Abstract
Biological disease-modifying antirheumatic drugs are recommended for use particularly in rheumatoid arthritis patients who had an inadequate response to methotrexate but they are costly. In this article, we estimated the number needed to treat and treatment costs associated with the use of baricitinib and adalimumab in moderate to severe rheumatoid arthritis patients who are inadequate responders to methotrexate. We used Efficacy data (American College of Rheumatology indices) from the RA-BEAM trial. For both DMARDs, the number needed to treat was calculated with reference to placebo, using the (American College of Rheumatology indices (ACR20, ACR50, and ACR70 response at 24 weeks)). Direct medical costs (therapy costs of medication) were the only costs considered in the base case analysis. However, administration and monitoring costs were included in the sensitivity analyses. Baricitinib had a lower cost per number needed to treat (ACR20: €10,846.08; ACR50: €12,942.72; and ACR70: €18,506.88) compared to adalimumab (ACR20: €21,124.45; ACR50: €23,945.22; and ACR70: €45,445.77). The most significant cost difference in favor of baricitinib was observed using ACR70 index (−€26,938.89). Sensitivity analysis confirmed these results. This analysis demonstrates that baricitinib represents a more cost-effective option in comparison to adalimumab in the treatment of rheumatoid arthritis patients with an inadequate response to methotrexate in Italy and based on Italian costs.
Keywords
Introduction
Rheumatoid arthritis (RA) is a chronic inflammatory autoimmune disease. It is characterized by a persistent synovitis, progressively causing cartilage destruction and bone erosion, leading to joint deformation and disability.1,2 RA patients may have highly variable patterns of radiologic progression over time. In any case, joint damage and functional status loss appear since the early disease stages.3,4 RA can potentially involve other organs, causing, for instance, severe respiratory and/or cardiovascular complications; for this reason, a higher mortality risk (as compared with general population) is associated with RA.5,6
On average, RA affects about 0.5%–1% of the adult population,1,2 with variations across countries (Japan 0.2%, The Netherlands 1%–1.5%, Scandinavia 4%, Spain 0.5%, and the United States 1%).7,8 According to an estimate provided by two regional investigations, the RA prevalence rate in Italy is approximately 0.48%. 9
Although RA management may include non-pharmacologic treatments and non-steroidal anti-inflammatory drugs and/or glucocorticoids, the first prescribed drugs are conventional synthetic disease-modifying antirheumatic drugs (csDMARDs), such as methotrexate (MTX). Aiming to optimize the clinical response and reduce long-term disability, the therapeutic approach in the latest years was directed toward prescribing biological disease-modifying antirheumatic drugs (bDMARDs).10–14 These drugs are, in fact, an efficacious treatment option, especially recommended for patients with a poor response to or intolerance of csDMARDs or for whom continued treatment with csDMARDs would be inappropriate.15,16
Baricitinib, a selective inhibitor of JAK1/JAK2 enzymes, is approved in over 40 countries including European countries, the United States, and Japan for the treatment of moderately to severely active RA in adult patients who have had an inadequate response to or intolerance to one or more disease-modifying antirheumatic drugs. This targeted synthetic disease-modifying antirheumatic drug (tsDMARD), administered orally, can be used in monotherapy or in combination with MTX. The efficacy and safety of baricitinib were assessed in four randomized clinical trials where more than 3000 patients were enrolled.17–20 One of the trials, the RA-BEAM study, was conducted in patients with moderately to severely active RA with an inadequate response to MTX. The trial compared baricitinib to placebo and to adalimumab (an inhibitor of tumor necrosis factor (TNF)-α) as the standard of care (SOC) bDMARDs for patients with moderately to severely active RA. 20 Overall, 1307 patients from 281 centers in 26 countries were randomized to receive (in addition to MTX) placebo, baricitinib 4 mg once daily, or subcutaneous adalimumab 40 mg every other week. At week 12, the primary endpoint ACR20 response (20% improvement in the disease activity) was achieved by a larger number of patients treated with baricitinib as compared both with placebo (70% vs 40%; p < 0.001) and adalimumab (70% vs 61%; p = 0.014). 20 The adverse events profile of baricitinib was similar to adalimumab. 20
Objective
Using data on efficacy from the RA-BEAM trial, 20 the present economic analysis assessed and compared the number needed to treat (NNT) and costs per NNT for baricitinib and adalimumab in adult patients with moderately to severely active RA with an inadequate response to MTX in Italy.
Materials and methods
The indices defined by the American College of Rheumatology (ACR) are among the most adopted tools in assessing the clinical response to a drug therapy administered in RA. They are composite scores that measure the treatment efficacy evaluating a defined number of disease activity variables (the “core set”), such as tender and swollen joints count, functional ability, acute phase reactants (erythrocyte sedimentation rate and C-reactive protein (CRP)), the judgment about the disease severity given by both physician and patient, and the judgment on the intensity of pain perceived by the patient.21,22 ACR indices are based on the relative variation of the core set variables between the respective initial and final values, classifying the response to treatment as a 20% improvement (ACR20), 50% improvement (ACR50), or 70% improvement (ACR70). 21
Clinical data
The RA-BEAM ACR20, ACR50, and ACR70 primary efficacy outcomes were used in this economic analysis. 20 The trial compared baricitinib versus placebo and versus adalimumab (each of the three in addition to the background therapy, MTX), in the treatment of patients with moderately to severely active RA with an inadequate response to MTX. With respect to both placebo and adalimumab, baricitinib showed higher response rates. Table 1 reports the respective ACR20, ACR50, and ACR70 responses at 24 weeks.
ACR20, ACR50, and ACR70 response rates (RA-BEAM trial at week 24 20 ).
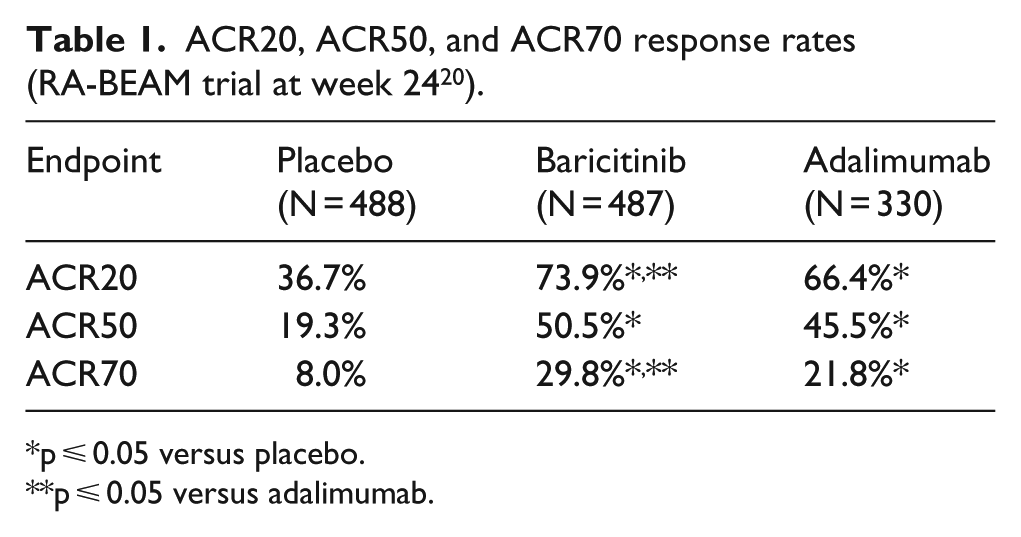
p ⩽ 0.05 versus placebo.
p ⩽ 0.05 versus adalimumab.
Evaluation technique
The costs per NNT of baricitinib versus placebo and that of adalimumab versus placebo were estimated.23,24 The NNT is the number of patients needing to be treated in order to get a therapeutic benefit—in our case, how many patients have to be treated with baricitinib (or adalimumab) as compared with placebo so that one additional response/patient can be gained (as assessed by the ACR20, ACR50, or ACR70 indices). The closer the value of the NNT is to 1, the more favorable the outcome. Cost per NNT for each drug was calculated by multiplying the NNT by the corresponding average treatment cost.
Time horizon
With the purpose of calculating the NNT of baricitinib and adalimumab versus placebo, the time horizon was set equal to the follow-up length for which efficacy data are available of all the three treatment options. 20 Although the RA-BEAM study is a 52-week trial, all patients treated with placebo switched to baricitinib at week 24. Therefore, due to switch in therapy, the economic analysis could not be conducted in data beyond 24 weeks, as the respective NNTs of baricitinib and adalimumab are calculated versus placebo. Nevertheless, on the basis of the published literature, 25 it can be argued that a 6-month period is enough to capture the most important differences in terms both of efficacy (NNT) and of treatment costs between the two drugs considered.
Analysis perspective
The perspective adopted in the economic analysis is the Italian National Health Service (NHS) perspective. Only direct medical costs related to the drug therapy (acquisition cost of baricitinib and adalimumab) were considered in the base case.
Costs of MTX, adverse events management, and administration and monitoring of drugs were not included in the analysis. Since MTX was the background therapy for all patients enrolled in the RA-BEAM study, 20 no cost for MTX administration was considered. Costs of adverse events management were also not considered because the baricitinib and adalimumab safety profiles found in the RA-BEAM study 20 are deemed similar. Although baricitinib and adalimumab have different administration routes (oral and subcutaneous, respectively), it was conservatively hypothesized, in the base case, that adalimumab is taken by patients at home—so charging no cost to the NHS, as is the case with baricitinib. Based on this assumption, administration costs for either drug were not considered in the analysis. Finally, monitoring costs were also not considered in the base case as no published literature exists on costs of monitoring between a bDMARD administered subcutaneously and a tsDMARD administered orally.
Treatment cost
In the RA-BEAM trial, 20 the dosage for adalimumab was 40 mg given subcutaneously every other week; for baricitinib, the dosage was 4 mg daily. The cost per pack used in the analyses corresponded to the ex-factory price net of any discounts by law (AIFA Resolution of 3 July 2006, Official Gazette no. 156 of 7 July 2006, and subsequent AIFA Resolution of 9 February 2007, Official Gazette no. 57 of 9 March 2007 and extensions thereof), excluding any other discounts agreed with NHS facilities.
Sensitivity analysis
The sensitivity analysis, conducted to assess the robustness of the base case results, is mainly focused on the efficacy data from the RA-BEAM trial. 20 In addition to cost per NNT using ACR indices, costs per NNT were calculated using the following secondary endpoints in RA-BEAM: DAS28 (Disease Activity Score for 28 joints)-CRP ⩽ 3.2, DAS28-CRP < 2.6, DAS28-ESR (erythrocyte sedimentation rate) ⩽ 3.2, DAS28-ESR < 2.6, SDAI (Simplified Disease Activity Index) ⩽ 11, SDAI ⩽ 3.3, CDAI (Clinical Disease Activity Index) ⩽ 10, and CDAI ⩽ 2.8. 20 The values of these endpoints at week 24 are shown in Table 2, respectively, for placebo, baricitinib, and adalimumab.
Further endpoints (RA-BEAM trial at week 24 20 ).
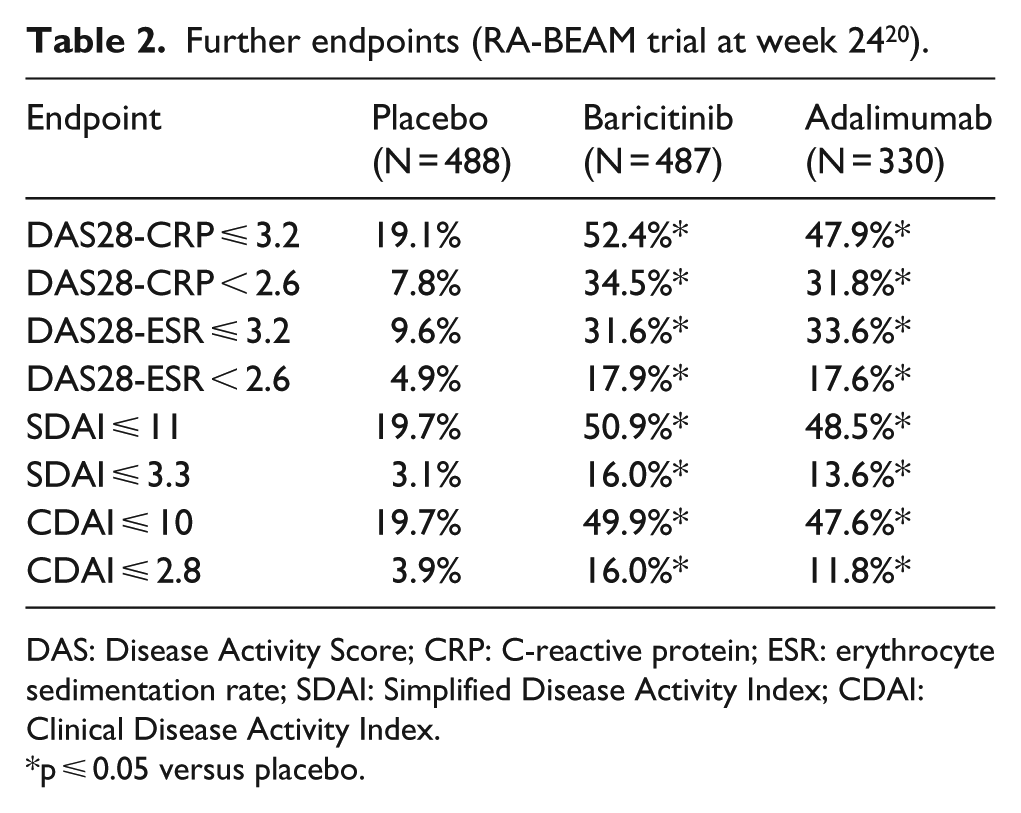
DAS: Disease Activity Score; CRP: C-reactive protein; ESR: erythrocyte sedimentation rate; SDAI: Simplified Disease Activity Index; CDAI: Clinical Disease Activity Index.
p ⩽ 0.05 versus placebo.
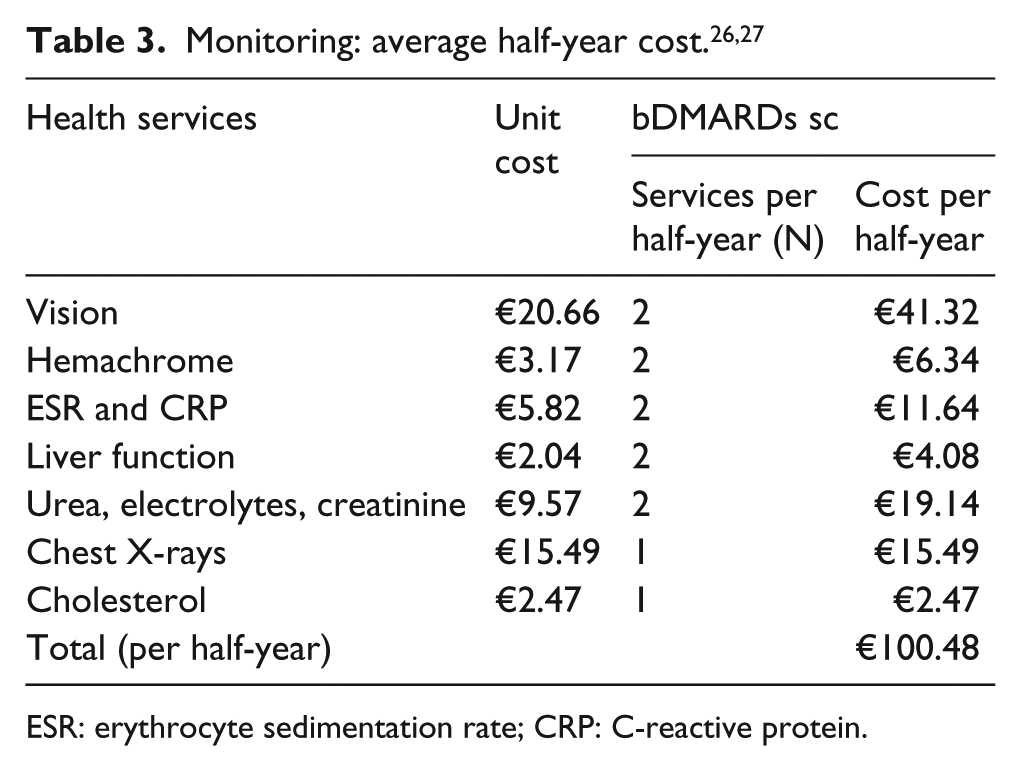
In addition, a scenario analysis was conducted where, in addition to the cost for drugs, costs borne by the NHS for administration and monitoring were included. The administration cost was charged to adalimumab only, since baricitinib is administered orally. Each administration activity (injection) was evaluated adopting as a proxy the tariff of €6.97 for a “subcutaneous or intramuscular injection or infusion of therapeutic or prophylactic substances” as reported in the national tariff list for specialist outpatient healthcare services (code 83.98). 26 Since an analysis assessing the economic impact of monitoring an oral tsDMARD is not available in the literature, the monitoring cost estimated for adalimumab in a recent investigation in Italy 27 was assumed for baricitinib. Table 3 reports the health resources consumption, the unit cost adopted to evaluate each service (national tariffs 26 ), and the average cost per 6 months used to evaluate the monitoring activity. 27
ESR: erythrocyte sedimentation rate; CRP: C-reactive protein.
Finally, a threshold analysis was conducted aimed at quantifying the relative price variation by which adalimumab is associated with lower costs per NNT as compared with baricitinib.
Results
NNT
The NNT for baricitinib and adalimumab versus placebo for ACR20, ACR50, and ACR70 are reported in Table 4. The data show, that for all three ACR indices, baricitinib has lower NNT values as compared with adalimumab.
Number needed to treat.
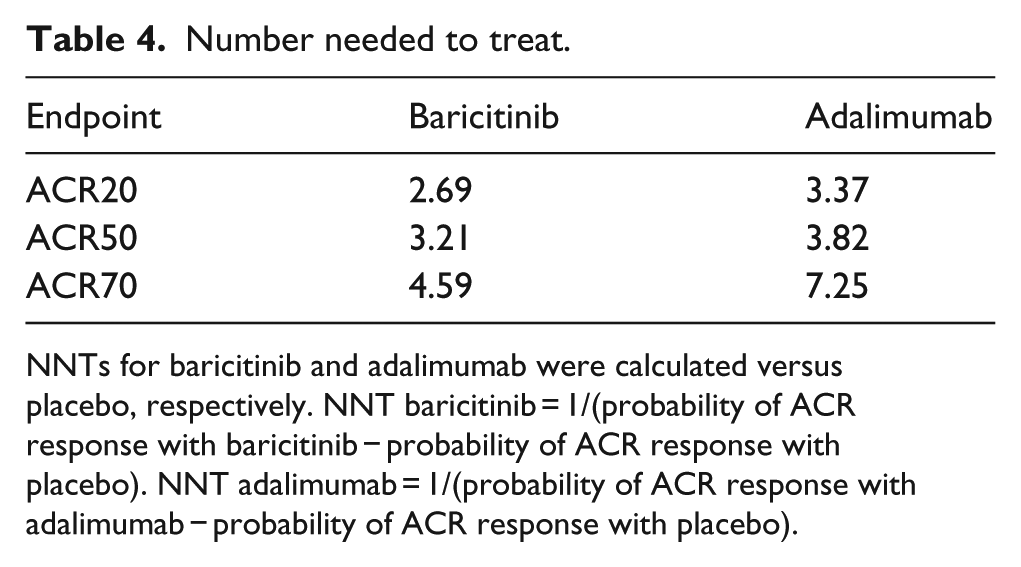
NNTs for baricitinib and adalimumab were calculated versus placebo, respectively. NNT baricitinib = 1/(probability of ACR response with baricitinib − probability of ACR response with placebo). NNT adalimumab = 1/(probability of ACR response with adalimumab − probability of ACR response with placebo).
Average treatment cost
The average 6-month cost per patient treated with baricitinib was €4032.00 (regardless of the type of packaging considered), which is lower than the analogous cost with adalimumab which was €6268.38 (with a pack of two pre-filled syringes). These costs refer to the pharmacological therapy cost only, based on the acquisition price and the administration dosage. As compared with adalimumab, baricitinib is associated with a significant cost saving of €2236.38 per patient treated for 6 months.
Cost per NNT
Figure 1 shows the comparison between baricitinib and adalimumab in terms of the average cost per NNT based on ACR20, ACR50, and ACR70, at week 24. With respect to the percentage of patients reaching an ACR response, baricitinib consistently has a lower average cost per NNT. Overall, cost differences between the two drugs are large and increase with the efficacy level measured with ACR indices. The highest advantage for baricitinib versus adalimumab is shown with ACR70 response (€26,938.89).
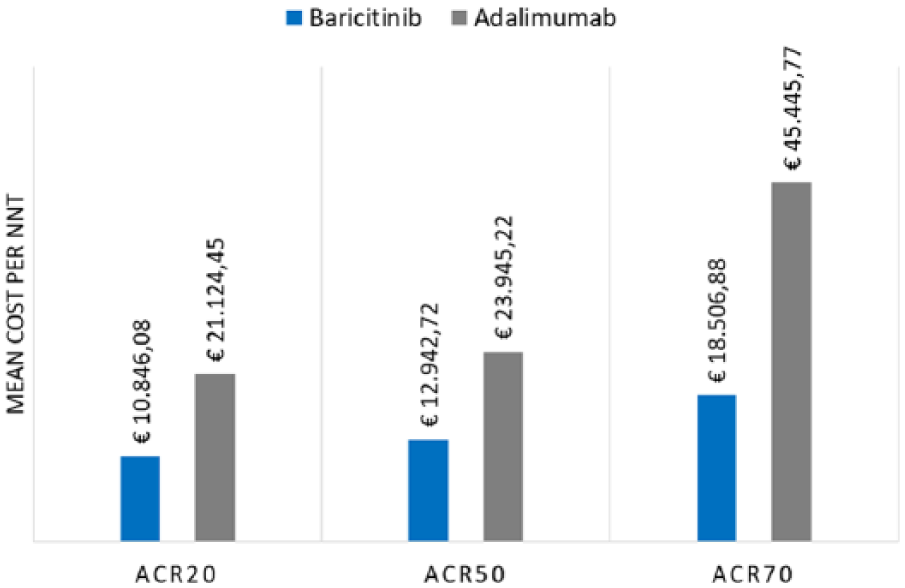
Mean cost per NNT.
Sensitivity analysis
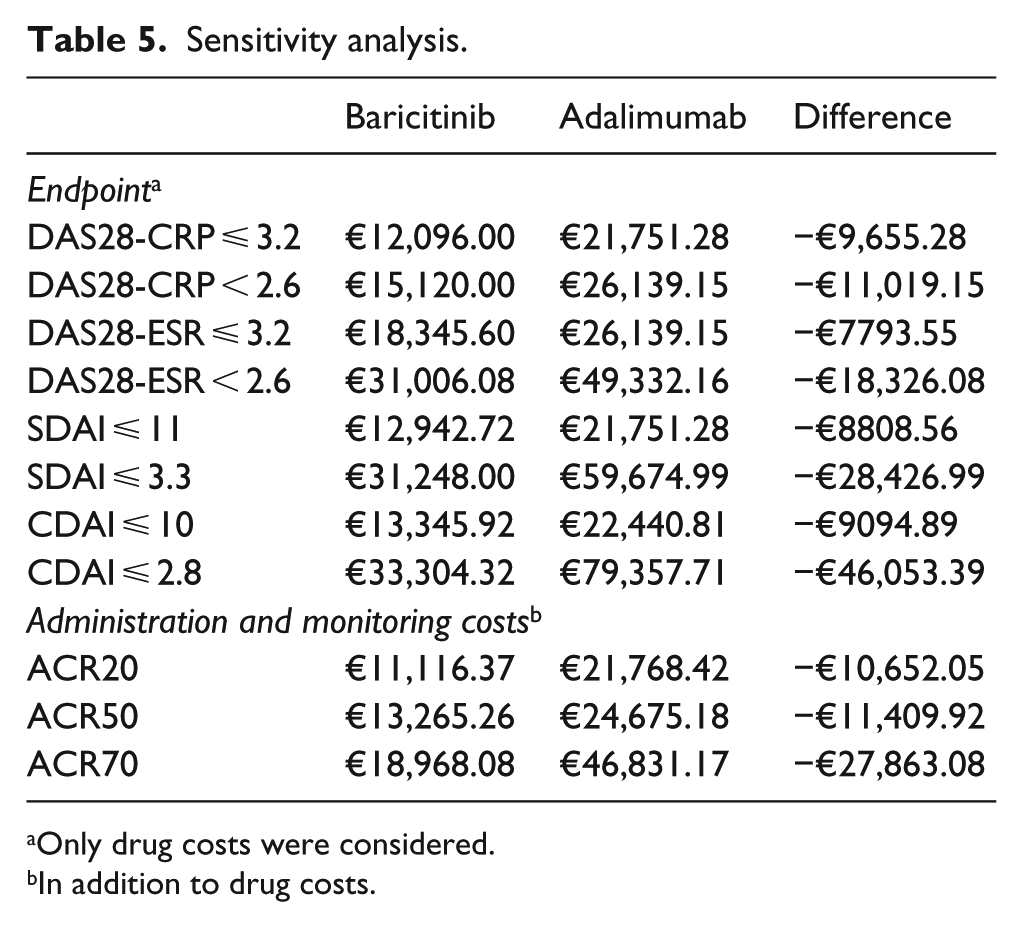
Table 5 shows the results of costs per NNT based on secondary endpoints (DAS28-CRP ⩽ 3.2, DAS28-CRP < 2.6, DAS28-ESR ⩽ 3.2, DAS28-ESR < 2.6, SDAI ⩽ 11, SDAI ⩽ 3.3, CDAI ⩽ 10, and CDAI ⩽ 2.8). The data show that, even on these secondary endpoints, baricitinib is always associated with a lower average cost per NNT compared to adalimumab.
Sensitivity analysis.
Only drug costs were considered.
In addition to drug costs.
Table 5 also reports the results from the scenario analysis where, in comparison to the base case, the costs for administration (adalimumab only) and for monitoring (baricitinib and adalimumab) were included in the analysis. The data show that baricitinib has a lower average cost per NNT than adalimumab in any of the comparisons conducted.
The results from the threshold analysis show that, independently from the ACR index adopted, reductions in adalimumab price were needed (−48.7% with ACR20, –45.9% with ACR50, and –59.2% with ACR70) for this bDMARD to become a cost-effective option in comparison to baricitinib.
Discussion
This analysis used NNT as an indicator to weigh the clinical benefits and costs associated with the use of baricitinib versus adalimumab in the treatment of moderately to severely active RA in adult patients with an inadequate response to methotrexate. The NNT was calculated with reference to the ACR20, ACR50, and ACR70 response rates from the RA-BEAM trial 20 and, together with drug costs, it was utilized to assess the cost-effectiveness (cost per NNT) of the two DMARDs. Independently from the ACR response index adopted (20/50/70), baricitinib was found to have a lower average cost per NNT in comparison to adalimumab. It should be noted that, overall, and as clinical improvement increases, the difference in costs per NNT between the two DMARDs increases, in favor of baricitinib. For example, the difference in cost per NNT was found to be €10,278.37 with the ACR20 index, and that increased to €26,938.89 with the ACR70 index, in favor of baricitinib (Figure 1).
When conducting economic analyses, it is often necessary to use clinical data provided by pooled analyses or by indirect comparisons (possibly implying non-similar populations, non-homogeneous clinical indicators, etc.). This analysis has the advantage to be based on a head-to-head comparison between baricitinib and adalimumab in the population of interest, hence not relying on indirect evidence on the comparison between these two drugs. In line with the RA-BEAM trial, 20 ACR responses were used in the analysis as efficacy measures for which NNT was calculated for the base case. This analysis was based on ACR responses evaluated from one head-to-head trial; further real-life data will be necessary to confirm these results.
In the sensitivity analyses, further secondary efficacy endpoints (e.g. DAS28, CDAI, and SDAI) were used to calculate the cost per NNT. Using the same methods adopted in the base case (ACR responses), the cost per NNT with baricitinib and adalimumab versus placebo was calculated for each secondary endpoint. All the analyses, whether in the base case or the sensitivity analyses, indicated that baricitinib is more cost-effective (lower average cost per NNT) than adalimumab.
For the economic evaluation of health technologies, national 28 and international29,30 guidelines recommend the use of a time horizon that is suitable to capture all major differences in terms of both outcomes and treatment costs. In this analysis, the choice of a 24-week time horizon was due to the follow-up time period for patients treated with placebo in the RA-BEAM trial, 20 where outcomes for those patients were recorded up to 24 weeks only (for baricitinib and adalimumab in RA-BEAM, the follow-up period was 52 weeks). Since the baricitinib and adalimumab NNT was calculated with respect to the response rate in the placebo arm, the analysis could not be conducted beyond 24 weeks. It is worth emphasizing here that, in the literature, 25 a 24-week time horizon is the most frequently used time horizon in the evaluation of drug efficacy in the treatment of RA. Given this, a 24-week time horizon for the cost per NNT analysis is assumed to be sufficient enough to capture major differences in efficacy and treatment costs between baricitinib and adalimumab. 31 The findings of this study need to be confirmed by future clinical observation and real-world analysis with longer time horizons.
In the scenario analysis (sensitivity analysis), monitoring and administration costs were considered in addition to drug costs (base case). This was done to reflect as much as possible costs in real-life clinical practice. With the inclusion of monitoring and administration costs, the difference in cost per NNT was even higher between the two drugs, favoring baricitinib due to its oral administration route, which can be managed by patients more easily and with less health resource consumption than those required by subcutaneous administration.
For both drugs, the ex-factory price was used in the base case analysis, net of temporary discounts by law excluding any other discounts agreed with NHS facilities. The threshold analysis, however, investigated which price would make adalimumab cost-effective in comparison to baricitinib. The results of this analysis showed that only with substantial price reductions (45.9%–59.2%) would adalimumab have costs per NNT that are lower than that estimated for baricitinib (assuming that the price of the latter would keep being equal to the value considered in the base case).
Conclusion
The results from this economic evaluation, based on the average cost per NNT, showed that baricitinib is a more cost-effective therapeutic option when compared with adalimumab—from the Italian NHS perspective—in the treatment of moderately to severely active RA in adult patients with an inadequate response to MTX.
Footnotes
Declaration of conflicting interests
R.R. declares no conflict of interest. S.A., V.R., W.F., J.P.C., and S.L. are full-time employees of Eli Lilly S.p.A.
Funding
This research was supported by an educational grant from Eli Lilly Italy S.p.A.