Abstract
In the present study, polylactic acid (PLA) was incorporated to ameliorate the rheological and mechanical properties of chitosan (CS)-based hydrogels. A novel injectable chitosan/polylactic acid (CS/PLA) nanofiber composite hydrogel, fabricated via the electrospinning technique, was developed for the sealing and repair of fistulas, thereby functioning as an innovative biomaterial for the treatment of tracheoesophageal fistula (TEF). Experimental data demonstrated that the CS/PLA composite nanofiber hydrogel exhibits superior mechanical properties, favorable rheological behavior, and prominent antimicrobial activity. At the optimal ratio, the compressive strength and tensile strength of CS/PLA composite nanofiber hydrogel is 43.7 MPa and 1.38 MPa, and the degradation rate is 68.9% after 10 days. Meanwhile, the antibacterial rates against E. coli and S. aureus CS/PLA composite nanofiber hydrogel reached 82.6% and 76.3%, respectively. In vitro experiments were performed to assess the biocompatibility and cell proliferation capacity of the composite hydrogel. The results revealed that the CS/PLA composite nanofiber hydrogel can effectively facilitate the proliferation and migration of target cells, while simultaneously exhibiting favorable biocompatibility. These findings indicate that the CS/PLA composite nanofiber hydrogel possesses considerable application potential in TEF repair, thereby offering an innovative and efficient biomaterial option for clinical intervention of TEF.
Introduction
Tracheoesophageal fistula (TEF) is an abnormal passage between the trachea and the esophagus caused by esophageal developmental defects or diseases.1–3 The presence of this fistula alters the normal physiological structure between the trachea and esophagus, leading to the entry of esophageal and gastric contents into the trachea through the fistula. In mild cases, this pathological condition may lead to recurrent pulmonary infections and hypoxic states; in severe cases, it can cause airway obstruction by ingested food, thereby posing a direct threat to the patient’s life. With the rapid advancement of intensive care medicine, the diagnostic rate of TEF has been significantly improved, and its pathogenesis as well as clinical therapeutic strategies have gradually become a research focus of increasing interest in the medical community. Therefore, determining the safest and most effective treatment for TEF has been a focus of research. Currently, common treatment methods include conservative treatment, surgical repair, and esophageal stent insertion, but each of these methods has its limitations.4–6 Conservative treatment is suitable for patients with mild cases or small fistulas. It can only alleviate symptoms and does not fully resolve the fistula issue. For most patients, conservative treatment cannot effectively prevent complications such as recurrent pulmonary infections, aspiration, or malnutrition, and it requires long-term monitoring and comprehensive management. 7 Surgical repair is the traditional treatment for tracheoesophageal fistula, particularly for more severe congenital and acquired cases of TEF. 8 However, surgical repair carries certain risks, especially in patients with underlying conditions or those in a critical state. The recovery period after surgery can be prolonged, and there may be risks of postoperative recurrence or complications, such as infections, fistula recurrence, or esophageal stricture. 9
In recent years, the use of biomaterials for sealing and repairing tracheoesophageal fistulas has garnered increasing attention in medical research and clinical practice. 10 This approach aims to provide an alternative to traditional surgical repair by utilizing advanced biomaterials that can effectively block the abnormal passage between the trachea and esophagus. Biomaterials, such as biodegradable polymers, hydrogels, and tissue-engineered scaffolds, have shown promise in promoting tissue regeneration, facilitating healing, and reducing the risk of complications associated with conventional surgery.11–13 Yunlang et al. 14 used 3D printing technology to fabricate a polycaprolactone (PCL) scaffold with a ring-like hollow alternating structure, and filled the hollows between the PCL rings with collagen sponges via casting and freeze-drying. This biomimetic scaffold was repopulated with chondrocytes in vitro, developing into tracheal cartilage. In vivo experiments showed significant deposition of tracheal cartilage and the formation of a biomimetic trachea that mimicked the structure and mechanics of the natural trachea. Long-segment tracheal replacement experiments conducted in a rabbit model demonstrated that the engineered biomimetic trachea led to satisfactory repair outcomes. Ni et al. 15 introduced lithium phyllosilicate (LP) nanoclay to improve the rheological and mechanical properties of chitosan-based hydrogels. They developed an endoscopically injectable nanocomposite shear-thinning hydrogel to seal and repair fistulas, presenting it as an innovative material for the treatment of TEF. Hydrogels due to their excellent plasticity, ability to control drug release, and tissue regeneration-promoting properties, have gradually emerged as one of the promising materials for TEF repair. In addition to effectively sealing the fistula, biomaterials such as hydrogels can also promote the healing of the surrounding tissues, supporting the natural repair process and reducing the risk of complications. 16
Chitosan (CS) is a natural polysaccharide with excellent biocompatibility, antimicrobial properties, and biodegradability. 17 Owing to these prominent biological and physicochemical properties, CS has been extensively applied in the medical field for the fabrication of diverse biomedical materials, including but not limited to bone regeneration scaffolds, artificial heart valve components, controlled drug delivery systems, and bioactive wound dressings.18,19 Liu et al. 20 designed and developed a bionic chitosan self-healing hydrogel that exhibits strain hardening properties under external force, which not only improves the mechanical strength of the material, but also maintains its properties after the material is damaged and self-healing at the site of a wound or crack. Deng et al. 21 developed a multifunctional double-layer composite hydrogel conduit based on chitosan, aiming to provide an innovative solution for peripheral nerve repair. This conduit not only promotes nerve regeneration but also has multiple functions, including guiding nerve growth and supporting the attachment and proliferation of nerve cells. However, chitosan itself has relatively weak mechanical properties, especially in its hydrated state, where the resulting hydrogels typically exhibit low mechanical strength and elasticity. 22 By combining it with Polylactic acid (PLA), a material known for its excellent mechanical properties, a composite gel can be obtained that provides better structural support. 23 This composite hydrogel material, combining the advantages of both chitosan and PLA, holds great potential for broader applications in the medical field.
In this work, the composite CS/PLA nanofibrous hydrogel fabricated by electrospinning for sealing and repairing tracheoesophageal fistula. The novelty lies in the fact that by precisely controlling the spinning parameters, the diameter and crystallinity of the fibers can be controlled. Eventually, the nanofibers are dispersed in the CS-based aqueous gel precursor solution, forming a “nanofiber suspension injectable system.” The mechanical properties, rheological behavior, and antimicrobial activity of the hydrogel were successfully enhanced via the composite modification strategy. In vitro experimental results indicated that the CS/PLA composite nanofiber hydrogel exhibited excellent biocompatibility, which not only effectively facilitated the proliferation and migration of target cells but also demonstrated prominent cell adhesion capacity and cell proliferation-promoting activity.
Experimental
Materials
D, L-lactide, zinc lactate, and dichloromethane were purchased from Shanghai Aladdin Biochemical Technology Co., Ltd. Chitosan (deacetylation degree ⩾95%), sodium carbonate (⩾99.5%), glacial acetic acid (⩾99.5%) and dichloromethane were purchased from China National Pharmaceutical Group Corporation (Sinopharm Group). Agar and Phosphate Buffered Saline (PBS) were purchased from Shanghai McLean Company. Escherichia coli (E. coli) and Staphylococcus aureus (S. aureus) were purchased from Chonghui (Beijing) Biotechnology Co., Ltd.
Preparation of PLA
PLA was synthesized via ring-opening polymerization (ROP) of D, L-lactide, using water as the initiator and zinc lactate as the catalyst. Specifically, D, L-lactide (5.4 g) was added to a dry round-bottom flask, followed by the introduction of a pre-determined volume of water (initiator) to regulate the degree of polymerization. After homogenization of the mixture, zinc lactate (5.4 mg, catalyst) was accurately weighed and incorporated into the flask. To establish an inert reaction environment, the round-bottom flask was purged with nitrogen gas. Subsequently, the flask was immersed in an oil bath preheated to 160°C and maintained at this temperature for 48 h to initiate and complete the ROP reaction. Post-polymerization, the crude PLA product was dissolved in dichloromethane to remove unreacted monomers and residual catalyst. The dissolved product was then precipitated in anhydrous diethyl ether to achieve phase separation of pure PLA. The resulting precipitate was collected via vacuum filtration and subjected to vacuum drying at 60°C for 12 h to eliminate residual solvents, yielding the final purified PLA sample. The final yield of purified PLA was 86.2 ± 2.3% (mean ± SD, n = 3 independent synthesis batches), calculated as (actual mass of purified PLA/theoretical mass of PLA) × 100%. The molecular weight was 68.5 kDa and it was determined via gel permeation chromatography.
Preparation of CS/PLA nanofibers
PLA was dissolved in dichloromethane to prepare a 15 wt% polymer solution, CS was dissolved in acetic acid to form a 5 wt% solution; both solutions were stirred until fully homogenized to ensure no undissolved polymer residues. Two sets of CS/PLA mixed solutions were prepared at volume ratios of 80:20 and 60:40 (CS: PLA). Each mixed solution was stirred using a magnetic stirrer for 3 h to achieve a homogeneous electrospinning precursor solution, eliminating phase separation between the two polymers. Each homogeneous precursor solution was loaded into a plastic syringe fitted with a stainless-steel spinneret. Electrospinning was conducted using a high-voltage power supply, with the applied voltage set to 14.5 kV and the distance between the spinneret tip and the grounded collector maintained at 10 cm. Under the high-voltage electric field, the polymer solution jet emerging from the spinneret tip was stretched and elongated into continuous fine fibers by electrostatic forces; solvent evaporation occurred during the jet’s flight to the collector, resulting in the formation of solidified fibers that were deposited onto the collector surface. The CS/PLA composite nanofibers prepared from the 80:20 and 60:40 (CS: PLA) precursor solutions were designated as CS/PLA1 and CS/PLA2, respectively.
CS/PLA composite nanofiber hydrogel preparation
To fabricate the hydrogel, the CS/PLA composite nanofibers need to be crosslinked to stabilize their three-dimensional (3D) network structure and enhance their water absorption capacity. The composite nanofibers are soaked in glutaraldehyde and a dilute ethanol solution is added. After reacting for 12 h, the fibers are washed with deionized water to remove any excess crosslinking agent. The nanofibers are then immersed in water, allowing water molecules to penetrate the crosslinked network, causing the fibers to swell and form a gel-like structure, ultimately resulting in the formation of the hydrogel. The schematic diagram of the preparation process is shown in Figure 1.

Schematic diagrams displaying the processes for the fabrication of composite nanofiber hydrogel and repairing tracheoesophageal fistula.
Scanning electron microscopy analysis (SEM)
The surface morphology of the fabricated composite nanofibers was observed using an FEI Quanta 250 FEG scanning electron microscope (SEM) at an accelerating voltage of 5 kV.
Thermogravimetric analyzer (TGA)
Thermal stability of the hydrogel samples was characterized using a NETZSCH STA 449C thermogravimetric analyzer (TGA) under a nitrogen atmosphere. The heating rate was 0.5ºC min–¹, with a temperature range from 30°C to 800°C, and the mass degradation of the samples was recorded during the heating process.
Fourier-transform infrared spectroscopy analysis (FTIR)
The freeze-dried hydrogel was analyzed using an infrared spectrometer in ATR mode to obtain its infrared spectrum Fourier Transform Infrared Spectroscopy (FT-IR), with a scanning range of 4000-500 cm–¹.
Swelling and degradation performance test of the hydrogels
Prepare the synthesized hydrogel sample, taking care to record the initial dry weight (W0) of the sample and soaked in a PBS (Phosphate Buffered Saline) (pH 7.4) solution at 37°C. At regular intervals, the hydrogel is taken out, the surface water is gently blotted with filter paper, and its weight is recorded on the 2nd, 4th, 8th, 12th, and 24th days (n = 3) . The swelling ratio of the hydrogel is calculated using the following formula:
Where SD (%) is the swelling ratio, W0 is the initial mass of the hydrogel, and Wt is the mass of the hydrogel after soaking in the solution. The result is obtained by averaging the values of three parallel samples and calculating the standard deviation.
After freeze-drying the hydrogel samples, weigh and record the initial mass. Then, immerse the samples in PBS solution (pH = 7.4) and place them in a shaker at 37°C. After certain time intervals, take out the hydrogel samples, rinse them 2–3 times with distilled water, then freeze-dry and weigh them on the 1st, 2nd, 3rd, 6th, and 10th days (n = 3). The in vitro degradation rate of the hydrogel is calculated using the following formula:
Where DR (%) is the swelling ratio, W0 is the initial mass of the hydrogel, and Wt is the mass of lyophilized hydrogel after degradation. The result is obtained by averaging the values of three parallel samples and calculating the standard deviation.
Mechanical property of the hydrogels
Stress-strain testing of the hydrogel was conducted using an electronic universal testing machine. The sample dimensions were 40 × 10 × 2 mm, with an initial gauge length of 20 mm and a stretching speed of 50 mm/min. Compression strength of the hydrogel was measured using a universal testing machine. The compression speed was set to 0.007 mm/s, and the load was applied at a strain rate of 10% per minute until the sample was compressed to approximately 10% of its original height. Stress-strain and compression strength were tested five times and the standard deviations were calculated, data are presented as mean ± SD (n = 5).
Rheological properties
Rheological properties of the hydrogel were tested using a TA HR10 rotational rheometer (USA). The frequency sweep test was conducted at 25°C, ranging from 1 to 100 rad/s, with a strain set to 1%. The sample thickness between plates was set to 2.0 mm to avoid edge effects. Excess sample was trimmed before testing, and a solvent trap was used to prevent dehydration during measurements.
Antibacterial experiment
The antibacterial experiment of the hydrogel was conducted using Escherichia coli and Staphylococcus aureus. The prepared hydrogel was mixed with 100 μL of bacterial suspension (1 × 108 CFU/mL). After incubation at 37°C for 1 h, the bacterial suspension of different groups was diluted and gently shaken in fresh Luria Bertani (LB) broth. The bacterial solution was then spread onto solid agar plates and placed in a biological incubator at 37°C for 12 h. The colony count was determined using the plate dilution method, and photographs were taken for recording. The bacterial suspension incubated in PBS was used as the control sample.
Cell experiment
Cytotoxicity of the hydrogel on L929 cells (mouse fibroblasts) was evaluated using the MTT assay. The hydrogel was first shaped into circular samples with a diameter of 5 mm and thickness of 1 mm, then washed three times with PBS and disinfection by ultraviolet (UV) irradiation for 45 min. The sterilized samples were placed in a 48-well plate and immersed in culture medium for 2 h before cell seeding. The cell seeding density was 1.5 × 104 cells/mL. The samples were then incubated at 37°C for 5 days, with medium changes every 2 days. At the designated time, MTT analysis was performed, and absorbance was measured at 450 nm using a UV spectrophotometer. The cell viability was calculated using the formula:
An is the absorbance value of the experimental group cells, and A0 is the absorbance value of the control group cells. All cell culture experiments were conducted with biological replicates (n = 3).
Results and discussion
Morphological properties
To verify the successful preparation of CS/PLA composite nanofibers via electrospinning, SEM was used to observe the morphology, diameter, and surface structure of the nanofibers. Scanning electron microscopy (SEM) images of CS/PLA composite nanofibers were showed in Figures 2(a) and (d), it can be observed that the morphology and structure of the CS/PLA composite nanofibers remain intact after crosslinking, indicating that this structure can enhance the strength of the chitosan membrane and prevent water absorption, swelling, and disintegration. 24 From the local magnified SEM images in Figures 2(b) and (e), as well as the fiber diameter distribution charts in Figures 2(c) and (f), it can be observed that the fibers exhibit uneven thickness, with an average size of approximately 600 nm. When the ratio of CS to PLA solution is 60:40, the size of the composite nanofibers slightly increases. This is due to the increased viscosity of the solution after the addition of PLA, which causes the electrospinning jet to become thicker.

Scanning electron microscopy (SEM) images of: (a, d) CS/PLA composite nanofibers, (b, e) partial enlarged detail, and (c, f) diameter profile CS/PLA composite nanofibers determined from SEM images using ImageJ software.
After swelling the composite nanofibers hydrogel, the sample was subjected to freeze-drying and subsequently characterized using SEM. As presented in Figure 3, the CS/PLA composite nanofibers hydrogel maintained an intact fibrous architecture post-swelling, this structural integrity confirms that the crosslinking network within the hydrogel effectively resists swelling-induced fiber fragmentation or collapse. Notably, a significant increase in fiber diameter was observed after swelling: quantitative analysis of SEM images revealed that the average fiber diameter increased from the pre-swelling value to approximately 1.2 µm. This diameter expansion is attributed to the absorption of water molecules by the hydrogel hydrophilic functional group, which drives the swelling of the fibrous matrix and leads to the relaxation of polymer chains within the fibers. The maintenance of fibrous structure alongside the measurable diameter increase further verifies the hydrogel balanced swelling capacity and structural stability.

SEM images of: (a) CS/PLA1 and (b) CS/PLA2 composite nanofibers after swelling and freeze-drying.
Thermal stability analysis
Thermal stability is an important index to evaluate the physical and chemical properties of hydrogels under high temperature conditions, which directly affects the service life and performance of hydrogels in practical applications. We measured the mass change of the composite nanofiber hydrogel during heating using Thermogravimetric Analysis (TGA), and Figure 4(a) shows the weight loss curve of the composite nanofiber hydrogel at different temperatures. From the figure, it can be observed that during the heating process from 30°C to 250°C, the mass of the composite nanofiber hydrogel gradually decreases due to the evaporation of water. When the temperature reaches 320°C, the mass of the hydrogel begins to decrease rapidly, indicating that CS and PLA start to degrade. The higher the PLA content, the faster the mass loss of the composite nanofiber hydrogel, suggesting that PLA degrades more rapidly at high temperatures around 300°C. Through TGA analysis, we successfully obtained information about the thermal stability of the composite nanofiber hydrogel during heating. The water evaporation at low temperatures and the thermal degradation characteristics at higher temperatures provide us with valuable data on its thermal behavior in practical applications.
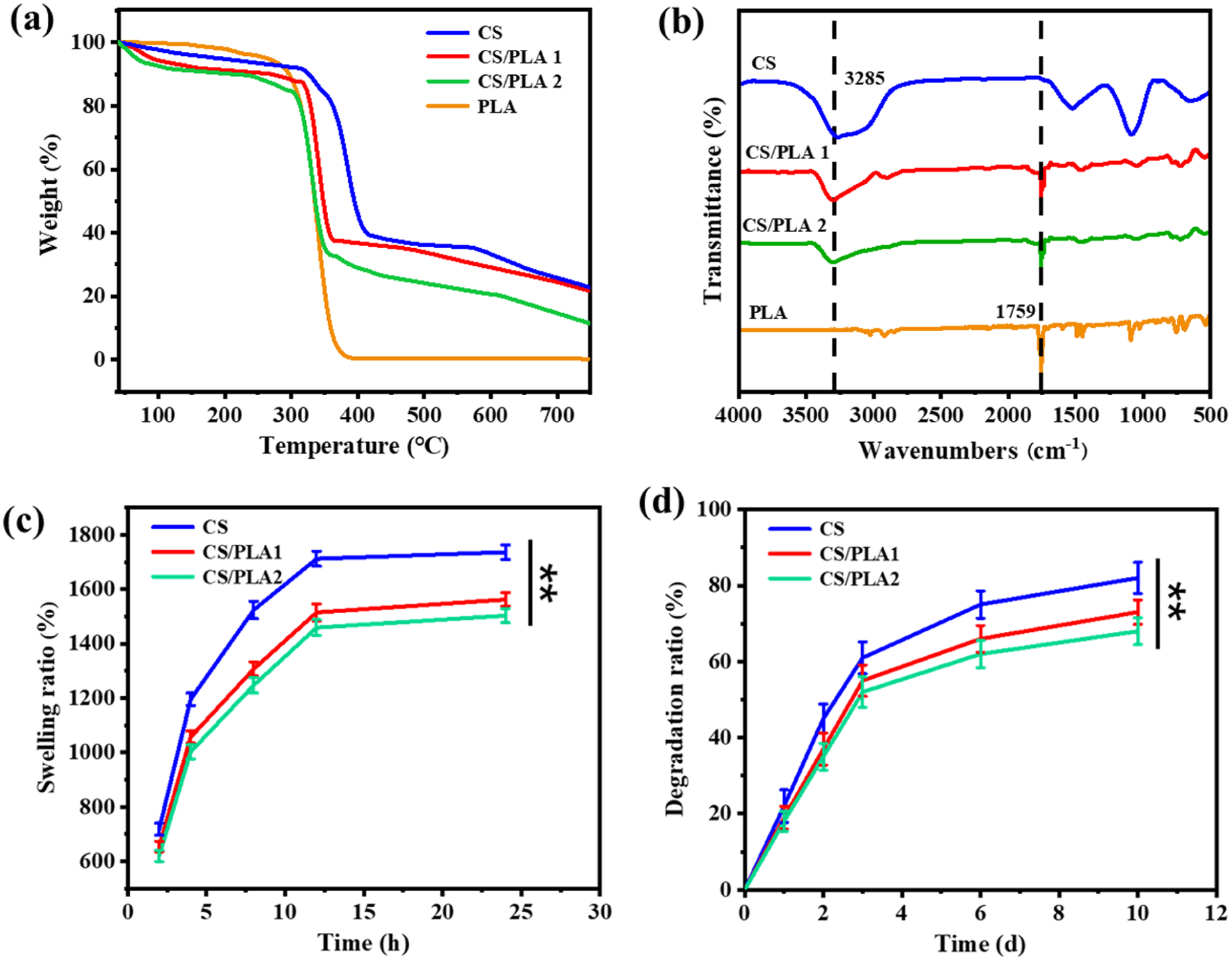
(a) TGA curves of CS, CS/PLA1, and CS/PLA2 composite nanofiber hydrogel, (b) FT-IR spectra of CS, CS/PLA1, and CS/PLA2 composite nanofiber hydrogel, (c) the swelling behavior of CS, CS/PLA1, and CS/PLA2 composite nanofiber hydrogel, (d) the degradation behavior of CS, CS/PLA1, and CS/PLA2 composite nanofiber hydrogel
FI-TR analysis of hydrogel
To analyze the molecular structure, crosslinking degree, and interactions between the components of the hydrogel, Fourier transform infrared spectra was used to test CS, CS/PLA1, CS/PLA2, and PLA as shown in Figure 4(b). It can be observed that CS exhibit characteristic absorption peaks at 1131 cm−1, 1532 cm−1, and a prominent broad band at 3285 cm−1, which corresponds to the stretching vibrations of the C–O stretching, N–H and O–H groups, respectively. 25 In PLA, characteristic absorption peak at 1759 cm−1, which is assigned to carbonyl groups in PLA. 26 In the FTIR spectra of CS/PLA1 and CS/PLA2, new absorption peaks appear around 1500 cm−1, corresponding to the C=O vibration of the crosslinking agent, indicating the occurrence of the crosslinking reaction.
Swelling and degradation behaviors analysis
The swelling behavior of hydrogels is an important factor affecting their repair performance. 27 As shown in Figure 4(c), PBS solution is used to simulate the human body environment for testing the swelling performance of hydrogels. It can be observed that the swelling of the three hydrogel samples reaches equilibrium after 15 h. The swelling rates of CS, CS/PLA1, and CS/PLA2 are 1730%, 1562%, and 1503%, respectively. With the increase of PLA content in hydrogel, the swelling rate of hydrogel also decreases, because PLA has strong hydrophobicity. This indicates that by incorporating PLA blocks into CS, the swelling properties of the hydrogel can be regulated, allowing for the adjustment of the physical properties of the hydrogel to meet the requirements for use in tracheoesophageal fistula repair.
The degradation and decomposition processes of the hydrogel under biological conditions were tested by simulating the human body environment. As shown in Figure 4(d), the degradation rate of the CS hydrogel is 81.7% after 10 days. Compared to the CS hydrogel, the CS/PLA hydrogel shows a reduced degradation rate, with the degradation rates of CS/PLA1 and CS/PLA2 being 72.6% and 68.9%, respectively. By introducing hydrophobic PLA into the hydrogel as a composite, the degradation rate of the hydrogel can be effectively reduced. The hydrogel prepared by Alves et al. 28 for organ repair degraded by more than 30% within 10 days, and its degradation performance was significantly better than theirs. This is because PLA has strong hydrophobicity, which reduces the penetration of water and slows down the degradation process of the hydrogel. Specifically, the hydrophobicity of PLA decreases the overall hydrophilicity of the hydrogel, reducing contact between water and the internal structure of the hydrogel, thereby delaying the degradation reaction. Therefore, by incorporating hydrophobic PLA and adjusting its ratio, the degradation rate of the hydrogel can be precisely controlled, optimizing its function in tissue repair. This ensures that the scaffold material provides appropriate support throughout the repair process and gradually degrades once the repair is complete.
Mechanical properties
The hydrogel material used for tracheoesophageal fistula repair indeed needs to have good mechanical properties to ensure its stability and effectiveness during the repair process. The compressive strength and stress-strain curves of the hydrogel were tested, as shown in Figures 5(a) and (b). It can be observed that the compressive strength of the three hydrogel types ranges between 37 and 44 MPa. The compressive strength of the CS hydrogel is 37.6 ± 1.86 MPa, while the compressive strength of the composite hydrogels shows a significant improvement. Specifically, the compressive strength of CS/PLA1 is 42.3 ± 1.62 MPa, and the compressive strength of CS/PLA2 is 43.7 ± 1.55 MPa. The incorporation of PLA into the hydrogel significantly enhances its compressive strength, thereby enabling it to maintain support in the repair area. This improved compressive strength helps prevent material failure due to external pressure. Figure 4(b) shows the stress-strain curve of the hydrogel. With the increase of PLA content in the hydrogel, the tensile strength of the hydrogel is significantly improved, from 1.04 to 1.38 MPa. However, the elongation at break of hydrogel decreased from 225% to 173% with the increase of PLA content. Zhao et al. 29 prepared a GelMA-SFMA-Al hydrogel for tissue engineering repair, and this hydrogel has a tensile strength ranging from 0.31 to 0.96 MPa. PLA molecules contain a large number of hydroxyl groups, which can react chemically with other groups to form strong intermolecular forces. At the same time, PLA interacts with CS through hydrogen bonding, interpenetrating the two components. This interaction increases the internal crosslinking density of the hydrogel, thereby enhancing the material’s mechanical properties.

(a) Compressive strength of CS, CS/PLA1, and CS/PLA2 composite nanofiber hydrogel, (b) stress-strain curves of CS, CS/PLA1, and CS/PLA2 composite nanofiber hydrogel, (c) oscillatory frequency sweep of CS, CS/PLA1, and CS/PLA2 composite nanofiber hydrogel, and (d) oscillatory strain sweeps of hydrogels were conducted at a frequency of 1 Hz.
Rheological characterization
Rheological properties of hydrogels play a crucial role in their practical applications, especially in situations where they need to simulate biological fluids or withstand external pressure. 30 As shown in Figure 5(c), in the entire frequency scanning range, the storage modulus G′ of the hydrogel is greater than the loss modulus G″, indicating that the hydrogel has a stable network structure, and the crosslinking points effectively restrict the movement of molecular chains, enabling the hydrogel to exhibit solid-like properties under shear or strain. As shown in Figure 5(d), when the strain exceeds its critical value by approximately 10%, the G′ value of the hydrogel decreases slowly with the increase in strain, indicating that the hydrogel has undergone a gel-solution transformation. Additionally, with the incorporation of PLA, the crosslinking network of the composite hydrogel becomes denser and stronger. This may be due to the formation of multiple non-covalent bonds between PLA and CS, which enhances the strength of the hydrogel network. 31
Preliminary injectability test
The injectability of the hydrogel is also characterized through actual injection operations. As illustrated in Figure 6, the injectability of the prepared CS/PLA composite hydrogel was verified using a 25-gauge (25G) hypodermic needle, an instrument with a standard inner diameter of 260 μm, which is consistent with the size of needles commonly used in minimally invasive clinical procedures, and manual pressure was applied to the syringe plunger under ambient conditions (25 ± 1°C). The hydrogel exhibited smooth, continuous flow through the needle lumen without any observable blockage, fragmentation, or excessive resistance. This result directly confirms that the hydrogel rheological properties are well-matched to the dimensional constraints of 25G needles: the absence of blockage indicates that no aggregated component are present in the hydrogel to obstruct the 260 μm inner diameter channel. Such injectability is critical for its potential clinical application, as it ensures the hydrogel can be precisely delivered to target defect sites via minimally invasive routes without requiring specialized injection equipment or risking needle occlusion.

Appearance image of injection of: (a) CS, (b) CS/PLA1, and (c) CS/PLA2 composite nanofiber hydrogel.
Antibacterial activity of hydrogel
To evaluate the antimicrobial effect of the composite nanofiber hydrogel, its antimicrobial activity against Escherichia coli (E. coli) and Staphylococcus aureus (S. aureus) was tested, and the control group used distilled water. Figure 7(a) to (c) show the antimicrobial effect of the hydrogel against E. coli and S. aureus, as well as the corresponding inhibition rate bar charts. Compared to the control group, a significant reduction in the colony count of E. coli and S. aureus was observed on the plates after treatment with the hydrogel. From Figure 7(b) and (c), it can be observed that compared to the antimicrobial activity of the original CS hydrogel, the CS/PLA composite hydrogel exhibits better antimicrobial performance. Specifically, CS/PLA1 and CS/PLA2 show inhibition rates of 79.6% and 80.3% against E. coli, and 80.7% and 82.6% against S. aureus, which are significantly higher than the CS hydrogel (with inhibition rates of 70.2% and 76.3%, respectively). This indicates that the incorporation of PLA effectively enhances the antimicrobial performance of the hydrogel, likely due to the synergistic interaction between PLA and CS, which improves the structure and functionality of the hydrogel, thereby increasing its ability to inhibit bacterial growth. The strong interactions between PLA and CS molecules not only improve the physical and chemical stability of the composite material but may also enhance its ability to disrupt bacterial cell membranes. 32 Additionally, the combination of the hydrophobic nature of PLA and the hydrophilic nature of CS optimizes the surface properties of the hydrogel, making it more effective in exhibiting antimicrobial activity when in contact with bacteria. Besides the significant inhibitory effects on E. coli and S. aureus, the improved antimicrobial properties of the composite hydrogel provide further potential for practical applications.

(a) The plate count results of various hydrogel against E. coli and S. aureus, respectively, (b) histograms of the E. coli viability rates, and (c) histograms of the S. aureus viability rates.
Cytotoxicity and biocompatibility of hydrogels
An ideal esophageal-tracheal fistula repair material should possess good biocompatibility. Therefore, the prepared hydrogel was co-cultured with mouse fibroblasts (L929) for 5 days, and cell viability was measured using the MTT assay to evaluate the cytotoxicity of the CS, CS/PLA1, and CS/PLA2 composite nanofiber hydrogel. As shown in Figure 8(a), after 120 h of culture, the cell viability in the CS, CS/PLA1, and CS/PLA2 composite nanofiber hydrogels were 92.8%, 96.5%, and 96.2%, respectively. During the test period, the cell viability for all samples remained above 90%, indicating that the CS, CS/PLA1, and CS/PLA2 composite nanofiber hydrogels showed no cytotoxicity. The high cell viability suggests that the hydrogel can provide a non-toxic environment that supports cell growth and proliferation, making it highly suitable for use as an esophageal-tracheal fistula repair material. Figure 8(b) exhibits the cell proliferation results, after 24 h of culture, no significant differences were observed in cell numbers between the groups. As the culture time increased, cell proliferation gradually increased. After 120 h of culture, the cell proliferation in the CS/PLA composite hydrogel group was approximately five times the initial number. The experimental results indicate that the CS/PLA composite hydrogel not only has good cell compatibility but also further promotes cell proliferation. The significant increase in cell proliferation suggests that the CS/PLA composite hydrogel might optimize the cell growth environment and enhance the interaction between the cells and the hydrogel, thus promoting cell activity and proliferation. As time progresses, cell proliferation accelerates, laying the foundation for its use in tissue repair and regeneration.

(a) The survival rate of L929 cells co-cultured with hydrogel and (b) the proliferation rate of L929 cells co-cultured with hydrogel.
Conclusions
In summary, CS/PLA composite nanofiber hydrogels suitable for TEF repair were successfully prepared using the electrospinning technique. The composite modification strategy effectively optimized the mechanical properties, rheological behavior, and antimicrobial activity of the hydrogel. In vitro experimental findings demonstrated that the CS/PLA composite nanofiber hydrogel possessed excellent biocompatibility, as evidenced by its capacity to facilitate target cell proliferation and migration, along with its prominent ability to support cell adhesion and proliferation. Compared to traditional repair methods, the CS/PLA composite nanofiber hydrogel showed significant advantages in repair efficiency and tissue recovery, particularly in terms of smaller trauma, faster recovery, and fewer complications. CS/PLA composite nanofiber hydrogel holds great promise as a material for TEF repair, providing an innovative and effective option for clinical treatment. At present, a key challenge lies in aligning the hydrogels degradation rate with diverse tissue healing rhythms and risks include local inflammation from degradation metabolites or complement activation by material components. Future in vivo validation of animal models will be conducted, which is crucial for evaluating factors such as long-term biocompatibility, tissue integration, and resistance to physiological stress. These are all key prerequisites for advancing to human clinical trials and promote its widespread clinical application.
Footnotes
Author contributions
Zhikui Luo: Conceived and designed the study, supervised the project, and contributed to manuscript writing and revision. Qiang Cao: Conducted data analysis and interpretation, and wrote the results section. Leilin Zhu: Performed experiments and contributed to data collection and methodology. Mengting Tan: Assisted in the literature review and contributed to data collection. Zhichao Xiao: Participated in data analysis. Junai Zhu: Contributed to the experiment design and data interpretatio. Xinyu Liu: Conducted critical review of the manuscript, particularly the results and discussion sections. Fusen Peng: Provided resources, and reviewed the manuscript for scientific accuracy. Ying Zhang: Contributed to manuscript drafting, provided intellectual input on the research framework, and revised the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Loudi Science and Technology Bureau [2022] 32 project, Hunan Provincial Natural Science Foundation of China (2024JJ9596).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available from the corresponding author.*



