Abstract
This analysis was aimed at estimating the cost per responder as measured by number needed to treat of ixekizumab as compared with other biologic drugs approved in Italy for the treatment of moderate-to-severe plaque psoriasis. The clinical efficacy was assessed in terms of number needed to treat, based on a network meta-analysis of published efficacy data as measured by Psoriasis Area and Severity Index response (PASI75, PASI90, and PASI100) for relevant biologic comparators. The cost was based on the number of administrations dispensed in the first (induction plus maintenance period) and the second (maintenance period only) year of treatment and the ex-factory price net of discounts of each biologic drug. The cost per responder was adopted as a cost-effectiveness indicator. Independent of the Psoriasis Area and Severity Index response (PASI75, PASI90, and PASI100) used and the year of treatment considered, the cost per number needed to treat for ixekizumab appeared consistently to be the lowest. For example, considering first-year costs and PASI75, the cost per responder for ixekizumab was €16,388, compared to adalimumab (€22,574), etanercept (branded original: €32,420; biosimilar: €21,432), secukinumab (€17,937), and ustekinumab (€20,014). The differences in the cost per responder between ixekizumab and the comparators increased when higher Psoriasis Area and Severity Index response levels were considered. This economic assessment confirmed that ixekizumab is a cost-efficient option from the perspective of the Italian National Health Service for the treatment of moderate-to-severe plaque psoriasis.
Keywords
Background
Psoriasis is an inflammatory chronic skin disease, characterized by redness (erythema), thickness and scaling of the skin, and by remittance/relapse phases of variable length.1–3 It affects men and women in similar proportions, while the onset age is variable—though the disease most frequently occurs in young-adults. 4
The typical cutaneous manifestations of psoriasis and its chronic course significantly interfere with the affected subjects’ quality of life (QoL), and can have a severe and sometimes disabling psychological and social impact. 5 In fact, a high prevalence of anxiety, depression, and alcohol abuse is observed in patients with psoriasis. 2 In addition to psychiatric comorbidities over the last few decades, psoriasis has been increasingly considered as a systemic disease, associated with several (rheumatologic, cardio metabolic, gastroenterological, etc.) comorbidities. In patients with severe psoriasis, a higher mortality risk (hazard ratio: 1.5; 95% confidence interval: 1.3–1.7) is found as compared to the general population.4,6 Affected men and women, respectively, die 3.5 and 4.4 years earlier than healthy controls, probably due to comorbidities, lifestyle (smoke and alcohol abuse), and administered therapies. 6
Psoriasis affects at least 100 millions of people all over the world, mostly Caucasians.4,7 Prevalence in Europe varies between 0.6% and 6.5%, and is highest in northern countries. 8 Psoriasis in Italy, with about 1.5–2.5 million cases, is one of the most common dermatological diseases, with a reported prevalence of 2.9%–3.1%9,10 and an incidence of 230–310 new cases per 100,000 persons/year. 11
Plaque psoriasis (PsO) is the most common form (80% ca. of all psoriasis patients).1–3 It is characterized by the presence of erythematous–desquamative lesions, located mainly at the extensor sites of the extremities (knees and elbows), the lumbosacral region and the scalp,1,2 often accompanied by burdensome symptoms such as itch and pain.3,12 In about 20% of cases, PsO is moderate to severe, defined by a Psoriasis Area Severity Index (PASI) score >10, or Body Surface Area (BSA) >10, or Dermatology Life Quality Index (DLQI) >10 according to the European S3-Guidelines and by the Italian guidelines SIDeMaST (Società italiana di Dermatologia medica, chirurgica, estetica e delle Malattie Sessualmente Trasmesse—Italian Society of Medical, Surgical, Cosmetic Dermatology, and Sexually Transmitted Diseases).13,14 A recent European consensus agreed to expand this definition of severity based on the presence of lesions in visible or so-called hard-to-treat areas like the face, genitals, palms, and soles which can reduce the patients’ QoL, even if PASI or BSA are <10.13,14
As compared with traditional systemic therapies, biologic drugs have significantly changed the treatment pathways in moderate-to-severe PsO. 15 Using biologic drugs, patients can now reach significant levels of skin clearance, associated with an improved QoL.16,17
Several biologic drugs are currently available in Italy for the treatment of moderate-to-severe PsO, including adalimumab, etanercept, infliximab (tumor necrosis factor (TNF)-alpha inhibitor), ustekinumab (interleukin-12/-23 inhibitor), and secukinumab (interleukin-17A inhibitor). Biosimilar drugs are currently available for etanercept and infliximab. AIFA (Agenzia Italiana del Farmaco—Italian Medicines Agency) has recently approved the reimbursement of ixekizumab, a new interleukin-17A inhibitor. In a series of clinical studies, ixekizumab has provided significantly higher efficacy in skin clearance compared with placebo, etanercept, and ustekinumab.18–20 Despite this clinical evidence, economic assessment of ixekizumab compared with other biologic therapies is currently unavailable regarding the Italian context. To address this gap for economic evaluation of ixekizumab versus other biologics indicated for the treatment of PsO, we calculated the cost per responder, based on the NNT (number needed to treat). The cost per responder combines clinical and economic dimensions and provides additional support to for cost-effective decision-making.
Objective
Adopting the Italian National Health Service’s (NHS) perspective (only direct medical costs were considered), this analysis was aimed at estimating the efficacy, the treatment costs, and the cost per responder of ixekizumab versus other biologic drugs approved in Italy for the treatment of moderate-to-severe PsO.
Materials and methods
The efficacy of approved systemic treatments for PsO treatment was assessed on the base of a series of outcome measurements taking into account the severity of symptoms. 14 PASI is one of the most frequently outcome measurement used; 14 it combines a qualitative (presence of lesions such as erythema, infiltration, and desquamation) and quantitative (proportion of affected area for each body region) skin disease assessment. The PASI score can vary between 0 (no lesions) and 72 (disease highest degree). In addition, reduction of PASI based on the score variation from an initial value is used to classify the response to treatment as an improvement of at least 75% (PASI75), or 90% (PASI90), or 100% (PASI100), respectively. 14
To assess the cost per responder of the biologic therapies approved in Italy for the treatment of moderate-to-severe PsO, the cost per NNT versus placebo was estimated.21,22 In general, the NNT is the number of patients in the experimental group who need to be treated in order to achieve one additional therapeutic benefit/responder patient (additional, that is, one more than the patients who would be successfully treated in the control group).
First, the NNT versus placebo of all biologic drugs was quantified using published study data (see the “Network meta-analysis” section). Second, multiplying the NNT by the corresponding treatment cost per patient, the cost per NNT of each biologic drug was evaluated.
Dosing considerations
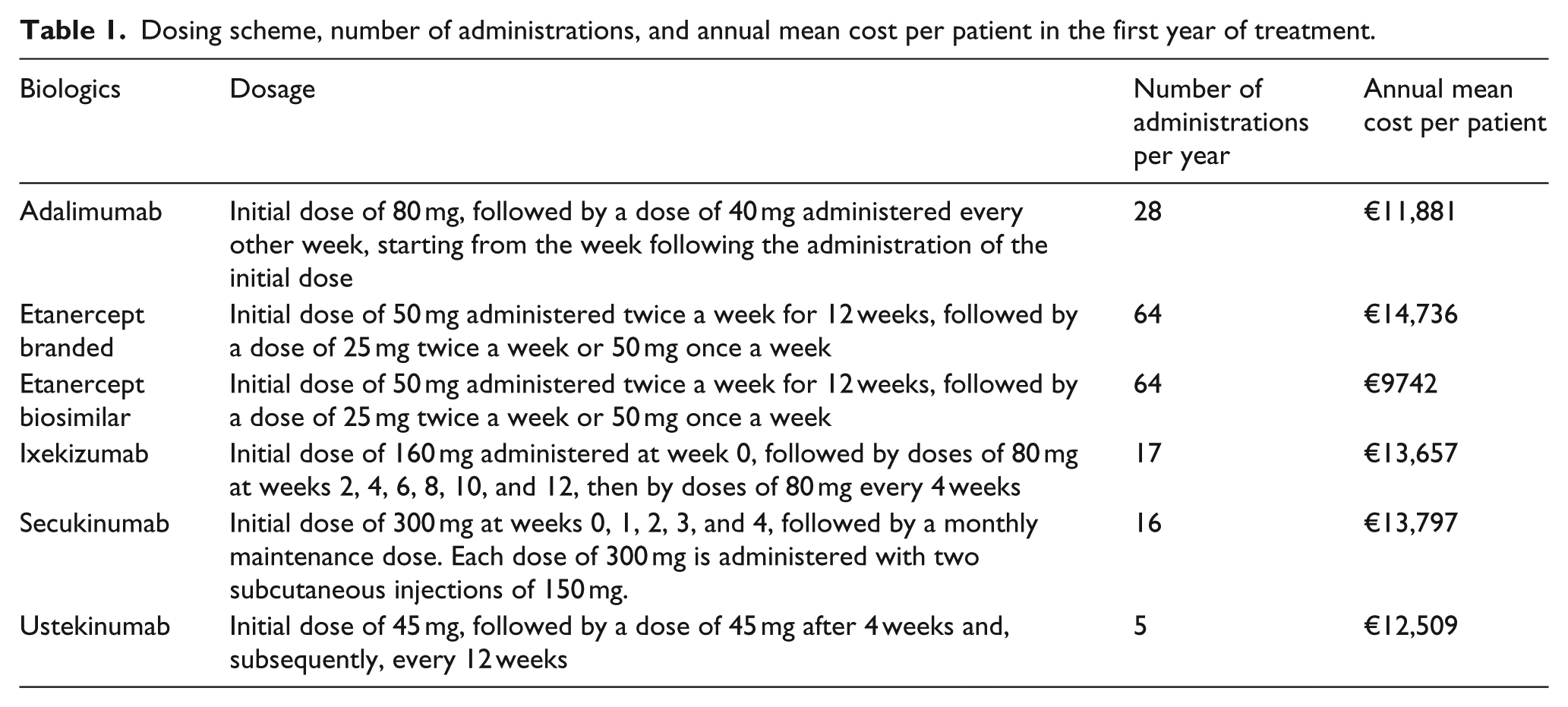
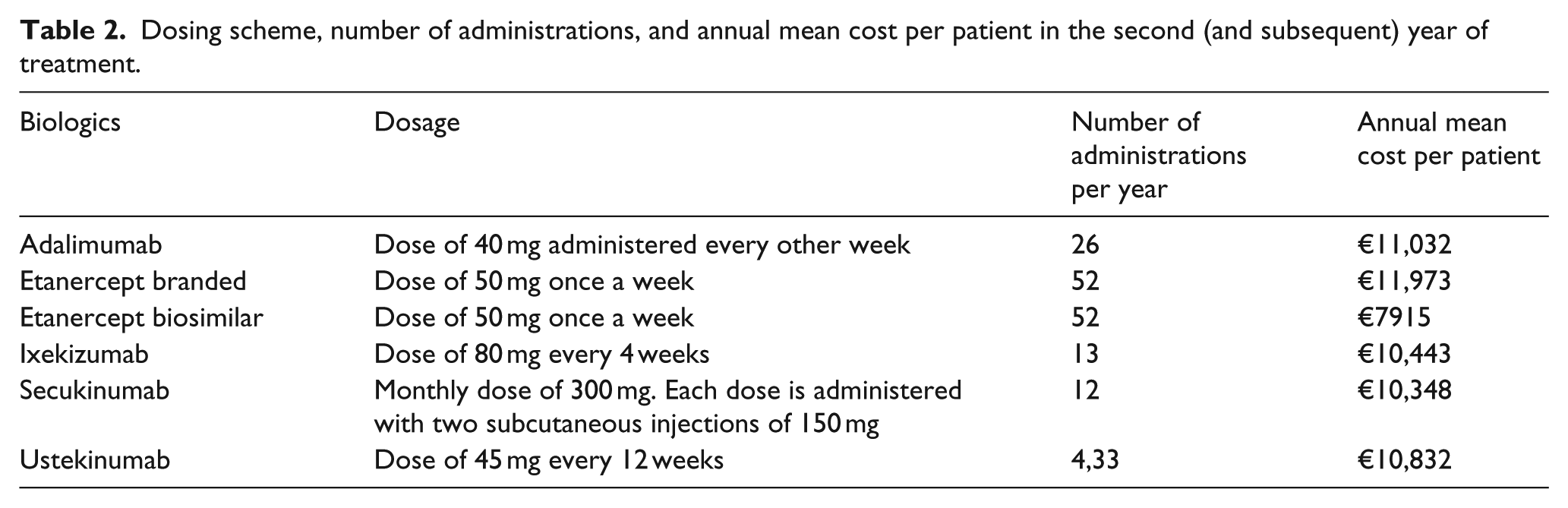
Based on the Summary of Product Characteristics (SPC), the dosing scheme of the biologic therapies considered changes between the first (induction plus maintenance period) and the second (or subsequent) year of treatment (maintenance period only). Therefore, to capture the most significant differences among the biologic drugs considered in terms of both efficacy (NNT) and treatment costs, we calculated the mean cost treatment referred (1) to the first year considering the induction and the maintenance dosing scheme (Table 1) and (2) to the second (or subsequent) year considering only the maintenance dosing scheme (Table 2).
Dosing scheme, number of administrations, and annual mean cost per patient in the first year of treatment.
Dosing scheme, number of administrations, and annual mean cost per patient in the second (and subsequent) year of treatment.
Network meta-analysis
This economic evaluation was based on the results of a recent cost analysis published in the United States, which evaluated the NNT versus placebo of adalimumab, etanercept, ixekizumab, secukinumab, and ustekinumab in the treatment of moderate-to-severe PsO, respectively. 23 Table 3 shows the NNT values considered. The NNT values, evaluated in the US cost analysis, 23 were obtained from a previous network meta-analysis (NMA) of PASI75, PASI90, and PASI100 data of published randomized clinical trials, 24 the results of which are presented in Table 4. The NMA was based on a systematic literature review (SLR) of published and gray literature, following guidelines from the Cochrane Handbook of Systematic Reviews and Interventions. 25 The SLR covered the period from January 1990 to November 2015 and included phase II, III, and IV randomized controlled trials (RCTs) of relevant conventional systemic and biologic therapies in moderate-to-severe PsO. The NMA was presented as a poster at the ISPOR 2016 (International Society for Pharmacoeconomics and Outcomes Research).
Number needed to treat per additional PASI75, PASI90, or PASI100 responder versus placebo for each of the evaluated biologics. 23
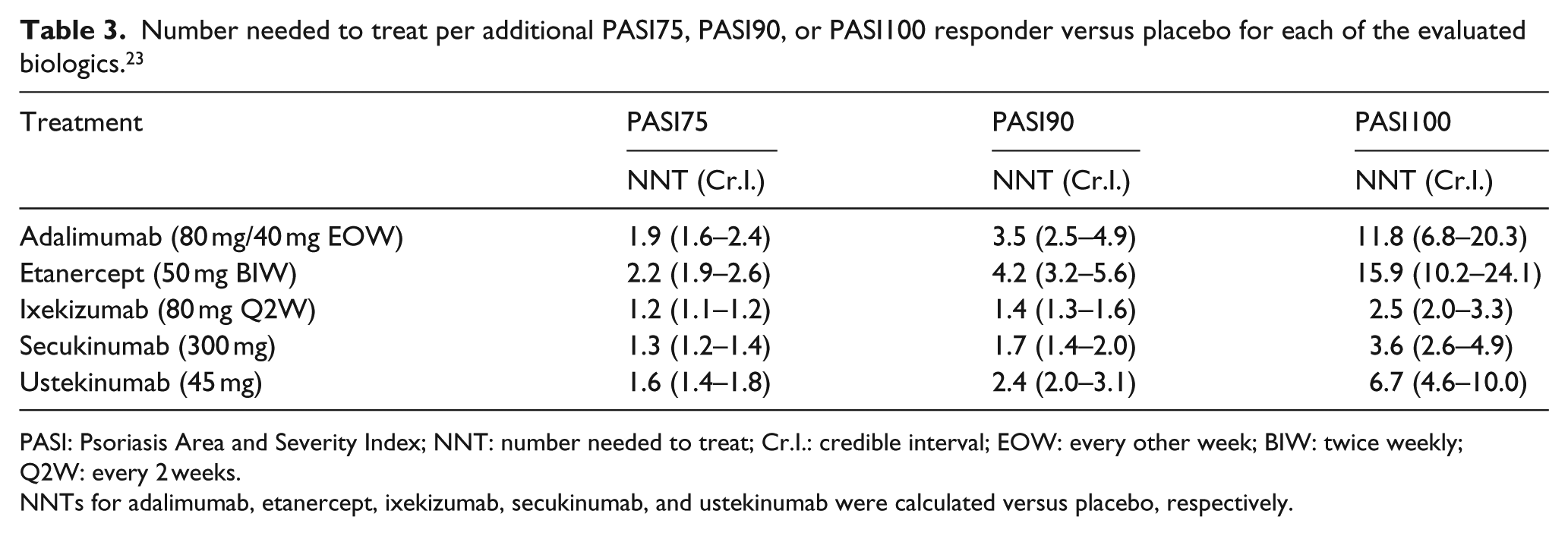
PASI: Psoriasis Area and Severity Index; NNT: number needed to treat; Cr.I.: credible interval; EOW: every other week; BIW: twice weekly; Q2W: every 2 weeks.
NNTs for adalimumab, etanercept, ixekizumab, secukinumab, and ustekinumab were calculated versus placebo, respectively.
Conditional probabilities of achieving PASI75, PASI90, or PASI100 response versus placebo for the evaluated biologics. 23
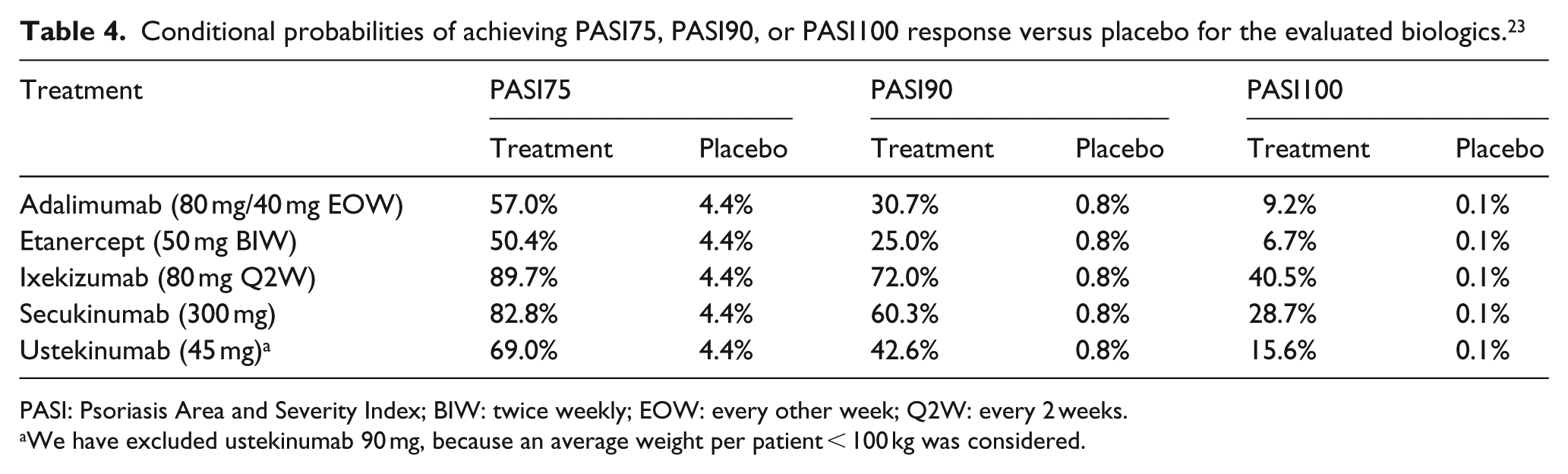
PASI: Psoriasis Area and Severity Index; BIW: twice weekly; EOW: every other week; Q2W: every 2 weeks.
We have excluded ustekinumab 90 mg, because an average weight per patient < 100 kg was considered.
We considered a longer time frame compared to the one reported in the NMA, which only looked at the first 12–16 weeks of therapy (induction period) of randomized clinical trials. 24 We hypothesized that response rates 24 and NNT values 23 will remain constant over the first and the second (or subsequent) year of treatment.
Treatments and costs
Of the biologic drugs currently available in Italy for the treatment of moderate-to-severe PsO, subcutaneous adalimumab, etanercept, ustekinumab, secukinumab, and ixekizumab were included in the analysis. Infliximab was not considered due to its different administration route (infusion). Biologics treatment costs were calculated for the total number of doses administered during the first (induction plus maintenance period) and the second years (maintenance period only) of treatment.
Calculation of costs of biologic therapies was based on the following assumptions: complete adherence to the indicated doses during the evaluated period, an ex-factory price per pack net of temporary discounts by law (AIFA Resolution of 3 July 2006, Official Gazette n° 156 of 7 July 2006, and subsequent AIFA Resolution of 9 February 2007, Official Gazette n° 57 of 9 March 2007 and extensions thereof), and any other discounts agreed with Italian NHS facilities.26,27
Administration and monitoring costs were not included in the analysis because, using the same route of administration (subcutaneous) for all biologic drugs, no difference in the consumption of resources was assumed.
Sensitivity analysis
A sensitivity analysis was conducted in order to evaluate the validity of the base case results 28 and was focused on the NNT values estimated by the US study. 23 All the NNT values were varied simultaneously to assess the impact of such changes on the mean cost per NNT. To this purpose, parameters were given the lower value of the respective 95% credible interval (Cr.I.) in a former round and the higher value in the latter (95% Cr.I. values are reported in Table 3).
Results
Cost per responder in the first year of treatment
Separately for each of the three PASI responses, Table 5 shows the mean cost per responder in the first year of treatment for each of the five biologics considered. In this analysis ixekizumab appears to be associated with the least mean cost per responder among all comparators.
Cost per responder in the first year of treatment.
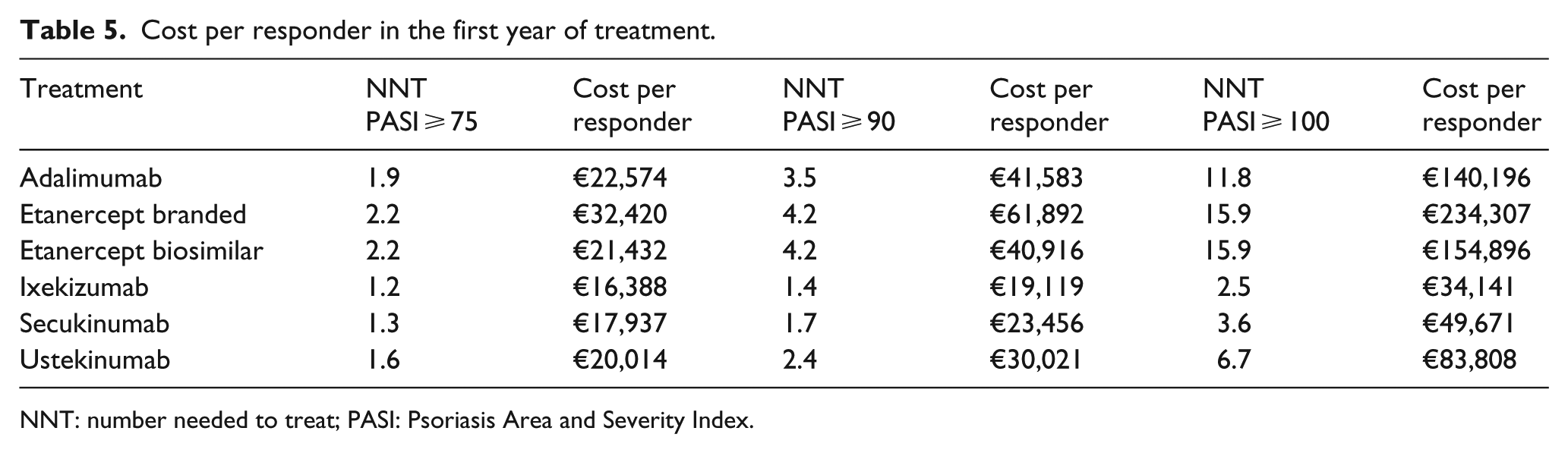
NNT: number needed to treat; PASI: Psoriasis Area and Severity Index.
Overall, the differences in the mean cost per responder between ixekizumab and the other biologic therapies increase with higher PASI thresholds. The observed differences between ixekizumab and the other biologic therapies were smallest compared to secukinumab and highest with etanercept. For PASI75, the difference in the mean cost per responder between ixekizumab and the other biologics ranges from €1549 (vs secukinumab) to €16,032 (vs branded etanercept) (Figure 1); for PASI90, differences range from €4336 (vs secukinumab) to €42,773 (vs branded etanercept) (Figure 2). Considering PASI100, cost differences range from €15,529 (vs secukinumab) to €200,166 (vs branded etanercept) (Figure 3).
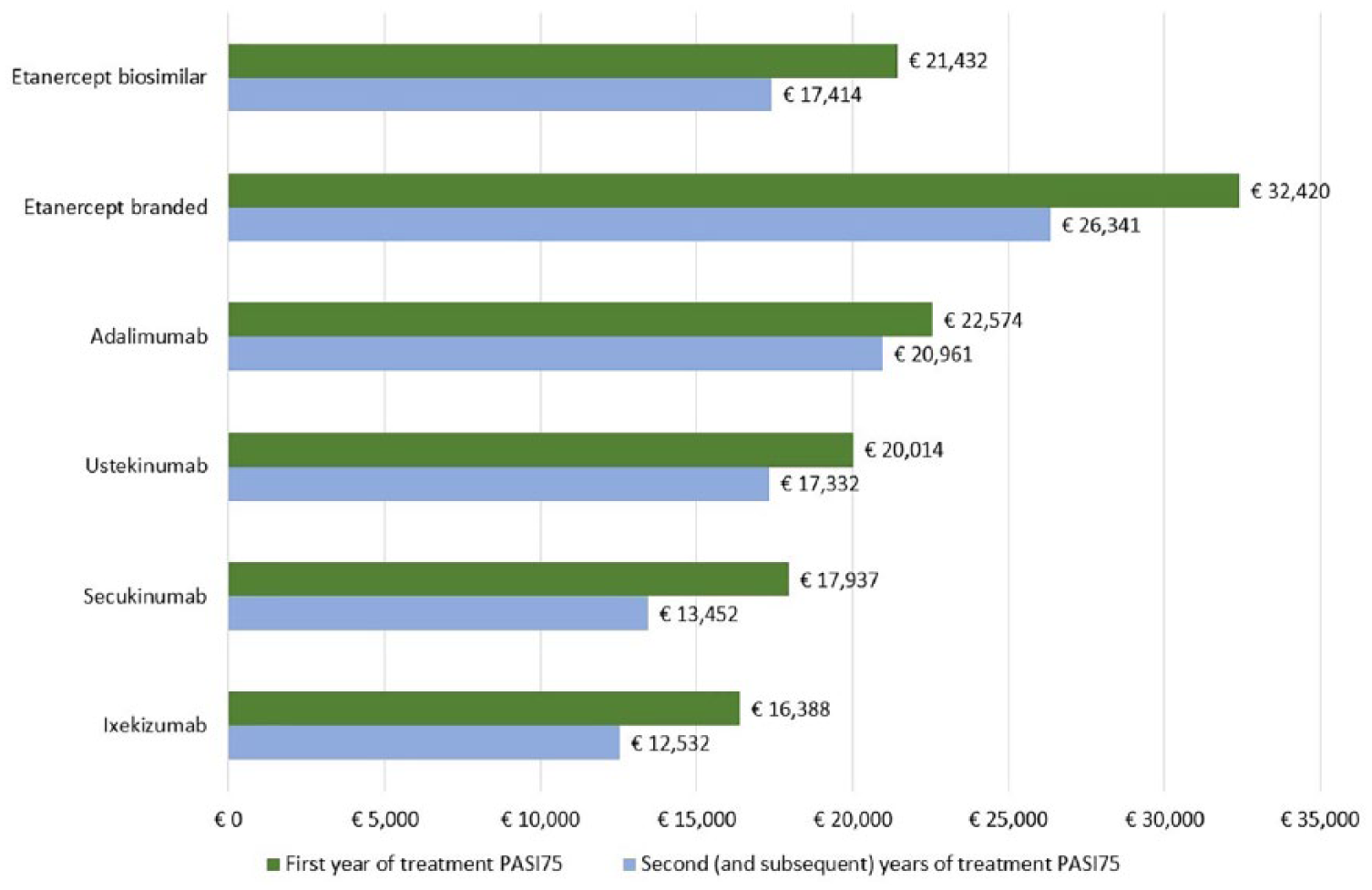
Cost per responder PASI75 in the first year of treatment (induction plus maintenance period) and in the second (and subsequent) year of treatment (maintenance period only) for each of the evaluated biologics.
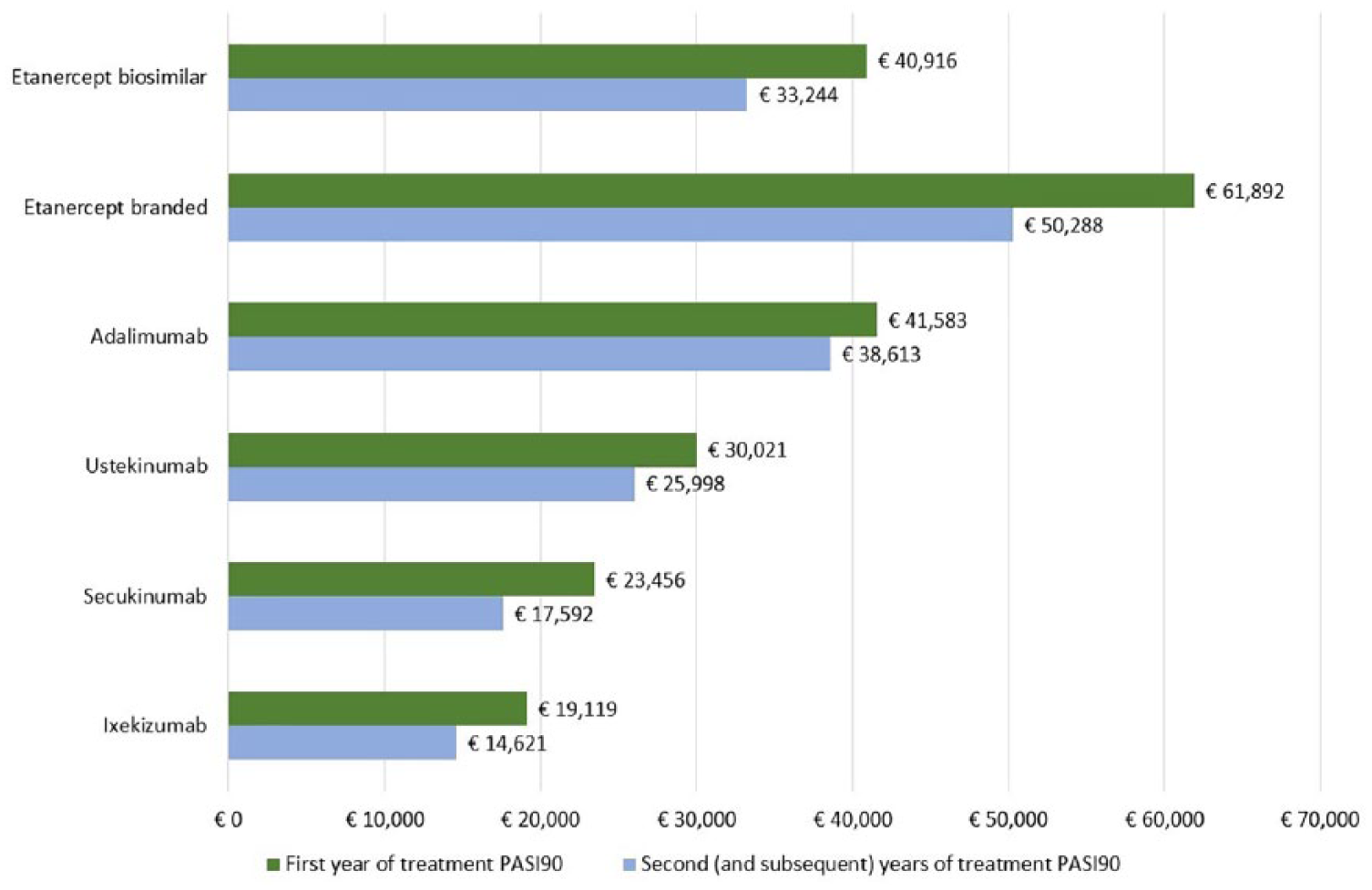
Cost per responder PASI90 in the first year of treatment (induction plus maintenance period) and in the second (and subsequent) year of treatment (maintenance period only) for each of the evaluated biologics.
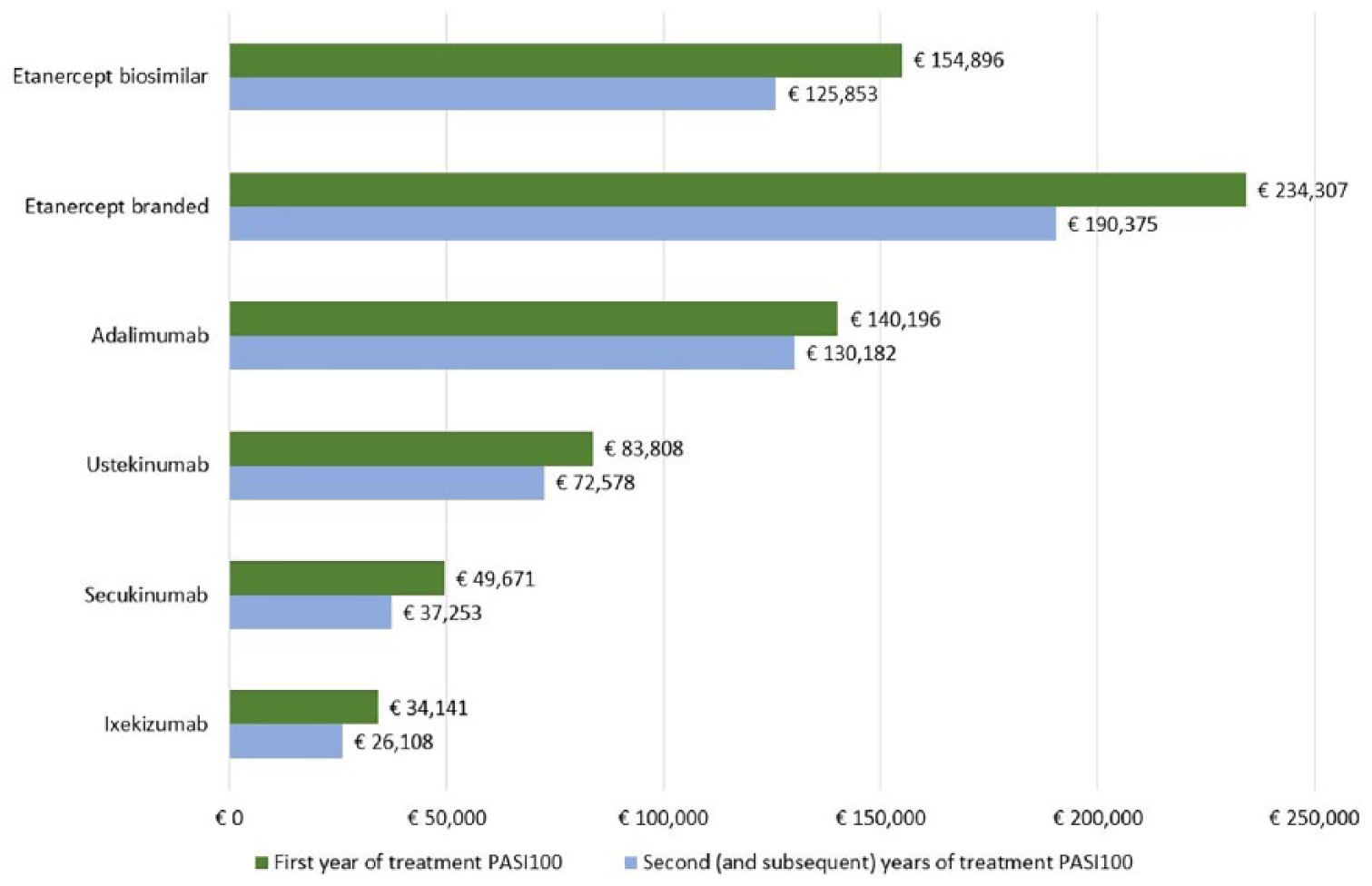
Cost per responder PASI100 in the first year of treatment (induction plus maintenance period) and in the second (and subsequent) year of treatment (maintenance period only) for each of the evaluated biologics.
Cost per responder in the second (and subsequent) year of treatment
Table 6 reports the mean cost per responder for each of the five biologics in the second (or subsequent) year of treatment across all PASI responses. In line with the results for first-year cost per responder, ixekizumab shows the lowest mean cost per responder. In line with the outcomes in the first year of treatment, cost differences in favor of ixekizumab versus its competitors increase with increase in the PASI response. Again, the smallest differences appear in comparison with secukinumab.
Cost per responder in the second (and subsequent) year of treatment.
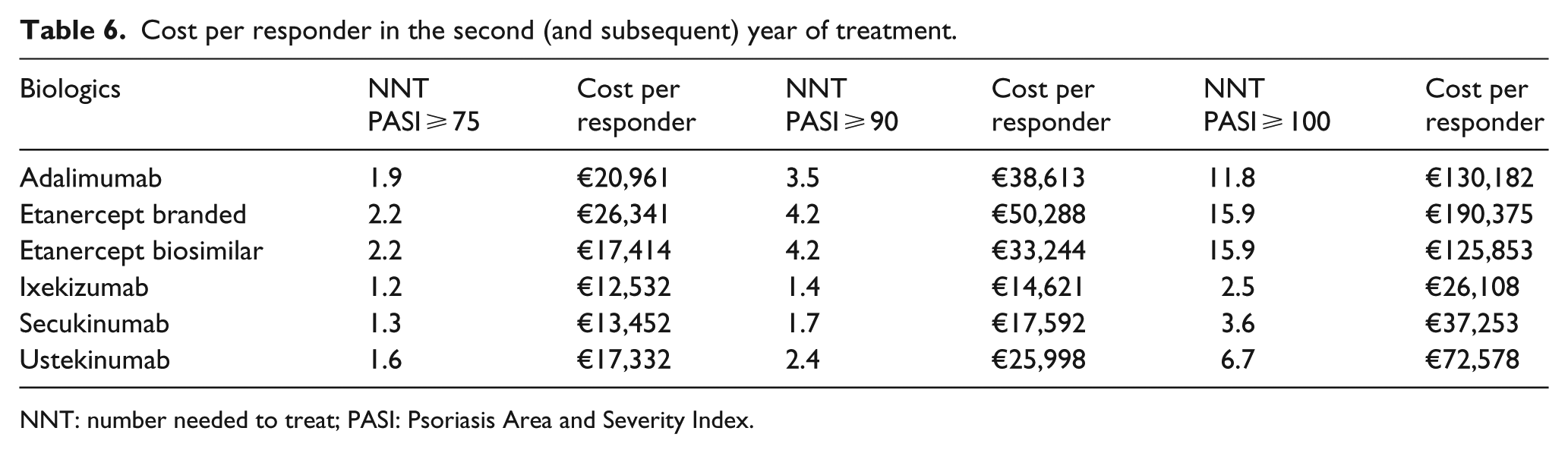
NNT: number needed to treat; PASI: Psoriasis Area and Severity Index.
Based on PASI75, the difference in the mean cost per responder in favor of ixekizumab ranges from €921 (vs secukinumab) to €13,809 (vs branded etanercept) (Figure 1). With PASI90, cost differences vary from €2791 (vs secukinumab) to €35,667 (vs branded etanercept) (Figure 2). For PASI100, the respective values range between €11,145 (vs secukinumab) and €164,267 (vs branded etanercept) (Figure 3).
Sensitivity analysis
Table 7 shows the results of the sensitivity analysis where, for re-evaluating the NNT values, the lower and upper limits of the respective 95% Cr.I. values were adopted. In all comparisons, ixekizumab remains the therapy with the least mean cost per responder.
Sensitivity analysis (95% credible intervals).
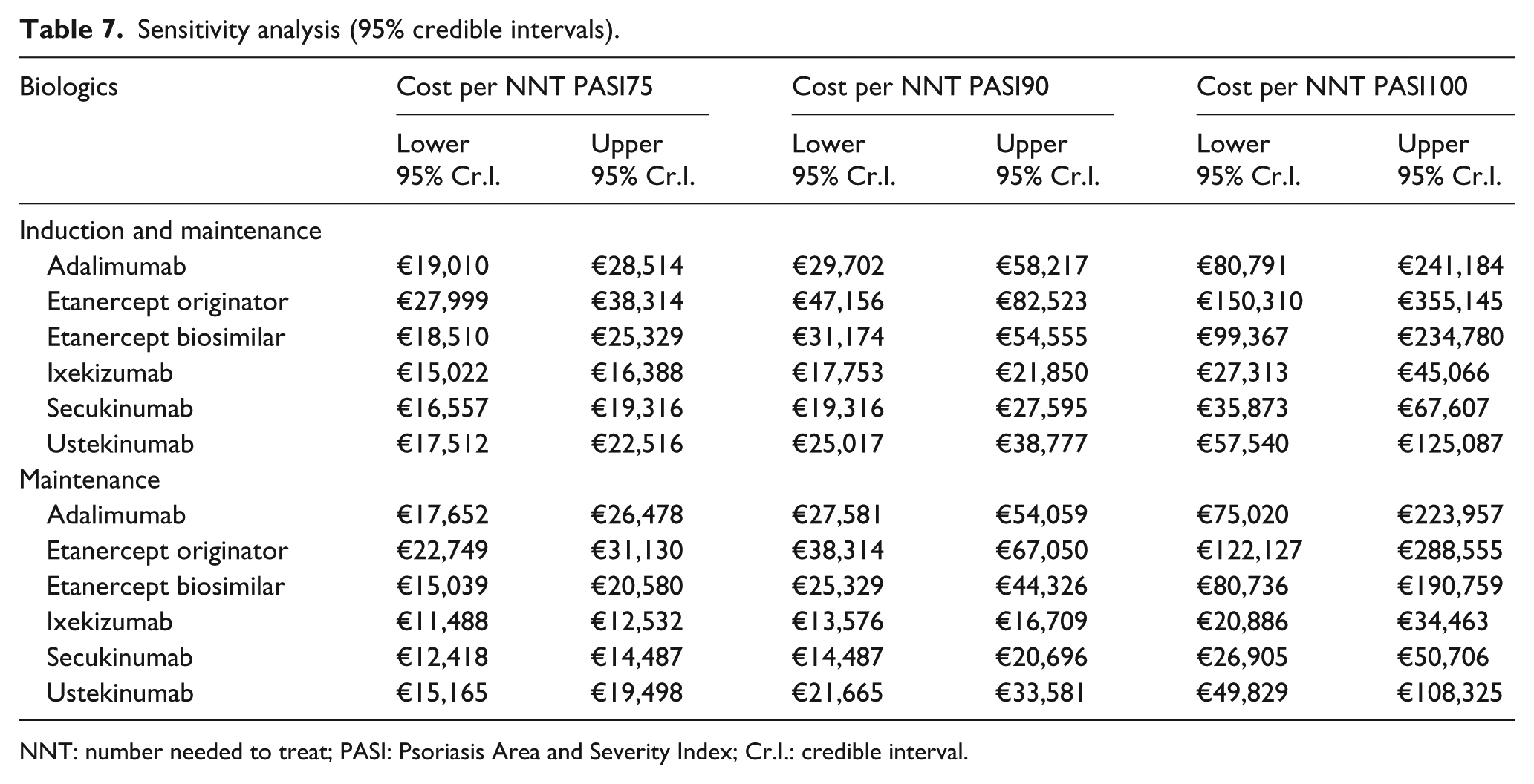
NNT: number needed to treat; PASI: Psoriasis Area and Severity Index; Cr.I.: credible interval.
Discussion
As new biologics are being approved, adding to the number of treatment options for the management of PsO, information on comparative treatment costs is increasingly important for decision-makers. NMA is a statistical approach to generate comparative efficacy data when head-to-head trials are not available for all the treatments considered.29,30 Using this method, Hartz et al. 24 recently highlighted the superiority of ixekizumab across all PASI response criteria (PASI75, PASI90, and PASI100) compared to placebo, secukinumab, ustekinumab, adalimumab, and etanercept. Based on the NNT values quantified in the US cost analysis, 23 the cost per responder (as compared to placebo) was calculated, as a combined indicator of both the clinical benefit and the treatment cost associated with each of the five biologic subcutaneous drugs approved for the treatment of moderate-to-severe PsO in Italy.
Independent of the PASI response criteria used to calculate the NNT, ixekizumab yielded the numerically lowest mean cost per responder, in both the first (induction plus maintenance period) and the second (maintenance period only) years of treatment. The results from this analysis show a more favorable difference between the mean cost per responder of ixekizumab and the mean cost per responder of the other biologic drugs evaluated, which increase with using more stringent PASI response criteria (PASI90 and PASI100). According to the European S3-Guidelines, reaching a PASI75 response has been accepted as the minimum clinically significant improvement.13,31,32 Important in this context is the impact of skin improvement on the patients’ health-related quality of life (HRQoL). In fact, achieving clear or almost clear skin (PASI90 and PASI100) has been shown to better correlate with HRQoL improvement and therefore should become the new standard therapeutic target.33,34
Our results are in line with a recent Spanish analysis where, based on the same methods, the highest cost-effectiveness (mean cost per responder) was found for ixekizumab versus adalimumab, etanercept, secukinumab, and ustekinumab in the treatment of moderate-to-severe PsO in both the first (induction plus maintenance period) and the following (maintenance period only) treatment years. 35
A limitation of this analysis is owed to the fact that efficacy results from a NMA and not from all head-to-head comparisons were used. However, a wide range of sensitivity analysis has confirmed NMA reliable to achieve solid results. 24
A second limitation refers to the long-term extrapolation of NMA’s short-term clinical outcomes. While for the NMA only the induction period (12–16 weeks) was considered, which prevented the assessment of the relative efficacy of ixekizumab (and other biologic drugs) over longer time periods, we assumed that response rates 24 and related NNT values 23 remain constant over the first and the second (or subsequent) years of treatment. Such assumption was made with the purpose of providing decision-makers with data for a comparison not limited to the induction period, but extended to a longer time horizon (at least 12–24 months) so that it can serve for drawing up spending budgets. Unfortunately, there are no published studies that could confirm 12- and 24-month effectiveness.
Although biologic drugs are associated with significant improvements in QoL for patients with PsO, 15 the current analysis did not take into account any measurement of HRQoL. Due to the same route of administration for all biologic drugs considered in this analysis, costs associated with administration and monitoring were excluded.
Conclusion
In this analysis, ixekizumab consistently emerged as the best treatment for moderate-to-severe PsO when compared to adalimumab, etanercept, secukinumab, and ustekinumab. This cost-per-responder assessment indicates ixekizumab as the most cost-effective treatment option from the perspective of the Italian NHS.
