Abstract
Background
The aim of this study was to develop a minimally invasive hydrogel system that can release strontium ions, an element that has been shown to increase osteoblast proliferation and prohibit bone resorption, in a controlled manner.
Methods
SrCO3 was selected as the salt of choice due to potential acid neutralization reaction between SrCO3 and degradation by-products of methoxy(polyethylene glycol)-co-poly(lactic-co-glycolic acid) (mPEG-PLGA): namely, lactic acid and glycolic acid. SrCO3 was incorporated into mPEG-PLGA hydrogel, and the system was assessed for gelation properties, drug release and biocompatibility.
Results
SrCO3 incorporation at hydrogel to SrCO3 ratios of 5:1, 3:1 and 1:1 (wt%) did not compromise the thermosensitivity of mPEG-PLGA hydrogels. Furthermore, incorporation of SrCO3 at 1:1 ratio prevented copolymer self-catalysis and decreased hydrogel weight loss from 85% to 61% in vitro after 30 days. During the 30-day time frame, zero-order strontium release was observed and was correlated to hydrogel degradation and acidity. The addition of SrCO3 also improved in vivo hydrogel biocompatibility, due to moderation of acidic microenvironment and amelioration of inflammatory response.
Conclusions
These results showed that the described system is suitable for the extended release of strontium and exhibits potential for localized treatment for osteoporosis or as a bone void filler.
Introduction
Early experimentation on strontium has identified its potential in promoting bone formation and mitigating bone resorption (1). Since 2004, strontium has been used in Europe for the treatment of osteoporosis, a progressive bone disease marked by decreased bone density and a greater chance of fractures. In vitro studies have shown that strontium inhibits the recruitment and activity of bone-resorbing osteoclasts while promoting osteoblastic proliferation, alkaline phosphatase production and subsequent mineralization (2-3-4). Clinically, strontium has been shown to decrease the likelihood of fractures in postmenopausal women by reinforcing the microarchitecture of bone to provide better biomechanical competence (3-4-5). Furthermore, variable salt forms of strontium, such as strontium chloride, carbonate and citrate, are widely available over the counter as nutritional supplements. However, compliance in daily oral intake of strontium compounds is poor, and oral administration often induces an increment in plasma drug concentration that is followed by sinusoidal drop. In addition, orally administered strontium may not effectively reach damaged tissues when local treatment is desired. In view of these aspects, the development of a local and sustained strontium delivery system that is minimally invasive and capable of achieving adequate tissue concentrations would effectively increase patient compliance and lower concerns regarding systemic effects.
In addition to osteoporosis treatment, strontium has also been incorporated into bone fillers by direct doping to promote osteoblast proliferation and precipitation of hydroxyapatite (6, 7). Strontium salts, such as strontium oxide, have been used together with bioactive glass to promote bone formation, while the addition of Sr2+ to a hydroxyapatite gel has been shown to up-regulate osteogenic genes (8, 9). Furthermore, coating of prosthetic devices with strontium allows the gradual release of strontium ions locally (10, 11). These methods have limitations, however, such as the quick dissociation of salt forms which results in the fast release of strontium ions or complicated processes that are required to coat materials with strontium. Nevertheless, the controlled release of strontium ions from a polymer/drug matrix has remained unexplored.
Hydrogels are 3-dimensional swollen networks that have been extensively studied for their applications in drug delivery (12). They typically act as depots where molecules can be incorporated and slowly eluted as a result of diffusion or hydrogel degradation. Biodegradable poly(lactic-co-glycolic acid) (PLGA) is one of the most attractive and commonly studied polymers approved by the US Food and Drug Administration for use in drug and tissue engineering applications. Most interestingly, diblock and triblock copolymers of poly(ethylene glycol) (PEG) and PLGA provide hydrogel systems that undergo gelation in situ due to their thermosensitivity (13-14-15). In the presence of water, PLGA degrades by hydrolysis of ester bonds to produce lactic and glycolic acids. While these acids are metabolized in the body, long-term exposure of cells to an acidic environment is one of the greatest limitations in the development and use of PLGA depots (16). Furthermore, self-catalyzed hydrolysis can cause the system pH to fall below 3 (17, 18). To resolve this shortcoming, buffering and neutralizing systems have been developed, such as the addition of buffers or neutralizers including hydroxyapatite, Mg(OH)2, amorphous calcium phosphate and sodium bicarbonate (19-20-21).
This study aimed to investigate the potential of methoxy (polyethylene glycol)-co-poly(lactic-co-glycolic acid) (mPEG-PLGA) hydrogel as a depot for strontium carbonate, which may dissociate in the presence of acid to release strontium ions. Strontium, in the form of strontium carbonate, was first incorporated with the previously established diblock mPEG-PLGA thermosensitive hydrogel. Theoretically, the hydrolysis of mPEG-PLGA and production of acidic by-products would dissociate SrCO3 to provide degradation-controlled release of strontium ions and simultaneous microenvironment pH moderation. Hydrogel degradation and strontium release were studied at various hydrogel to SrCO3 ratios. Lastly, hydrogels were injected subcutaneously to monitor their in situ gelation and their effects on the surrounding tissue in terms of inflammatory response.
Material and Methods
Materials
Methoxy poly(ethylene glycol) (molecular weight [MW] = 550), DL-lactide, glycolide, ε-caprolactone, stannous 2-ethyl hexanoate, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium (MTT) reagent, stannous 2-ethyl hexanoate, chloroform-d, trypsin–ethylenediaminetetraacetic acid (EDTA), 1,6-diphenyl-1,3,5-hexatriene (DPH), Live/Dead cell double-staining kit and strontium carbonate were obtained from Sigma-Aldrich (St. Louis, MO, USA). Dimethyl sulfoxide (DMSO) and tetrahydrofuran (THF) were acquired from Echo Chemicals (Taiwan). Phosphate-buffered saline (PBS) at pH 7.4, Dulbecco's modified Eagle's medium (DMEM) and fetal bovine serum (FBS) were obtained from Gibco (Carlsbad, CA, USA).
Synthesis of mPEG-PLGA
mPEG-PLGA was synthesized by ring-opening polymerization of DL-lactide and glycolide with hydroxyl-terminated mPEG in the presence of stannous 2-ethyl hexanoate as the catalyst. A detailed synthesis procedure has been described previously (14). Briefly, 8.01 g of mPEG, 16.67 g of DL-lactide and 3.8 g of glycolide were heated to 160°C until homogenous and fully melted, then 0.45 μL of the catalyst was added to start an 8-hour reaction. The product was then dissolved in DMSO and dialyzed (MW = 1,000) to obtain the final product. Structure and molecular weight of the copolymers were verified by 1H nuclear magnetic resonance (1H-NMR) on a Varian Unity Inova 500 NMR system and by gel permeation chromatography (GPC) on a JASCO (RI-2031, PU-2080) system.
Gelation Characteristics
The test tube inversion method was used to construct the solution-gel profile. Briefly, 200 μL of the solution was equilibrated at 4°C for 10 minutes, and the temperature was increased to 60°C at a rate of 0.5°C/min. At each temperature, the test tube was inverted, and the temperature at which the solution turned into gel was recorded as the sol-gel point. The gel-sol point was taken as the temperature where mechanical integrity was lost with gentle agitation. Dynamic mechanical analysis was conducted using a TA Instrument AR2000 EX system with 40-mm parallel plate geometry and geometry gap of 50 mm. A test sample (700 μL) was loaded onto the prechilled plate, and a temperature sweep between 4°C and 45°C was conducted at 2.2°C/min (2% strain, 1 rad/s). The storage modulus (G’) was recorded as a function of temperature.
Degradation of Hydrogel and Strontium Release
Briefly, 20% mPEG-PLGA solution was mixed with strontium carbonate at different ratios to provide systems with final polymer to drug ratios (wt%) of 1:0 (G1S0), 5:1 (G5S1), 3:1 (G3S1) and 1:1 (G1S1). A sample volume of 1 mL was used, and hydrogels were equilibrated at 37°C for 1 hour prior to the addition of 9 mL of prewarmed PBS as the incubation medium (pH 7.4). Samples were incubated at 37°C with gentle rotating motion of 100 rpm. At designated time points, pH, residual weight and strontium release were assessed. Residual weight was calculated according to the equation: Residual gel% = (Wt-Wsr)/Wo, where Wt is the weight of the system after incubation, Wsr is the weight of SrCO3 remaining in the system and Wo is the total initial weight of the system. Strontium concentration was measured by inductively coupled plasma–mass spectrometry (ICP-MS) using a Perkin Elmer SCIEX Elan 5000 system.
In Vitro Cell Compatibility of System
For the cell compatibility tests, 293T cells were used. When grown to 80% confluency, cells were trypsinized and seeded in 48-well plates at a density of 5 × 103 cells/well. The cells were cultured overnight, washed twice with warm PBS and treated overnight with 1 mL of the incubation medium withdrawn on selected time points. Upon removal of the incubation medium, cells were washed twice with warm PBS and 500 μL of MTT reagent (2.5 mg/mL) was added to sample wells. After 2 hours, the solution was aspirated, and 1 mL of DMSO was added to dissolve the formazan crystals. Absorbance was read at 570 nm using a BioTek Synergy HT plate reader.
To visualize the viability of encapsulated cells, cell-laden hydrogels were stained with Live/Dead cell double-staining kit which renders viable cells green (calcein AM) and dead cells red (ethidium). Dyes were prepared according to the manufacturer's protocol and added to sample wells for 20 minutes prior to washing and observation with a Leica DMI3000 fluorescent microscope.
In Vivo Compatibility
Animal study was approved by the Animal Experiment Committee of Chang Gung University, Taoyuan, Taiwan, and conducted in accordance with the Guide for the Care and Use of Laboratory Animals. All materials were UV-irradiated overnight to ensure sterility. Sprague Dawley rats weighing approximately 400 g were used, and the animals were anesthetized with 1%-3% isoflurane prior to the procedure One milliliter of each hydrogel solution was injected subcutaneously into the dorsal region of the back. At 1, 2 and 3 weeks, the animal was euthanized, and hydrogels and their surrounding tissues were removed and fixed with formaldehyde followed by standard embedding and deparaffinization. Inflammation response was evaluated by hematoxylin and eosin (H&E) stain.
Results and Discussion
Copolymer Characterization and Preliminary Gelation test
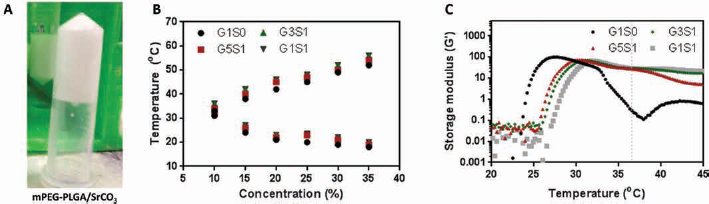
The synthesized mPEG-PLGA copolymer had a molecular weight (MW) of 1,945 and polydispersity index (PDI) of 1.07 as determined by GPC. Structure verification was conducted via 1H-NMR as shown in Figure 1. Aqueous solutions of the mPEG-PLGA copolymer (MW = 550-1,405) have been shown previously to undergo thermosensitive gelation in the concentration range of 15%-40% (22). The gelation mechanism can be described as the sudden aggregation of micelles which provides a dense bulk hydrogel structure when both the critical gelation concentration (CGC) and critical gelation temperature (CGT) are reached. Preliminary study revealed that the addition of SrCO3 at the polymer to drug ratio of 1:1 (wt%) did not compromise the thermosensitivity of the mPEG-PLGA copolymer solution (Fig. 2A). Owing to its reactivity in an acidic environment, described by the equation 2 H+ + SrCO3 → Sr2+ + CO2 + H2O, SrCO3 is capable of releasing its strontium ion in an acidic microenvironment. A 20% mPEG-PLGA copolymer solution prepared in PBS buffer exhibited a pH value of 4.37 which, while hostile to cells, might allow the release of strontium ions. Therefore, theoretically, when incorporated into acidic mPEG-PLGA hydrogels, SrCO3 can release its strontium ion while neutralizing acidic by-products. Other salt forms of strontium are readily available, such as strontium oxide (SrO), strontium chloride (SrCl2) and strontium sulfate (SrSO4); however, SrO reacts exothermically with water, while SrCl2 and SrSO4 are incapable of neutralizing the environmental pH. Therefore, SrCO3 was adopted as a suitable salt for the described system.
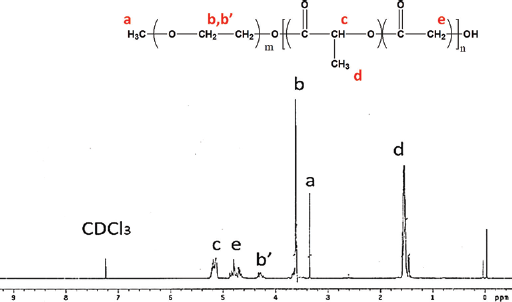
1H nuclear magnetic resonance of methoxy(polyethylene glycol)-co-poly(lactic-co-glycolic acid) (mPEG-PLGA) in chloroform-d. Results indicated a LA/GA ratio of 76:24, which is in agreement with feed. The presence of characteristic peaks of lactic acid (CH-C
(
Hydrogel Properties
A sol-gel transition profile was constructed for hydrogel: SrCO3 mixtures at various ratios, as shown in Figure 2B. The CGC was 10% regardless of the amount of SrCO3 added, which indicates that the incorporation SrCO3 did not compromise general gelation ability. A slight upward shift was noted in the gelation window after the addition of SrCO3. It is known that in response to temperature change, amphiphilic thermogelling solutions undergo a solution to gel to syneresis transition. In this study, syneresis occurred at 38°C and 42°C for 15% and 20% mPEG-PLGA solutions, respectively. To prevent premature syneresis with extended culture, a 20% copolymer concentration was selected for the remainder of the study. A previous study has shown that a 20% mPEG-PLGA solution effectively entraps insoluble hydroxyapatite (HAP) to provide a composite graft at nearly a 1:1 ratio without sacrificing the gelation character (23, 24). Therefore both soluble and insoluble molecules can be incorporated into these hydrogel systems to a certain extent without sacrificing gelation ability.
The storage modulus of the hydrogels at a copolymer concentration of 20% was recorded as a function of temperature as shown in Figure 1C. An increase in the storage modulus of G1S0 occurred at approximately 20°C, which is in agreement with the sol-gel point obtained from the test tube inversion method. The addition of SrCO3 decreased the maximum storage modulus from 99.5 to 66.14 Pa for G1S1. However, with further temperature elevation, the G’ of the blank hydrogel decreased significantly, starting from 33°C, while the G’ of SrCO3-laden hydrogels decreased only slightly. This could be attributed to the composite structure formed by nanoparticles and the hydrogel which reduced system sensitivity to temperature elevation.
Degradation of Hydrogel and in Vitro Release of Sr2+
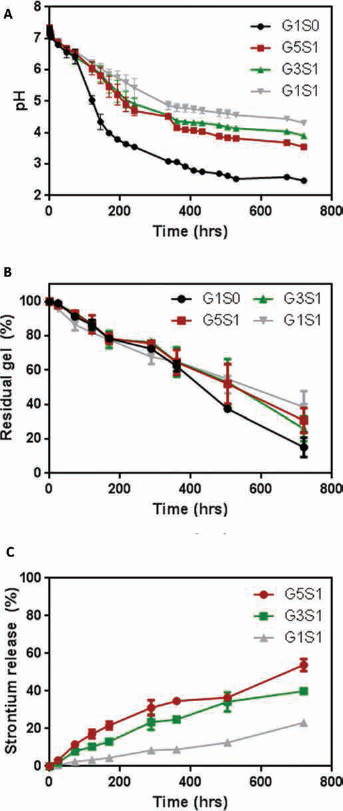
Changes in medium pH and hydrogel weight were monitored over time as shown in Figure 3A, B. Degradation of PLGA has been shown to decrease local pH and induce inflammatory reactions in surrounding tissues (2).
(
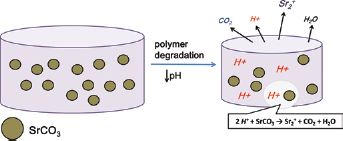
The release of Sr2+ from hydrogel through reaction of SrCO3 with acidic byproducts of PLGA to produce CO2, H2O, and Sr2+.
Generally, PLGA degrades through random scission of ester bonds, resulting in water infusion and subsequent bulk degradation. The medium pH of mPEG-PLGA hydrogel decreased significantly starting on day 3, and dropped to 2.5 at the end of day 14. The addition of SrCO3 at a ratio of 1:1 (G1S1) resulted in a more gradual decrease in pH and a final pH of 4.3 at the end of day 14. Since ester hydrolysis is catalyzed by acidic and basic reactions, a decrease in the medium pH self-catalyzes hydrogel degradation. Various strategies have been used to moderate PLGA degradation, such as controlling LA/GA ratio or the addition of nanoparticles (25, 26). mPEG-PLGA hydrogel without SrCO3 lost 84.8% of its original mass after 30 days of incubation. On the other hand, addition of SrCO3 significantly retarded degradation starting from day 21, and only 61.6% of the original hydrogel mass was lost on day 30. In this study, SrCO3 acted similarly to buffering nanoparticles, serving both to scavenge hydrogen ions and interfere with water diffusion. Under these conditions, degradation of the hydrogel was adequately controlled. Figure 4 depicts the neutralization reaction within the hydrogel system.
(
A method to determine the concentration of strontium has been developed using ICP-MS (27). To elucidate the release mechanism and kinetics, results were analyzed using the zero-order equation and Ritger-Peppas equation as shown below (28).
In this equation, n is the release exponent, k is the release rate constant and Mt/M∞ is the fraction of drug released at time t.
As shown in Figure 3C, a linear relationship between fraction of drug released and time, confirmed that the hydrogel system obeyed zero-order release kinetics. The release of Sr2+ was negatively correlated to SrCO3 concentration and was dependent on hydrogel degradation and medium acidity. On day 30, approximately 53.9% (21.5 ± 1.3 mg) of the Sr2+ was released from G5S1, while only 23.2% (9.3 ± 0.2 mg) was released from G1S1. In this particular system, liberation of Sr2+ was only possible through the depletion of hydrogen ions and creation of CO2 and H2O. Therefore, the pH of the system played a crucial role in the release of Sr2+ from the hydrogel, and no initial burst release was observed. The system with a high polymer to drug ratio of G5S1 was poor in buffering ability, and the acidic microenvironment pH prompted more Sr2+ to be released. Fitting of the Ritger-Peppas empirical model is shown in Table I and provided further verification that the release of Sr2+ was non-Fickian (n>0.5) and dependent on scaffold degradation. The currently approved oral dose form of strontium delivers approximately 680 mg of elemental strontium daily, of which 25% (170 mg) is absorbed through the gut (29). Therefore, the strontium doses provided by the hydrogel should raise no toxicity concerns. Furthermore, it is generally accepted that strontium is incorporated into the bone through 2 phases: an initial rapid uptake into new bone, and a long-term process exchange into old bone (30). Since the incorporation of strontium is also dependent on the length of exposure, a sustained release system would increase the likelihood of strontium incorporation into bones.
Kinetics data of Sr2+ release from hydrogels (Ritger-Peppas model)
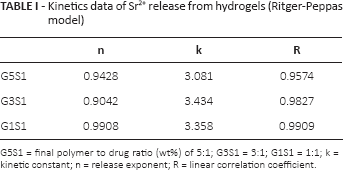
G5S1 = final polymer to drug ratio (wt%) of 5:1; G3S1 = 3:1; G1S1 = 1:1; k = kinetic constant; n = release exponent; R = linear correlation coefficient.
Biocompatibility: In Vitro and in Vivo
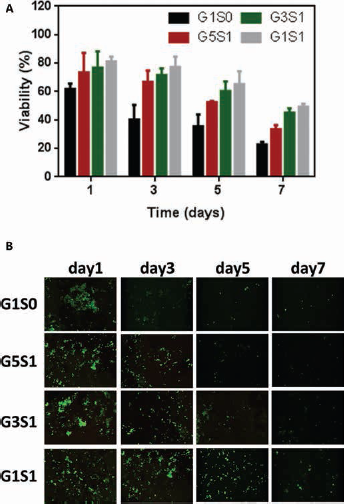
The potential effect of the accumulated acidic by-products on proximate cells was investigated by treating 293T cells with the release medium. A decrease in viability was noted when the amount of incorporated SrCO3 decreased, as shown in Figure 5A. A previous study showed that a reduction in medium pH from 7.3 to 5.6 immobilized all cell components (31). Significant toxicity was noted in the G1S0 group, where cell viability decreased to 61.9% on day 1. On the other hand, cells retained up to 77.3% viability in the G1S1 release medium up to 3 days. Live/Dead staining revealed that the cells were metabolically active at different time points (Fig. 5B). However, with extended culture, the number of viable cells decreased in all groups and most notably in G1S0 and G5S1. The pH of the release medium of G1S0, G5S1 and G1S1 on day 7 was 4.06, 5.49 and 5.7, respectively. Thus, a direct relationship between the pH of the incubation medium and cell metabolism was established. Only a few cells stained red, because nonviable 293T cells became nonadherent and were washed off during the washing step of the staining protocol. Nevertheless, this result is in agreement with the results of the MTT viability test.
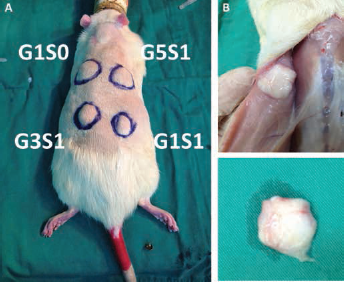
Formation of hydrogel network after subcutaneous injection of various hydrogel systems. After 3 weeks of culture, G1S1 hydrogel retained integrity under the skin.
Despite its general acceptance as a biocompatible polymeric material, PLGA has been shown in some cases to cause in vivo inflammation and poor tissue development (32). Several approaches, including addition of synthetic and natural particles to PLGA scaffolds, have been demonstrated to moderate inflammatory response (33, 34). To confirm in vivo thermosensitive gelation of the system and evaluate the inflammatory response of the surrounding tissue, prepared polymer/drug solutions were injected subcutaneously. As shown in Figure 6, all systems were injectable through a 25-gauge needle and resulted in noticeable lump formation. Large numbers of inflammatory cells were observed at week 1, reflecting acute inflammatory response that is typical after the introduction of foreign material. (Fig. 7). Chronic inflammatory response was noted on week 3 in tissues surrounding G1S0 and G5S1 due to the continuous acidification that is associated with fast resorption of the hydrogel. G3S1 and G1S1 maintained gel integrity up to 3 weeks, and scant inflammatory cells were observed. This may be attributed to the delay of hydrogel degradation, allowing unreacted acid by-products to be eliminated through the regular metabolic process, which prevented their accumulation. Therefore, the described system circumvented a major limitation of PLGA depots to provide greater biocompatibility in vivo.
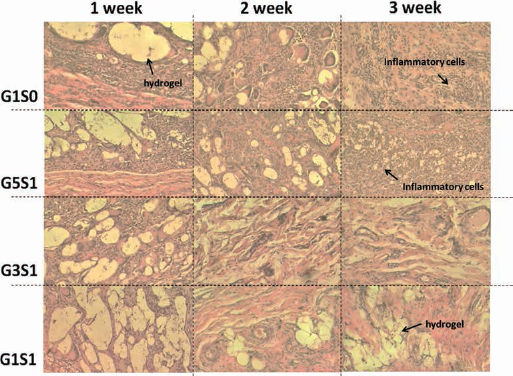
Bright-field microscopic images of hydrogel and surrounding tissues removed at designated times after hematoxylin and eosin (H&E) staining.
Conclusion
Although PLGA can undergo hydrolysis and be metabolized in the physical environment, its by-products of lactic acid and glycolic acid lower the local pH, which in turn induces an inflammatory reaction. This specific character may be used to drive reactions that require acids. In the event of combining PLGA with strontium carbonate, the small molecule drains local protons and neutralizes the pH while releasing strontium ions, with carbon dioxide and water as the final by-products. This reaction significantly improved in vitro cell compatibility and ameliorated in vivo inflammatory response due to milder microenvironment condition. Degradation controlled reactions allowed slow and controlled release of strontium ions, which in their free form would leak immediately from the hydrogel system after preparation. The results herein suggest that an injectable thermosensitive strontium carbonate hydrogel exhibits potential as a minimally invasive depot for the sustained release of strontium ions in the treatment of various bone-related ailments.
Footnotes
Acknowledgements
We would like to thank the Center for Advanced Molecular Imaging and Translation, and Expensive Advanced Instrument Core Laboratory at Chang Gung Memorial Hospital, Linkou, for their help in this project.
Financial support: This work was supported by grants CMRPG3D0611 and CMRPG3E0071 from Chang Gung Memorial Hospital.
Conflict of interest: None of the authors has any financial interest related to this study to disclose.

