Abstract
Multiple sclerosis progressively impairs patients’ ability to independently perform activities of daily living, reduces working capacity, and negatively affects social interactions and relationships, imposing a cost for the society. The aim of this study was to explore the impact on society of treatment of multiple sclerosis with natalizumab in Italian clinical practice. A prospective, observational study was conducted in 24 specialized centers throughout Italy. Direct and indirect costs, as well as the health-related quality of life of patients undergoing treatments, were estimated, while societal impacts were determined using a cost-utility approach. Non-medical direct and indirect costs accounted for 55.7% of the total cost prior to treatment and up to 47.5% after treatment with natalizumab. From the social perspective, greater medical direct costs for second-line drug treatment are nearly offset by savings on non-medical direct costs and by greater productivity, resulting in a +0.5% incremental cost. Assuming a societal perspective, the first year of treatment with natalizumab in real-world clinical practice results in an incremental cost of €2814.8/quality-adjusted life year gained. Multiple sclerosis imposes a considerable burden on patients, their families, and caregivers, stressing the importance of considering the societal perspective in the appraisal process. Treatment with natalizumab shows a noteworthy benefit in social terms.
Keywords
Introduction
Multiple sclerosis (MS) is a chronic immune-mediated demyelinating disease of the central nervous system (CNS) affecting more than 2 million people worldwide 1 and representing the most common non-traumatic cause of neurological disability in young adults. 2
MS progressively impairs patients’ ability to independently perform activities of daily living (ADLs). 3 Furthermore, it has been shown to reduce working capacity 4 and negatively affects social interactions and relationships, 5 thus leading to a progressive deterioration in health-related quality of life (HR-QoL), which inversely correlates with the accumulation of disability. 6 In addition, people with MS increasingly require continuous assistance from formal or in most cases informal sources (i.e. from family members or friends) for personal care and help in the execution of ADLs, which in turn affects caregivers’ productivity and quality of life (QoL) as well.7,8
MS not only has a major impact on the lives of individuals but also represents a relevant socioeconomic burden, due to its onset during young adulthood, when the productivity of individuals is the highest and the need for continuous care over the entire life span.9–11
Natalizumab (Tysabri®) is approved in the European Union for adult patients with relapsing–remitting multiple sclerosis (RRMS) with high disease activity despite treatment with a disease-modifying therapy (DMT) or with rapidly evolving severe disease. 12
The efficacy of natalizumab in reducing the frequency of clinical and radiological manifestations and delaying the progression of disability in patients with RRMS was demonstrated in the AFFIRM study, a 2-year, randomized, placebo-controlled study13–16 and confirmed over the longer term in clinical practice in cohorts of RRMS patients, most of whom had previously failed therapy with ⩾1 DMT.17–19 Some evidence suggests that the use of natalizumab may result in lower on-treatment disease activity when eligible patients start treatment at lower grades of physical disability, as measured by the Expanded Disability Status Scale (EDSS) score.17,20 Another observational study suggested that, in patients with evidence of clinical disease activity during treatment with interferon (IFN) beta or glatiramer acetate (GA), starting treatment with natalizumab induced better control of disease activity compared with switching between IFNs and/or GA, 21 although no conclusion on the relative efficacy of these DMTs can be drawn, due to the absence of controlled comparative studies.
The analyses of incremental cost-effectiveness of natalizumab published so far largely reported favorable results for this drug compared to other approved treatments for RRMS.11,22–27 In particular, natalizumab was shown to be cost-effective compared to IFN beta and GA in the United Kingdom, regarding costs per quality-adjusted life year (QALY) gained22,28 and cost per relapse avoided.23,25 Of note, the results of another study in Sweden and the United States also indicated that natalizumab is cost-effective compared to fingolimod, in terms of costs per relapse avoided. 26
However, all these analyses utilized economic models where efficacy inputs for natalizumab and comparators were derived either from the clinical studies in RRMS14,23–26 or from national registries.22,28 To the best of our knowledge, no prospective evaluation performed entirely on patient populations treated within the setting of clinical practice has been published yet. In addition, no cost-consequence data exist on treatments for RRMS in the Italian real-life setting.
This prospective observational study aimed to evaluate the direct and indirect costs as well as the HR-QoL of RRMS patients before and after treatment with natalizumab in Italian clinical practice setting. The societal impact was determined using a cost-utility approach.
Methods
Ethics statement
The study was conducted in compliance with the Declaration of Helsinki and current regulations for observational studies. Approval was obtained from the Institutional Ethical Committee of the Coordinating Center, the San Camillo-Forlanini Hospital, Rome, and of all participating sites. This trial was registered in the National Registry of Observational Studies of Agenzia Italiana del Farmaco (AIFA). All patients provided written informed consent before entering the study.
Study design
The prospective, observational study enrolled adult RRMS patients consecutively followed in 24 specialized MS centers throughout Italy and eligible to receive natalizumab according to current criteria for reimbursement of treatment in clinical practice.29,30 The Italian centers started the enrolment between the end of 2010 and the beginning of 2011, for 6 months. At the time of enrollment (baseline), the decision to start treatment with natalizumab had already been taken by the treating physician, and all patients had already received one dose of natalizumab. Enrolled patients were observed for 1 year.
Data collection
The data were collected using patients’ and physicians’ questionnaires. At the time of enrolment (baseline), questionnaires were administered using the computer-assisted personal interviewing (CAPI) method, including both subjective patient assessments and objective physician-assessed measures. Those completed by patients included demographic, socioeconomic, resources used, and HR-QoL data. Those completed by neurologists involved the main elements of clinical evaluation (i.e. EDSS, relapse). At the end of the follow-up period, patients and physicians were asked to repeat the questionnaires, and information was collected about any treatment combinations. The reasons for withdrawal, where applicable, were collected.
Direct and indirect cost data
Resources used, related to medical/not medical direct and indirect costs, were identified and measured by patients’ and physicians’ questionnaires prepared ad hoc and validated by a board of clinicians and pharmaco-economists.
Direct medical costs included all resources used for RRMS treatment: hospitalization, day hospital and/or outpatient services, specialist visits, laboratory tests, diagnostic procedures, relapses, and drugs.
These costs were quantified using the Italian Health Service list of charges.31,32 Direct non-medical costs took into consideration the cost of traveling to attend visits and the cost necessary for help in performing household chores. The latter were calculated using the effective minimum hourly wage of €7.2, according to the Italian Institute of Social Security criteria (Istituto Nazionale della Previdenza Sociale (INPS)). For travel costs, Italian Automobile Club (Automobile Club Italia (ACI)) vehicle running cost data for a medium-sized hatchback were used, assuming a unit cost per km of €0.37.
The number of days away from work was also recorded in order to calculate the loss of productivity. The average number of days of reduced work ability was also recorded. A record was also made of the number of days the patients were unable to make full use of their leisure time and those on which they required help from their family members. Indirect costs were quantified using a human capital approach. All the costs refer to the year 2016 or last available.
HR-QoL data
HR-QoL was elicited using the European Quality of Life Questionnaire (EuroQoL or EQ-5D version 3L) administered to patients at baseline and after 1 year of follow-up. EQ-5D is a widely used generic QoL questionnaire that assesses how patients fare in five domains (mobility, self-care, usual activities, pain/discomfort, and anxiety/depression). UK social tariffs were applied to derive utility values.
Statistical analysis
The significance of the differences in the mean values between the enrolment (baseline) and follow-up periods was assessed using the paired samples t-test. More specifically, normality was analyzed using the Kolmogorov–Smirnov test and the homogeneity of variance using Levene’s test. When normality was refused, non-parametric one-sample tests were performed.
Cost-utility analysis
The cost-utility analysis was conducted by comparing total benefits and total costs before and after the treatment with natalizumab from the Italian societal perspective. The results are reported as incremental cost-effectiveness ratio (ICER) per QALY gained. The ICER was calculated as the difference between the estimated costs divided by the difference between the QALYs produced before and after the treatment with natalizumab. An ICER value not exceeding €30,000 per QALY was considered cost-effective. 33
Sensitivity analysis
Univariate deterministic and probabilistic sensitivity analyses were performed on the ICER calculation. In the deterministic analysis, a 10% change in the individual cost items (hospitalization, specialist visit, laboratory test, rehabilitation therapy, drugs, and indirect costs) was hypothesized.
Probabilistic sensitivity analysis was performed, assuming Gamma distribution for the costs and beta distribution for the QALYs.
Results
Baseline data
A total of 176 patients were enrolled; of these 147 completed the 1-year observation, while 29 withdrew from the study. The analyses included the 147 patients for whom data at baseline and at 12 months were available.
The mean age of patients was approximately 38 years (median 38.0 years). The mean age at first symptoms of disease was 28.5 years (median 27 years). More female patients were included, 59.2% of the total cohort (Table 1).
Characteristics of the enrolled cohort.

GA: glatiramer acetate; IFN: interferon.
At baseline, the majority of enrolled patients was treated with IFN beta (60%) or GA 20 mg (22%) (Figure 1).
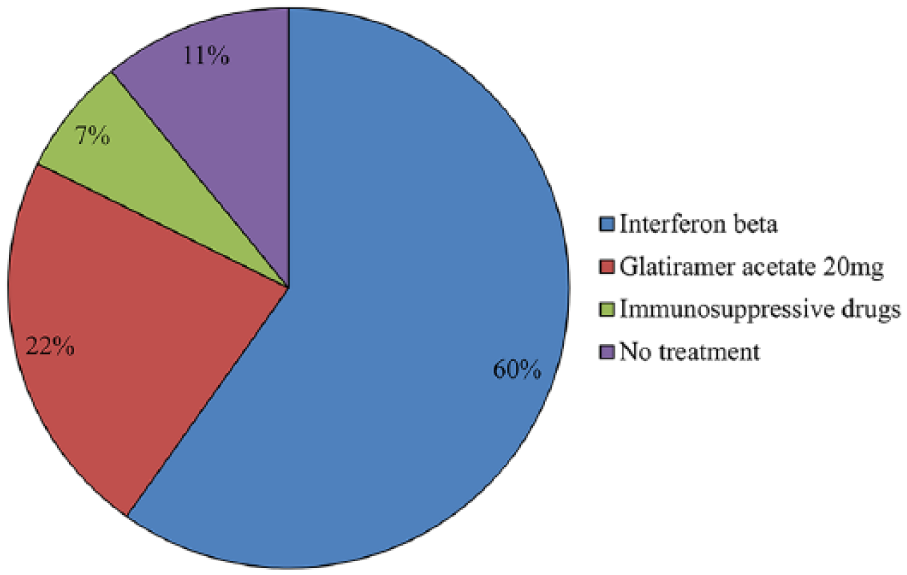
Pharmacological therapy at baseline.
Clinical data
Between the start of treatment (baseline) with natalizumab and the end of the follow-up period (12 months), all subjective and objective measurements of clinical status improved. Patients’ mean EDSS score dropped from 3.33 (standard deviation (SD) 2.16) to 2.98 (SD 2.94) (p < 0.001) (Table 2).
Clinical benefits (EDSS) and quality of life (utility).

EDSS: Expanded Disability Status Scale.
The significance of the differences in the mean values between the baseline and follow-up periods was assessed using the paired samples t-test.
The proportion of patients with at least one clinical relapse was decreased by 94.1% between baseline and the annual follow-up.
QoL data
During the 1-year observation period, there was a general improvement in HR-QoL, with a mean gain in utility of 0.04 per patient (p < 0.001) (Table 2).
Cost data
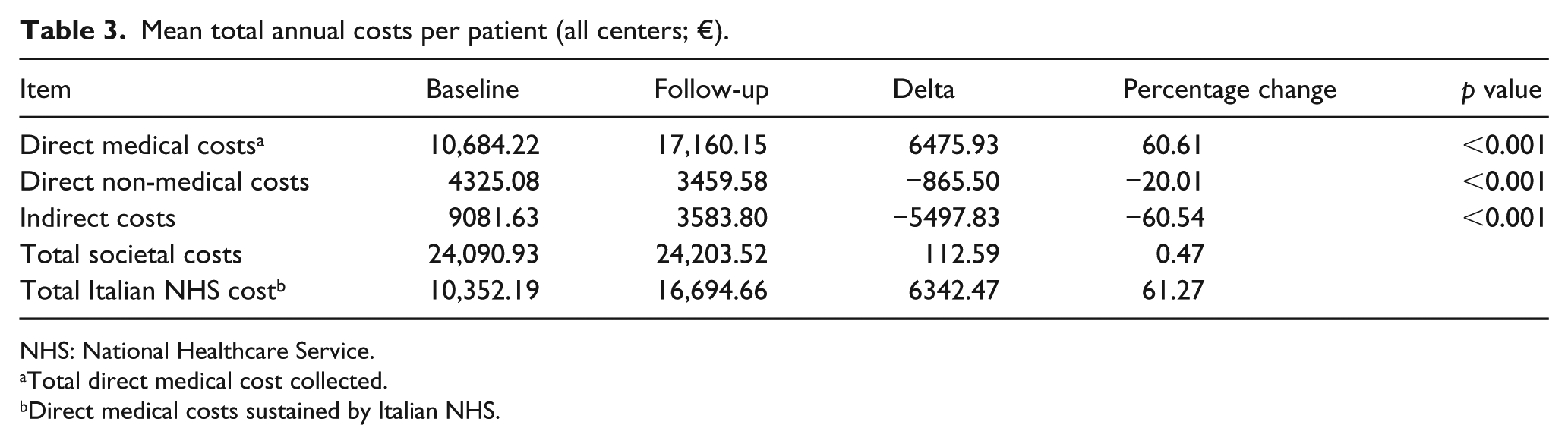
At baseline, the patients entering the study had accumulated mean direct medical costs (expressed on a 2016 basis) related to RRMS in the perspective of Italian National Healthcare Service (NHS) of €10,352.2: 12.8% for hospitalization, 1.3% for specialist visits, 0.6% for rehabilitation therapies, 7.3% for laboratory tests and diagnostic procedures, and 78.0% for drugs.
Following the treatment with natalizumab, the same costs rose by 61.3% (Table 3) due to the higher acquisition cost of the drug (+95.8%), whereas some components of this costs dropped, particularly the cost of hospitalization (–81.9%) and rehabilitation therapies (–68.5%).
Mean total annual costs per patient (all centers; €).
NHS: National Healthcare Service.
Total direct medical cost collected.
Direct medical costs sustained by Italian NHS.
The direct medical costs incurred by the patient increased from €332.0 to €465.5, leading to a direct medical total cost (reimbursed and not by the Italian NHS) of €10,684.22 and €17,160.15, respectively, before and after natalizumab treatment (Table 3). The difference between before and after natalizumab treatment was €6475.9 on an annual basis (p < 0.001).
The total direct non-medical costs amounted to €4325.1 per year at baseline and €3459.6 after 1-year treatment with natalizumab, corresponding to a 20.0% reduction (Table 3). This occurred mainly because patients required an average of 5.9 days of assistance per month in performing their household chores before enrolment, and only 4.7 days of assistance per month after treatment with natalizumab. The average annual cost for assistance in performing household chores amounted to €4079.3 at baseline and €3228.7 after 1-year follow-up, that is a 20.9% decrease.
Indirect costs included the value of lost productivity by the patient and family/friends due to the patient’s illness or treatment. At baseline, patients claimed a mean of 48.2 days of lost work each year for RRMS-related problems. The loss of productivity by friends and relatives was relatively limited, being a mean of 17.3 days per year. Following treatment with natalizumab, patients claimed a mean of 17.5 days of lost work per year for RRMS-related problems. The loss of productivity of friends and relatives fell to 8.4 days a year.
Following the human capital approach, the productivity loss calculation was based on the net national annual income per capita. This gives an estimate of annual indirect cost per patient of €9081.6 at baseline and €3583.8 at the end of follow-up; a 60.5% reduction in indirect costs (p < 0.001) (Table 3).
Assuming the Italian societal perspective, on an annual basis, the costs at baseline were €24,090.9 per patient, of which 44.3% were direct medical costs, 18.0% were direct non-medical costs, and 37.7% were indirect costs. In the observation year, the costs are equal to €24,203.5 per patient, of which 70.9% were medical direct costs (due to higher acquisition cost of natalizumab vs. drugs used at baseline), 14.3% were non-medical direct costs and 14.8% were indirect costs. Thus, the total annual cost per patient increased by €112.6, after treatment with natalizumab, assuming a societal perspective (Table 3).
Cost-utility analysis and sensitivity analysis
The cost-utility analysis considered the total costs (direct and indirect) and the total benefits in terms of QALYs before and after the treatment with natalizumab from the Italian societal perspective. The results are reported as ICER for QALY gained.
The first year of treatment with natalizumab in the Italian clinical practice setting results in an incremental cost of €2814.8 per QALY gained (Table 4), a value that appears to be well below the considered acceptability threshold, calculated as €30,000 per QALY gained. 34 Furthermore, it refers to a very short observation period.
Incremental costs for first year of treatment with natalizumab.
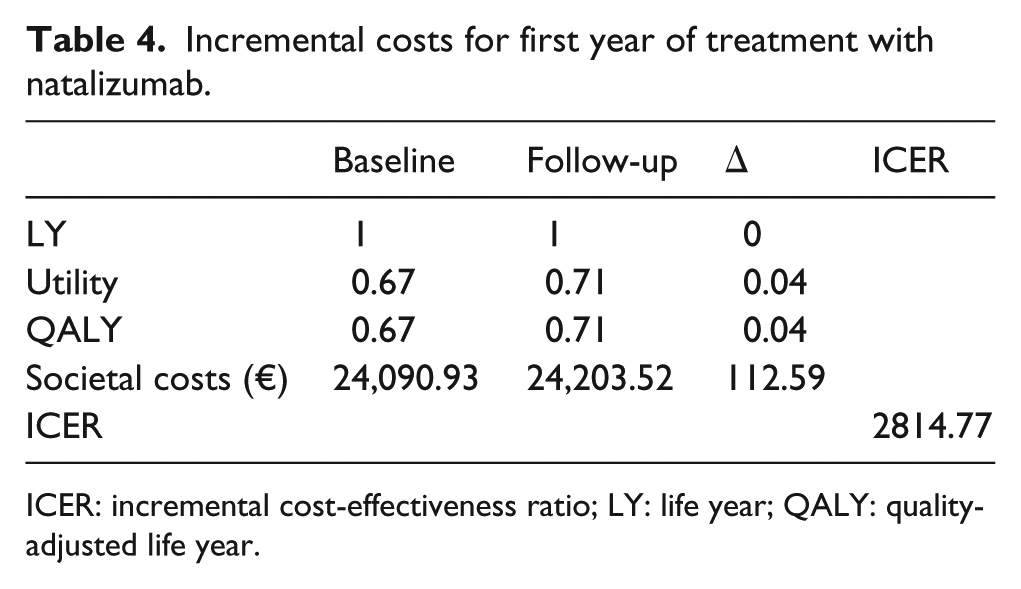
ICER: incremental cost-effectiveness ratio; LY: life year; QALY: quality-adjusted life year.
The results of the univariate sensitivity analysis are shown in the Tornado diagram (Figure 2). The analysis focused on the change of cost data shows that the incremental cost for the first year of treatment with natalizumab always remains below the €30,000 per QALY threshold (Figure 2). The ICER appears robust to variation in input parameters, ranging from a minimum of €16,283.7 to a maximum of €21,834.2 per QALY gained.
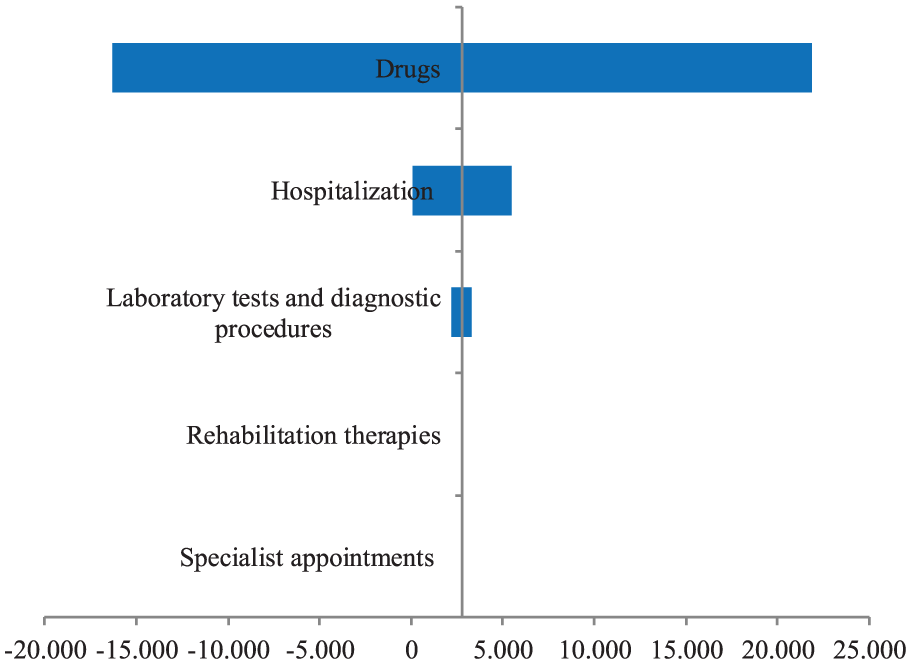
Tornado diagram: univariate sensitivity analysis.
The probabilistic sensitivity analysis indicates that natalizumab in the first year of treatment is cost-effective with a 99.9% probability, assuming a willingness to pay €30,000 per QALY gained and the societal perspective (Figure 3).
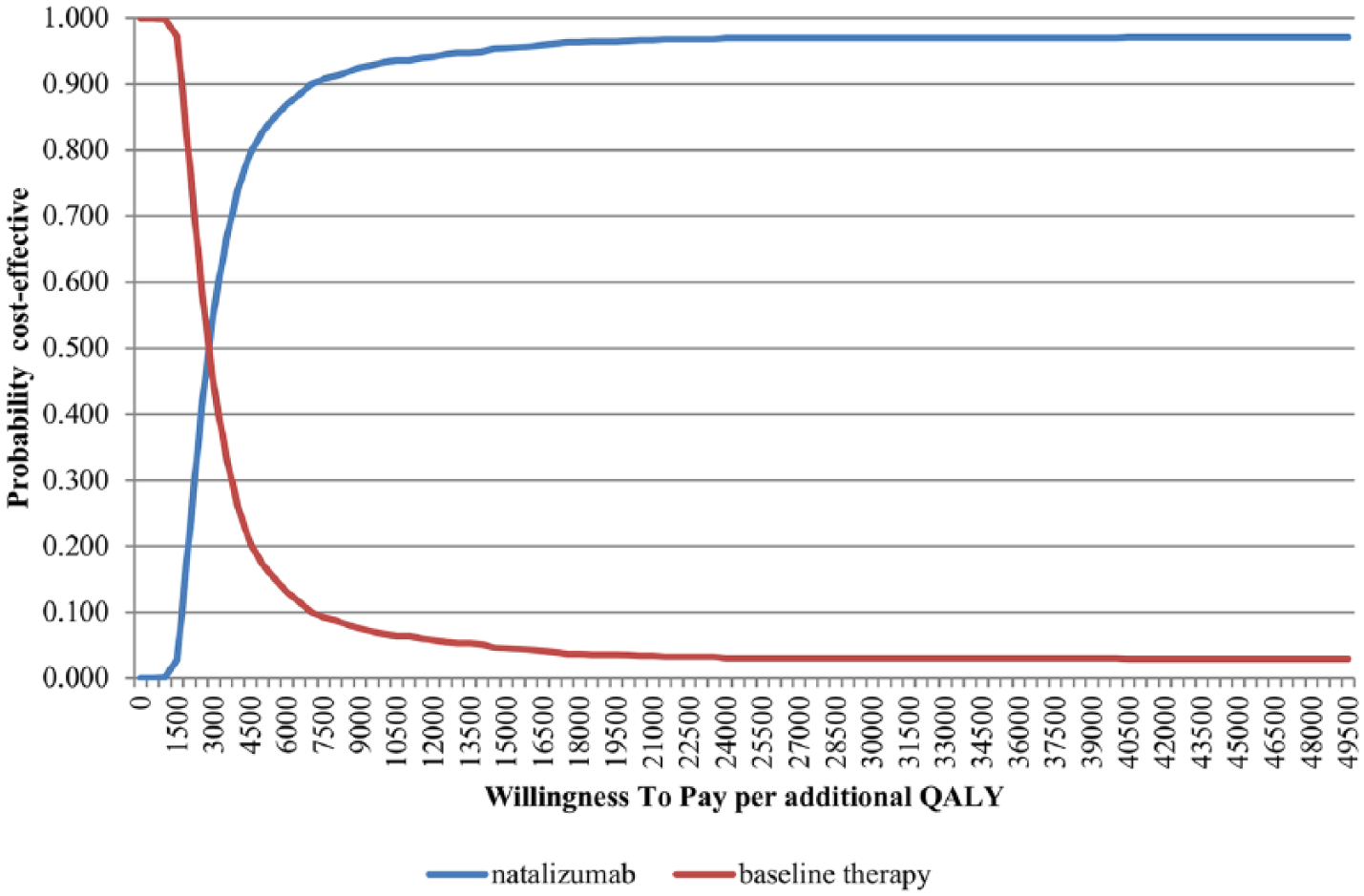
Cost-effectiveness acceptability curve (CEAC).
Discussion
The sample enrolled in this study is a representation of RRMS patients eligible for natalizumab therapy according to current criteria for reimbursement of treatment in Italian clinical practice. 30
After 1 year of follow-up with natalizumab treatment, positive and significant clinical benefits can be discerned. The average EDSS score was reduced from 3.33 at baseline to 2.98 at the end of 1-year follow-up, and the number of patients with at least one clinical relapse was reduced by 94.1% between baseline and follow-up.
Regarding the HR-QoL, the EQ-5D utility score was improved from 0.67 at baseline to 0.71 at follow-up with a mean gain in utility of 0.04 per patient.
In the case of costs, it should be noted that besides an increase in those related to the drug acquisition, treatment with natalizumab was associated with substantial savings on other direct and indirect costs. Direct costs borne by the Italian NHS for hospital admissions, specialist visits, rehabilitation treatment, and laboratory and diagnostic testings were reduced by 81.9%, 30.3%, 68.5%, and 29.3%, respectively.
Assuming the societal perspective, greater acquisition costs for second-line drug treatment are nearly offset by the savings achieved on non-medical direct costs (–20.0%) and by greater productivity (–60.5% of indirect costs) with an incremental total social cost of +0.5%.
The EQ-5D utility mean score recorded for patients who enrolled in this study was higher at baseline than that reported in the Italian cohort of patients with the same EDSS level in the cost of illness study by Kobelt et al. 34 (0.67 vs. 0.62). This difference could be explained by a number of factors basically related to the sample of the patients analyzed by Kobelt et al. For example, the higher average age (46 vs 37 years), the higher average age at the diagnosis (38 vs 31 years), and the higher proportion of females (65% vs 59%). Furthermore, in Kobelt et al.’s 34 study, 35% of the enrolled patients had the relapsing–remitting form of MS and 51% had a progressive form of MS (11% “Don’t know” and 3% “No answer”), whereas this study enrolled all patients with RRMS.
Concerning the utility difference between baseline and 1-year follow-up (from 0.67 to 0.71), this is in line with or greater than other studies in the literature.28,35
In terms of costs, some differences were recorded when comparing with some already published data. At the baseline, the cost data estimated by this study are lower than those of the cost of illness study conducted in Italian patients by Kobelt et al.: 34 the difference (€24,091 vs €38,845) can be explained considering that the sample of patients enrolled by Kobelt et al. had a higher average age and especially a greater baseline EDSS score, which supports the trustworthiness of the data we have measured. In fact, the annual social cost of a patient with a similar level of EDSS in the two studies appears in line.
At the annual follow-up, we do not have an Italian benchmark, but the results for Swedish patients in the cost-effectiveness study by Kobelt et al. 28 are aligned with those measured in this study (–5.5%).
The cost-utility analysis, conducted with the observational study results, provided evidence that treatment with natalizumab is a cost-effective option in RRMS patients eligible from a clinical perspective. In fact, assuming a societal perspective, the first year of treatment with natalizumab in this real-world clinical practice study results in an incremental cost of €2814.8 per QALY gained. This value is well below the considered acceptability threshold (€30,000/QALY). Univariate and probabilistic sensitivity analyses confirmed the robustness of the results.
Results also showed that pharmacological expenditure represents a relevant cost for the Italian NHS, but there are other cost components contributing to healthcare and social costs. In fact, although acquisition costs with natalizumab are substantially higher, the treatment produced savings which nearly offset the increase in direct medical cost, thus making the natalizumab treatment a cost-effective alternative.
Limitations of this study
The main limitation of our analysis was the short observation period which did not allow for a comprehensive analysis of the persistency of the treatment effect (and consequently constancy of costs).
In interpreting the results of this study, it should be observed that the improvements of the benefits were derived from an observational study lacking a control group and do not take into account the worsening patients encountered when these did not go through second-line drug treatment. Nevertheless, despite that the estimated results may underestimate the actual figures, we have recorded a substantial overlapping with those achieved in the aforementioned studies.28,35
In addition, as this is an observational study, with no control group, calculation of ICER would not be perfectly comparable with that of previous analyses based on randomized controlled trial evidence. However, our calculation could be considered a conservative hypothesis, assuming that without switching to natalizumab, the patients’ state of health and costs incurred would remain constant.
Finally, due caution should be used for the transferability of the results based on Italian costs and clinical practice.
Conclusion
To the best of our knowledge, this article for the first time presents evidence on societal costs of treatment with natalizumab in the Italian clinical practice setting, therefore contributing to the economic assessment of the societal impact of the drug.
Direct non-medical costs and indirect costs account for nearly 55.7% of the total costs prior to the treatment with natalizumab (of which 37.7% represents productivity loss). The prescription of natalizumab gives rise to a 47.5% drop in these costs. These savings compensate for the increase in drug cost, resulting in a mere €112.6 incremental cost.
The results confirm the significant social impact of MS, including the importance of direct non-medical costs and productivity loss, therefore highlighting the importance of adopting a broad societal perspective in economic evaluation.
Finally, the study shows that the correct use of natalizumab is cost-effective in Italian clinical practice and therefore in “real” Italian patient populations.
Footnotes
Acknowledgements
We thank Ray Hill, an independent medical writer, who provided English language editing and journal styling prior to submission on behalf of Health Publishing & Services Srl and funded by Biogen Italia. TyPE Investigators: Amato M.P., Bertolotto A., Cargnelutti D., Centonze D., Comi G., Cottone S., Deotto L., Florio C., Gallo P., Ghezzi A., Granella F., Malentacchi G.M., Mancardi G. L., Marrosu G.M., Morra V.B., Montanari E., Nichelli P., Savettieri G., Tedeschi G., Tola M.R., and Trojano M.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: B.P. has received grants from ARIAD, MSD Italia, Pfizer, Zambon Roche, Sanofi Pasteur MSD, Servier, Sigma Tau, Stroder, and Teva. F.S. has received grants from Abbott, Bayer, Biogen Idec, Biomarine, BMS, Boehringer-Ingelheim, Celgene, Daiichi Sankyo, Edwards, Eli Lilly, Genzyme, GlaxoSmithKline, Janssen Cilag, Johnson & Johnson, Lundbeck, Medac, MSD Italia, Medtronic, Novartis, Novo Nordisk, Obi, Otsuka, Pfizer, Roche, Sanofi Pasteur MSD, Servier, Sigma Tau, Stroder, and Teva. R.C. has received fees as a speaker or member of advisory board for Biogen, Teva, Novartis, Sanofi Aventis, and Genzyme. S.F. and L.S. are employees of Biogen Italia. G.B.Z. has received fees as a speaker or member of advisory board for Bayer, Biogen, Merck Serono, Teva, Novartis, and Genzyme. C.G. has received research grants from Teva. He has received fees as a speaker or member of advisory board for Bayer Sheering, Biogen, Merck Serono, Teva, and Novartis.
Funding
This study was financially supported by Biogen Italia (Milan, Italy).
