Abstract
Multiple sclerosis is the most frequent cause of non-traumatic neurologic disability in young adults with over 3400 newly diagnosed cases annually in Italy. The research objective was to assess the cost-effectiveness of alemtuzumab in comparison with other disease-modifying therapies in the management of relapsing-remitting multiple sclerosis, from a payer perspective in Italy. A Markov model was created to assess cost-effectiveness of alemtuzumab in comparison with subcutaneous IFN β-1a, natalizumab and fingolimod. Treatment effects were derived from a network meta-analysis. Economic input included cost of therapies, their administration and follow-up, cost of adverse events and cost of relapse. Data on health care resource utilization and their costs were retrieved from published sources. Cost-effectiveness was measured as incremental cost (€, 2017) per quality-adjusted life year while the robustness of the results was demonstrated in sensitivity analyses. Over a lifetime horizon, alemtuzumab yielded more quality-adjusted life years and less costs compared to the other disease-modifying therapies in all base-case analyses. Treatment with alemtuzumab yielded an incremental quality-adjusted life years of 1.62, 1.03 and 1.36 with savings of €4312, €81,562 and €54,067 versus IFN β-1a, fingolimod and natalizumab, respectively. Results on the multiple cost-effectiveness acceptability curve showed alemtuzumab carries the highest likelihood of being below the accepted willingness-to-pay threshold (€40,000) compared to IFN β-1a, natalizumab and fingolimod. Based on the current analysis, alemtuzumab is likely to be cost-effective versus IFN β-1a, natalizumab and fingolimod in the treatment of relapsing-remitting multiple sclerosis patients in Italy.
Introduction
Multiple sclerosis (MS) is a chronic immune-mediated neurologic disease characterized by inflammation, demyelination and axon degeneration. 1 MS is the most frequent cause of non-traumatic neurologic disability in young adults (primary onset between 20 and 40 years of age). MS has a heterogeneous clinical presentation and it is classified into four phenotypes based on the severity and frequency of relapse and disease evolution (i.e. progression): relapse-remitting MS (RRMS), primary progressive MS (PPMS), secondary progressive MS (SPMS) and progressive-relapsing MS (PRMS). Approximately 85% of patients present with RRMS at disease onset, and around two-thirds of them progress to SPMS. 1
The MS population affects approximately 2.8 million individuals worldwide, 2 with a reported female-to-male ratio of 2:1. 3 The estimated median prevalence in Europe is 100–190/100,000, affecting around 600,000 people. 4 In Italy, there are approximately 114,000 people affected by MS with over 3400 new diagnoses each year, based on 2017 data from the Italian Association of Multiple Sclerosis (AISM). 4
MS poses a significant socioeconomic burden associated with the cost of treatment, detriment in patients’ work productivity and increased need for supportive care in the advanced stages of the disease. 5 In the early years following diagnosis, the cost of pharmaceuticals constitutes the largest share of the total costs of MS (29%–82% of all costs), while in later years, costs associated with loss of productivity and informal care (17%–67%) comprise the largest share of the expenditure. 6 Total annual costs of MS in Europe are estimated to reach €14.6 billion. 5 Several researchers have investigated costs associated with MS in Italy.7–10 The average cost of diagnosis is estimated to be €1236/patient and is due to hospital and day hospital admissions, and instrumental and laboratory investigations in the ambulatory setting. 9 In patients with mild severity (Expanded Disability Status Scale (EDSS) 0–3.5), direct medical and rehabilitation costs were estimated at €9949/year/patient; these costs were derived through a cost of illness study by Berto et al. based on data from 8326 Italian MS patients. 9 The pan-European study on Treatment Experience, Burden and Unmet Needs (TRIBUNE) found the mean per patient cost of MS management increases substantially to €22,461 (SD = €13,799) per patient/year when productivity losses associated with sick leave and early retirement are considered, as well as patient out-of-pocket expenditures.7,11 Mean annual costs were reported to be in the range of €8322–€41,327 (±€31,263)/patient in patients with moderate MS severity (EDSS 4–6.5) and from €6485–€39,592 (±€37,785)/patient in patients with severe disease (EDSS 7–9.5).7,9
As there is no current curative therapy for MS, disease management is focused on reducing risk of relapses and disability progression. 12 Disease-modifying therapies (DMTs) may reduce the development of central nervous system (CNS) lesions as well as frequency of exacerbations, and hence delay physical and cognitive deterioration. 1 The first DMTs were approved in the 1990s (injectable therapies with IFN β). 12 In recent years, a new generation of therapies have become available with a higher efficacy profile and a favourable impact on the patient’s Quality of Life (QoL).
Alemtuzumab is a humanized monoclonal antibody licenced for treatment of adult patients with RRMS with active disease confirmed by clinical or imaging examination. 13 A sustained treatment effect was demonstrated during clinical investigation and in the pivotal phase 3 randomized, dose-blinded, active-comparator trials in more than 1000 RRMS patients (CARE-MS I and CARE-MS II).14,15 Recent findings from the CARE-MS extension programme (median follow-up of 6 years) indicated durable efficacy (magnetic resonance imaging (MRI) outcomes, reduction in brain volume loss and improvement in preexisting disability) in the absence of continuous treatment beyond the initial two treatment courses.16–18 During 6 years of follow-up, the majority of patients did not receive additional alemtuzumab courses and their clinical and MRI disease activity remained low. 19
At the time of writing, alemtuzumab, with its long-term biological effect, is the only treatment reimbursed in Italy for active RRMS disease with a sustained treatment effect and convenient administration: only two initial treatment courses, with up to two additional treatment courses, if needed (second treatment course to be administered 12 months after the first treatment course; third or fourth course, if needed, to be administered no less than 12 months after the prior treatment course). 13 Alemtuzumab is currently licenced for adult patients with RRMS with active disease defined by clinical or imaging features. 20 Alemtuzumab is recommended in the management of RRMS patients by health-technology assessment (HTA) and decision-making bodies across several jurisdictions.21–23 In Italy, alemtuzumab, like fingolimod and natalizumab, is reimbursed in24–26
RRMS patients with active disease despite prior treatment course with at least one disease-modifying agent therapy
RRMS patients with rapid evolving severe disease 26
Of note, the EMA (European Medicines Agency) has recently recommended the immediate suspension and recall of the MS therapy daclizumab beta following 12 reports of serious inflammatory brain disorders worldwide, including encephalitis and meningoencephalitis. For this reason, daclizumab was not included in this analysis. 27
The aim of this research was to assess the health and economic outcomes associated with alemtuzumab therapy in comparison with the active comparator of the clinical programme CARE-MS (IFN β-1a 44 µg) and the other DMTs currently reimbursed in Italy in the same RRMS patient population as alemtuzumab (natalizumab and fingolimod).
Methods
A Markov model was developed to assess long-term clinical outcomes and costs of RRMS management associated with alemtuzumab in comparison with IFN β-1a 44 µg, natalizumab 300 mg and fingolimod 0.5 mg. Health effects were expressed as quality-adjusted life years (QALYs), while costs were expressed in Euro (€, 2017). Cost-effectiveness analysis and use of QALYs were selected as the most appropriate to assess outcomes of a chronic condition associated with reduced life expectancy and elevated morbidity, such as RRMS. The model structure was based on the previously published School of Health and Related Research (ScHARR) model, developed to evaluate cost-effectiveness of four DMTs in patients with RRMS and SPMS. 28 Model health-states were based on the Kurtzke 29 EDSS, 0–9 scale (Part I: Supplementary Appendix). EDSS 10 (MS-related death) was not accounted for as an individual health-state and mortality was captured separately, to allow for an increasing risk of mortality by age. Given the chronic nature of the disease, the model followed yearly cycles and evaluated effects and costs over the lifetime horizon (i.e. 50 years). The cohort of RRMS patients was distributed across the health-states (EDSS 0–9) at model initiation, allowing patients to progress/improve (transition to a health-state with a higher/lower EDSS score, or progress to SPMS), remain in the same health-state or die. Model schematic is shown in Figure S1 (Supplementary Appendix).
Italian evidence was used wherever possible in order to appropriately reflect treatment patterns in the Italian context. Inputs related to disease, treatment and adverse event (AE) management were aligned with local data and validated with clinical expert judgement. Model assumptions were consistent with those of the cost-effectiveness model published within the UK HTA authority that resulted in a positive recommendation by National Institute for Health and Care Excellence (NICE). 23 Costs and effects were discounted at an annual 3.5% rate, in line with the UK NICE23,30 recommendations and hence, alemtuzumab cost-effectiveness model previously submitted to the UK HTA authority. This approach was deemed appropriate as it already had precedents in published Italian literature.31,32 The analysis was performed from the national payer perspective (SSN, Servizio Sanitario Nazionale) in Italy.
Model population
The modelled patient population female to male ratio (1.9), and mean age of the cohort at the start of the model (34 years) were similar to the pooled alemtuzumab clinical trials cohorts (CARE-MS I and CARE-MS II). The CARE-MS I and II cohorts were deemed comparable to those in pivotal studies of comparator treatments, in terms of share of RRMS patients in the cohort (⩾80%), female to male ratio, mean age and mean EDSS score at baseline (Table S2, Supplementary Appendix).
Health effects
Data on natural disease history provided information on how MS patients progress in disease severity in the absence of treatment. These data were derived from the two large MS-population databases: the British Columbia Multiple Sclerosis (BCMS) 33 database and the London Ontario dataset (observational registry with longitudinal data collection on MS patients during 1972–2000 period) for the EDSS states where transitions were not available (EDSS 7–9). The BCMS database covers >80% of the MS population in British Columbia and includes disability worsening data, recorded at the individual patient-level for over 28 years of prospective follow-up. Use of BCMS data was deemed appropriate as it was consistent with NICE considerations on modelling of MS natural history. 34 Natural history of RRMS was captured as a probability of being in any of the defined health-states of the Markov model [EDSS scores] at a given point in time (Table S3, Supplementary Appendix, provides matrices for transitions based on MS natural history).
Patients entered the model on active treatment with a DMT and moved on to best supportive care (BSC) after DMT withdrawal. The cohort of RRMS patients was distributed across EDSS health-states, based on the distribution of the pooled alemtuzumab cohorts in the CARE-MS I and CARE-MS II trials at baseline (Table 1).
Overview of the EDSS health-states (distribution of patients at baseline, mean utility per health-state and mean cost of MS per health-state). a
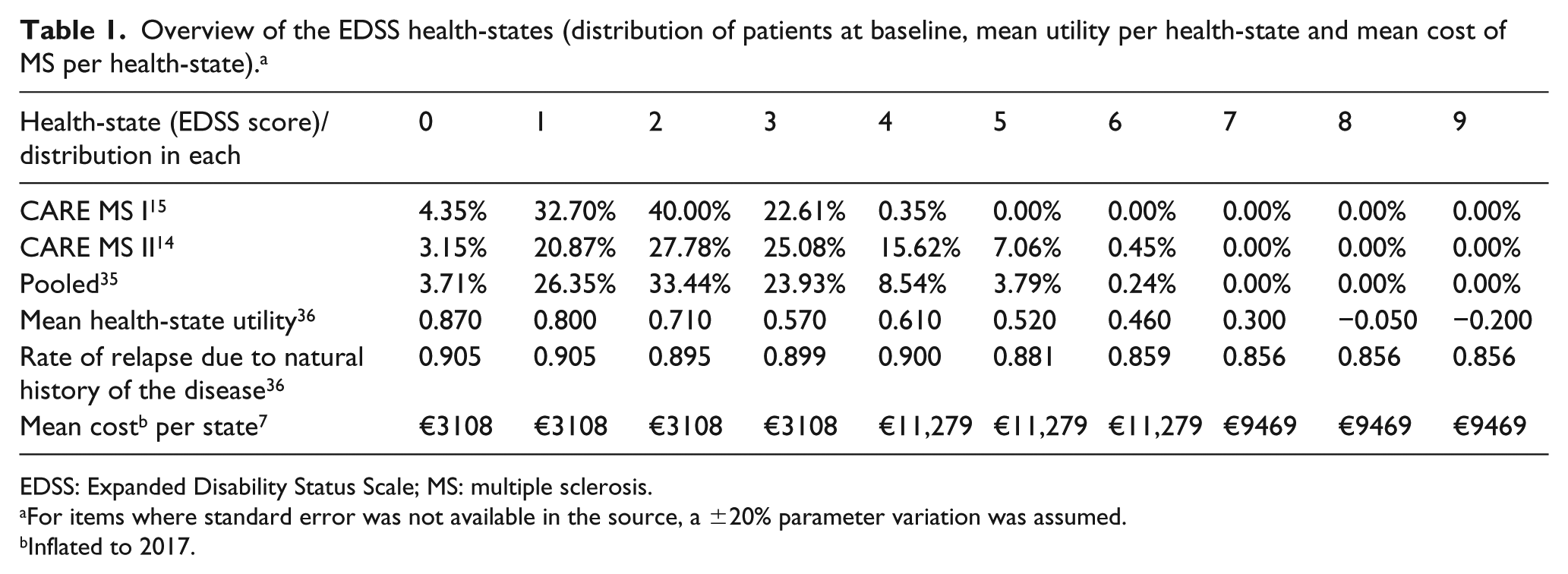
EDSS: Expanded Disability Status Scale; MS: multiple sclerosis.
For items where standard error was not available in the source, a ±20% parameter variation was assumed.
Inflated to 2017.
Disease progression was simulated using three key parameters: disability progression, incidence of relapse and mortality:
Disability progression: Table S3, Supplementary Appendix, shows transition matrices based on the natural history of RRMS. Progression to SPMS was not influenced by pharmacological treatment based on information from the London Ontario database (data from BCMS were not available; Table S4, Supplementary Appendix). Treatment effect on disability progression was derived from the network meta-analysis (NMA). 37 An overview of hazard ratios (HRs) for respective treatments versus placebo is provided in Table 2.
Incidence of relapse: The rate of relapses modelled in the economic analysis was EDSS state-dependant. Due to the lack of relapse data from the London Ontario or alternative registries, a literature search was conducted. Relapse rates were modelled based on the information from Held et al., 38 as this study was based on a larger sample size and offered more recent evidence in comparison to the other reviewed sources. The relationship between relapse rate and time since diagnosis was informed from the regression analysis from Patzold et al. 39 Relative risk of relapse versus placebo for each treatment was derived from the NMA, as reported in Table 2.
Mortality: Mortality was not impacted by treatment. It was associated with population age and sex based on data from the Italian National Institute of Statistics (ISTAT). It was adjusted by mortality risk associated with increased EDSS scores 40 (Table S5, Supplementary Appendix).
Efficacy parameters based on the NMA.38.
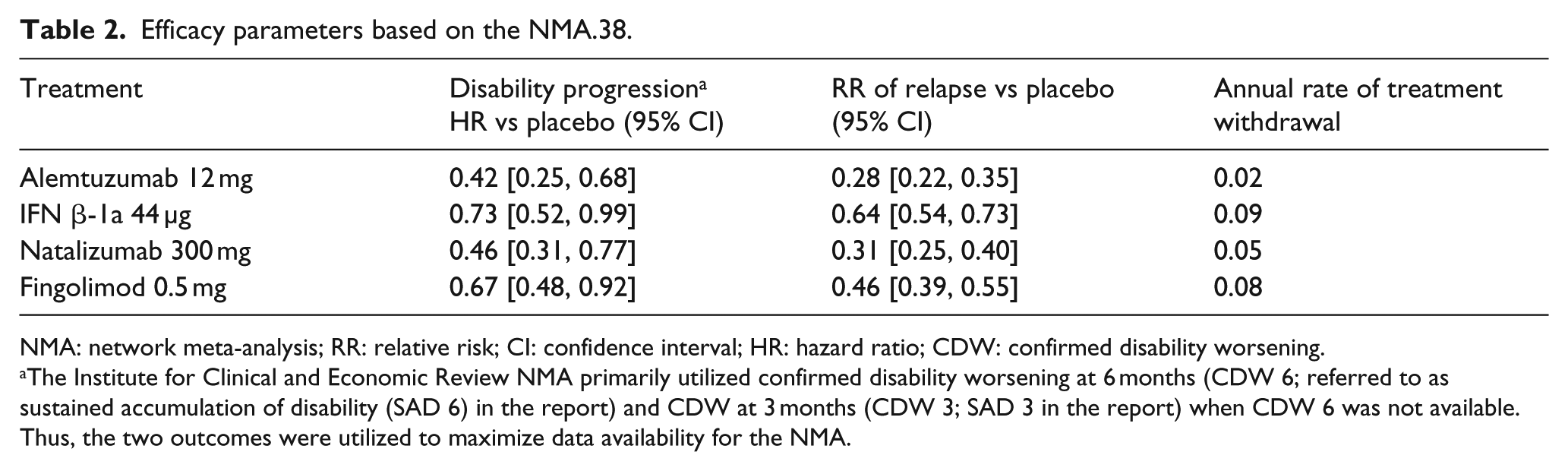
NMA: network meta-analysis; RR: relative risk; CI: confidence interval; HR: hazard ratio; CDW: confirmed disability worsening.
The Institute for Clinical and Economic Review NMA primarily utilized confirmed disability worsening at 6 months (CDW 6; referred to as sustained accumulation of disability (SAD 6) in the report) and CDW at 3 months (CDW 3; SAD 3 in the report) when CDW 6 was not available. Thus, the two outcomes were utilized to maximize data availability for the NMA.
Transition probabilities were contingent on patients’ EDSS score and constant over time. At any time, patients experienced a fixed rate of progression or relapse depend-ing only on their current health-state (i.e. EDSS score). Treatment effect on natural disease history was captured as a reduction in disability worsening and reduction in the frequency of relapse. With no head-to-head data for comparator therapies available, evidence from a NMA conducted by the Institute for Clinical and Economic Review on DMTs for RRMS 37 was utilized to inform comparative clinical effectiveness for the model (Part IV, Supplementary Appendix, provides details on the NMA).
The annual rate of treatment withdrawal was derived from the NMA (Table 2). Due to the sustained effect of alemtuzumab long after administration, the impact of withdrawing alemtuzumab is assumed to be nil. 41 This assumption was in line with the cost-effectiveness analysis from the NICE technology appraisal of alemtuzumab [TA312]. 23
AEs
This cost-effectiveness model included AE with at least 4% difference between intervention and control treatment (comparator) and those included in the prior HTAs of DMTs in RRMS population. Table 3 provides overview of the frequency and assumed duration of the AE included in the model. Furthermore, the model includes the frequency of treatment effect on relapse severity as reduction of AE related to diseases progression. This was accounted for as relative treatment effect on proportion of patients with a relapse which leads to hospitalization (Table 4).
AEs associated with DMT in the model: frequency (in year 1 and subsequent years post treatment initiation), associated disutility and cost per event. a
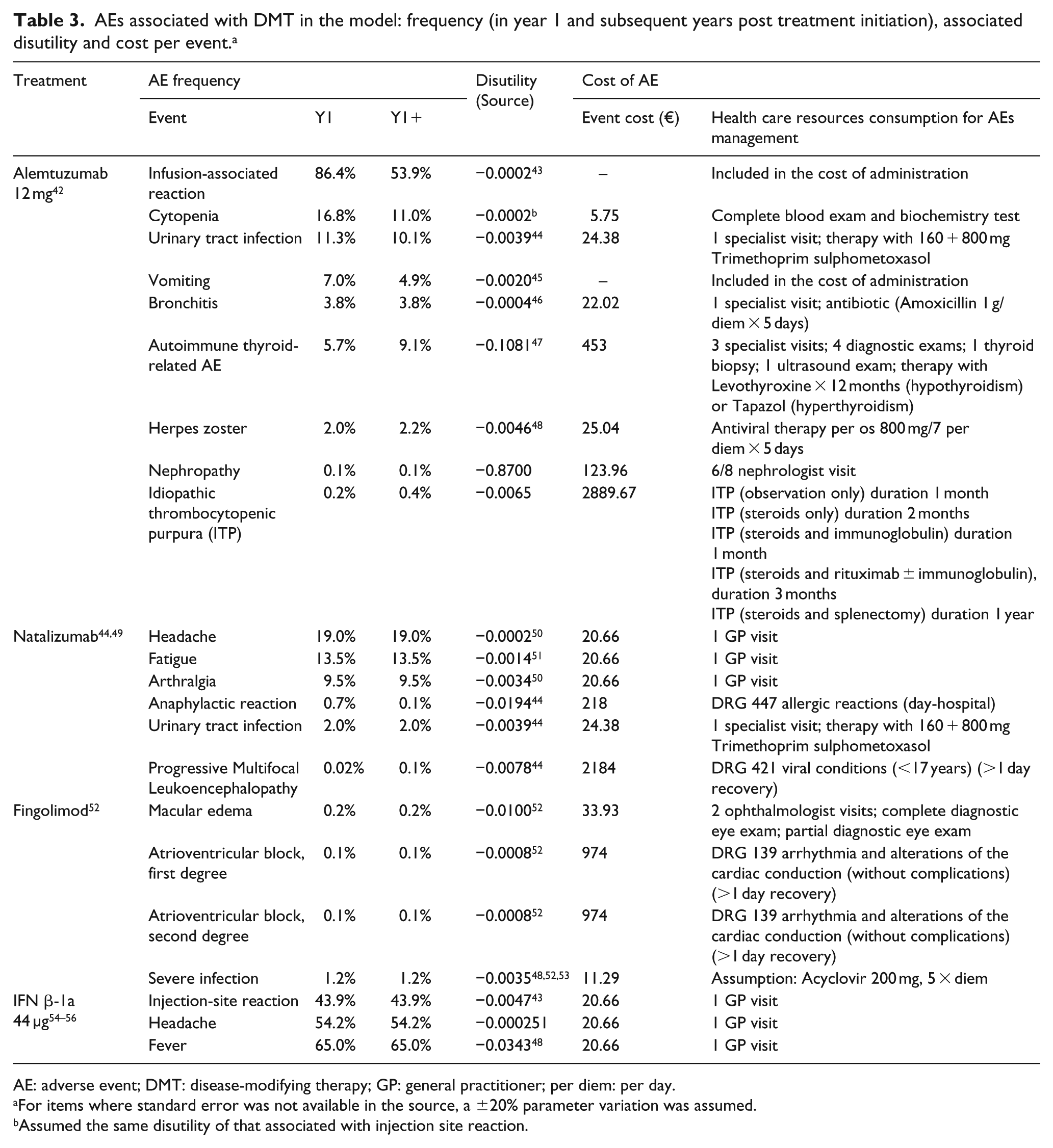
AE: adverse event; DMT: disease-modifying therapy; GP: general practitioner; per diem: per day.
For items where standard error was not available in the source, a ±20% parameter variation was assumed.
Assumed the same disutility of that associated with injection site reaction.
Relative treatment effect on proportion of patients with a relapse which leads to hospitalization. a
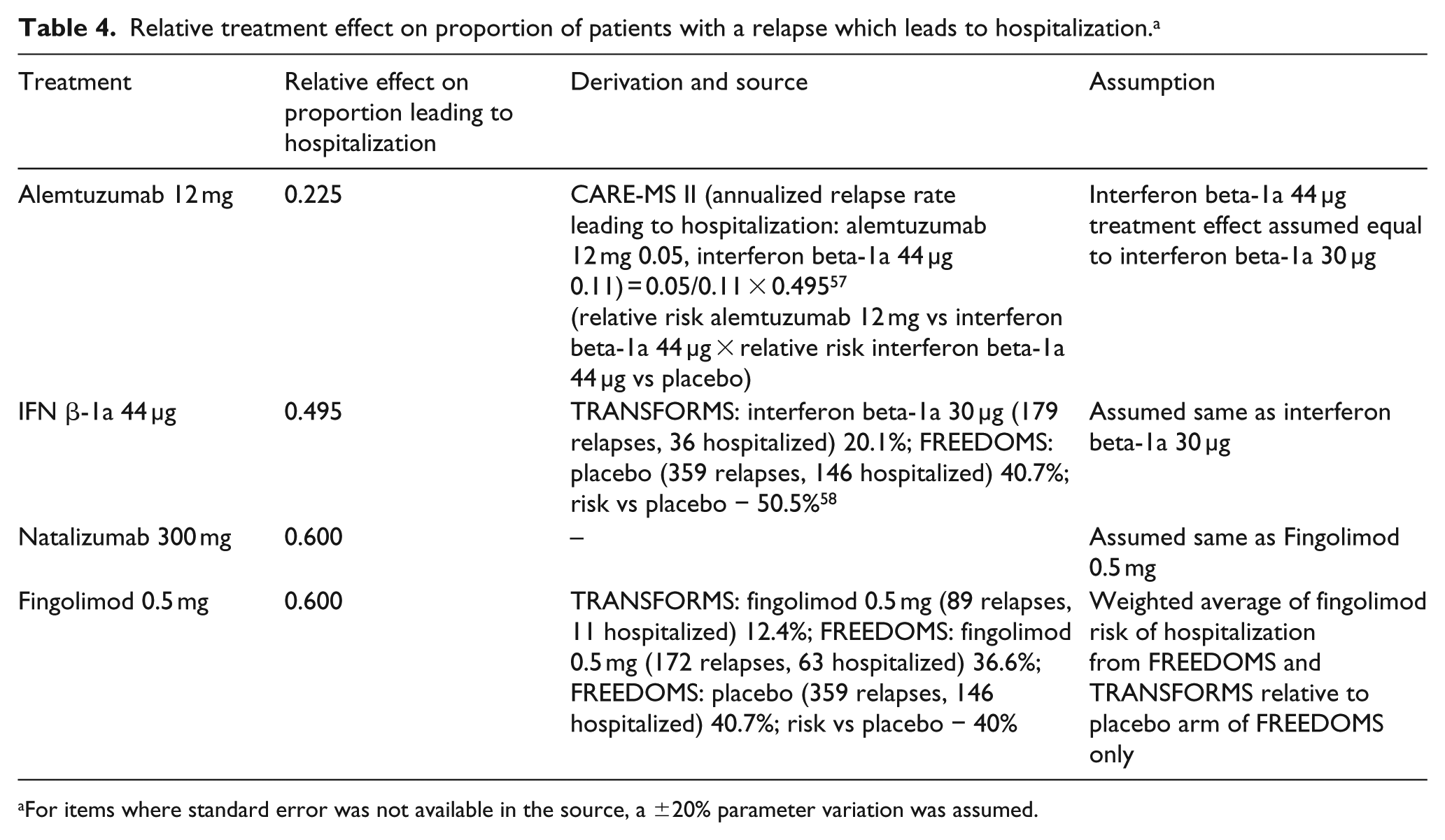
For items where standard error was not available in the source, a ±20% parameter variation was assumed.
Health utilities
The international literature was searched and reviewed for information on utilities in the MS population specifically in Italy. Due to the absence of Italian data, evidence on health utilities was derived from a UK population-based survey of MS patients and their caregivers. 36 The European Quality of Life – 5 Dimensions (EQ-5D) utility scoring system was applied, with respondent domain scores converted to a single utility weight using the UK value set. 36 Health utilities were applied based on disease severity (EDSS score) (Table 1). Each AE and relapse episode was associated with a utility decrement (i.e. disutility), applied for the duration of the AE or relapse. Utility decrement at relapse was assumed to last for 3 months and thereafter yearly values were calculated and applied in the model. Disutilities of 0.236 and 0.071 were associated with episodes of relapse leading to hospitalization and relapse not leading to hospitalization, respectively. 36
Resource use and costs
The model accounted for the costs of therapies (drug acquisition and administration costs and treatment follow-up), cost of AE management and cost of relapse with and without hospitalization (Tables 5 and 6). The international literature was searched and reviewed to retrieve average annual costs associated with EDSS severity. Several Italian studies were reviewed and Italian data on health care resource consumption from the international cost-of-illness study TRIBUNE in MS were used. 7 The study included n = 251 MS patients from Italy (N = 1261 in Europe 11 ) that largely matched patients’ baseline characteristics from the CARE-MS trials (mean age = 37 years, >50% with no or limited disability, and only 1.6% with EDSS ⩾ 7). 7 Frequency of specialist visits, frequency of diagnostic follow-up exams associated with disease management, treatment follow-up and management of AEs in the RRMS population were derived based on the disease management guidelines from the Emilia Romagna region of Italy from 2016, 59 and from expert opinion.
Drug acquisition and administration costs (€, 2017). a
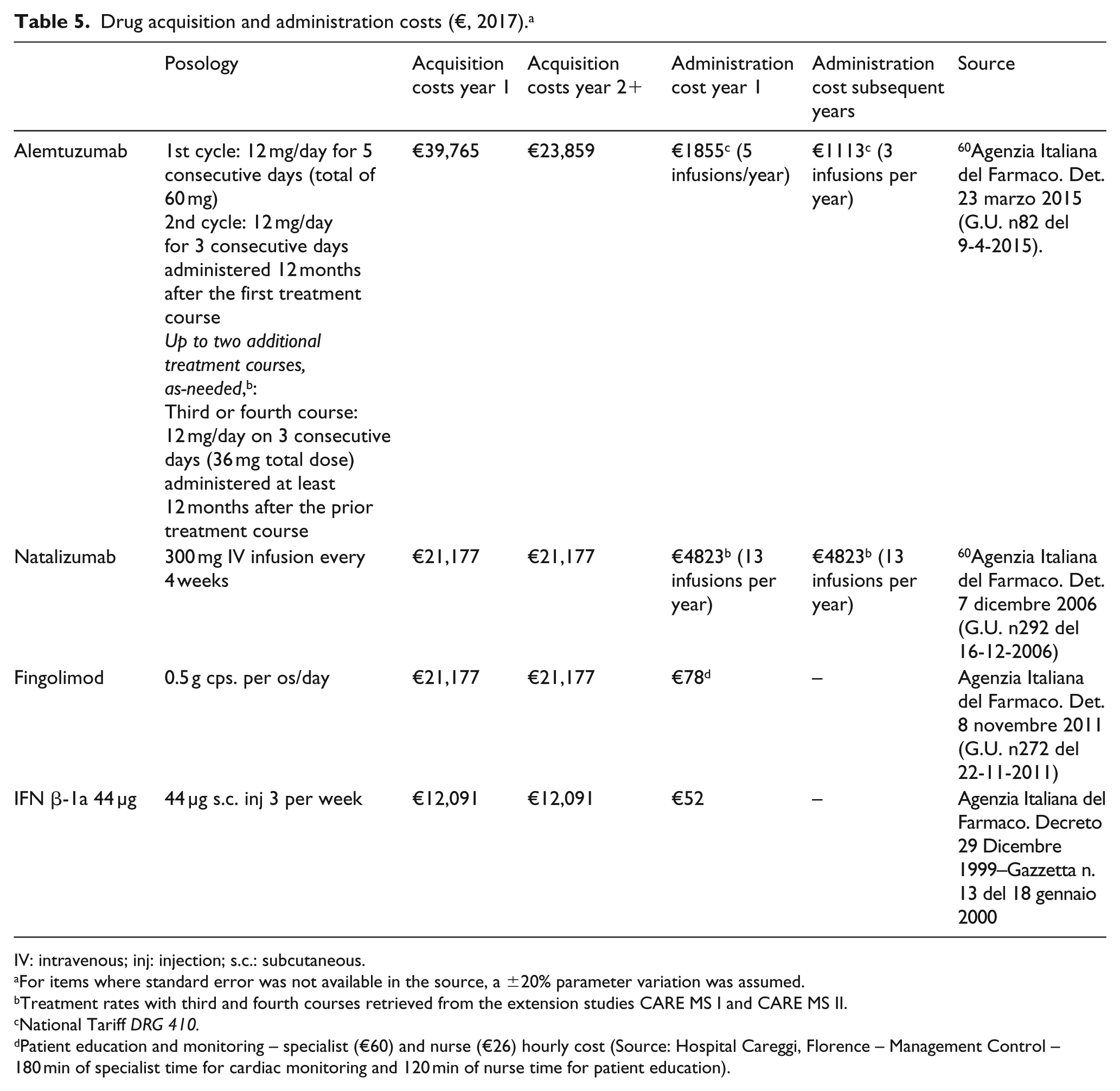
IV: intravenous; inj: injection; s.c.: subcutaneous.
For items where standard error was not available in the source, a ±20% parameter variation was assumed.
Treatment rates with third and fourth courses retrieved from the extension studies CARE MS I and CARE MS II.
National Tariff DRG 410.
Patient education and monitoring – specialist (€60) and nurse (€26) hourly cost (Source: Hospital Careggi, Florence – Management Control – 180 min of specialist time for cardiac monitoring and 120 min of nurse time for patient education).
Resource use and cost associated with treatment follow-up.
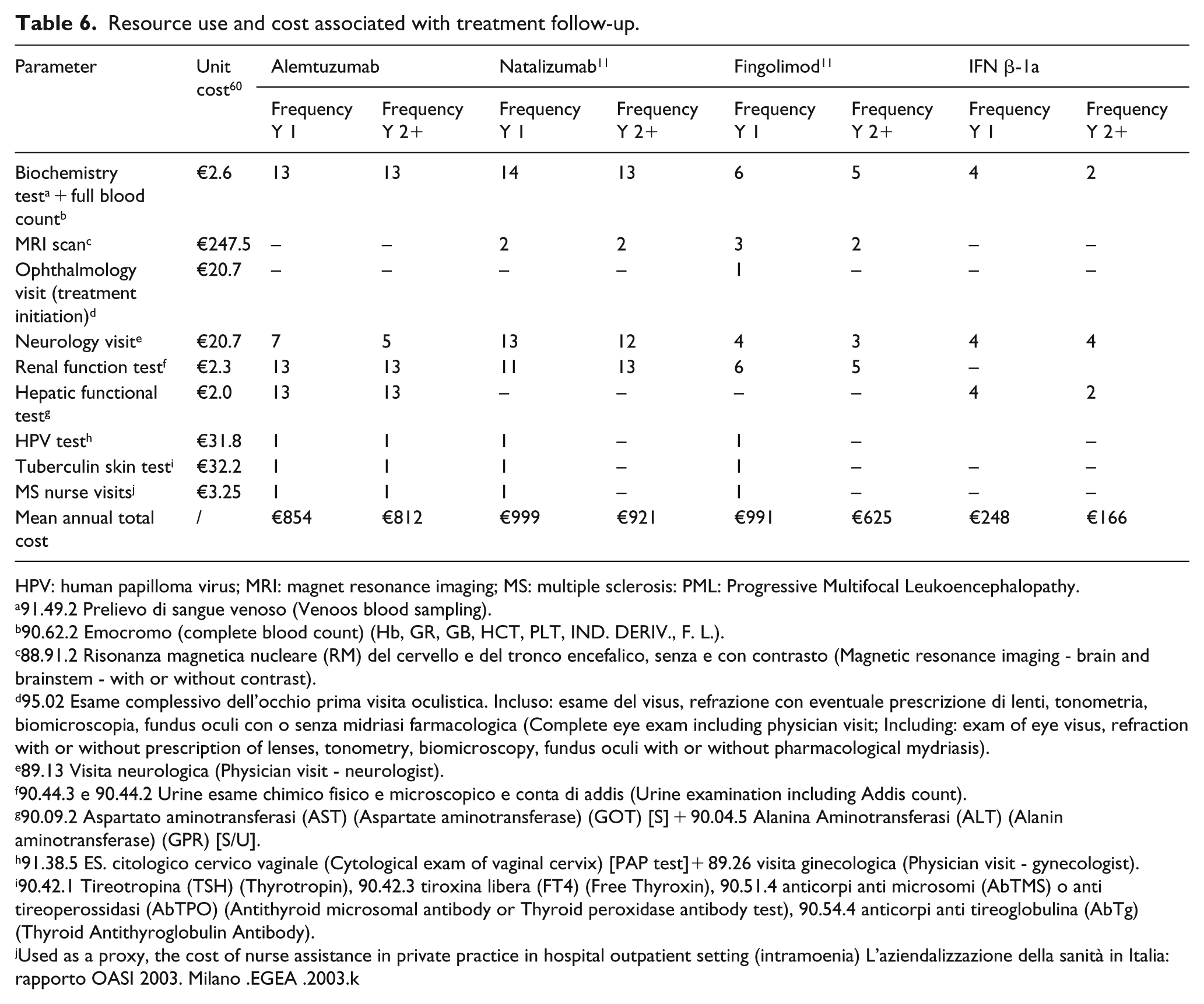
HPV: human papilloma virus; MRI: magnet resonance imaging; MS: multiple sclerosis: PML: Progressive Multifocal Leukoencephalopathy.
91.49.2 Prelievo di sangue venoso (Venoos blood sampling).
90.62.2 Emocromo (complete blood count) (Hb, GR, GB, HCT, PLT, IND. DERIV., F. L.).
88.91.2 Risonanza magnetica nucleare (RM) del cervello e del tronco encefalico, senza e con contrasto (Magnetic resonance imaging - brain and brainstem - with or without contrast).
95.02 Esame complessivo dell’occhio prima visita oculistica. Incluso: esame del visus, refrazione con eventuale prescrizione di lenti, tonometria, biomicroscopia, fundus oculi con o senza midriasi farmacologica (Complete eye exam including physician visit; Including: exam of eye visus, refraction with or without prescription of lenses, tonometry, biomicroscopy, fundus oculi with or without pharmacological mydriasis).
89.13 Visita neurologica (Physician visit - neurologist).
90.44.3 e 90.44.2 Urine esame chimico fisico e microscopico e conta di addis (Urine examination including Addis count).
90.09.2 Aspartato aminotransferasi (AST) (Aspartate aminotransferase) (GOT) [S] + 90.04.5 Alanina Aminotransferasi (ALT) (Alanin aminotransferase) (GPR) [S/U].
91.38.5 ES. citologico cervico vaginale (Cytological exam of vaginal cervix) [PAP test]+ 89.26 visita ginecologica (Physician visit - gynecologist).
90.42.1 Tireotropina (TSH) (Thyrotropin), 90.42.3 tiroxina libera (FT4) (Free Thyroxin), 90.51.4 anticorpi anti microsomi (AbTMS) o anti tireoperossidasi (AbTPO) (Antithyroid microsomal antibody or Thyroid peroxidase antibody test), 90.54.4 anticorpi anti tireoglobulina (AbTg) (Thyroid Antithyroglobulin Antibody).
Used as a proxy, the cost of nurse assistance in private practice in hospital outpatient setting (intramoenia) L’aziendalizzazione della sanità in Italia: rapporto OASI 2003. Milano .EGEA .2003.k
Because of a lack of published evidence on the cost of an MS relapse in Italy, the cost of one relapse episode not leading to hospitalization was derived based on primary data from the hospital centre Careggi in Florence using a micro-costing method and accounting for the cost of specialist visit, 1 MR scan and 15-day treatment with cortisone preparations (including costs of nursing and costs of disposable materials), which was €477/episode. The cost of a relapse episode leading to hospitalization was modelled based on the national tariff for DRG 13 ‘Multiple sclerosis and cerebral ataxia’, in-patient admission (DRG 13–National tariff €1417). 60
Ex-factory drug prices calculated with net of mandatory deductions (i.e. −5%/−5%) were retrieved from the Italian official journal (Gazzetta Ufficiale – G.U.). The cost of ambulatory visits was based on the National tariffs. 60 Table 6 provides a complete overview of the resource use and cost data used in the model as well as underlying assumptions and sources.
Analyses
Health effects and costs were modelled for a lifetime horizon (assumed 50 years for a chronic condition) and all outcomes and effects were discounted using an annual 3.5% rate based on NICE recommendations. 61 Therapies were modelled in line with the prescribing criteria of RRMS therapies in Italy (Nota AIFA 65: IFN β-1a prescribed for RRMS EDSS 0–5.5 and DMT discontinued in case of progression to SPMS). 62 The overall costs and utilities of each treatment were calculated by multiplying the number of patients in each state at the beginning of each cycle by the corresponding utility and cost values, and then summing across health-states and model cycles. Mean per patient cost was derived by dividing the total mean cost by number of alive patients at the end of each cycle. Estimated mean costs are contingent on patient transition across health-states (based on EDSS score) and time spent at each health-state.
Parameter uncertainty was assessed by means of deterministic one-way sensitivity analysis (OWSA) and probabilistic sensitivity analyses (PSAs).
Key model parameters were varied independently over a plausible range determined by the standard error of each variable. For parameters where a measure of uncertainty was not available, the range was estimated using an arbitrary ±20% variation around the point estimate. For PSA, parameters were assigned a distribution (Table S7, Supplementary Appendix) based on the underlying data and in line with good modelling practice. 63 PSA was performed by varying key model parameters simultaneously and randomly within their probability distribution. It was conducted using 1000 Monte Carlo iterations. The Incremental Cost-Effectiveness Ratio (ICER) was contrasted against the unofficial acceptability range (€25,000–€40,000) as proposed by Fattore G per Gruppo di lavoro Associazione Italiana di Economia Sanitaria (AIES). 64
In addition, cost-effectiveness of alemtuzumab in RRMS population was assessed in ad hoc scenario analyses:
Discount rate variation 0%/5% for costs and outcomes
Time horizon alteration: 15 years
Societal perspective including productivity losses due to short-term absence and reduced working time/income were derived from the TRIBUNE study 7
Based on expert opinion, some regional centres in Italy may implement more frequent follow-up schedule in RRMS management than that reported by the guidelines for specific patient populations. Thus, frequency of treatment follow-up visits in this scenario analysis was based on clinical practice of an Italian large regional MS centre (University Hospital Orbassano, Piedmont) (Table S6, Supple-mentary Appendix).
Internal validity was confirmed by comparing the distribution of patients across EDSS states with that from the pooled CARE-MS I and CARE-MS II trial populations at year 2.
Results
Treatment with alemtuzumab was more effective than other assessed therapies, with accrued incremental benefits over a lifetime in the range of 1.03–1.62 QALYs in comparison with natalizumab and IFN β-1a, respectively. Alemtuzumab cohort yielded the lowest cumulative number of relapses over a lifetime (a cumulative for both relapses that do not require hospitalization as well as those which require hospitalization) (Figure S5 Supplementary Appendix). Cumulative number of relapses over a lifetime in alemtuzumab cohort was 27.86, whereas it was 28.66 in fingolimod cohort, 29.25 in natalizumab cohort and 30.64 in IFN β-1a cohort.
Alemtuzumab showed cost-savings in comparison with all other assessed DMTs (Table 7).
Cost-effectiveness model results for lifetime horizon.
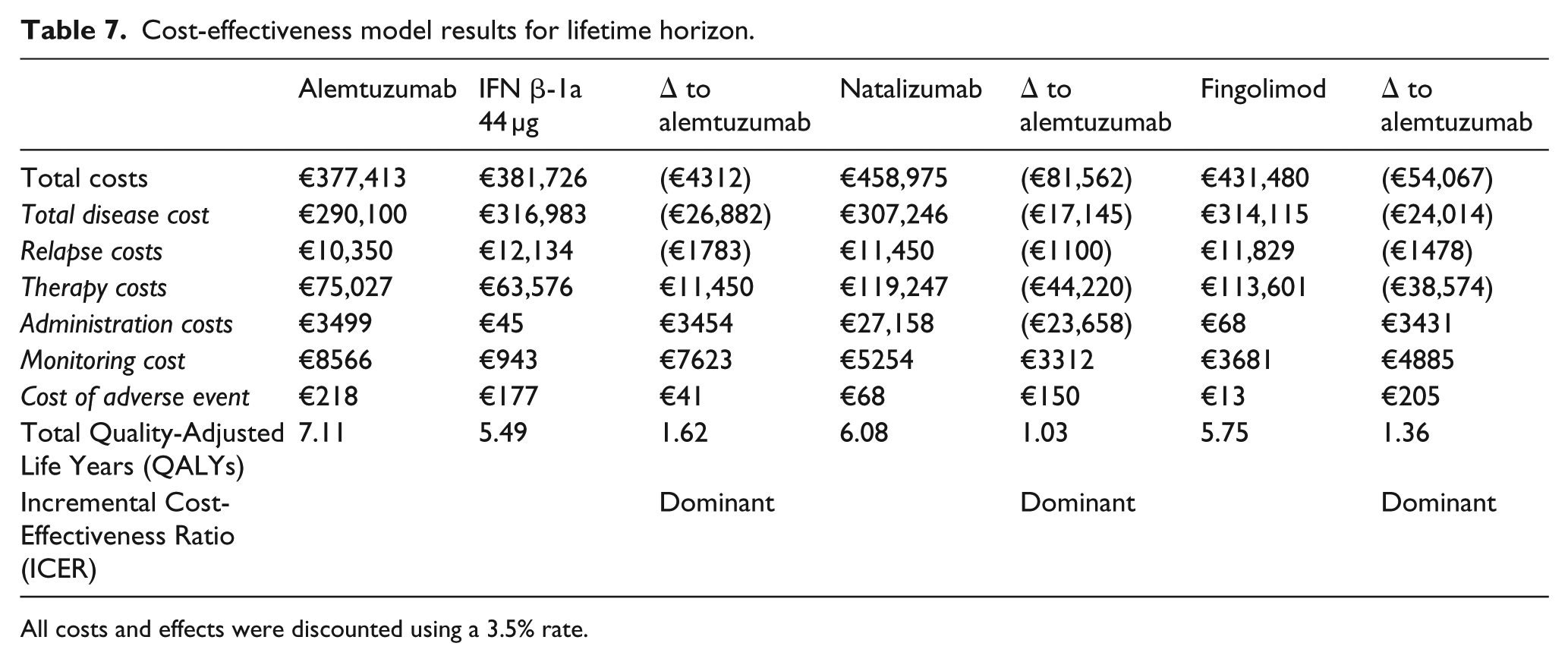
All costs and effects were discounted using a 3.5% rate.
In terms of the cost-effectiveness comparison, alemtuzumab was dominant (yielding better outcomes at reduced cost) in comparison with all assessed DMTs in the base-case analysis.
A multiple cost-effectiveness acceptability curve (CEAC) was constructed, which showed that the ICER of alemtuzumab carries the highest likelihood of being below a commonly accepted willingness-to-pay (WTP) threshold in Italy (€40,000/QALY) (below WTP in 52% of replications), compared to the other evaluated therapies (IFN β-1a in 49%, fingolimod in 6% and natalizumab in 3% of replications) (Figure 1).
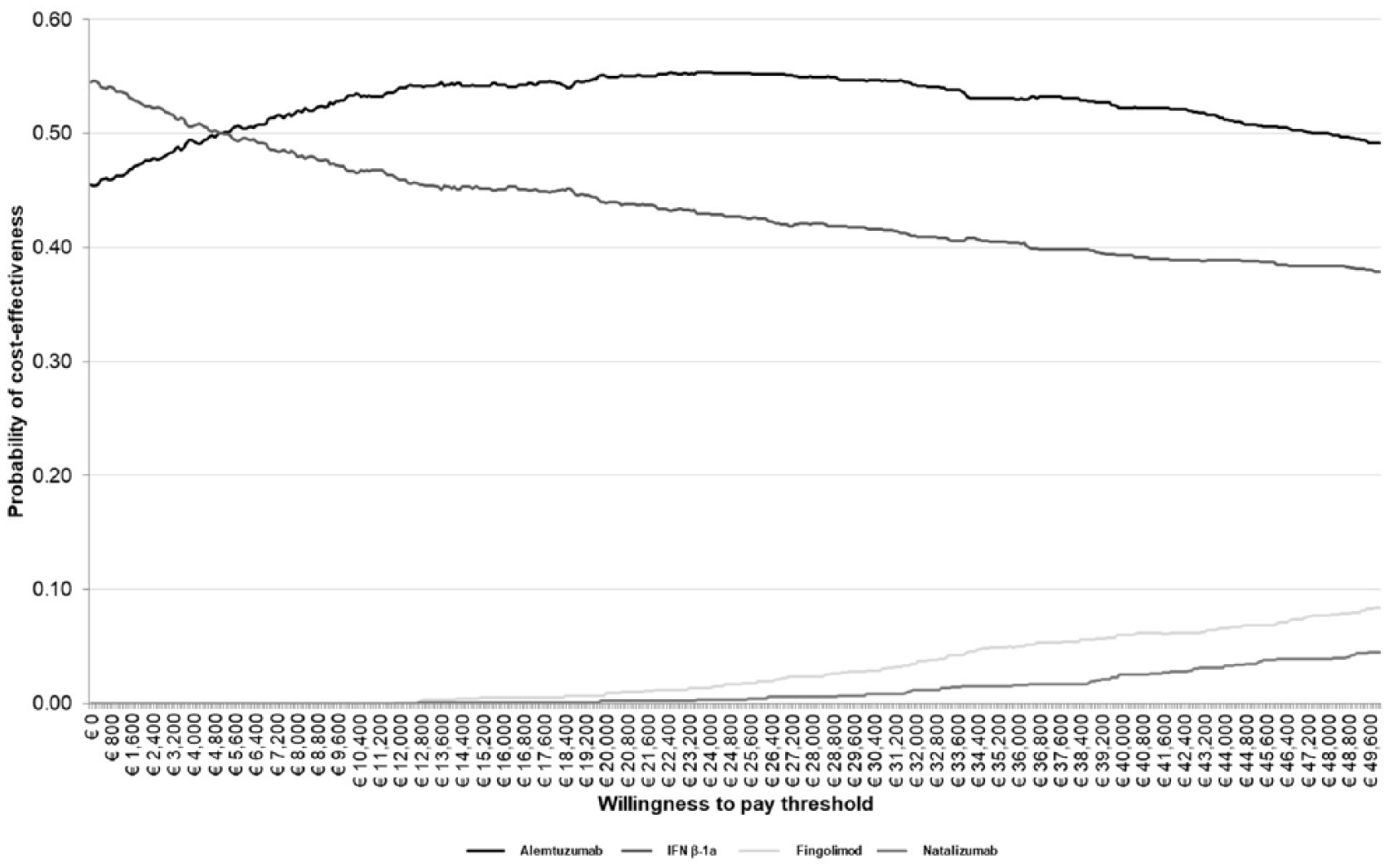
Cost-effectiveness acceptability curve for alemtuzumab versus other disease-modifying therapies using a lifetime horizon.
Results of the deterministic sensitivity and scenario analyses are presented in a Tornado chart (Figure 2) displaying results of those analyses that had ⩾±20% impact on the base-case ICER. Alteration of the annual discount rate and time horizon had the greatest impact on the base-case ICER. In addition, results were sensitive to the variation of disability progression HR of alemtuzumab and comparator treatments and change in comparator acquisition costs. However, alemtuzumab dominated IFN β-1a, fingolimod and natalizumab across all analyses. Results were somewhat sensitive to alteration of frequency of alemtuzumab follow-up visits with an impact of 6% and 15% on the base-case ICER in comparison versus natalizumab and versus fingolimod, respectively.
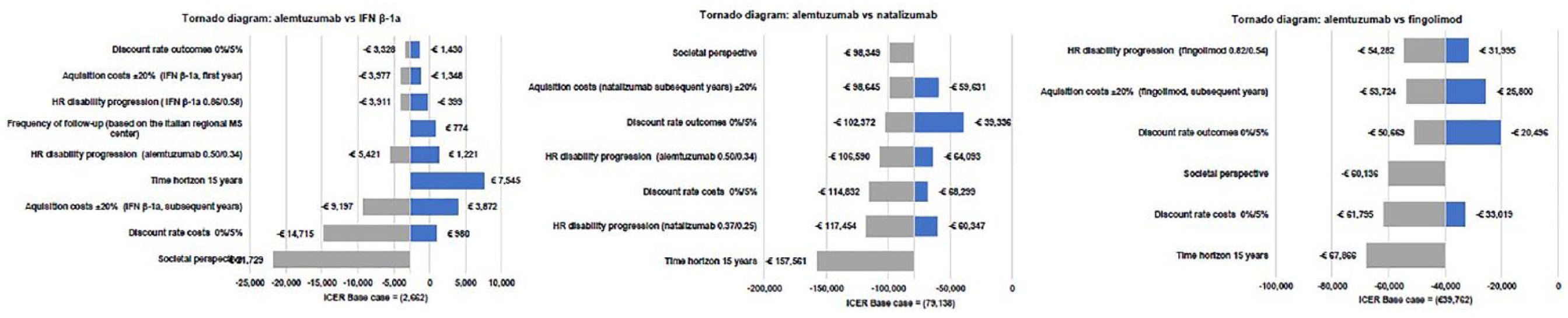
Results from the one-way sensitivity analysis.
Following the societal viewpoint, alemtuzumab treatment was the dominant option in comparison to IFN β-1a, natalizumab and fingolimod with ICERs up to 3 times lower than in the base-case (Figure 2).
Discussion
DMT may reduce the development of neural lesions as well as frequency of exacerbations and hence delay physical and cognitive detriment in RRMS patients. 1 DMTs require intravenous, oral, subcutaneous or intramuscular administration with variable frequencies, depending on treatment. At present, alemtuzumab, due to its unique mode of action and prolonged therapeutic effect, offers sustained treatment benefit with a convenient administration schedule (two initial treatment courses with up to two additional treatment courses if needed (second treatment course: 12 months after the prior treatment course; third or fourth course: at least 12 months after the previous treatment course)). The efficacy of alemtuzumab versus active comparator (SC IFN β-1a) was established through a comprehensive clinical development programme. Evidence from the extension trials indicated durable treatment effect in the absence of additional treatment after the initial two courses.16–18 Given the high costs of lifetime MS management associated with DMT, it is important to demonstrate the impact of these therapies on long-term health effects including QoL, and hence prove their value in the context of substantial disease management costs. This work aimed to assess lifetime costs and effects associated with the use of alemtuzumab in the management of RRMS patients in Italy in comparison with the direct comparator from the CARE-MS trials (SC IFN β-1a) and in comparison, with the other DMTs currently reimbursed in Italy for the same patient population as alemtuzumab (i.e. natalizumab and fingolimod).
This analysis suggests that alemtuzumab is a cost-saving option in the lifetime management of RRMS in Italy. Total treatment costs were largely driven by the underlying cost of disease management and drug therapy costs. Treatment with alemtuzumab was estimated to be less expensive than with IFN β-1a, fingolimod or natalizumab: this was driven primarily by lower disease management costs relative to the other treatments, with considerably less cost associated with relapse.
Our results show patients treated with alemtuzumab experience more QALYs (increment of 1.62 over IFN β-1a), more years without the use of walking aids and fewer relapses than all comparators included in the base-case analyses. This confirms that alemtuzumab is more effective than those treatments in terms of preventing relapse and progression to higher EDSS states, in all base-case analyses. Although the cost of treatment follow-up for alemtuzumab was higher than with other analysed DMTs, alemtuzumab remained a cost-saving treatment option in comparison with IFN β 1a, fingolimod and natalizumab even after increasing the annual frequency of follow-up visits to match what is in some cases at Local Health Unit level within the Italian SSN. Alemtuzumab remained a cost-saving option in comparison with IFN β-1a, fingolimod and natalizumab across most (>80%) sensitivity and scenario analyses.
Several works assessed the cost-effectiveness of DMTs from the Italian perspective;65–67 however, none assessed long-term effects and costs of alemtuzumab in RRMS patients. Dashputre et al. 68 and Montgomery et al. 69 assessed alemtuzumab among other DMTs from the perspective of the third-party payer in the United States and the perspective of the National Health Service in the United Kingdom. Dashputre and colleagues assessed alemtuzumab therapy in comparison to IFN 1β over the 2-year time horizon; their analysis showed alemtuzumab is a cost-effective treatment option in the United States with an accrued ICER of $25,276 per relapse avoided at 2 years (a much shorter time horizon than the present study). Montgomery et al. evaluated use of alemtuzumab in comparison with fingolimod varying the discount price of fingolimod across various patient access scheme scenarios in the United Kingdom. Although fingolimod resulted in lower acquisition costs across all tested scenarios, those advantages were offset by lower disease management costs associated with fewer relapses in the modelled cohort treated with alemtuzumab. 69
As any research, this cost-effective model has several limitations primarily stemming from the lack of evidence. In the lack of head-to-head comparison, the model was based on a published NMA. In addition, in the absence of local Italian tariffs for MS relapse management, a micro-costing approach was used. Despite these limitations, sensitivity analyses demonstrated results were robust across most (>80%) sensitivity and scenario analyses. Although the model designed allowed for a pairwise comparison of MS therapies, a multiple CEAC was constructed for a comparative analysis. At the time of manuscript submission, model programming features allowed for an OWSA around base-case ICER, while we acknowledge that an OWSA around net monetary benefit results may have provided additional information.
Alemtuzumab was shown to be a cost-saving treatment option in comparison to IFN β -1a 44 µg, natalizumab and fingolimod in the management of RRMS patients in Italy, notwithstanding its posology with a maximum of two additional treatment courses, if needed, after the initial treatment of two courses. The current study suggests that alemtuzumab is a preferable treatment option in the management of active or highly active RRMS in Italy.
Supplemental Material
Supplemental_Material – Supplemental material for The cost-effectiveness of alemtuzumab in the management of relapse-remitting multiple sclerosis in Italy
Supplemental material, Supplemental_Material for The cost-effectiveness of alemtuzumab in the management of relapse-remitting multiple sclerosis in Italy by Sanja Stanisic, Antonio Bertolotto, Patrizia Berto, Paolo Di Procolo and Julia Morawski in Global & Regional Health Technology Assessment
Footnotes
Declaration of conflicting interests
Antonio Bertolotto is the Principal Investigator of the Project # RF-2013-02357497 ‘Improving therapeutic appropriateness of Multiple Sclerosis treatments using biological approaches to personalize therapy and save pharmaceutical spending’ supported by the Servizio Sanitario Italiano, Ricerca Finalizzata.
Funding
The study was supported by Sanofi SpA. through an unrestricted grant to Certara Italy Srl, Via G.B. Pirelli, 27 - Milano. No interferences occurred in carrying out the project and in writing the manuscript that is the sole responsibility of the authors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
