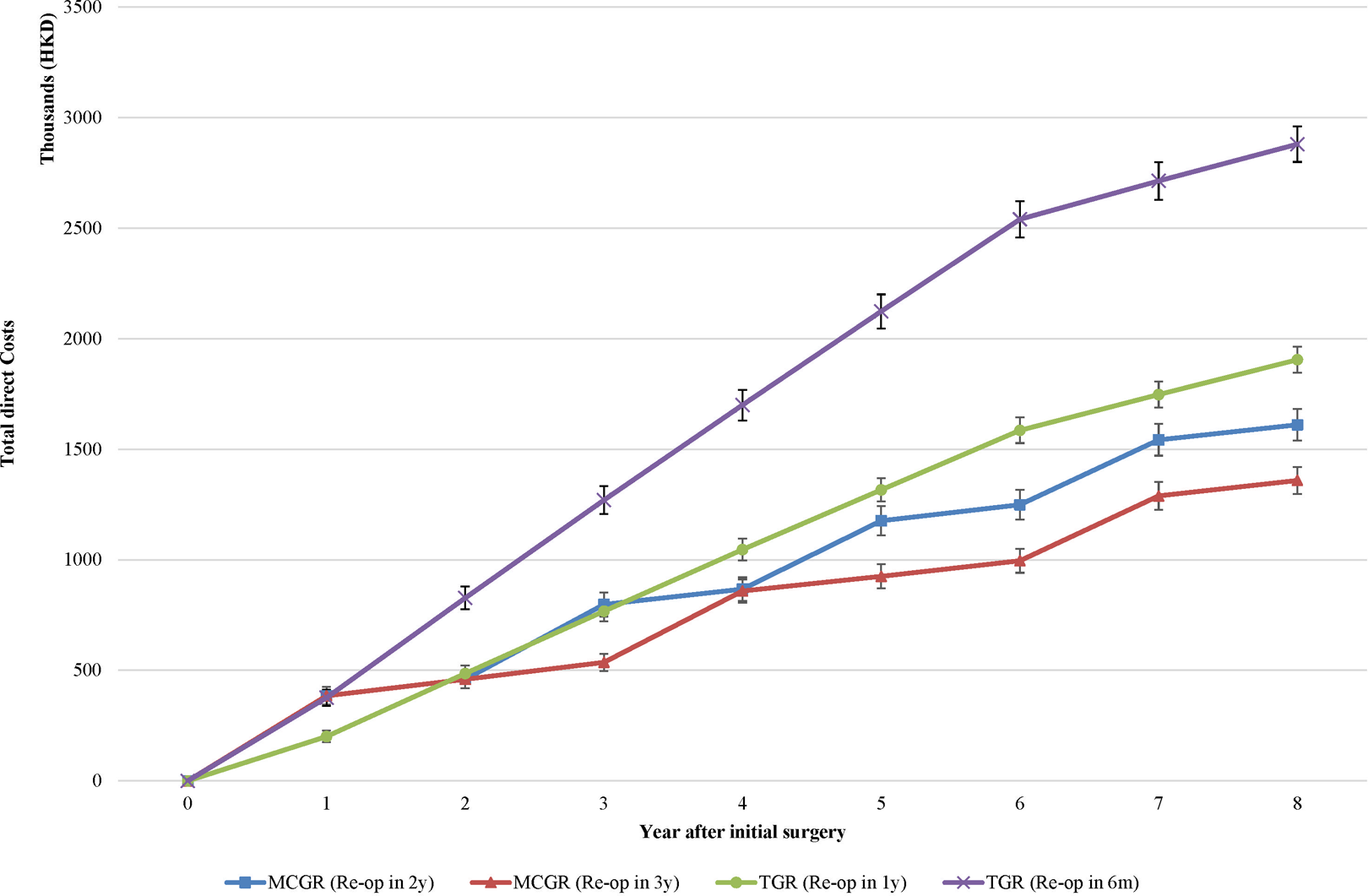
Direct costs for each strategy from year 1 to 8 after initial surgery in sensitivity analysis varying time horizon. Error bar refers to the mean ± 1 SD of direct costs by microsimulation.
Discussion
This cost-analysis study has significant implications on the future of EOS management. Despite the high cost of the MCGR device, the overall resource consumption is less than that of the TGR in this simulation but only if the entire duration of treatment spans more than 4 years. The cost of the MCGR dwarfs any individual consumable for TGR treatment but the cumulative cost of more frequent surgeries over the course of an 8-year treatment is overall costlier. This finding holds true even with more frequent exchanges of the MCGR (every 2 years). Furthermore, our findings are in line with the international literature related to cost comparison between MCGR and TGR groups. At the health technology assessment submitted to UK National Institute for Health and Care Excellence (NICE), 30 the MCGR generates cost savings of GBP12,077 per patient when compared to TGR over 6 years, while the MCGR results in cost saving of US$40,000 when compared to TGR over 5 years from the US payer’s perspective. 31
The health-care system in Hong Kong is a government-based public platform where the government covers all costs incurred at a flat rate. Hence, it is important for our model to analyze the costs to health care and patient separately. This is visibly different from the health-care systems represented in France, 22 United Kingdom, 24 and the United States. 23 It is also important to note that unlike Charroin et al. 22 and Rolton et al., 24 our model is based on an entire treatment plan for an EOS patient from initial surgery to skeletal maturity. In addition, with our longer experience with the MCGR device, this model is more realistic in terms of the timing of rod exchange and the costs incurred for each complication.
The cost saving threshold for both MCGR strategies compared to the TGR occurs at 4 years after initial surgery in our model. Beyond this point, each groups’ cumulative cost continue to increase at its own projection, with the TGR remaining higher than MCGR for the rest of the treatment period. This takes into account a consistently higher annual indirect cost of MCGR than TGR at all times. Hence, the cost-saving benefit of MCGR is sustainable and remains true in the long run as supported by this model. Also, it is worth noting that MCGR exchanged at every 3 years becomes the most cost-saving approach from year 3 onward, and it continues to be the least costing approach until the end of treatment. Upon further examination of the MCGR groups, the differentiation of costs between a 2-year and 3-year exchange strategy is most definite and is best illustrated 5 years into the patient’s treatment where the 2-year strategy overtakes a 3-year strategy in cost. This suggests that a substantial period of treatment with the MCGR is needed to be cost saving comparing to the TGR.
The two major cost items in EOS management as evidenced in this model are implant cost and intensive care stay. Regardless of possible interregional variations in the cost of the MCGR, the device is still an expensive cost unit and thus interval rod exchanges are a significant impact to the overall cost. The main reason for interval MCGR exchange is the maximum capacity of 4.8 cm distracted length allowed in the rod’s housing unit. There has been discussion of whether retraction of the distracted length and extending the construct length with an additional rod and rod connectors can avoid the purchase of a new MCGR. However, whether this affects the outcome of successful distraction or the complication rate is unknown and thus not a factor included in this cost model.
Despite the high cost of the MCGR, the costs of operative consumables and postoperative intensive care stay were higher for the TGR and thus the balance was maintained. Partly contributed by the similarly high complication rate of MCGR treatment, differences in cost between TGR and MCGR are not as apparent until the fourth year of treatment. Nevertheless, the reported MCGR complication rates used in our analysis are only based on limited evidence supported by case series. 25 These studies are limited in sample size and follow-up duration. Since the MCGR has only been in use for less than a decade, long-term follow-up studies are lacking, and we expect more refined complication rates to be available for future cost-analysis studies. Although larger scale studies have reported the complication rates of the TGR 2 (81/140: 58%) and MCGR 32 (11/26: 42.3%) separately, the estimates in these studies provide very indirect comparisons between the two techniques. Direct comparisons are preferred over unadjusted indirect comparisons with estimates from separate studies. 33,34 Hence, without a meta-analysis incorporating all direct and indirect comparisons, the current study by Teoh et al. 25 is the best available evidence for our purposes. Despite the high complication rates used for analysis, from the authors’ experience, the complication rate of the MCGR is no less than TGR over the entire course of the EOS treatment. Hence, having similar TGR and MCGR complication rate parameters for our model creation is a more realistic representation than the previous cost analyzes. 22 –24
MCGR aside, calculations of other implant costs were based on references provided by Medtronic® for the CD Horizon® Legacy™ instrumentation system. Although the cost is subject to variations between different countries of origin and users with different instrumentation strategies, this is the cheapest system available to us and can help limit the effect of implant variability on our model. Despite differences in accumulative costs with user variations, this system is used for both our TGR and MCGR strategies and thus should still be a viable cost comparison for others to use as a reference regardless of the health-care system.
One of the major interuser variations in the MCGR is the frequency of distractions that can cause a rift between the costs to the public health-care system and to the patients/family with each year of growing rod treatment. While our model suggests that the MCGR is cost saving compared to the TGR in terms of accumulative costs and monetary expenses with respect to the health-care system, a higher cost is incurred by the patient and family. The yearly patient costs with MCGR treatment may be almost 4 times the patient cost with TGR. This is likely caused by the increased indirect costs of time away from work for parents, clinic visits, and transportation. At the end of the 8-year treatment period, the accumulative institutional cost is reduced 1.9 times, whereas the patient cost increased by 3.1 times when comparing a 2-year MCGR exchange strategy with a 6-monthly TGR distraction strategy. Yet, this is based on the authors’ practice of performing outpatient distractions on a monthly basis. There are other users who perform distractions every 3-monthly, 4-monthly, or even 6-monthly. It is reasonable to expect this cost to be reduced with less distraction frequencies. However, our setting in Hong Kong allows for monthly distractions as patients need to travel only relatively short distances to the hospital for these distraction procedures. In other regions, such as those who include care for the rural population, the travel time may inhibit the availability of a monthly distraction service. It may also be possible to provide these patients with their own External Remote Controller (ERC) if high distraction frequency is considered.
As it is not feasible for these patients to travel far for frequent distractions, the effect of increased travel expenses will be countered by reduced distraction frequency. As such, we expect follow-up visits, time off from work, and transportation to be of minor impact on the overall cost. Furthermore, even with travel expenses controlled, the overall cost-saving results of the MCGR is unlikely to change. The only difference will be the point where the accumulative costs of MCGR and TGR intersect, which may occur earlier with reduced follow-up visits and time off work. This indicates that the frequency of distractions does not significantly influence the overall cost. Whether the cost-effectiveness is reduced with less distractions however is unknown and should be addressed in future work. This relationship is nevertheless only in monetary value, and it is important for clinicians to realize that the overall impact on patients cannot be weighed by a cost figure. Although more clinic visits are required for MCGR treatment, the risks of repeated surgeries can be minimized as well as the possible mental burden of repeated hospitalizations, surgery, and subsequent rehabilitation for these vulnerable patients.
Several limitations need to be reported in this study. Despite the robust cost analysis performed in the study, it may be subject to management variations in other countries and with different implantation systems and distraction methodology. It is also important to emphasize that our model is a government-based public health-care system that is different from insurance-based systems. However, the results generated from our model is a pure and unbiased unit for unit representation of the actual costs incurred separately by the health-care system and the patient. Moreover, it is based on a wide breadth of cost unit parameters with an appropriate complication rate comparison between the TGR and MCGR. Hence, it is applicable for referencing in other settings. The evidence regarding complication rates with the MCGR is scarce however and should be revisited in the future. Hence, we can only base our risk calculation on the limited available evidence and the authors’ experience. There may be some criticism over inclusion of MCGR fracture as a possible complication. A change in the welding procedure of the actuator from a pulsed laser to a continuous laser may have dealt with the weak points in the rod that led to these fractures. However, this change was only made in 2012 and further follow-up data are required to verify this. In addition, health services utilized in each year for both groups were not supported by real-life observational or clinical trial data. Nevertheless, most of the episodes reported are based upon the experience of the authors who have the longest experience of managing EOS patients with the MCGR, including subjects who have completed the entire length of treatment from implantation until maturity. The information regarding the time spent on treatment and transportation were also based on the experience of the authors. There are some concerns over the use of costing-analysis design, also named cost-minimization analysis, wherein the health benefits and effectiveness have not fully been accounted for in this model. Negative impacts due to surgical complications were accrued to direct costs but not transferred to the loss of health effectiveness in term of quality of life. Equal clinical efficacy between two groups was the key assumption of our model and widely adopted in other costing models because of the lack of good quality comparative evidence. 30
Conclusions
This simulation cost-analysis study suggests that the MCGR is cost saving compared to the TGR, and this is based on an 8-year treatment duration from the perspectives of health-care provider and society, bringing important health service implications to decision makers. Although the accumulative cost of the MCGR is more than the TGR within 1 year of implantation, the TGR is costlier at year 2 but its cost-saving potential is only observed from year 4 onward due to the increased frequency of surgery. Thus, the MCGR has a modest cost-saving benefit as compared to TGR in the management of EOS patients. Based on the available evidence and the authors’ long-term experience in managing EOS patients with MCGR, this model is a more accurate and detailed representation of the actual cost incurred with regard to the breadth of cost unit parameters, rod exchange timing, and complication costs. This model has helped to investigate the cost-saving benefit of MCGR and has taken into account the perspectives of the clinicians, parents, and patients and the health-care system as a whole. With improved knowledge of the expectations with long-term use of the MCGR with regard to complication and outcomes, better constructed cost-effectiveness studies can be performed.
Footnotes
Author contribution
Carlos King Ho Wong and Jason Pui Yin Cheung contributed equally to this study and are co-first authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
