Abstract
Tendon injuries present a significant clinical challenge due to their limited natural healing capacity and the mechanical demands placed on these tissues. This review provides a comprehensive evaluation of the current strategies and advancements in tendon repair and regeneration, focusing on biomaterial innovations and scaffold design. Through a systematic literature search of databases such as PubMed, Scopus, and Web of Science, key studies were analyzed to assess the efficacy of biocompatible materials like hydrogels, synthetic polymers, and fiber-reinforced scaffolds in promoting tendon healing. Emphasis is placed on the role of collagen fiber architecture, including fiber diameter, alignment, and crimping, in restoring the mechanical strength and functional properties of tendons. Additionally, the review highlights emerging techniques such as electrospinning, melt electrowriting, and hybrid textile methods that allow for precise scaffold designs mimicking native tendon structures. Cutting-edge approaches in regenerative medicine, including stem cell therapies, bioelectronic devices, and bioactive molecules, are also explored for their potential to enhance tendon repair. The findings underscore the transformative impact of these technologies on improving tendon biomechanics and functional recovery. Future research directions are outlined, aiming to overcome the current limitations in scaffold mechanical properties and integration at tendon-bone and tendon-muscle junctions. This review contributes to the development of more effective strategies for tendon regeneration, advancing both clinical outcomes and the field of orthopedic tissue engineering.
Keywords
Method
This review was organized using a structured approach to ensure comprehensive coverage of current advancements in tendon repair and regeneration. The primary databases utilized for the literature search included PubMed, Scopus, and Web of Science. The search strategy employed a combination of relevant keywords, including “tendon repair,” “biomaterials,” “scaffold design,” “hydrogels,” “tendon regeneration,” and “regenerative medicine.” The time frame for the literature search ranged from 2010 to 2023 to focus on recent innovations and significant contributions to the field. The inclusion criteria emphasized studies that presented innovations in biomaterials, scaffold design, and regenerative approaches for tendon repair. Exclusion criteria ruled out studies that were not available in English or lacked robust clinical or experimental data. Articles were reviewed for their contributions to advancements in biomaterials, scaffold mechanics, and tissue engineering strategies relevant to orthopedic applications.
Introduction
Tendons are crucial components in the musculoskeletal system, as connective tissues that link muscles to bones, and they are known for their impressive tensile strength and ability to transfer the mechanical force generated by muscle contractions to bones and facilitate movement. Tendon primarily consists of highly organized collagen fibers. The hierarchical structure of these tissues is critical for their performance. The collagen fibers—mainly type I—are grouped into fascicles, which are the primary structural units, and these fascicles are further composed of smaller subfascicles (Figure 1). The diameter of these collagen fibers typically ranges from 1 to 30 μm, while fascicles can be 50–1000 μ, and subfascicles are 15–400 μm.1 –3 The collagen fibers within the fascicles are surrounded by the endotenon, a loose connective tissue containing fibroblasts, which facilitates the sliding of fascicles against one another during movement. The entire tendon is then enveloped by the epitenon, another connective tissue sheath, which further encases the dense collagenous tissue (Figure 1).4 –6 This complex organization allows for the tendon tissues to withstand the mechanical forces and stresses they encounter during body movements. The interfibrillar matrix, rich in proteoglycans, glycoproteins, and water, fills the spaces between the collagen fibers and is critical for the viscoelastic properties of the tissues, contributing to their ability to absorb compressive forces and resist deformation.7 –10 It’s noteworthy that the composition and the proportion of these components can vary between different tendons, reflecting their specialized functions and locations in the body.8,11 For instance, the Achilles tendon, which bears a significant load with each step, has a different composition and structure compared to the less burdened tendons in the hand (Table 1). 12

The basic structure of a tendon. 13
Generalized values for tensile strength, water content, modulus, and collagen content for major tendons. It’s important to note that these values can vary based on several factors, including the method of measurement, the location within the tendon, age, and activity level of the individual.
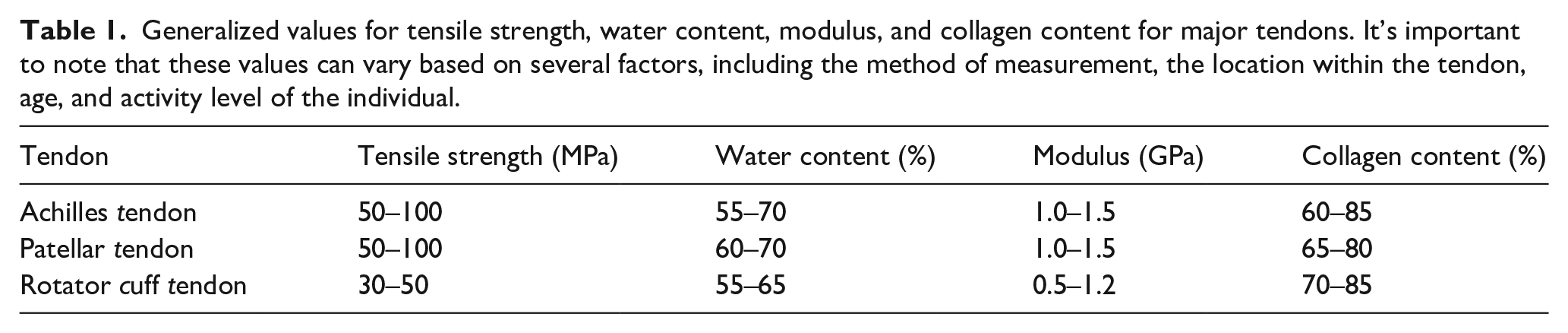
Tendon ruptures are significant clinical problems that can lead to substantial pain, disability, and healthcare costs. These injuries occur when the tensile load applied to the tendon exceeds its intrinsic strength, leading to partial or complete tears14,15 (Figure 2). Factors contributing to such ruptures include acute trauma, overuse, aging, and degenerative changes within the tissue.16,17 For athletes and physically active individuals, sudden, excessive force or incorrect movements can lead to acute ruptures, while repetitive stress over time is a common cause of overuse injuries. Systemic diseases such as rheumatoid arthritis or metabolic conditions like diabetes can also predispose individuals to higher risks of these injuries.18,19 The global incidence of tendon injuries is hard to pinpoint precisely due to varying reporting standards and healthcare practices worldwide. However, in Western populations, it is estimated that such injuries are relatively common, particularly among the athletic and aging populations.20,21 The economic burden of tendon repair is substantial. The economic burden of tendon injuries is significant, with the global incidence of tendon ruptures estimated at 80–90 cases per 100,000 inhabitants annually, accounting for 6–7 million cases per year. This burden is further amplified by prolonged recovery periods and high rates of re-rupture, contributing to disability and lost productivity. For example, 6% of flexor tendon repairs require reoperation, with workers’ compensation patients being 63% more likely to undergo reoperation.22,23 Direct costs include surgery, hospital stays, physical therapy, and medications, while indirect costs encompass lost wages, productivity, and long-term disability expenses. Moreover, the long-term recovery and rehabilitation process, which can last from several months to over a year, adds to the indirect costs and personal burden.24 –28

The mechanical properties of the tendon (Tendon stress-strain curve). 13
The repair process of the injured tendons is a complex biological sequence that involves both intrinsic and extrinsic healing mechanisms. Upon injury, an acute inflammatory response is initiated, characterized by the recruitment of platelets and inflammatory cells to the site of damage. These cells release cytokines and growth factors that are essential for the subsequent stages of healing. The proliferative phase follows, during which tenocytes begin to synthesize and lay down new extracellular matrix, including collagen fibers, to form a repair tissue. The final phase of healing is remodeling, where the initially disorganized collagen fibers become aligned along the lines of mechanical stress, improving the tensile strength of the repaired tissue. This phase can take several months and is crucial for restoring the functional properties of the tendon.29 –31 However, the repaired tissue often fails to regain the biomechanical properties of the uninjured tissue, resulting in a predisposition to re-injury. Throughout the repair process, mechanical loading is of paramount importance. Controlled mobilization and physical therapy are typically employed to facilitate proper collagen fiber alignment and to prevent adhesions and scar tissue formation. However, the balance between loading and overloading the healing tissue is delicate and individualized rehabilitation protocols are required to optimize recovery.26,32,33
Advances in regenerative medicine, including the use of stem cells, growth factors, and tissue engineering, are being explored to enhance the repair process and to develop strategies that more closely restore the native structure and function of injured tendons.26,34,35 Despite these innovations, the complexity of tendon repair presents ongoing challenges, and continued research is critical to improve patient outcomes (Table 2).
The surgical treatment of tendon injuries is often a consideration when conservative measures, such as physical therapy and bracing, fail to restore function or when the tendon has sustained a complete rupture. Surgical intervention aims to reapproximate the torn ends of the tendon or to reconstruct the damaged segment, providing mechanical strength and stability to facilitate healing and functional recovery.27,36 When considering surgical options, primary repair of the tendon is often preferred when feasible. This involves directly suturing the torn ends of the tendon together, which is typically advantageous due to the preservation of the original tissue’s biomechanical properties. However, this approach is sometimes limited by the quality of the remaining tendon tissue and the extent of the injury. In cases where primary repair is not possible or advisable, alternative surgical options include the use of graft materials for tendon reconstruction.37,38 Autografts, taken from the patient’s tissue, are considered the gold standard due to their biocompatibility and lower risk of immune rejection. They maintain similar biomechanical properties to the native tissue, which is beneficial for functional outcomes. However, they do come with disadvantages, such as donor site morbidity and limited availability of graft material.39,40
Decellularized xenografts, derived from animal tissues that have been processed to remove cellular components, present another option. These grafts provide a scaffold for the patient’s cells to repopulate and can reduce the risk of disease transmission compared to traditional xenografts. Nevertheless, there may still be a risk of an immune response, and their biomechanical properties may not match those of native human tissue as closely as autografts do.41 –43 Allograft scaffolds, sourced from human donors, offer the advantage of avoiding donor site morbidity while providing a scaffold that is biologically similar to the patient's tissue. They can be readily available in various sizes and shapes, making them suitable for extensive injuries. However, allografts carry a slight risk of disease transmission, and there may be variability in graft quality. Additionally, the potential for an immunogenic response exists, although it is reduced compared to xenografts44 –46 (Table 3).
The surgical treatment options for tendon injury.

Bioelectronic therapies are emerging as promising approaches for the treatment of tendon injuries, leveraging the body’s innate healing mechanisms and advancing the field of regenerative medicine. One area of innovation within bioelectronic therapies involves the use of electrical stimulation to promote tendon healing. Electrical stimulation can enhance cellular activities such as migration, proliferation, and differentiation, which are crucial for tissue regeneration. Research has demonstrated that low-frequency electrical stimulation can effectively modulate inflammatory responses and accelerate tissue repair, making it a valuable tool in the management of tendon injuries. Additionally, the integration of bioelectronic devices with advanced biological treatments such as stem cell therapy is gaining traction. For instance, the use of exosomes derived from mesenchymal stem cells has shown significant potential in tendon repair due to their ability to modulate the immune response and stimulate tissue regeneration. These exosomes, when combined with bioelectronic stimulation, could enhance the therapeutic outcomes by promoting a more conducive environment for tendon healing. 49 Another promising area involves the use of non-coding RNAs (ncRNAs) in conjunction with bioelectronic therapies. NcRNAs have shown significant promise in tendon injury treatment by regulating gene expression and cellular processes critical for tendon healing. Their integration with bioelectronic devices could lead to novel therapeutic strategies that enhance the natural healing processes of tendons. 51 In addition, Peniche et al. 52 explored the use of non-coding RNAs, such as siRNAs and miRNAs, as molecular tools to modulate gene expression during tendon healing. Their work demonstrated the ability of siRNAs to reduce fibrosis by silencing genes involved in collagen type III production, while miRNAs like miR-29 and miR-124 showed significant roles in promoting collagen type I synthesis and reducing scarring. These findings are supported by another study that highlight the therapeutic potential of miRNAs in tendon injury, especially through the regulation of key signaling pathways such as TGF-β and SMAD3. 53 Furthermore, A study by Fernández-Yague et al. 54 explores the development and application of a self-powered piezo-bioelectric device designed to regulate tendon repair processes through the modulation of mechanosensitive ion channels. This device, composed of a piezoelectric collagen-analogue scaffold made from poly (vinylidene fluoride-co-trifluoroethylene) (PVDF-TrFE), harnesses the electrical fields generated by physiological motion to stimulate tendon-specific signaling pathways. The study demonstrates that electromechanical stimulation using this device promotes tendon repair in vitro and in vivo, particularly by enhancing collagen synthesis, maintaining tendon-specific gene expression, and inhibiting the formation of non-tenogenic tissues such as cartilage and bone. The findings suggest that this innovative approach could offer a significant advancement in the treatment of tendon injuries by providing a means to control and enhance the natural healing processes through bioelectrical cues.
Fibrous materials for Tendon repair
Biocompatible synthetic materials, such as polyester-based Dacron, and polytetrafluoroethylene (PTFE)-based Gore-Tex, have been employed in the repair of tendon ruptures with varying degrees of success.55,56 Dacron, a durable and versatile polyester, has been used in reconstructive surgeries due to its strength and stability. 57 Gore-Tex, known for its expanded PTFE composition, has been favored for its inert properties and reduced tissue reactivity. 58 However, despite their initial mechanical strength and the ease with which they can be shaped and used, synthetic materials generally possess disadvantages when compared to biological grafts. One of the main limitations is their lack of biological integration, which can lead to a foreign body reaction characterized by chronic inflammation and fibrosis. Over time, this can compromise the material’s structural integrity and potentially lead to graft failure.35,59 Furthermore, these materials do not have the same viscoelastic properties as native tendons, which can result in an inadequate biological response to mechanical stress and strain during the healing process. Complications associated with the use of synthetic materials in tendon repair include synovitis, persistent inflammation, and the potential for material fatigue and wear, which can result in the release of wear debris and subsequent osteolysis. There is also an increased risk of infection, which can be particularly challenging to manage due to the potential for biofilm formation on the synthetic surface. In some cases, the material may fray or rupture, necessitating revision surgery.60 –62
Key factors in the design of biomaterials for tendon repair and regeneration
An ideal engineered scaffold for tendon repair and regeneration must satisfy a series of stringent criteria to ensure successful outcomes. Primarily, the scaffold must be biocompatible to avoid eliciting an adverse immune response upon implantation. 63 Its biomechanical properties should align closely with those of native tendon tissues, possessing the requisite tensile strength, elasticity, and flexibility to support physiological loads and facilitate the transmission of mechanical cues essential for healing.64,65 The scaffold should also be biodegradable, with a degradation rate that matches the rate of new tissue formation, thus providing temporary mechanical support while gradually transferring load to the emerging native tissue.35,66 From a biological standpoint, the scaffold should promote cellular attachment, proliferation, and differentiation. This can be accomplished through the incorporation of biochemical cues and growth factors that mimic the native extracellular matrix (ECM) and cellular environment.2,67 Furthermore, the scaffold’s architecture should be porous to support vascularization and nutrient diffusion, which are critical for cell survival and tissue integration.35,68 The design of the scaffold should also consider ease of delivery and the potential to be pre-seeded with cells or to recruit endogenous cells once implanted. Understanding the intricate architecture at both the macroscopic and microscopic levels of the tendon is crucial for the engineering of scaffolds aimed at tendon repair and regeneration, as these structures must mimic the native organization to restore function effectively.
Traditionally, tissue engineering approaches have relied on the triad of a suitable biocompatible scaffold, the introduction of appropriate cell lines, and the application of growth factors to foster a better biological response and facilitate the complex process of tissue regeneration.69,70 This orchestrated interaction aims to create an environment conducive to tissue integration and healing, mirroring the native tissue’s mechanical and biological properties. However, the application of growth factors has been met with challenges, including issues of stability, controlled delivery, costs, and potential side effects. Consequently, scientists have been investigating alternative material-based strategies to circumvent these limitations. One such strategy is the development of composites or surface coatings that incorporate bioactive inorganic micro- or nanoparticles within a polymer matrix.71,72 These composites can elicit specific cellular responses through the controlled release of biologically relevant ions such as strontium, zinc, silicon, and copper, which can influence processes such as cell adhesion, proliferation, differentiation, and neovascularization, without the direct introduction of growth factors73 –76 (Table 4). By doing so, they attempt to recreate the biochemical cues naturally present in the ECM. This approach seeks not only to replicate the biophysical characteristics of the tendon tissues but also to provide the necessary biochemical signals to foster endogenous tissue regeneration, potentially offering a more controlled and sustainable therapeutic avenue for tendon repair.
The role of different ions in tendon repair and regeneration.

Hydrogel in tendon regeneration
Hydrogel has emerged as a promising material for tendon regeneration due to its unique properties and biocompatibility. 89 In the field of regenerative medicine, a major challenge lies in developing scaffolds that can mimic the native ECM and provide a supportive environment for tissue growth and repair. Hydrogels, which are three-dimensional networks of hydrophilic polymers, possess a high water content and mechanical properties that closely resemble soft tissues.90,91 When used in tendon regeneration, hydrogels can serve as a biomimetic platform for cell adhesion, proliferation, and differentiation. They can also provide a controlled release of growth factors and bioactive molecules to promote tissue healing. Furthermore, the porous structure of hydrogels allows for nutrient and oxygen diffusion, facilitating cellular activities and tissue regeneration in vitro and in vivo experiments90,92 (Figure 3).
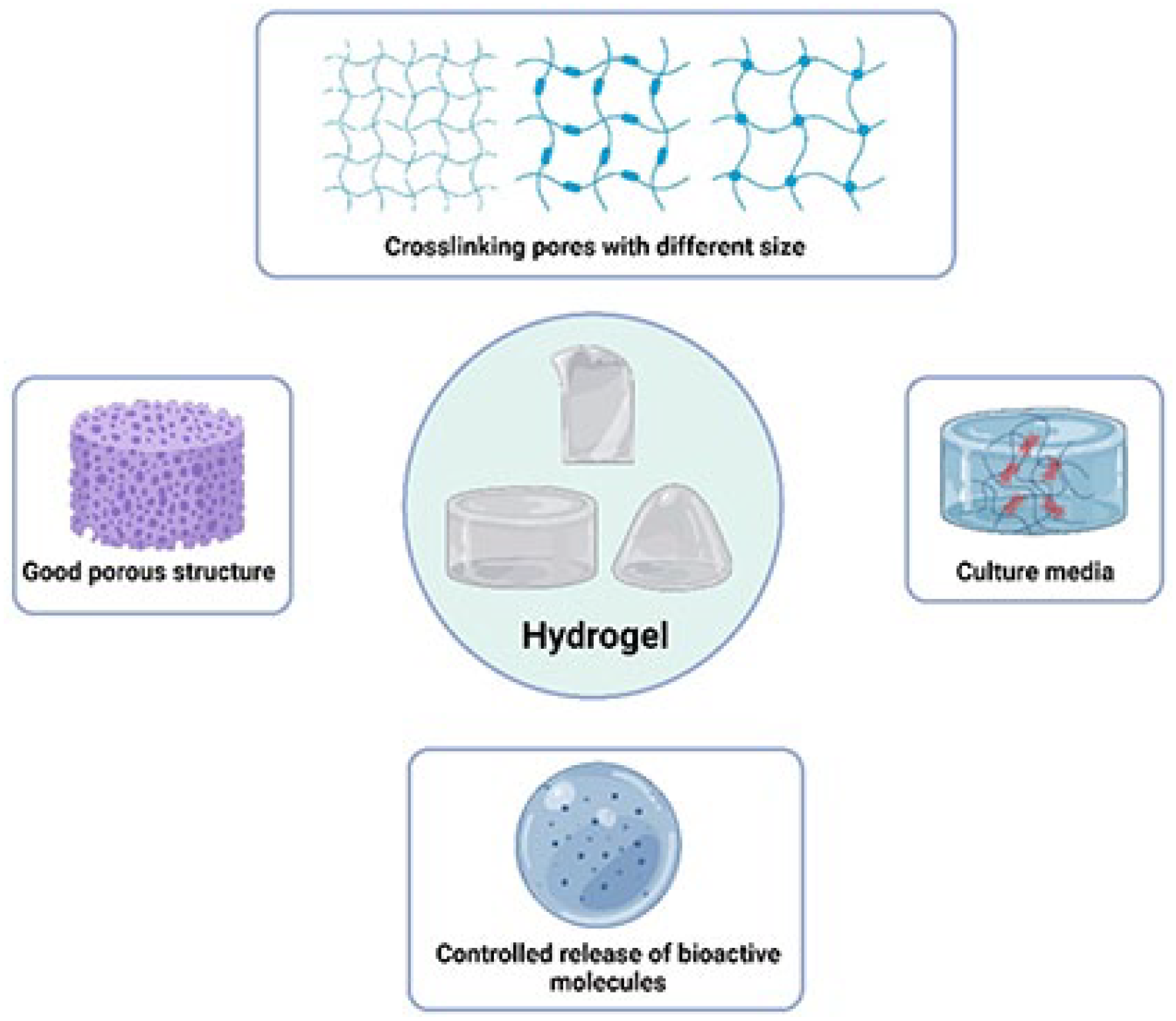
General features and characteristics of the hydrogel.
In recent years, there has been a surge of interest in developing hydrogel systems using natural polymers for various biomedical applications. These studies have focused on harnessing the unique properties of natural polymers, such as chitosan, collagen, fibrin, alginate, gelatin, and hyaluronic acid, to create hydrogels with enhanced biocompatibility and bioactivity. 93 Natural polymers offer several advantages compared to synthetic polymers, including their abundance, biodegradability, and ability to mimic the native extracellular matrix. 94
Chitosan is a deacetylated derivative of chitin, which is found in the exoskeleton of crustaceans. Its biocompatibility and biodegradability, along with its inherent antibacterial properties, make it a promising material for tendon repair. Chitosan-based hydrogels can be designed to encourage fibroblast proliferation and alignment, which are essential for the synthesis of organized tendon ECM.95,96 Collagen-based hydrogels provide a natural environment for cells, which is conducive to the regeneration of these tissues. They can facilitate the alignment of newly formed collagen fibers, thereby improving the mechanical strength and functionality of the repaired tissue. 59 Fibrin hydrogels can serve as a temporary scaffold that is eventually replaced by the body’s tissue. They support cellular infiltration and provide a matrix for the deposition of new ECM components during the healing process. 97 Gelatin is a denatured form of collagen that retains the ability to support cell adhesion and growth. Gelatin-based hydrogels can be crosslinked to alter their physical properties and degradation rates, making them versatile materials for tissue engineering applications.98,99 A study investigates the use of three-dimensional pure bovine gelatin scaffolds for healing significant tendon defects, using a rabbit Achilles tendon model. The research found that the gelatin scaffold facilitated superior tendon repair compared to untreated control model, as evidenced by improved weight-bearing, reduced adhesion formation, and better collagen organization in the treated tendons. 100 Biomechanical testing also showed that tendons treated with the gelatin scaffold had higher load-bearing capacity and stiffness, indicating a stronger and more functional repair. The scaffold was completely resorbed and replaced by new tendon tissue, suggesting that gelatin scaffolds could be a promising option for surgical reconstruction of large tendon injuries. 100 Alginate is a polysaccharide derived from brown seaweed, known for its gel-forming properties in the presence of divalent cations. 101 Therefore, alginate hydrogels can encapsulate cells and growth factors, maintaining a moist wound environment and facilitating cell-mediated tissue repair.102,103 Furthermore, Hyaluronic acid (HA) is a naturally occurring polysaccharide in the ECM and is particularly abundant in soft connective tissues. 104 HA hydrogels provide excellent biocompatibility and can be modified to support the slow release of growth factors, enhancing the healing of tendons89,105(Table 5).
The characteristics and roles of natural polymer hydrogel in tendon repair and regeneration.

However, despite the advantages of natural hydrogel-based scaffolds, these scaffolds still face the critical challenge of matching the load-bearing capacity of native tendons. While they facilitate the formation of new tissue, the mechanical strength of hydrogels is typically less robust initially, and they do not provide the necessary support immediately after implantation. This limitation necessitates a gradual rehabilitation protocol or the use of additional support structures to protect the repair site during the early stages of healing until the scaffold is sufficiently integrated and replaced by new, mechanically competent tissue.
Fiber-reinforced hydrogels (FRH) are being developed for effective tendon repair, combining sufficient strength for structural support with biological activity to facilitate proper integration with the ECM.113,114 Recent advancements in biomaterials have focused on the potential of FRH. In these composites, the integration of fibers significantly improves various mechanical properties, including toughness, modulus, and strength, particularly along the fiber alignment. This enhancement mirrors the improvements seen in other composite materials like fiber-reinforced composite structures and fiber-reinforced concrete.115–117 Typically, the production of fibers or fiber scaffolds involves methods such as electrospinning, and more advanced techniques like melt-electrowriting are employed for greater structural control.115,117 –119 Following their production, these fibers are coated or submerged in a precursor solution for gel formation, which is then cross-linked. To shape the final structure of the FRH, a mold can be utilized before the crosslinking process begins. An alternative approach involves using a layer-by-layer assembly method, wherein constructs of FRH are constructed with alternating layers of electrospun fiber mats and a slender hydrogel layer.115,117 To effectively fabricate FRH, the techniques and synthesis procedures must be refined to ensure compatibility between the hydrogel and fiber fabrication processes. Important factors, including the choice of solvents, the temperature during synthesis, and the properties of the interface between the fiber and hydrogel, must be carefully considered and adjusted for optimal results. The degree of interaction between the fiber and the hydrogel matrix can lead to synergistic improvements in mechanical properties. Additionally, this interaction may result in changes such as limiting the swelling capacity of the hydrogel and modifying its Poisson ratio.120,121 Nevertheless, the FRH compositions documented in existing literature often utilize materials that are not ideally suited for positive biological interactions. These include substances like epoxy-based gels or stainless-steel fibers. Consequently, they lack biological characterization and do not have the necessary aligned structure to be effectively used as tendon scaffolds.120,121
In the development of FRH for tendon repair and regeneration, structures have been created using aligned, electrospun polycaprolactone fibers combined with coatings of gelatin gels or chitosan–hyaluronic acid, as well as chitosan–collagen gel reinforced with electrospun polylactic acid fibers.115,122 While these constructs have shown potential in supporting cell growth and alignment, their tensile strengths are typically below 5 MPa.100,115
The impact of collagen fiber architecture on the tendon regeneration
Fiber diameter
The diameter of collagen fibers is a fundamental aspect influencing the biomechanical and biological properties; including gene expression and cell behaviour of tendon tissues.64,123 The biomechanical properties of tendons are directly tied to the diameter of their collagen fibers. Larger fiber diameters are typically associated with increased tensile strength. This correlation is crucial since tendons are predominantly subjected to tensile forces. The larger fibers are more capable of withstanding these forces, reducing the risk of injury under normal and high-stress conditions, such as in athletic activities or heavy physical labor.7,30,124 Furthermore, the diameter of these fibers also influences the stiffness of the tissue. Stiffer tendons, resulting from thicker collagen fibers, can store and release more energy efficiently. This characteristic is particularly beneficial in activities requiring explosive movements, such as jumping or sprinting.15,125 On a biological level, the diameter of collagen fibers can affect cellular activities within tendons. Thicker fibers may provide a more robust framework for cellular attachment and proliferation, which is essential for tissue repair and regeneration. 123 Cardwell et al. 126 investigate the influence of electrospun fiber diameter and alignment on the differentiation of mesenchymal stem cells (MSCs) into tendon/ligament lineage. The study fabricated mats with varying fiber diameters—small (<1 μm), medium (1–2 μm), and large (>2 μm)—and orientations (random or aligned) using electrospinning of a poly (esterurethane urea) (PEUUR) scaffold. It was found that fiber diameter had a more significant impact on cellular behavior than fiber alignment. Erisken et al. 123 found that the greatest expression of collagen I and tenomodulin occurred on fibers with a diameter of 1.8 μm, in contrast to the lesser expressions on fibers with smaller diameters. This aspect is especially significant during the healing process following an injury, where the reformation of collagen fibers of appropriate diameter is crucial for restoring the original strength and functionality of the tissue. Conversely, thinner collagen fibers confer greater flexibility to the tendons. This flexibility is essential for the range of motion required in joints. Tendons with thinner fibers can more readily adapt to the stretching and twisting movements involved in various physical activities, from daily tasks to athletic endeavors. The diameter of collagen fibers is not static and can change due to aging or pathological conditions. For instance, aging increases collagen fiber diameter, leading to stiffer tendons. This change can reduce elasticity and increase susceptibility to injuries. Pathological conditions like tendinopathies can also alter fiber diameter, impacting tissue function and health.127,128
Fiber alignment
The alignment of collagen fibers within tendon tissues plays a pivotal role in their biomechanical functionality and overall health. This aspect of collagen fiber architecture is integral to how these tissues respond to mechanical forces and maintain structural integrity. 32 Collagen fibers in tendons are typically aligned in parallel formations. This orientation is essential for the primary function of these tissues: transmitting tensile forces from muscle to bone. The parallel alignment allows these fibers to work collectively, providing maximal strength in the direction of force application.129,130 This anatomical arrangement is particularly efficient in resisting tensile loads while minimizing damage or deformation. Proper alignment of collagen fibers contributes to the biomechanical efficiency of tendons.130,131 When fibers are well-aligned along the axis of force, the tissue can handle more significant loads without excessive stretching or risk of injury. This efficiency is crucial in dynamic and high-impact activities, where rapid force transmission is essential for performance and safety. The alignment also affects the elastic properties of these tissues. While tendons are not highly elastic, their limited elasticity is optimized by the aligned collagen fibers. This arrangement allows for a degree of stretch and recoil, providing a balance between flexibility and rigidity. It’s a critical feature for absorbing and releasing energy during movement, such as running or jumping.51,132 Injury or disease can disrupt the alignment of collagen fibers, for example, in tendon injuries; the healing process often involves the formation of scar tissue, which may not replicate the original, orderly alignment of collagen fibers. This misalignment can lead to reduced strength and flexibility, increasing the risk of re-injury and potentially leading to chronic issues. Therefore, therapeutic interventions often aim to promote the re-establishment of proper fiber alignment during the healing process.33,133 It’s also noteworthy that the degree of alignment can vary among different tendons, influenced by their specific functional demands. For instance, tissues subjected to more unidirectional forces generally exhibit more linear fiber alignment compared to those under multi-directional stresses. On the other hand, A study demonstrates that tendon fibroblasts cultured on aligned polycaprolactone (PCL) fiber mats maintain a more favorable phenotype and exhibit less degenerative extracellular matrix remodeling compared to those on randomly oriented fibers, especially when subjected to inflammatory conditions. 133 Sensini et al. highlighted the potential of hierarchical electrospun scaffolds made from poly-L-lactic acid (PLLA) and collagen blends, designed to replicate the multiscale organization of tendons, ranging from collagen fibrils to tendon fascicles. These scaffolds promote significant cellular alignment and elongation, especially when subjected to dynamic stretching conditions (Figure 4). 134 In another study, electrospun PLLA/collagen scaffolds were designed to closely mimic the biomechanical properties of tendon fascicles, demonstrating favorable mechanical strength, stiffness, and durability. These scaffolds maintained structural integrity after crosslinking and immersion in phosphate-buffered saline, with high-resolution X-ray computed tomography (XCT) confirming the retention of fiber alignment and biomimetic architecture. Furthermore, in vitro studies showed increased metabolic activity of fibroblasts, indicating excellent biocompatibility and the potential for enhanced ECM production.135,136

(a) The production, structure, and cellular behavior of electrospun hierarchical scaffolds designed for tendon and ligament regeneration. The scaffolds, composed of aligned nanofibers, were cultured with fibroblasts under static and dynamic conditions. Imaging techniques, including (c) high-resolution X-ray tomography (XCT), (d) scanning electron microscopy (SEM), (b) fluorescence microscopy, and (e) histology, were used to assess fibroblast morphology and alignment. The results show that static cultures promote circumferential alignment of fibroblasts, while dynamic conditions induce axial alignment and elongation of the cells along the scaffold fibers. 134
Fiber crimping
Fiber crimping in collagen fibers refers to the wave-like, zigzag pattern observed in the structure of tendons. This unique feature of collagen fiber architecture has a profound impact on the biomechanical behaviour and biological properties of these tissues.137,138 The crimped structure of collagen fibers endows tendons with a combination of strength and elasticity. When these tissues are subjected to normal physiological loads, the crimps unfold, allowing the fibers to stretch.137,138 This mechanism provides a degree of elasticity which is crucial for the efficient functioning of tendons. It enables the tissue to absorb and dissipate forces, particularly during high-impact or dynamic activities or rapid directional changes. This feature is vital in repetitive motions, where it helps to reduce fatigue and wear on the tissues. The ability of crimped fibers to store and release energy efficiently contributes to the overall resilience and durability of tendons and ligaments. 139 The pattern and degree of fiber crimping can be altered in response to injury, overuse, or aging. Changes in crimping can lead to decreased elasticity and altered biomechanical properties, potentially increasing the risk of further injury.140,141 During the healing process, the restoration of the original crimp pattern is a key factor in regaining the full functionality of the tissue. However, this restoration can be challenging, as scar tissue formation often results in less organized and less effective crimping patterns.139,142 Interestingly, the crimping pattern of collagen fibers can adapt to changes in mechanical stress. Regular physical activity and specific types of exercises can influence the degree and pattern of crimping, potentially enhancing the strength and resilience of tendons. 143 This adaptive response is an important consideration in exercise regimens and rehabilitation protocols designed to strengthen or repair these tissues.
Advanced collagen fibrous architecture techniques
Conventional techniques used to manufacture different architectural scaffolds for tendon regeneration fail to create a fibrous structure like the native ECM of the tendons. Therefore, in recent years, different fiber formation techniques have been investigated, and the most adapted one is the solution electrospinning (SES) 1 (Table 6). SES involves using an electric field to draw out fine fibers from a polymer solution. This process can produce fibers with diameters ranging from a few nanometers to several micrometers, closely resembling the size scale of natural ECM fibers, but still suffer from patterning control limitation and reproducibility issues. Thus, many studies have modified the SES process to enhance the control over fibber patterning by applying rotating mandrels, parallel electrodes, and sacrificial materials.144 –147 A study indicates a new SES setup containing two parallel ceramic magnets and copper electrodes causes the aligned PCL nanofibers collection in multi-layers, in addition, the fabricated aligned scaffolds increase mechanical properties, collagen alignment, and tendon gene expression. 148 Another study targeted the improvement of control over fiber geometry by the combination of SES and rotating mandrel of water-soluble polyethylene oxide (PEO) and poly L-lactide (PLL). 145 Furthermore, the application of postspinning thermal and sacrificial fiber population treatment showed higher PLLA nanofibers crimped morphology and scaffold porosity. 145
The recent fiber patterning technologies and fiber scaffold characteristics used in tendon regeneration.

Yu et al. 149 developed a tri-layered nanofibrous scaffold composed of PLGA integrated with doxycycline, collagen, and bupivacaine for the repair of ruptured Achilles tendons in rates. Using an electrospinning technique, they successfully incorporated these biomolecules to enhance the scaffold’s healing properties, structural support, and pain management capabilities. The study demonstrated that these scaffolds provided a sustained release of the drugs over 28 days and significantly improved the mechanical strength of the repaired tendons compared to untreated controls. In addition, the histological analysis revealed a higher density of collagen I and improved tendon organization, suggesting the scaffold’s potential effectiveness in clinical applications for tendon repair. 149 Therefore, electrospinning is a versatile technique that allows for the fabrication of nanofibrous scaffolds designed to mimic the hierarchical structure of tendon tissues, replicating their mechanical and functional properties across different scales. At its core, electrospinning utilizes a high-voltage electrostatic field to stretch a polymer solution into fine fibers, which can be deposited in various configurations, such as mats, bundles, or 3D structures. These fibers, which range from nanometers to micrometers in diameter, closely resemble the collagen fibrils found in tendons, providing a biomimetic structure that enhances cellular attachment and tissue regeneration. This is particularly important at the smallest scale, where nanofibers can replicate the collagen fibrils that provide tendons with their tensile strength (Figure 5).150,151 To accurately mimic tendon tissue, fiber alignment is a key factor. In tendons, collagen fibers are aligned in parallel to the direction of tensile load, and electrospinning allows for this alignment through the use of a rotating drum collector or patterned surfaces. This strategy not only enhances the mechanical properties of the scaffold but also promotes the alignment of cells and ECM production, which are critical for effective tissue regeneration.152,153 Moreover, electrospinning can be used to bundle aligned fibers into larger hierarchical structures that replicate the fascicles found in tendons. These fascicles are formed by grouping collagen fibers, which are then arranged into subfascicles and larger tendon units. Researchers have successfully created these bioinspired hierarchical assemblies, improving both the mechanical strength and the biological functionality of the scaffolds.150,154 In addition to replicating the internal structure of tendons, electrospinning can also mimic the outer layers, such as the epitenon and endotenon, which protect tendons and facilitate their movement. Multi-layered electrospun scaffolds can be designed to replicate these protective layers, providing structural integrity while allowing for nutrient exchange and cellular infiltration.153,155 Through these strategies, electrospinning creates scaffolds that not only replicate tendon architecture but also offer mechanical properties and biological cues essential for tendon repair and regeneration.152,156 Astudy by Ramos et al. 157 investigates the potential of insulin-functionalized electrospun nanofiber matrices, with and without bone marrow-derived mesenchymal stem cells (MSCs), to enhance tendon repair in a rat Achilles tendon injury model. The researchers developed and implanted these bioactive matrices into transected tendons and evaluated the outcomes over 8 weeks. The study found that matrices functionalized with insulin, especially when combined with MSCs, significantly increased collagen I and III synthesis and improved collagen alignment in the repaired tendons. However, the mechanical properties of the repaired tendons did not match those of the native tendon, indicating that while the insulin-functionalized scaffolds improved biochemical markers of healing, they did not enhance the tensile strength of the tendons. 157

Recently, electro-hydrodynamic fiber printing methods, including melt electrowetting (MEW), have shown promise in overcoming these challenges through the precise and consistent deposition of microscale fibers. 158 Typically, this involves extending a single fiber onto a collector plate controlled by a computer, which moves horizontally with a static extruder nozzle. To create 3D structures, this process meticulously stacks fibers one after the other in sinusoidal, angled, or straight shapes.158,159 Numerous studies have successfully printed scaffolds made of PCL and poly (e-caprolactone-coacryloyl carbonate) with a crimped structure. This is achieved by adjusting the collector speed and the mass flow of the polymer jet.158,160 These studies demonstrate that the tensile stress-strain characteristics of MEW fiber scaffolds can be effectively controlled by altering the degree of fiber crimping. Notably, fibers with crimp angles in the range of 10%–13% have been shown to produce regions that closely resemble those found in natural collagenous tissues, such as tendons and ligaments.158,160
A study introduced a novel method to adjust the morphology of fibers in MEW, enabling the creation of angled fiber patterns. These microfibers showed impressive compliance under dynamic tensile stretching, making them particularly promising for soft tissue engineering, especially in tendon engineering. The diameter of fibers produced by MEW typically falls between 5 and 50 μm.161,162 This range aligns with that of human collagen fibers, although it’s notably larger than the finer dimensions achieved through solution-based electrospinning. The ideal target would be to mimic the diameter of collagen fibrils, which are key in the topographical environment sensed by tenocytes in tissues and are crucial for preventing phenotypic drift. However, achieving these smaller diameters representative of natural collagen fibrils remains a challenge.
A major challenge in MEW lies in achieving controlled deposition of non-straight fibers to create highly ordered, large-volume 3D scaffolds, especially those with thicknesses exceeding 1 mm.53,163 Experimental studies have highlighted the impact of residual charges on the printed fibers, which can interfere with precise fiber stacking. This issue, although more pronounced in MEW, is also a common challenge across various spinning techniques, where electrical charge dynamics can limit scalability. 164 To address this, researchers are exploring hybrid methods that merge spinning techniques with textile technologies. This innovative approach typically involves converting mono- or multi-fibers, initially produced by MEW, SES, or wet spinning, into volumetrically significant structures.146,153 This is achieved using various textile methods, such as braiding, knitting, or weaving. Such hybrid fabrication techniques enable the combination of different natural and synthetic polymers, like PCL /chitosan/cellulose nanocrystals, silk fibroin/bromogelatine, PCL/PLA (polylactic acid), and others into yarns.153,165 These yarns can then be crafted with various braided or twisted angles, offering versatility in scaffold design. This approach not only overcomes the limitations posed by residual charges but also expands the potential applications of these fibers in creating more complex and functionally diverse 3D structures.153,164 PCL nanofiber yarns were initially produced using SES onto a specially modified rotating mandrel collector. Following this, these nanofiber yarns were combined with PLA multifilaments through a weaving textile strategy. The resulting woven composite fabrics closely resembled the highly organized collagen structure found in native tendons in terms of structure, size, and organization. 164 The mechanical properties of these woven composites were notable: they exhibited an ultimate tensile strength (UTS) of approximately 10 MPa and a tensile modulus of around 60 MPa. 164 In an innovative hybrid strategy focused on improving tensile mechanical properties, researchers crafted 3D fibrous scaffolds with a distinctive design. The core of these scaffolds was composed of aligned SES PCL nanofibers. Surrounding this core was a single-shell film embedded with elongated pores, created through laser perforation. The scaffolds thus formed exhibited both high porosity and mechanical characteristics similar to tendon tissues, demonstrating a UTS of about 56 MPa and a tensile modulus in the region of 375 MPa. 166 The strategic layering of a nanofiber core and a laser-perforated shell not only bolstered the scaffolds’ mechanical strength but also enhanced their anisotropic and waveform microstructure. This configuration significantly boosted the expression of key phenotypic markers linked to tenogenesis, such as tenascin, type-I collagen, and decorin. These markers play a vital role in the formation and repair of tendons. When these structured scaffolds were compared with control samples that had non-anisotropic and randomly aligned fibers, they demonstrated a significantly greater capability in fostering characteristics typical of tendon-like tissues. 166 Although synthetic fibrous scaffolds exhibit favorable mechanical characteristics, they face notable challenges, including poor cell adhesion and the risk of chronic inflammation in vivo. To address these issues, recent research has focused on creating fibrous scaffolds using the primary components of tendon ECM. In research, a method combining rotating electrophoretic compaction with macromolecular alignment was used to create aligned type-I collagen threads. These threads were then skillfully pin-woven to form scaffolds that were highly porous about 81% and suitably dimensioned 20 mm × 5 mm × 1.5 mm. 167 The resulting collagen scaffolds displayed a tensile modulus and UTS comparable to those of natural tendon tissues. Additionally, the specific weaving pattern of the collagen scaffolds replicated the crimp-like configuration typical of tendons, effectively facilitating the production of cartilage oligomeric matrix protein, collagen, and tenomodulin. This was particularly notable when the scaffolds were used in conjunction with differentiated MSCs, without requiring the addition of growth factors. 167
Tendon’s junctions
The Osteotendinous and Myotendinous Junctions serve as vital transitional areas located at the tendon’s extremities. These junctions play a crucial role in anchoring the tendon to the bone and muscle. Their unique structure facilitates a gradual transition in material composition, reducing the risk of stress concentrations and potential failure at the interface between different tissue types.173,174 The Osteotendinous junction is characterized by a four-zone continuum, each with its own specific mix of cell types, proteoglycans, collagen types, and varying degrees of mineralization. The tendon zone primarily consists of type I collagen and a limited number of proteoglycans, inhabited predominantly by tenocytes and fibroblasts. Next, the fibrocartilage region is rich in collagen types II and III, and a higher concentration of proteoglycans, housing resident chondrocytes. This region transitions into the bone through a mineralized interphase known as the tidemark. Finally, the bone itself is mainly composed of mineralized type I collagen and is the home to osteoblasts and osteoclasts.174–178 The myotendinous junction is characterized by a complex interweaving of muscle’s actin filaments and tendon’s collagen fibrils. This intricate connection is further strengthened by a basement membrane, which is notably abundant in laminin and collagen type IV. This structure not only ensures a robust linkage between muscle and tendon but also facilitates effective force transmission during muscle contractions.175,179 The creation of an ideal synthetic graft for tendon repair and regeneration necessitates addressing the critical challenge of fixation at both the bone and muscle interfaces. A synthetic tendon graft's efficacy is limited by its most vulnerable component. Therefore, it’s imperative to foster optimal tissue integration at the bone and muscle junctions of the synthetic tendon. 59
Given the distinct repair and regeneration requirements of different tissue types, which demand specific levels of stiffness, biomaterial composition, and architectural design for effective cellular activity, a graft with a uniform structure would fail to adequately support bone tissue integration at one end, tendinous tissue in the middle, and muscle integration at the other end. 180 To overcome this, a heterogeneously structured graft, which exhibits regional variations along its length, could be more effective. Such a design would facilitate cell differentiation, appropriate proliferation, and tissue growth, potentially leading to the formation of enthesis-like structures with diverse cellular populations around the implant. This variability could be achieved through techniques like developing graded polymer-inorganic composites, thereby altering both mechanical and biological properties. For instance, nanofibrous constructs with a graded calcium phosphate content can induce a linear, dose-dependent response in both stiffness and cellular proliferation, such as with murine osteoblasts. Additionally, varying fiber compositions (PCL, PLLA) and architectures (aligned and random within the same construct) can be achieved by modulating electrospinning parameters.181 –183
One promising method to regenerating the enthesis is the development of multiscale and multimaterial scaffolds that mimic the structural and functional properties of the native tissue. A notable example is the work of Micalizzi et al. 184 who combined electrospinning and 3D bioprinting to fabricate scaffolds with a gradient architecture that mirrors the transition from tendon/ligament to bone. These scaffolds demonstrated optimal mechanical properties, comparable to those found in native tissues, and supported the differentiation of MSCs into tenogenic and osteogenic lineages. The ability of these scaffolds to promote cellular adhesion, proliferation, and differentiation across different phenotypes makes them promising candidates for clinical applications aimed at repairing tendon or ligament injuries at the bone insertion site (Figure 6). In another study, Olvera et al. 185 explored the spatial presentation of tissue-specific ECM components along electrospun scaffolds to replicate the bone-ligament interface. By functionalizing different regions of the scaffold with ligament-ECM and cartilage-ECM, the researchers were able to direct MSC differentiation toward ligamentous or chondrogenic phenotypes, depending on the ECM present. This method mimicked the natural transition from the collagen-rich ligament to the fibrocartilage and bone regions, promoting spatially defined cell differentiation. Furthermore, the addition of a mineralized layer in the cartilage region facilitated the endochondral ossification process, crucial for recreating the bone attachment site. Recent advances in auxetic scaffolds—structures that expand laterally when stretched—are also contributing to enthesis regeneration. Sensini et al. 186 developed biomimetic nanofibrous scaffolds that exhibited auxetic behavior, a characteristic mechanical property of the natural enthesis. These scaffolds, made from a poly(L-lactic) acid and collagen blend, featured a region-specific arrangement of random and aligned nanofibers to mimic the structural gradient of the enthesis. Using synchrotron imaging techniques, the researchers were able to visualize the strain distribution across the scaffolds, showing that the auxetic behavior helped reduce stress concentrations at the tendon-bone interface. This auxetic design not only enhanced the mechanical performance of the scaffolds but also supported the differentiation of MSCs into both tendon and bone phenotypes, offering a promising solution for enthesis repair. Another study was presented by Alkaissy et al., 187 who combined electrospun filaments with 3D-printed structures to create biphasic scaffolds designed for rotator cuff repair. This scaffold mimicked the transition from soft tendon to hard bone by embedding electrospun polydioxanone (PDO) filaments within a 3D-printed PCL block. The scaffold supported the growth of tendon cells on the soft component and bone cells on the hard component, effectively replicating the functional gradient of the enthesis. The mechanical properties of this biphasic scaffold were optimized to withstand the high forces experienced at the tendon-bone junction, and the biological evaluation showed promising results for enhancing the outcomes of rotator cuff repairs, which often fail at the enthesis. In terms of creating more sophisticated scaffolds, Criscenti et al. 188 designed a triphasic scaffold (TPS) that mimicked the different regions of the bone-ligament interface. The scaffold consisted of a 3D fiber-deposited PCL structure and an electrospun polylactic co-glycolic acid mesh, which together created a gradient of mechanical and biological properties. The scaffold demonstrated region-specific biological responses, with increased metabolic activity and differentiation potential in both bone and ligament regions. This triphasic design allowed for a continuous transition between soft and hard tissues, facilitating the integration of the scaffold with both bone and ligament cells. Additionally, Camarero-Espinosa et al. 189 introduced a tri-phasic fibrous scaffold to mimic the structure of the ACL. This scaffold featured a wavy fiber arrangement in the intermediate zone, which replicated the characteristic “toe region” of the ACL, where the wavy collagen fibers dissipate loads before transferring them to the bone. The mechanical properties of the scaffold closely resembled those of the native ACL, and in vivo studies showed that the scaffold promoted cell infiltration and the deposition of organized ECM.

Multi-scale structural characterization of the enthesis scaffold, covering both macro- and nano-level features. (a) region that mimics the T/Ls behavior made of electrospun PLGA (ePLGA), (b) interface region composed of PCL extruded onto the PLGA structure (ePLGA+3DPCL), and (c) region that mimics the bone tissue characteristics made of PCL (3DPCL). (d, f, g–i) SEM and (e) microscopic images reveal the alignment and interaction of fibers in the scaffold, highlighting its ability to replicate the anisotropic structure crucial for tissue regeneration. 184
Regarding the myotendinous junction, Ladd et al. 181 introduce a co-electrospun dual scaffolding system designed to replicate the mechanical gradients present in the native MTJ. The authors created a scaffold composed of PLLA)/collagen and poly(ε-caprolactone) (PCL)/collagen, spun onto different regions of the scaffold to represent the stiff tendon and the compliant muscle, respectively. By tailoring these mechanical properties, they were able to produce a scaffold that mimicked the gradual transition seen in native MTJs, where muscle tissues have lower stiffness and tendons have higher stiffness. Mechanical testing revealed that the scaffold exhibited region-specific strain profiles, much like the native MTJ, and supported cell attachment and myotube formation, which are essential for functional muscle-tendon integration. Nevertheless, Sensini et al takes a different method by designing bi-material nanofibrous junctions to mimic the MTJ. They used electrospinning to create aligned nanofibers composed of polyurethane (PU) to simulate muscle-like properties and nylon 6.6 to simulate tendon-like properties. The study explored two types of junction geometries; flat and conical. Through mechanical and structural analyses using scanning electron microscopy and micro-computed tomography, they demonstrated that conical junctions more closely mimicked the natural MTJ’s structure, which features a gradual transition between the muscle and tendon tissues. Mechanical testing showed that the conical junctions exhibited stiffness and elastic modulus values similar to the natural MTJ, and these junctions could withstand cyclic loading without breaking. 190 Finally, a study focused on the development of an in vitro model of the MTJ using aligned electrospun PCL fibers. In their study, human myoblasts and tenocytes were co-cultured on these fibers, and cyclic strain (10%) was applied to simulate the biomechanical environment of the MTJ. The combination of mechanical strain and co-culture significantly enhanced cell elongation and upregulated the expression of MTJ-specific markers such as collagen type XXII and paxillin, which are critical for muscle-tendon integration. The study also showed that co-cultured cells produced higher levels of these MTJ markers compared to cells cultured separately, highlighting the importance of cellular interactions and mechanical cues in promoting the formation of a functional MTJ. 191
Conclusion
The exploration and advancement in the field of tendon repair and regeneration represent a significant stride in orthopedic medicine and biomechanical engineering. The intricate understanding of tendon structure, from their collagenous architecture to the critical junctions, has laid the groundwork for innovative therapeutic approaches. The pursuit of an ideal synthetic graft, capable of mimicking the native biomechanical and biological properties of tendons, has been a focal point of recent research. The future of tendon repair and regeneration hinges on the successful integration of biomaterial science with an intricate understanding of tendon physiology and pathology. The development of heterogeneous grafts, which cater to the specific requirements at different junctions (bone, tendon, and muscle), appears promising. These grafts aim to achieve optimal tissue integration, supporting diverse cellular populations and facilitating appropriate mechanical and biological responses. Innovations such as graded polymer-inorganic composites and nanofibrous constructs, offering variability in stiffness and biological properties, are at the forefront of this research. Moving forward, the challenge remains to refine these technologies to ensure not only their biomechanical compatibility but also their biological efficacy. The integration of these materials with the native tissue without eliciting adverse immune responses or complications like delamination and stress concentration points under physiological conditions is crucial. Additionally, the scalability of these solutions, their long-term viability, and their adaptability to various clinical scenarios will be key factors determining their success.
Footnotes
Acknowledgements
Not applicable.
Authors’ contributions
Writing – original draft: AA, YD, and XZ; Validation: WZ, SHAE, and VGK; Writing – review and editing: HL, WZ, SHAE, and SA; Supervision: HL, and SA; Data collection: AA, YD, VGK, and MHAHA.
Availability of data and materials
The dataset supporting the conclusions of this article is included in this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.

