Abstract
Study Design
Multicentre comparative analysis of explanted Spine Magnetically Controlled Growing Rods (MCGRs).
Objectives
MAGEC X, the latest commercially available generation, was recalled in 2020 due to the risk of post-implantation separation of an actuator end-cap component. Currently, the supply of all MAGEC rods was temporarily suspended in the UK and the EU. Objective of this study is to compare the performance of the MAGEC X MCGR to the earlier MAGEC 1.3 design iteration, by means of retrieval analysis.
Methods
Fifteen of both MAGEC X and MAGEC 1.3 rods were consecutively collected from five different hospitals following removal surgery and matched by time to removal. Clinical and implant data was collected for all MCGRs. Analysis comprised visual assessments of external damage, plain radiograph evaluations, force and elongation testing, MAGEC X end-cap torque testing and disassembly. Mann-Whitney U tests were used to statistically compare groups.
Results
Rod distraction reached in vivo was significantly higher in the MAGEC 1.3 (
Conclusion
MAGEC X showed similar performance results than the previous design iteration MAGEC 1.3. End-cap component loosening was observed, with no major consequences on the internal mechanism.
Introduction
Early Onset Scoliosis (EOS) is a spinal pathology defined as a curvature of the spine ≥10° in the frontal plane with onset before 10 years of age. 1 Several treatment options have been proposed, distraction-based systems being the most commonly used for severe cases. 1 Magnetically Controlled Growing Rods (MCGRs) are distraction-based systems that use an external magnetic distraction system aimed at avoiding repeated surgery for rod lengthening, otherwise necessary with other systems.1-5
The most commonly used MCGR is the MAGnetic Expansion Control rod (MAGEC), manufactured by NuVasive (Nuvasive Specialised Orthopaedics, San Diego, CA). The first prospective patient series using MAGEC rods was published in 2012.
6
and since then 7 design iterations have been commercially available, the latest (still implanted) being MAGEC 1.3, 2.0,2.1 and MAGEC X, first used in mid-2017. Some design modifications have been implemented in the latest design iteration (MAGEC X) compared to the MAGEC 1.3, end-cap component introduction being the most important one. While MAGEC 1.3 and previous design iterations used an O-ring seal component to isolate the internal mechanism from the surrounding tissues, MAGEC X features a threaded end-cap component with 2 O-rings (Figure 1). Different sealing design between MAGEC 1.3 and MAGEC X rods. On top, a photograph of retrieved implant. On the bottom, a schematic diagram of the cross section of the corresponding rod. A MAGEC 1.3 implants uses an O-ring seal to isolate the internal mechanism from the outside, while B MAGEC X adopts a threaded end-cap component with 2 O-ring seals, one between the threaded end-cap component and the housing tube and one between the telescopic rod and the end-cap component.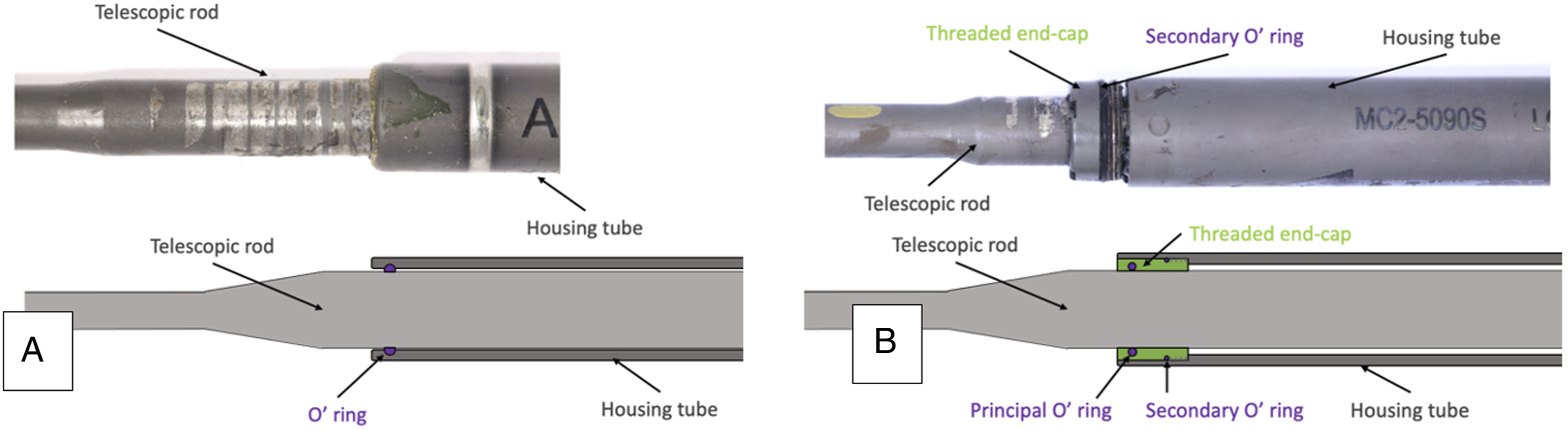
Recently, concerns regarding the risks associated with the use of MCGRs have been raised. The latest design iteration (MAGEC X) was recalled in 2020 following an Urgent Field Safety Notice (FSN) 7 describing a .5% probability of post-implantation separation of an actuator end cap component. On 1st April 2020, a second FSN was issued by the manufacturer, voluntarily suspending the supply of all MAGEC rods to the UK. The Medicines & Healthcare products Regulatory Agency (MHRA, UK) on the same day released an Medical Device Alert (MDA) advising surgeons not to implant MAGEC rods until further notice in the UK and Republic of Ireland. On 5th April 2021, NuVasive published a company statement communicating the temporary suspension of the CE mark in the European Union due to evidence gaps for the MAGEC system. 8 This statement followed a FSN voluntarily issued by the manufacturer in December 2020 informing healthcare providers that the notified body DQS Medizinprodukte GmbH (“DQS”) which delivered the CE marking, was undertaking an audit of the MAGEC System. 9 The FSN states that implant malfunctioning in vivo can manifest as locking pin breakage, O-ring seal failure, metal wear debris and failure of the rod to distract.
We aimed to better understand the clinical performance of the MAGEC X through the analysis of retrieved implants and a comparison with MAGEC 1.3 rods.
Materials and Methods
This study investigated all retrieved MAGEC X rods (n = 15) collected at our centre; these were removed from 10 patients after a median (range) time of 15.5 (1-34) months in situ. At our retrieval centre, we had a large number of consecutively collected MAGEC 1.3 rods and recruited the first 15 that matched the time to removal of the MAGEC X implants; 22.5 (1-29) months,
Figure 2 presents our study design. Flowchart showing the study design.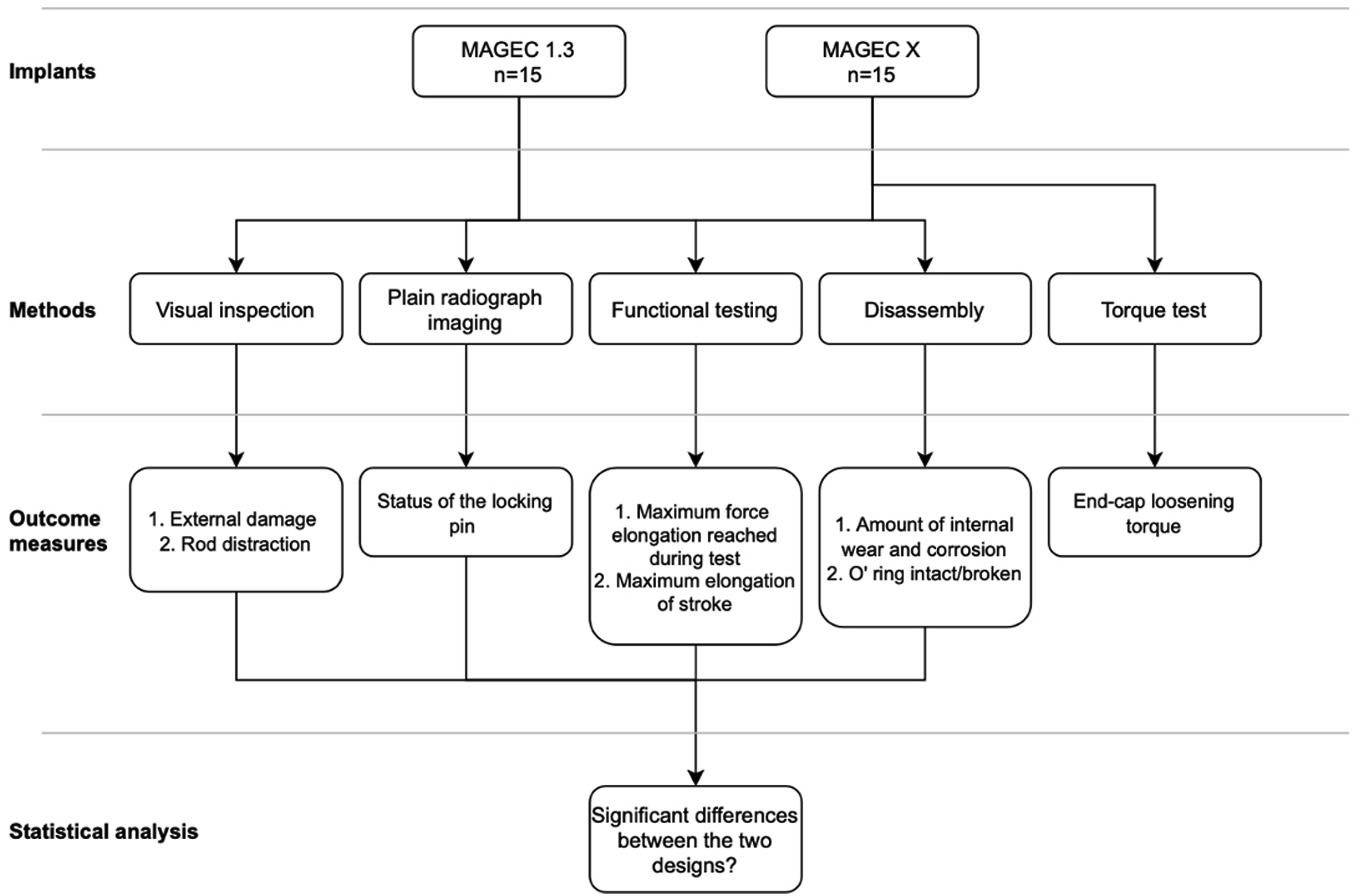
Clinical data (age at implantation surgery, gender, time to removal, reason for revision) and implant data (rod generation, length size, diameter size, configuration) were recorded for all MCGRs. Rod distraction was measured as the distance between the housing tube opening and the furthest growth mark from that point. Magnetically Controlled Growing Rods can be implanted in double or single rod configuration. Clinical data was presented and analysed relative to the patient (double rod constructs considered as one), while implant and retrieval data relative to the MCGR (double rod constructs analysed separately).
All patients provided informed consent for their implants and associated clinical data to be investigated at our implant centre. This research was approved by London-Riverside REC: Implant Study - 07/Q0401/25.
Visual Inspection
The condition of the implants was documented using a Canon 6D DSLR camera and a Canon 100 mm L lens, while total rod length and distracted portion length were also recorded (Figure 3). Microscopic analysis was also performed using a Keyence VHX-700F (Keyence Co., Japan) light microscope. (A) Division of the rod in sections for grading surface damage and lengths of the rod at retrieval. Rod distraction was measured as the distance between the housing tube opening and the furthest growth mark from that point; (B) Identification of quadrants for left and right rod.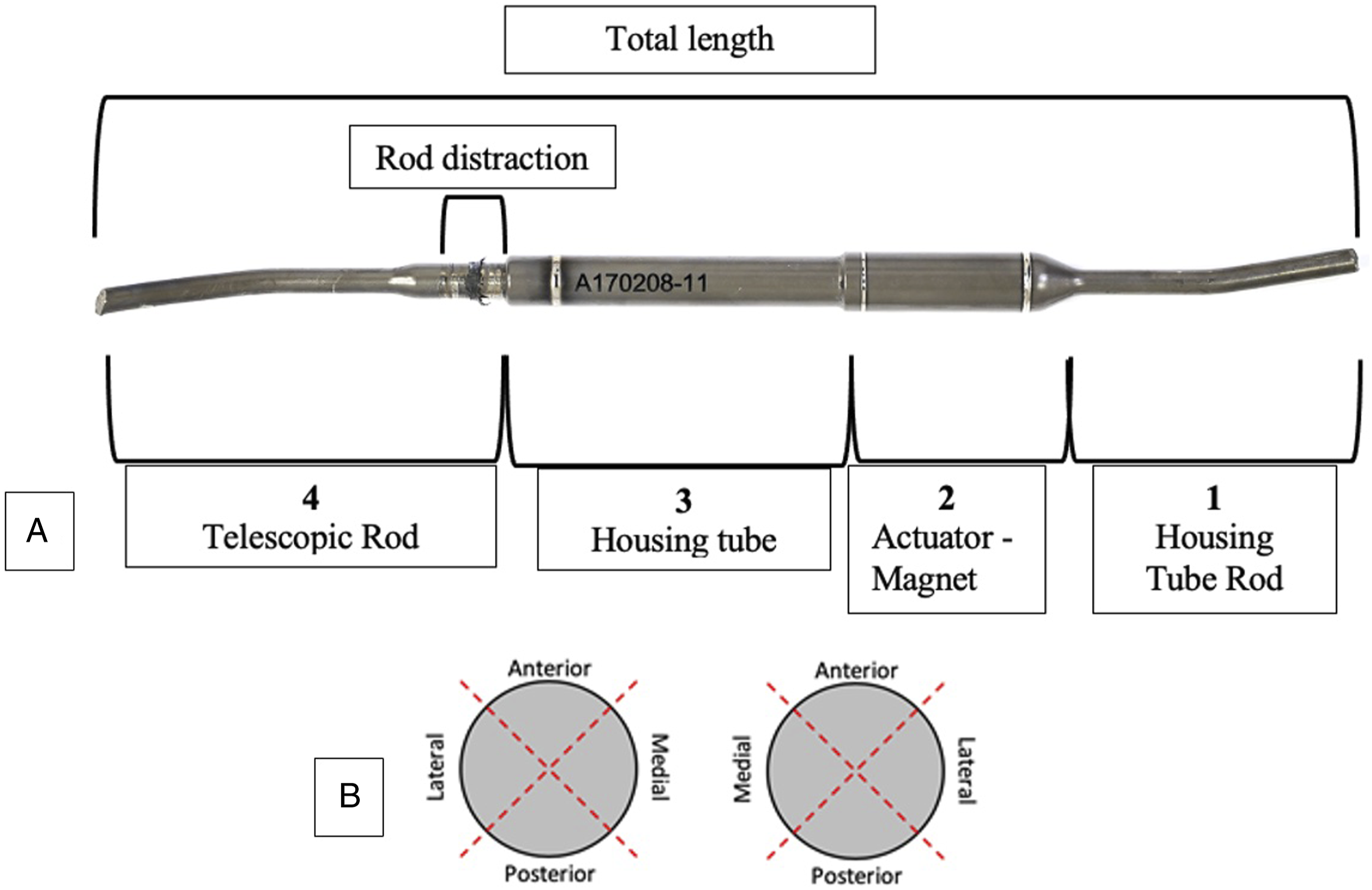
The presence and severity of external surface damage was assessed using a semiquantitative method, considering its type, area/magnitude and location. This involved grading wear, galling, corrosion, scratching and mechanical damage on a scale from 0 to 3, where: a. 0 = absent b. 1 = less than 25% of the area affected c. 2 = between 25% and 75% d. 3 = more than 75% of the area affected.
The rod was divided into 4 sections along its length, which included the housing tube rod, actuator magnet, housing tube and telescopic rod (Figure 3), and scores were assigned to each quadrant of these sections.
Plain Radiographs
Plain radiographs were captured of each retrieved rod in order to visualise the state of the internal mechanism and identify rods in which a fracture of the locking pin had occurred.
Imaging was performed using high energy X-ray scans (Samsung GC85 A, Samsung Electronics) in Computed Radiography (CR) mode, using a scintillator area detector.
Functional Testing
Magnetically Controlled Growing Rods can be detracted (elongated) or retracted (shortened) using a manual distractor or an External Remote Controller (ERC) that is used clinically to do so in vivo. A manual distractor was used to determine their elongation of stroke (difference between the maximum and minimum rod distraction lengths). Uniaxial force testing was then performed on rods that showed no evidence of pin fracture and could be manually retracted/detracted. This was achieved using a Chatillon DFE2-500 Digital Force Gauge, threaded adaptors and a spring block, all mounted on a force test fixture. The MCGRs were initially retracted to 10 mm of elongation (in order to avoid compromising the internal mechanism), placed through an ERC gap block and connected at either end to the spring block and the force gauge using adaptors. The gap block held the ERC on top of the magnetic portion of the MCGRs as they were being distracted (elongated), until 3 consecutive stalls were detected. The test duration, peak force reached and difference in length between the initial and final test configuration (elongation in test) were recorded.
MAGEC X end Cap Torque Test
A torque test was performed on the end-cap component of MAGEC X rods in order to evaluate the strength of this seal (experimental setup in Figure 4). This was performed using a STAHLWILLE 1/4 in a Square Drive Electronic Torque Wrench (1-20Nm). The peak torque required to achieve endcap loosening was recorded. Experimental setup for the loosening torque test. The rod was anchored to the worktable using a chuck clamp and a G-clamp. A custom-made connector was then used to grip the end-cap component and the torque wrench.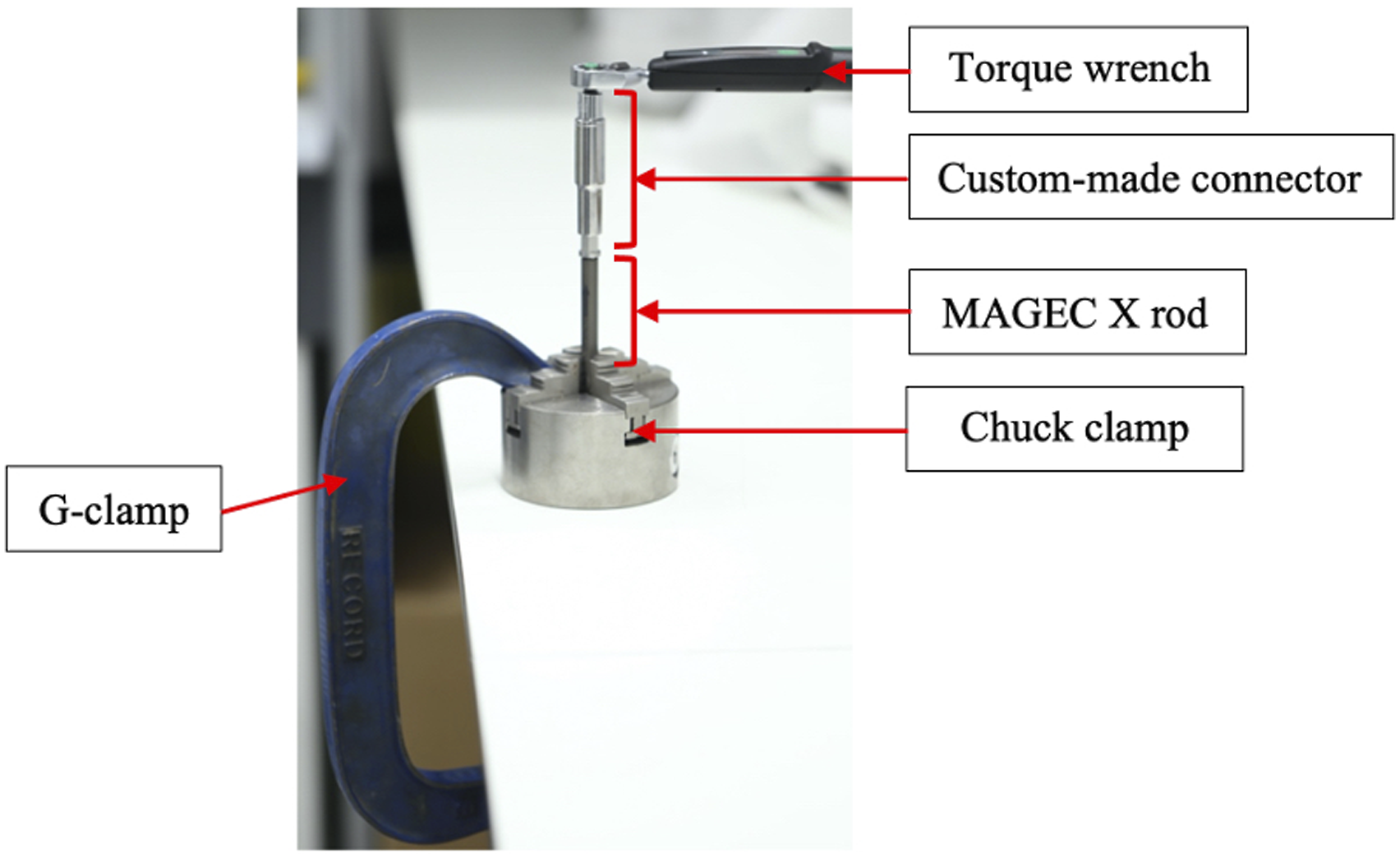
Disassembly
Firstly, MCGRs with evident pin fracture and/or not retractable were disassembled to study the mechanisms of failure leading to a non-functional implant. Rods were sectioned in correspondence to the laser welds, exposing the magnet-internal screw junction. Implants were then categorized as functional, with a pin fracture, or unable to lengthen due to a locked junction: internal screw and telescopic rod, telescopic rod and housing tube, seal between telescopic rod and housing tube.
Secondly, in order to retrieve the O-ring and evaluate its state, functional implants were also sectioned, enabling the evaluation of their internal state. This was done by means of visual assessment, scoring internal telescopic rod wear and corrosion (as described in the “visual assessment” section). State of the O-ring (principal and secondary O-ring in the case of MAGEC X implants) was recorded for all implants where it could be retrieved.
Statistical Analysis
Statistical analysis was performed using SPSS (IBM SPSS Statistics for Mac, Version 27.0). Statistical significance was considered for
Potential or Perceived Conflict of Interest
Two surgeon authors (ST and MS) use the devices investigated in this study in their clinical practice. The senior author (AH) receives institutional level funding from the manufacturer to independently collect and analyse the devices investigated in this study.
Results
Clinical and Implant Data
Clinical and implant data and results from the retrieval analysis. Numerical results are shown as medians (range). Clinical data was analysed using the patient specific data (rather than the rod specific data), and the implant data considered double rods as single entities. P-values are reported for the difference in medians between the two rod generations.
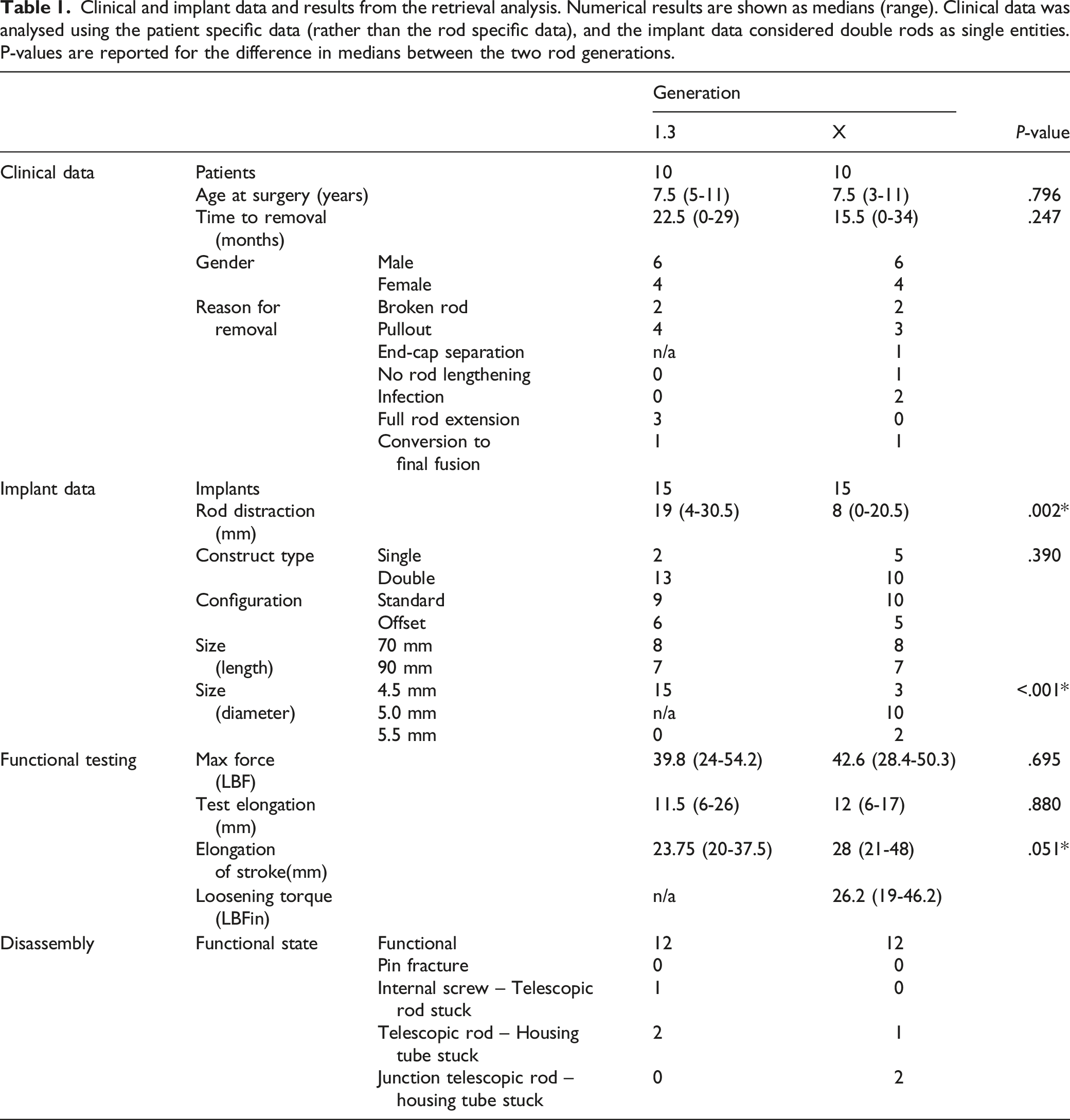
No statistically significant difference was found in the clinical and implant data between the 2 rod generations.
Visual Inspection
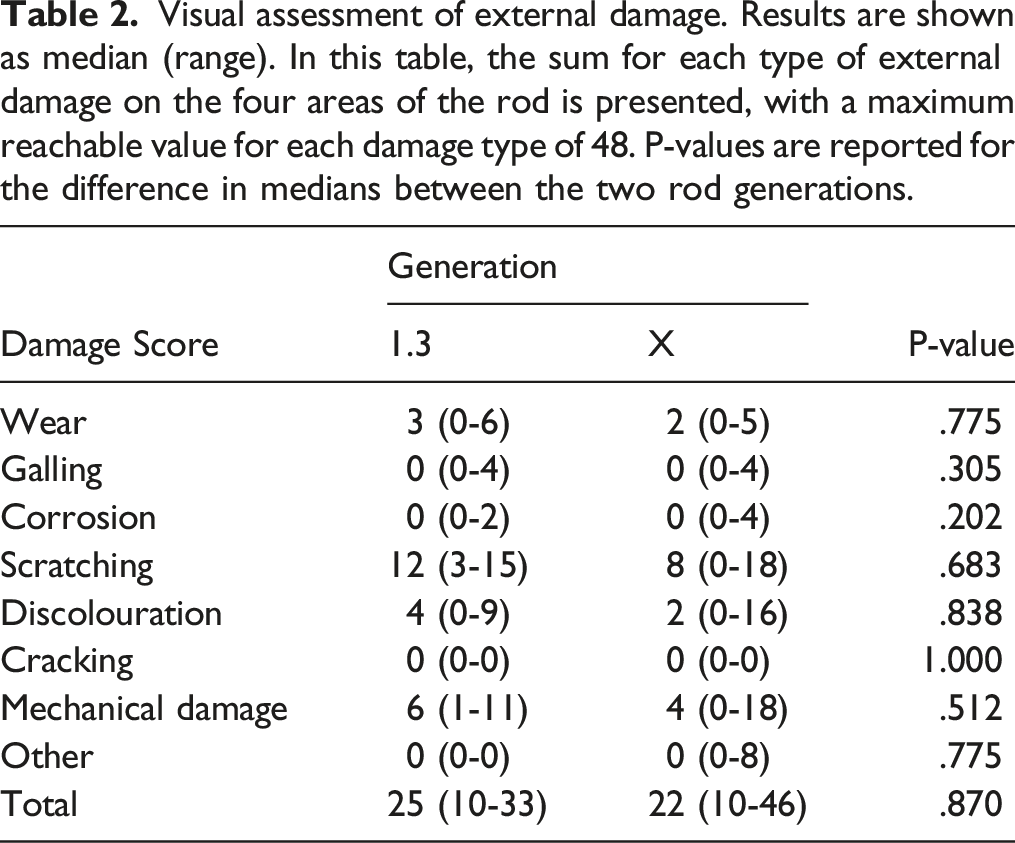
Visual assessment of external damage. Results are shown as median (range). In this table, the sum for each type of external damage on the four areas of the rod is presented, with a maximum reachable value for each damage type of 48. P-values are reported for the difference in medians between the two rod generations.
Plain Radiographs
No pin fracture was observed on plain radiographs of the retrieved implants, which was confirmed during disassembly.
Functional Testing
Twelve out of fifteen rods in each group were able to distract (i.e., functioning normally). The median maximum force generated by functional MCGRs was 42.6 (28.4-50.3) lbf for the MAGEC X, compared to 39.8 (24.0-54.2) lbf for the MAGEC 1.3,
MAGEC X End Cap Torque Test
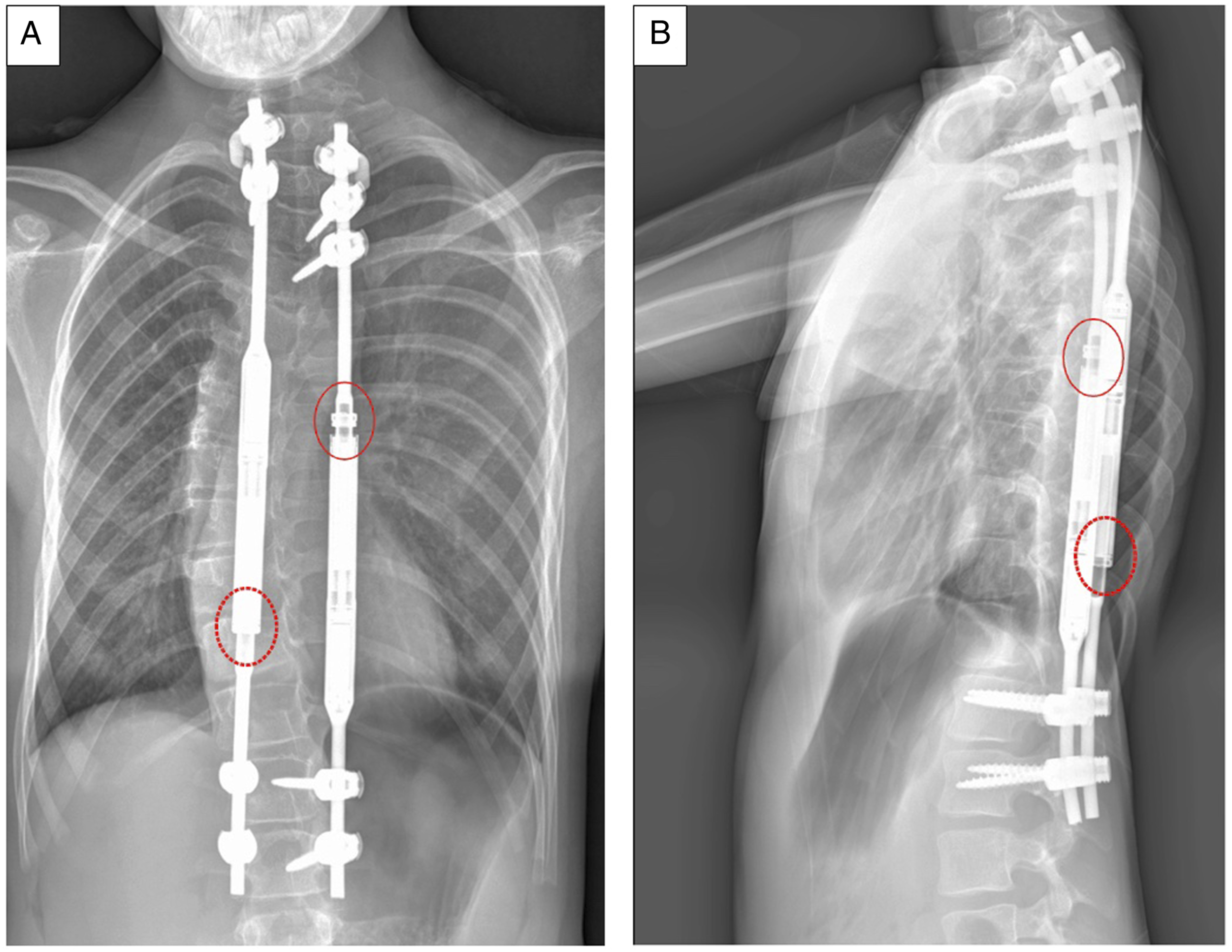
One implant was found to have a loose end cap component (Figure 5), which was also its clinical reason for removal (Figure 6). A torque test was successfully performed on 10/15 MAGEC X rods and their median peak loosening torque was 24.9 (19.0-46.2) LBFin. In 4 MAGEC X constructs the end-cap component could not be loosened with the torque wrench. Two of these implants were able to retract and detract, while the other 2 were unable to detract. End-cap component separation in one retrieved implant. Pre-removal planar radiograph at 1 year follow-up showing evident end-cap loosening. Circled in red: loosened end-cap component. Circled with dashed line: well-fixed end-cap component. A) Frontal view B) Lateral view.
Disassembly
Three implants from each group were unable to retract/detract. In the MAGEC X group, more implants were found to be locked at the junction between the housing tube and telescopic rod (2/15 rods) and one implant showed evident corrosion on the whole housing tube-telescopic rod area. The MAGEC X implant with evident end-cap component loosening did not show any evident sign of internal wear or corrosion.
The inability to generate relative motion between housing tube and telescopic rod (due to corrosion) was the main reason why MAGEC 1.3 were not distractible (2/15 implants), while in one case the housing tube and the internal screw could not be unscrewed.
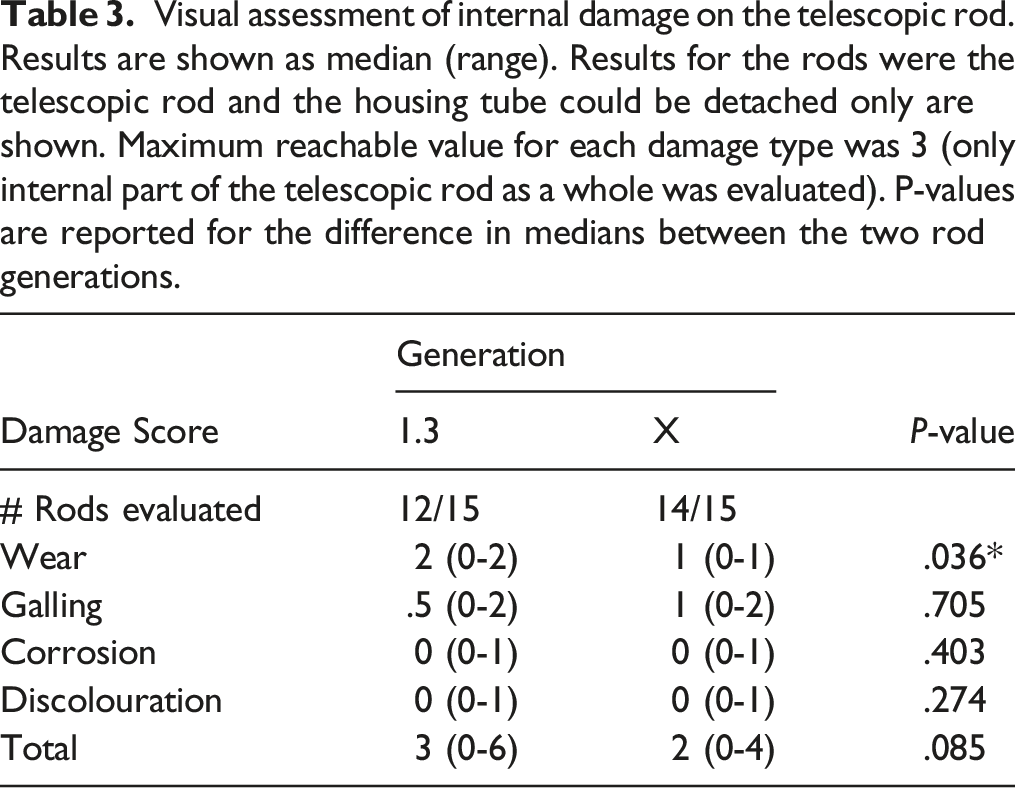
Visual assessment of internal damage on the telescopic rod. Results are shown as median (range). Results for the rods were the telescopic rod and the housing tube could be detached only are shown. Maximum reachable value for each damage type was 3 (only internal part of the telescopic rod as a whole was evaluated). P-values are reported for the difference in medians between the two rod generations.
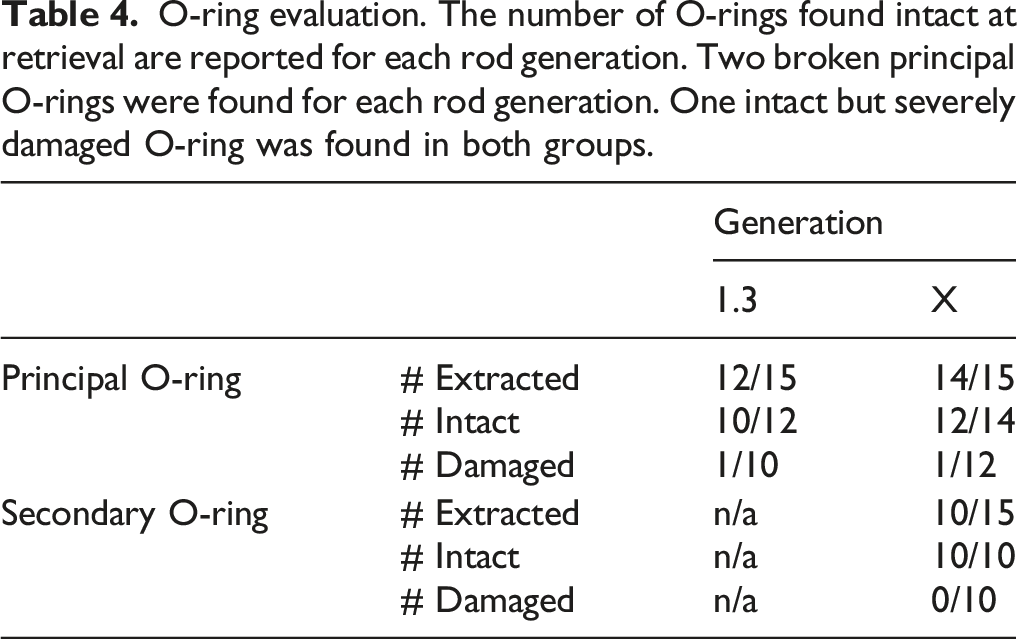
O-ring evaluation. The number of O-rings found intact at retrieval are reported for each rod generation. Two broken principal O-rings were found for each rod generation. One intact but severely damaged O-ring was found in both groups.
Discussion
This is the first study to report retrieval findings of the MAGEC X implant. We found MAGEC X performance similar to the one of MAGEC 1.3. We also found some variability in the end-cap component loosening torque, with one implant being explanted because of evident end-cap component loosening in vivo. Nevertheless, no evidence of lower performance of the MAGEC X rod design was found in this study.
In order to perform an equitable comparison between the 2 rod design iterations, only MAGEC 1.3 implants with a time to removal less than 34 months (maximum time to removal of a MAGEC X implant recorded at our retrieval centre) were included. Time to removal has been found to significantly influence retrieval findings, including wear rate, 10 force generated by the implant, 11 pin fracture, 12 and implant functionality. 13 Excluding implants with longer follow-up led to an overall relatively high implant-related reason for removal (in 13/20 patients).
Implant disassembly and mechanical testing can induce pin fracture or damage the internal components, thus an initial visual/radiological assessment was performed. Locking pin fracture, in particular, has been a subject of debate since the Nuvasive MAGEC Rod Urgent FSN, released on June 25, 2019. 14 All implants included in this study were manufactured after that date, while no pin fracture was observed.
Functional testing results for the 2 rod generations were comparable with the standard stated by the manufacturer for functional rods (42 lbf). 11 The better performance in the elongation of stroke test, combined with similar results in maximum force produced by the magnet, suggests that MAGEC X housing tube and telescopic rod have a higher relative range of movement at retrieval compared to MAGEC 1.3, with the same force produced by the magnetic mechanism.
End-cap component loosening was the reason for MAGEC X implants to be recalled by the manufacturer in February 2020. This particular design feature was not present in previous design iterations. To our knowledge, this study is the first to assess end-cap component loosening on retrieved MAGEC X rods and provide numerical values of torque. In the implant with evident end-cap loosening, any evidence of internal wear or corrosion at disassembly was similar to those without a loose end-cap, and both O-rings were intact and in good state. In this particular example, a loosening of the end-cap did not appear to increase any internal damage to the rod, compared to those with a fixed end-cap. This single data point however cannot be used to draw conclusions about the impact of this type of failure on damage to the internal components of the implant. We acknowledge this low sample size as a limitation of the study. Due to the nature of the testing, we were unable to perform repeatability tests of the loosening torque, which we acknowledge as a limitation; all testing was however performed by a single examiner experienced in retrieval analysis.
It was previously suggested that the relative motion between the telescopic rod and the housing tube, combined with the loads exerted on the implant, might cause body fluids to enter the internal mechanism provoking corrosion and wear debris formation. 15 The resulting increase in friction between the telescopic rod and the housing tube components was thought to impair the implant’s ability to distract and result in an increase in mechanical wear. This mechanism was supported by our retrieval findings at disassembly with evidence of black debris observed within the rods that was consistent with those reported in a previous study. 15
Visual assessment of the internal mechanism of functional and non-functional rods revealed a slightly higher wear rate in the MAGEC 1.3 group (
Overall, 12/15 implants (80%) were found to be still functional at retrieval in both the MAGEC X and MAGEC 1.3 cohorts. We suggest that further investigations on the surgical/patient factors affecting the performance of MCGRs are needed in order to explain the relatively high complication and revision rate of this system.
Conclusion
This is one of the first studies to present comparative retrieval findings of the latest generation of MCGRs (MAGEC X). We found no evidence of pin fracture in MAGEC X rods nor in the control group, MAGEC 1.3. There was comparatively less evidence of internal damage in the MAGEC X designs, despite a variability in the measured loosening torque of the end-cap component. One MAGEC X was revised due to evident end-cap component loosening, however the implant was found to be functional at mechanical testing and there was no greater sign of internal damage at disassembly.
Footnotes
Authors' Contributions
All authors contributeed to the Study Design/Data Acquisition/Analysis, Involved in Draft of Work/Revision, Approved Version to be Published, and Agree to be Accountable for all Aspects of the Work.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Authors HH and AH receive institutional support from NuVasive to independently collect and analyse retrieved MAGEC rods. Authors MS, ST and EB use MAGEC rods in their clinical practice.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Authors HH and AH receive institutional support from NuVasive to independently collect and analyse retrieved MAGEC rod. There was no funding directly for this study.
Ethics Approval
This research was approved by London-Riverside REC: Implant Study - 07/Q0401/25.
Consent to Participate
All patients provided informed consent for their implants and associated clinical data to be investigated at our implant centre.
Consent for Publication
All patients provided informed consent for their implants and associated clinical data to be investigated at our implant centre.
