Abstract
Abdominal aortic aneurysm (AAA), defined as a permanent and often asymptomatic dilatation of the abdominal aorta, poses a significant threat of rupture with high mortality, yet lacks effective pharmacological interventions for stabilisation or regression. Current surgical options are invasive or require strict anatomical suitability, leaving patients with small aneurysms under passive surveillance. This critical gap underscores the urgent need for novel therapeutic strategies. Nanomedicine has emerged as a promising approach, offering solutions for targeted drug delivery, precise imaging, and dynamic monitoring of AAA progression. This review comprehensively analyses recent advances in nanomaterial-based systems for AAA management. We classify and discuss key nanocarriers—inorganic nanomaterials, organic nanomaterials, and hybrid nanomaterials—highlighting their unique design strategies, targeting mechanisms, and therapeutic functions. Specific applications include the targeted delivery of anti-inflammatory agents, modulation of matrix metalloproteinase activity via siRNA delivery, protection of vascular smooth muscle cells (VSMCs) from oxidative stress and apoptosis, and strategies for in situ vascular repair and regeneration. Furthermore, the role of nanomaterials in enhanced diagnostics and intelligent sensing for rupture risk prediction is explored. Despite encouraging preclinical results, challenges regarding long-term biosafety, translatability from rodent models to human pathophysiology, and optimisation of hemodynamic delivery remain significant hurdles. Future directions involve closed-loop theranostic systems, artificial intelligence integration, hemodynamic-optimised nanoparticle design, and exploring gene-editing nanocarriers. This review concludes that engineered nanomaterials hold substantial potential to transform AAA management from passive monitoring to active prevention and precision therapy, paving the way for future clinical translation.

Keywords
Introduction
Clinical challenges of abdominal aortic aneurysm (AAA)
AAA is defined as a permanent dilatation of the abdominal aorta characterised by an aortic diameter ⩾1.5 times the diameter of the aorta at the level of the renal arteries (minimum diameter of approximately 30 mm).1–5 The disease is characterised by insidious progression6–8 and high morbidity and mortality.9–11 AAA causes ∼170,000 deaths annually worldwide. 12 Abdominal aneurysms are usually asymptomatic until catastrophic rupture occurs. 13 If rupture of an AAA occurs outside the hospital, the mortality rate approaches 90%. 14 The pathological process involves multidimensional molecular mechanisms: chronic inflammatory response drives monocyte/macrophage infiltration, which activates the family of matrix metalloproteinases (MMPs) through the secretion of pro-inflammatory factors such as IL-6 and TNF-α, leading to excessive degradation of elastin fibres and collagen; overproduction of reactive oxygen species (ROS) in the setting of oxidative stress induces vascular smooth muscle cells (VSMCs) via the MAPK/NF-κB pathway (VSMC) apoptosis and phenotypic transformation; abnormal expression of calcification-related proteins (e.g. BMP-2, OPN) triggers calcium salt deposition in the vascular wall, accelerating the decline of aortic wall mechanical properties.15–21
Typical treatments for aortic aneurysms are open surgery and aortic endovascular repair for high-risk patients with a diameter >5.5 cm,22–26 but there are obvious limitations. For example, open surgery is highly invasive and postoperative recovery is slow; endoluminal repair, although minimally invasive, requires strict anatomical conditions and carries a higher risk of complications such as postoperative endoleak and stent migration.27–32 For patients with small AAA diameters, surgery may instead aggravate the medical and psychological burden of patients.33,34 More critically, there is no effective pharmacological intervention for small unruptured aneurysms with a diameter of 3–5 cm, and surgery can only be delayed by regular monitoring.35–37 This ‘passive waiting’ strategy not only aggravates the psychological and economic burdens of patients, but also fails to stop the uncontrollable progression of the aneurysm. In patients with asymptomatic aneurysms, current treatment is limited to regular monitoring with imaging, control of cardiovascular risk factors, and use of statins and antiplatelet therapy. There is no effective pharmacological treatment that can limit the progression of AAA or avoid rupture.38,39 Currently, aortic diameter is the only marker of rupture risk, and when its value is greater than 5.5 cm, the need for surgical repair is decided.9,40–42 Therefore, the development of early screening and new treatments is crucial. This review discusses the applications and advances of nanomaterials in the treatment of abdominal aortic aneurysms. It was conducted by analysing literature from 2018 to 2024 using subject terms such as ‘Nanomaterials in Abdominal Aortic Aneurysm’, ‘Targeted Therapy’, ‘Smart Vascular Repair’, ‘Drug Delivery’, ‘Disease Modelling’, ‘Drug Delivery’, ‘Disease Modelling’, and ‘Clinical Applications’.
The rise of nanomedicine
Nano-sized particles or nanoparticles have emerged as promising tools for a wide range of applications in drug delivery, diagnostics, agriculture, cosmetics, and several other biotic and abiotic fields.43,44 In the field of medicine, nanotherapeutic systems mainly refer to 1–100 nm-scale carrier systems consisting of liposomes, polymeric micelles, dendrimers, etc., which are loaded with drug molecules by physical encapsulation or chemical coupling to form a composite system with spatial targeting capabilities.45–47 The potential of nanoparticles in tumour therapy has been widely recognised due to their ability to integrate multifaceted functions into a single nanosystem.48–50 Nanoscience offers the possibility of more targeted imaging and drug delivery systems for the screening and treatment of abdominal aortic disease, which can reduce systemic toxicity, by modifying with ligands or biofilms in order to deliver therapeutic agents targeted to the lesion site.51–56 Compared to conventional contrast agents, nanoparticles offer extended blood circulation time, controlled biological clearance pathways and specific molecular targeting capabilities.57–60 Due to their modifiability and biocompatibility, NPs have been reported as AAA imaging agents in several studies.33,61–63 Currently, the development of nanomedicine therapeutics is still immature, and only limited efficacy has been observed in the clinic when administered systemically, which may be attributed to the rapid clearance and poor targeting of drugs.64–66
The multidimensional modulation ability of nanomaterials provides a new paradigm for precise intervention in AAA, and its clinical translation potential is expected to fill the gap of current drug therapies and promote the transformation of vascular surgery from ‘passive repair’ to ‘active prevention and treatment’.
Classification of nanocarriers for targeting AAA
Nanotherapeutic carrier systems for AAA have been developed into three major technology branches—inorganic nanomaterials, organic nanomaterials and hybrid nanomaterials—each of which has demonstrated different therapeutic advantages through unique design strategies.
Inorganic nanomaterials: Structural stability and multimodal diagnosis and treatment
Among the inorganic nanomaterials, interleukin-1 receptor antagonist (IL-1Ra) was loaded into Mn-doped Mesoporous Silica Nanoparticles (MMSN) to form a complex (IL-1Ra@MMSN) for targeting macrophage-mediated chronic aortic inflammation in to treat AAA. In vivo experiments were conducted to observe the distribution of IL-1Ra@MMSN (5-FAM labelled) in the AAA tissues of mice. The distribution of IL-1Ra@MMSN (5-FAM labelled) was observed by fluorescently labelling the IL-1Ra@MMSN (5-FAM labelled). The results demonstrated that IL-1Ra@MMSN exhibited augmented fluorescent signals in AAA lesion tissues, while no significant signals were observed in normal abdominal aortic tissues. This finding indicates that IL-1Ra@MMSN exhibits effective in vivo targeting capabilities and accumulates specifically at AAA lesion sites. Furthermore, IL-1Ra@MMSN led to a substantial reduction in the prevalence of AAA (from 83% to 17%) and a concomitant attenuation of the severity of AAA in the Angiotensin II-induced AAA model in mice. 67 In parallel, matrix-bound gold nanoparticles (GNPs) undergo enzyme-responsive degradation in the AAA microenvironment by surface modification of MMP-2/9 substrate peptides. Its high X-ray attenuation coefficient (5.2 × 10³ HU-cm³/g) enhanced CT imaging resolution by 40%, and an imaging histology-based rupture risk prediction model could be constructed by dynamically monitoring the retention time of GNPs in the vessel wall (AUC = 0.87). The findings of the present study demonstrated a positive correlation between the accumulation of EL-GNPs and the extent of elastin degradation. The EL-GNP signals exhibited greater intensity in regions characterised by severe elastin degradation, suggesting their capacity to target AAA lesions with precision. In addition, the investigators discovered that the signal intensity of EL-GNPs exhibited a negative correlation with aortic burst pressure, as determined by means of micro-computed tomography (micro-CT) and aortic burst failure testing. This finding indicates that the targeted signal of EL-GNPs could serve as a potential indicator for assessing the risk of AAA rupture. 68 In addition, using a metal-organic framework (MOF), RAP@PFB integrated pentaethylene dicarboxyglucose (PGG) and rapamycin (RAP) into a MOF structure, achieving significant drug loading capacity and colloidal stability. PGG (polyphenolic antioxidant) synergistically interacts with RAP (mTOR inhibitor) in the pathogenesis of AAA, which co-regulates key cell types, including macrophages and smooth muscle cells.
In an in vitro validation of the targeting ability, by performing experiments in RAW264.7 macrophages, the investigators found that RAP@PFB was able to significantly scavenge intracellular reactive oxygen species (ROS) and inhibit the secretion of inflammatory factors, as well as promote the polarisation of macrophages from M1-type (pro-inflammatory) to M2-type (anti-inflammatory). The results of the present study suggest that RAP@PFB is able to modulate the inflammatory response, resulting in a targeted therapeutic effect on aneurysmal aortic dilatation lesions. RAP@PFB exhibited concentration-dependent scavenging of multiple free radicals and was superior to PGG or RAP alone, further confirming its ability to target free radicals from AAA lesions.
In addition, the in vivo targeting ability was further validated in the calcium chloride (CaCl2)-induced AAA rat model by tail vein injection of RAP@PFB that had been labelled with Cy5.5 fluorescence. The investigators observed significant accumulation of fluorescent signals at the AAA lesion site, whereas there was virtually no fluorescent signal at the normal aortic site. This finding suggests that RAP@PFB has the capacity to target specifically the sites of AAA lesions. Through the implementation of dynamic monitoring of fluorescence intensity, the investigators ascertained that the accumulation and retention time of RAP@PFB at the AAA lesion site exceeded 24 h, thereby substantiating its targeting capability. 69
Organic nanomaterials: Bio-adaptation and dynamic regulation
Among the organic nanomaterials, a polyethylene glycolated lipid nanoparticle (PEGylated LNP) has been applied to deliver JNK siRNA. in cytokine-activated rat aneurysmal smooth muscle cell (EaRASMC) culture, JNK siRNA-loaded PEGylated LNP successfully inhibited JNK2, which in turn reduced MMP2 expression, while upregulating the gene expression of key proteins such as elastin and lysyl oxidase (LOX), which enhanced elastin homeostasis and helped to prevent the progression of small AAA. In cellular experiments, JNK2 gene expression was significantly silenced and reduced by 70% after 5 days of LNPs treatment. This demonstrated that LNPs were able to effectively deliver siRNAs into target cells and achieve gene silencing. JNK2 protein expression was reduced by 35%, and MMP2 protein expression was reduced by 35% after 5 days of LNPs treatment. This finding further corroborates the targeted therapeutic effect of LNPs in inhibiting the activity of elastin-degrading enzymes. 70 Polymeric micelles of statins have also been used for the treatment of AAA in a rat model. The micelles were shown to inhibit aortic aneurysm expansion in a dose-dependent manner, demonstrating a significant therapeutic effect. In addition, in the AAA model, the group receiving micellar injections exhibited reduced macrophage infiltration, as well as reduced matrix metalloproteinase-9 (MMP-9) activity. The therapeutic effect of AAA model demonstrated that in the elastase-induced AAA rat model, the diameter of AAA treated with drug-loaded micelles (PS/m) was significantly smaller than that of the control group treated with PBS. The following data is indicative of the PBS-treated group: The mean ± standard deviation of the measurement was 6.41 ± 0.43 mm, and the PS/m2 mg/kg treated group was observed to show a significant increase. The mean ± standard deviation of the measured parameters was 4.78 ± 0.43 mm, with a PS/m of 5 mg/kg in the treated group. The mean ± standard deviation of the PS/m measurement was 10 mg/kg in the treated group: The mean measurement was found to be 3.63 mm ± 0.66 mm. 71 The application of biomimetic nanocarriers has provided new ideas for the treatment of vascular diseases, and a novel biopremedicine nanoplatform (BUD-l-Arg@PSA) co-loaded with budesonide (BUD) and levarginic acid (l-Arg) via polysialic acid (PSA) can target activated endothelial cells and modulate vascular endothelial function and inflammatory responses, resulting in normalisation of the vasculature. The nanocomplexes referenced in the article have been shown to enhance vascular endothelial function and suppress inflammatory responses by modulating the endothelial-type nitric oxide synthase (eNOS)/NO signalling pathway and impeding the NF-κB signalling pathway. These mechanisms are also critical for the treatment of AAA, which are characterised by endothelial dysfunction and chronic inflammation (Figure 1). 72

The schematic diagram of the structure and function of the BUD-l-Arg@PSA biopharmaceutical nanoplatform.
Hybridised nanomaterials: Functionally integrated and intelligently driven
As for hybrid nanomaterials, the novel GOx-driven Janus nanomotor significantly enhanced biocompatibility and targeting of thrombi by asymmetrically immobilising glucose oxidase (GOx) on the surface of platelet membrane-encapsulated polymeric nanomotor in conjunction with urokinase fibrinogen activators (uPAs) to take advantage of the camouflaging properties of platelet membranes (Figure 2). 73 Nano-enzymatic complexes exhibit high sensitivity to hepatitis B virus core protein-modified nanomedicine EVMS@R-HNC containing RGD sequences for targeting AAA lesions. The RGD sequences specifically bind to the integrin αvβ3, which is highly expressed on macrophages and smooth muscle cells in AAA, resulting in dual targeting. In the Angiotensin II-induced AAA mouse model, EVMS@R-HNC demonstrated a significant reduction in aortic dilatation, a decrease in the incidence of AAA, and an amelioration of pathological changes in the aortic wall. 74

The diagram illustrates the mechanism of action of the GOx-driven Janus nanomotor.

Nanomaterials in AAA treatment
Critically, nanocarriers are engineered to precisely intercept pivotal pathological signalling cascades driving AAA progression, with prominent examples including the inhibition of the nuclear factor-kappa B (NF-κB) pathway to quench chronic inflammation and cytokine storms (e.g. TNF-α, IL-6) via systems like RAP@PFB and BUD-L-Arg@PSA which block IκB kinase (IKK) and prevent NF-κB nuclear translocation; the targeted suppression of MMP activity, particularly MMP-2 and MMP-9, through siRNA delivery platforms (e.g. TPNs-siRNAs) to halt excessive extracellular matrix (ECM) degradation and preserve vascular structural integrity; and the mitigation of oxidative stress-induced VSMC apoptosis and phenotypic switching, often linked to the MAPK/NF-κB pathways, utilizing antioxidant nanotherapeutics (e.g. HA-SOD2 mRNA NPs, RAP@PFB) that scavenge ROS and bolster cellular defenses.
The treatment of AAA is challenging due to the complexity of the pathology (e.g. inflammation, matrix degradation, oxidative stress). Nanomaterials have emerged as an emerging strategy to break through the bottleneck of conventional treatment due to their targeted, controlled release and multifunctional synergistic effects. The core applications of nanomaterials in AAA therapeutics are systematically described below in the context of breakthrough studies in the last 2 years.
Targeted delivery of anti-inflammatory drugs
Activation of inflammatory factors (e.g. TNF-α, IL-6) and NF-κB signalling pathway is the core mechanism driving vascular wall damage in the pathological process of AAA, 75 so ameliorating vascular damage with targeted anti-inflammatory drugs is a promising research direction. Wang’s team developed a self-assembled core-shell nanodrug delivery system (RAP@PFB). This MOF-based nanodrug delivery system targeted AAA foci by loading rapamycin, which significantly inhibited macrophage pro-inflammatory polarisation (M1-type), while inducing anti-inflammatory (M2-type) macrophage repolarisation, and reduced the expression of inflammatory factors such as IL-6 and TNF-α. This study used a CaCl2-induced AAA rat model to evaluate the in vivo targeting of RAP@PFB. The system had a rapamycin load of up to 53.8% and was released responsively in the lesion microenvironment, achieving a dual effect of anti-inflammation and tissue protection. The researchers measured the drug release behaviour of RAP@PFB in the presence of hydrogen peroxide (H2O2). The results demonstrated that drug release was further accelerated under H2O2 conditions, with a cumulative release of approximately 58.6% after 24 hours (h). Thus, RAP@PFB exhibited ROS-responsive drug release characteristics. To determine the ROS scavenging efficacy of RAP@PFB, the researchers initially employed the 2,2-diphenyl-1-picrylhydrazyl (DPPH) assay. The results revealed that RAP@PFB supplemented with 50 μM propyl gallate (PGG) achieved 80% scavenging of 0.5 mM DPPH within 5 min. Furthermore, the scavenging capacity of RAP@PFB against various reactive oxygen and nitrogen species (RONS), including H2O2, hydroxyl radical (•OH), superoxide anion radical (•O2⁻), and nitric oxide radical (•NO), was evaluated. The results indicated concentration-dependent scavenging of all tested radicals. In summary, RAP@PFB demonstrated significant ROS scavenging capability, highlighting its potential utility as an effective therapeutic agent against AAA by alleviating oxidative stress-induced vascular damage (Figure 3). 69 Vascular diseases characterised by vascular endothelial dysfunction combined with inflammation are associated with many fatal diseases, Wang et al. developed a polysialic acid nanocomplex (BUD-L-Arg@PSA) to promote vascular normalisation by simultaneously modulating vascular endothelial dysfunction and inflammation. BUD is a classical glucocorticoid anti-inflammatory drug that inhibits IKK mediated NF-κB nuclear translocation, blocking downstream pro-inflammatory cytokine release (TNF-α, IL-6). L-Arg is a substrate for eNOS, which promotes the production of NO. NO improves the function of vascular endothelium and inhibits the proliferation and migration of smooth muscle cells. BUD-L-Arg@PSA is a dual prodrug minimal nanoplatform built by co-loading BUD and l-Arg through polysialic acid (PSA). It can inhibit inflammation and improve vascular function by targeting inflammatory endothelial cells and dually regulating NF-κB and NO/eNOS pathways. In a mouse model of atherosclerosis, BUD-l-Arg@PSA reduced atherosclerotic plaque load and improved vasodilation. This represents a revolutionary therapeutic strategy for inflammatory vascular diseases. 72

The diagram illustrates the drug delivery strategy of the RAP@PFB system.
Based on the anti-inflammatory peptides designed for AAA pathogenesis in nanomaterials modified by targeting ligands, manganese-doped mesoporous silica nanoparticles (MMSN) were designed to target macrophage-mediated chronic aortic inflammation with surface-loaded interleukin-1 receptor antagonist (IL-1Ra) to achieve precision therapy for AAA. 67 Additionally, by targeting key enzymes that degrade elastin, pentaacetylglucose-loaded nanoparticles therapy was able to reverse the progression of AAA while suppressing the expression of inflammatory and immune-related markers. 76
A pH-responsive and fluorescence-guided nanocarrier system (HPC-1@DOX) has been developed for targeted drug delivery based on environmentally responsive design. The HPC-1@DOX fluorescence-guided system is a pH-responsive carrier based on an amphiphilic cationic polymer (HPC-1), which can stably encapsulate doxorubicin (DOX) under normal physiological environment, and the structural deassembly was triggered by amination in the AAA inflammatory subregion (pH 6.5–6.8), with a drug release rate of 88% within 72 h; its innovation lies in the integration of near-infrared fluorescent moiety (Cy7.5) to achieve real-time monitoring of drug distribution in the tumour (with a signal-to-noise ratio of up to 14:1), and optimisation of the pore size of the carrier through molecular dynamics simulation (2.6 nm) to ensure the high loading capacity of DOX (2.6 nm). High loading (16.2 wt%) with controlled release kinetics (burst release rate <8%) of DOX, this system is capable of improving drug release efficiency significantly in acidic environment, a property that makes it promising for AAA therapy, targeting the localised acidic inflammatory region of the aortic aneurysm. 77 Nanoparticle drugs designed for targeted therapy based on reactive oxygen species (ROS) responsive design achieved precise drug release in the inflammatory microenvironment by loading rapamycin and combining with integrin-targeting peptides and macrophage membrane artefacts, effectively inhibiting the expansion of AAA (Figure 4). 78
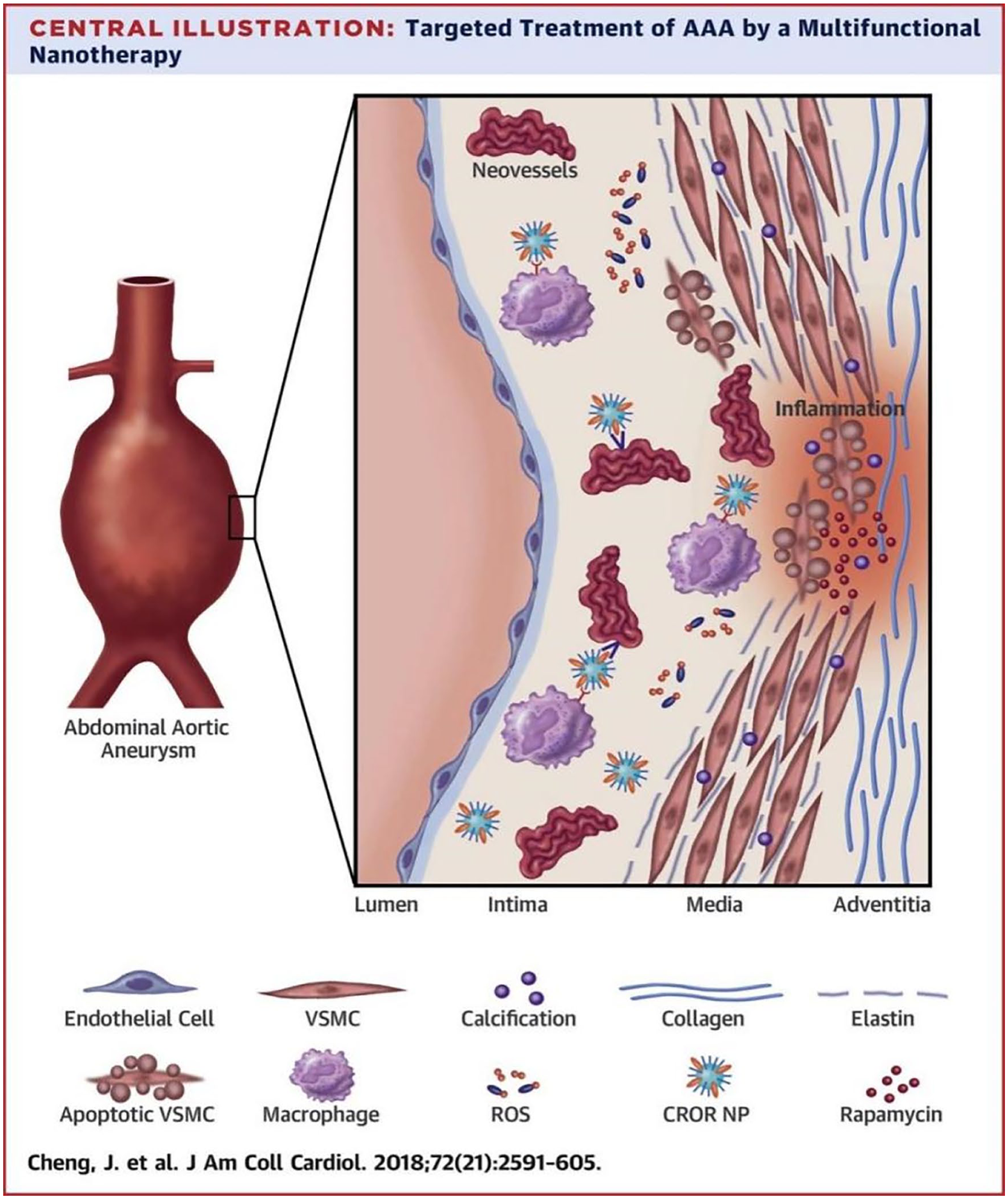
Targeted therapeutic strategy of nanoparticle drugs based on reactive oxygen species.
Nanoparticle-targeted regulation of MMPs activity
Overexpression of MMPs also affects the AAA case process, leading to degradation of the ECM, which disrupts the structural integrity of the vessel wall and thus promotes aneurysm formation and dilatation. Therefore the activity or expression of MMPs can be inhibited to slow down the development of AAA. MMP genes can be silenced by targeted delivery of siRNAs. Therefore, influencing the expression of the MMP gene is a very promising approach. Wang developed TPNs-siRNA system to simultaneously deliver two siRNAs targeting MMP-2 and MMP-9. Tea polyphenol nanoparticles (TPNs), as a polybioactive substance, attenuates oxidative stress and inflammation, and promotes M1–M2 repolarisation in macrophages. TPNs-siRNAs protect siRNAs from biodegradation, promote intracellular siRNA transfection, facilitate lysosomal escape, and release siRNAs to silence MMP-2 and MMP-9. The cytotoxicity of TPNs-siRNA was evaluated using RAW264.7 macrophages and VSMCs. Cell viability remained above 90% across the concentration range of 10–100 μg/mL. Thus, TPNs-siRNA demonstrated biocompatibility. The results showed that TPNs-siRNA treatment significantly reduced the levels of each RONS in the cells, highlighting its ability to effectively scavenge free radicals and alleviate oxidative stress in LPS-stimulated RAW264.7 cells. An AAA model was established by periadventitial application of CaCl2 solution to the abdominal aorta. This model recapitulated several key pathological features observed in human AAA, including significant aortic calcification, inflammation, oxidative stress responses, MMP activity, elastin degradation, and VSMC apoptosis. Compared to the limitations of conventional siRNA delivery systems that are susceptible to nuclease degradation, this nanocarrier prolongs the intracellular action of siRNAs through antioxidant and anti-inflammatory synergistic effects, and animal experiments showed that TPNs-siRNAs efficiently accumulated and prolonged their retention time in aneurysm tissues of AAA mice, and specifically targeted macrophages and VSMCs, which are critical to the pathogenesis of AAA (Figure 5). 79 An injectable supramolecular nanofibre system based on peptide amphiphilic molecules (PA) has been applied to target AAA for diagnosis and treatment. The PA molecules were constructed using solid-phase peptide synthesis by integrating four targeting peptide sequences (targeting fracture elastin, MMP-2, and MT1-MMP) designed from X-ray crystallography data, and the optimised formulations that could be self-assembled into nanofibres were screened by modulating the molar ratio of the targeting PA to the diluted PA (25%–95%). The results showed that PAs were targeted to all three AAA biomarkers (MMP-2, MT1-MMP, and fragmented elastin) at the aneurysm site. Among these nanofibers, the PAs targeting MMP-2 exhibited the strongest targeting ability. Compared to conventional approaches, PA materials exhibit significant advantages: they possess exceptional chemical stability, streamlined manufacturing processes, and eliminate the need for complex nanocarrier labelling with antibodies. Notably, the supramolecular self-assembly-derived PA nanofiber architecture provides high-density, orderly arranged targeting sites. This unique geometric configuration substantially enhances the loading efficiency and binding capacity for targeting molecules (e.g. matrix metalloproteinase-2, MMP-2), outperforming traditional nanocarriers. Experimental evidence confirms the superior in vivo targeting capability of this nanofiber structure toward type IV collagen over spherical counterparts. Furthermore, this study successfully overcomes inherent limitations of antibody-based elastin-targeting strategies—including the bulky size, structural complexity, and susceptibility to denaturation and deactivation of antibody molecules—providing an optimised solution for targeted therapies (Figure 6). 80

Schematic diagram of TPNs-siRNAs materials.

Schematic diagram of X-ray crystallography for treatment with peptide amphiphilic molecules.
Furthermore, it has also been shown that rod-shaped nanoparticles (high aspect ratio) are more likely to penetrate the diseased endothelial layer than spherical particles and enter the middle layer of the vessel wall through an extravasation mechanism, resulting in the efficient delivery of MMP inhibitors or gene editing tools to target cells. 81
Nanoparticles protect smooth muscle cells
In the treatment of AAA, anti-oxidative stress is also one of the key strategies to delay disease progression and protect vascular structural integrity. Oxidative stress accelerates AAA deterioration by promoting VSMCs apoptosis. Yan et al. developed a p5RHH amphiphilic peptide-based nano-delivery system to form stable nanoparticles of approximately 55 nm (HA-SOD2 mRNA NPs) by binding SOD2 mRNA to functionalised hyaluronic acid (HA). The delivery system significantly enhanced the uptake efficiency of therapeutic mRNA by macrophages through a CD44 receptor targeting mechanism and demonstrated excellent aortic targeting in in vivo experiments, achieving sustained SOD2 protein expression in the aortic wall for 4 weeks after only three intravenous injections. The nanoplatform offers several advantages over conventional antioxidant therapies. The unique HA encapsulation design of the nanoparticles conferred long-term stability (4 weeks of storage at 4°C with full functionality); the mitochondrial targeting sequence precisely enhanced the subcellular antioxidant efficacy of SOD2, which reduced iNOS and nitrotyrosine levels by 68% and 72%, respectively; additionally, the dual-disease model validation showed that it not only inhibited elastase-induced AAA dilatation (28% reduction of aortic diameter ), but also significantly increased the aneurysm rupture survival rate from 45% to 87.5% in the TGFβ blockade model, providing a strong experimental basis for clinical translation. 82 In addition, the RAP@PFB system also plays a role by piggybacking both rapamycin and galloylglucose to reduce ROS-induced inflammatory factor release by blocking the NF-κB pathway and protect VSMCs from oxidative stress-mediated apoptosis and calcification. It not only scavenges a variety of reactive oxygen radicals (e.g. –OH, H2O2), but also inhibits SMC calcification and apoptosis, and reduces pathological remodelling of the vessel wall. 69
In situ vascular repair and regeneration
The application of nanofibrous scaffolds in vascular remodelling shows great potential, especially in promoting vascular endothelialisation, improving vascular function and tissue regeneration. Electrostatic spinning technology enables the preparation of scaffolds with high specific surface area and nanoscale fibre structures that mimic the topology of the natural ECM. 83 For example, nanofibrous scaffolds of polycaprolactone and gelatin composites can provide a good environment for cell attachment and growth, and promote VSMC migration and proliferation. It was shown that nanofibre scaffolds loaded with vascular endothelial growth factor were able to recruit endothelial progenitor cells and accelerate the regeneration of the vascular endothelial layer. This scaffold promotes vascular endothelial layer repair by mimicking the structure and function of the ECM and providing support for EPC attachment and differentiation. This scaffold shows good biocompatibility and tissue repair ability in animal models, and has important potential for application in tissue engineering and regenerative medicine (Figures 7 and 8). 83

Schematic diagram of nanofiber scaffold for vascular remodelling.
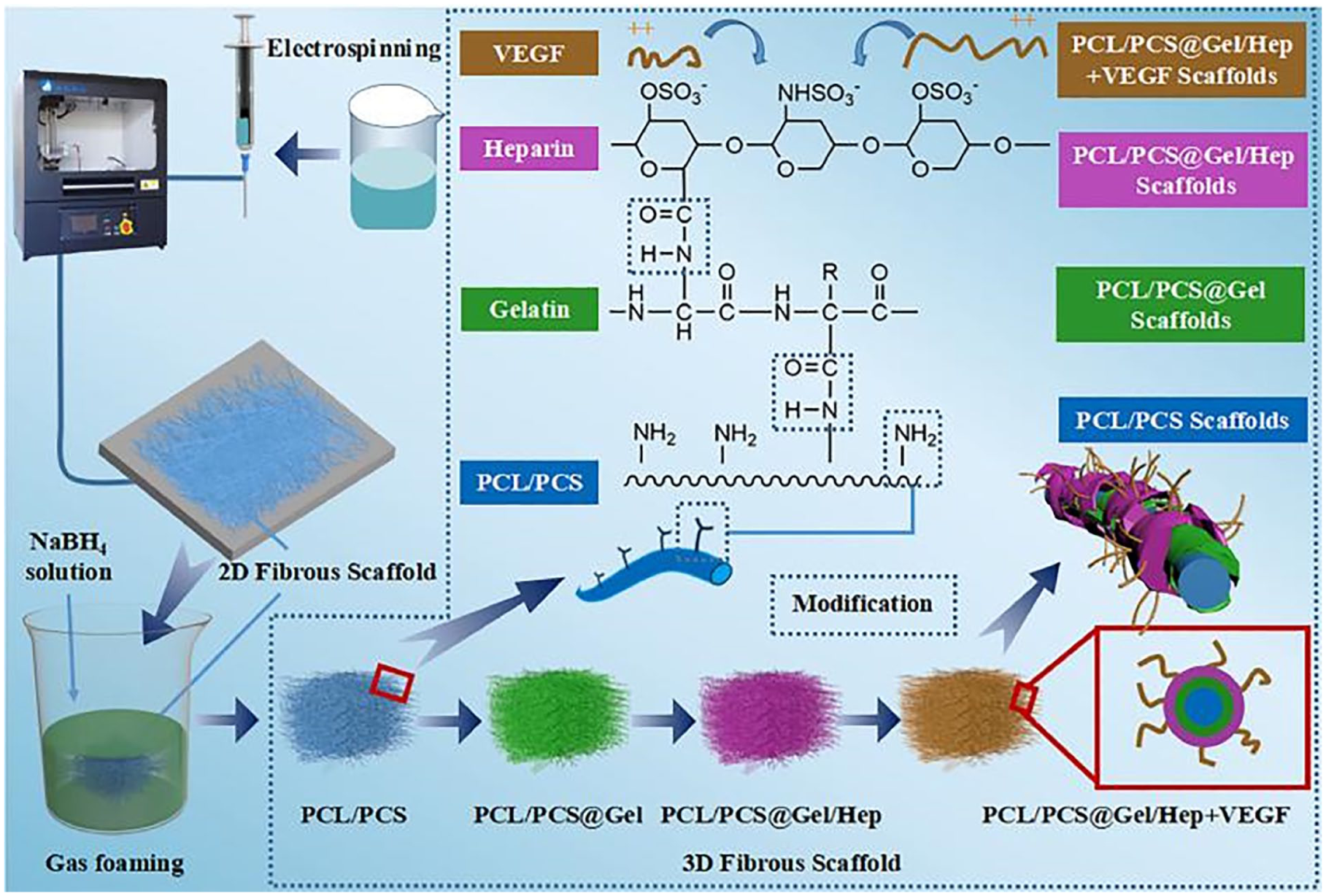
The diagram illustrates the preparation process of nanofiber scaffolds.
Wang et al. developed a magnetically responsive chemotactic nanomotor delivery system for dual-targeted therapy of AAA by coupling mesenchymal stem cell exosomes with superparamagnetic iron oxide nanoparticles (SPION)-embedded mesoporous silica nanocarriers loaded with catalase (CAT). The system uses an external magnetic field to guide the nanomotor to penetrate the high blood flow resistance and enrich in the aneurysm region, and activates the CAT catalytic reaction through the local high H2O2 microenvironment in the AAA lesion to drive the nanomotor towards the vascular wall in a convergent motion, which enhances the exosome delivery efficiency by 3.5-fold compared with the conventional method. This delivery strategy not only significantly promotes elastic matrix regeneration (1.6-fold increase in elastin deposition and improved cross-link maturation), but also synchronously inhibits matrix metalloproteinase activity (50% down-regulation of MMP2) and scavenges excess reactive oxygen species (60% reduction in ROS), while visualising the treatment with the magnetic resonance imaging capability of SPION, providing a multimodal solution for precise AAA repair (Figure 9). 84
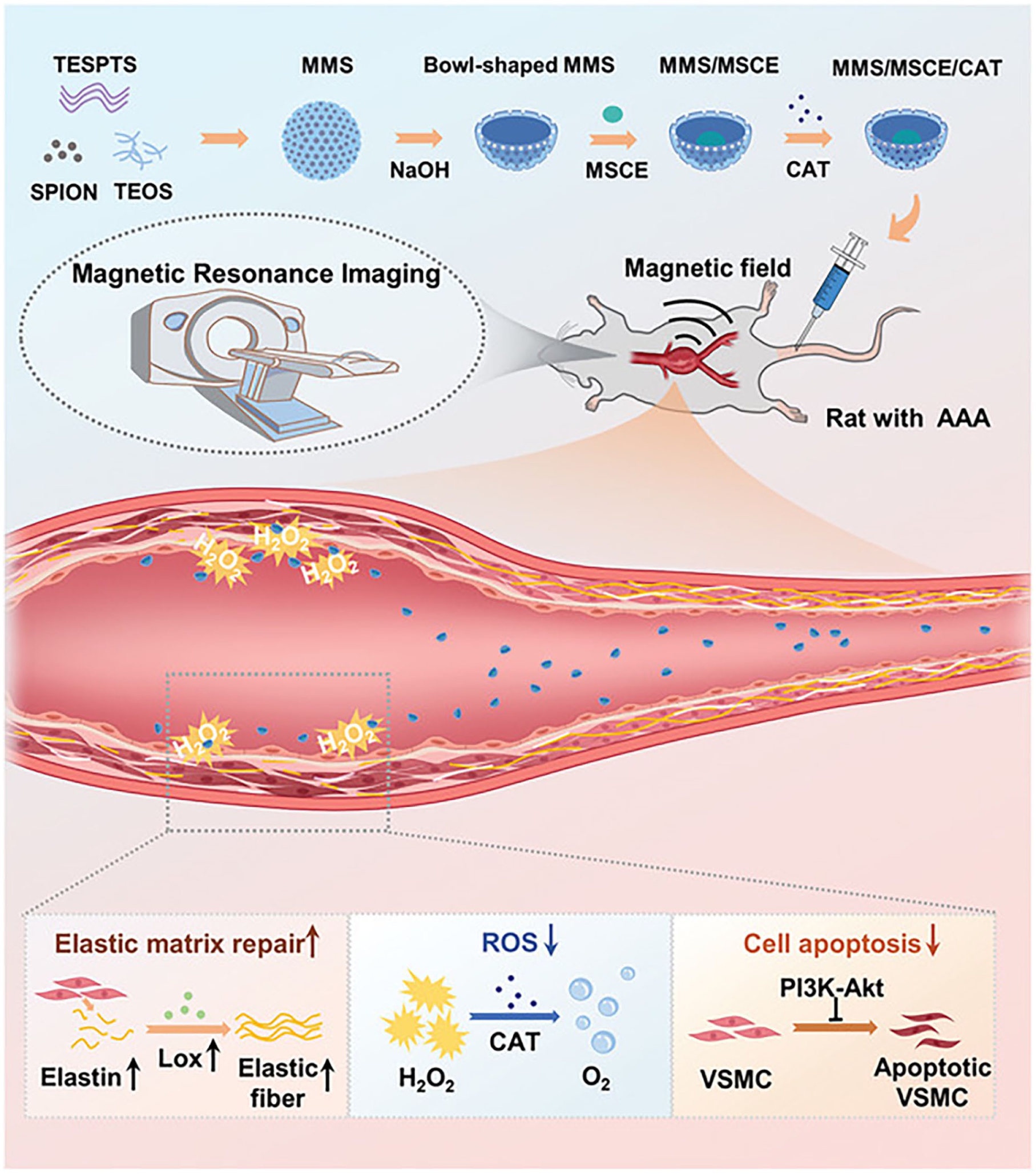
The diagram illustrates the mechanism of action of the magnetically responsive chemotactic nanomotor delivery system.
Although rodent models have significantly advanced our understanding of AAA pathogenesis and nanomaterial-based therapies, they possess inherent limitations that hinder clinical translation. These models often fail to fully replicate the complexity of human AAA, including the presence of intraluminal thrombus, calcification, and chronic inflammation, which are critical features of human disease.28,85 Additionally, differences in hemodynamics, vessel size, and immune responses between rodents and humans can affect the biodistribution and efficacy of nanomaterials. To overcome these limitations, large animal models such as porcine AAA models are increasingly recommended due to their anatomical and physiological similarities to humans, including comparable aortic size and hemodynamic profiles.86,87 Vascular organoids and ex vivo human tissue models also offer promising alternatives, as they allow for the study of human-specific cellular interactions, ECM remodelling, and immune responses in a controlled environment. 88 These models can better recapitulate the human AAA microenvironment and provide a more accurate platform for evaluating the safety and efficacy of nanomedicines before clinical application.
Nanomaterials in AAA diagnostics and monitoring
Molecular imaging and microenvironment sensing
The detection of early structural changes in the vasculature, such as microcalcifications, is crucial for early diagnosis and prediction of diseases. Advanced molecular imaging techniques and probes play a vital role in this process. For instance, the two-photon ratiometric fluorescent probe BH-Vis has been designed to monitor dynamic changes in the cell plasma membrane (CPM) and peroxynitrite (ONOO⁻) in real time. This probe offers enhanced sensitivity and specificity, with good biocompatibility and specific targeting ability to the CPM. 89 In the context of inflammation imaging, Ly6G and Cy7 are two critical components of a multimodal imaging agent designed to target neutrophils, which are key players in the inflammatory response. Ly6G (Ly6G antibody) is a specific marker for neutrophils, enabling precise targeting of these immune cells that infiltrate sites of inflammation, such as in AAA. By coupling Fe₃O₄ nanoparticles with Ly6G antibodies and Cy7, researchers have developed a highly sensitive multimodal imaging agent (Ly6G NPs) for magnetic particle imaging (MPI) and fluorescence imaging (FLI). This agent leverages the exceptional sensitivity of MPI to SPION and the fluorescence capabilities of Cy7 to achieve high-resolution detection of neutrophil infiltration and monitoring of AAA severity in a mouse model. Ly6G NPs not only enhance the sensitivity and specificity of neutrophil detection but also provide a comprehensive understanding of the inflammatory microenvironment. This multimodal approach allows for the simultaneous assessment of neutrophil activity and the overall inflammatory state, offering a powerful tool for both research and clinical applications. 90
Intelligent sensing and dynamic early warning
The limitations of current AAA monitoring methods, which often rely on periodic imaging or catheterisation, highlight the urgent need for continuous, non-invasive monitoring solutions. The arterial stiffness sensor, a fully implantable, battery-free, soft platform developed using printed nanomaterials, represents a significant advancement in this field. This wireless sensor utilises flexible piezoelectric nanoarrays comprising lead zirconate titanate nanowires with a diameter of 80 nm, which are capable of detecting strain changes as small as 0.1% in the blood vessel wall. Operating on a self-supplied energy design, it harnesses up to 1.2 mW/cm² of energy density through the conversion of mechanical energy from blood vessel pulsation, thus eliminating the need for batteries. Coupled with radio-frequency identification technology, the sensor can transmit vessel stiffness data in real time at a sampling rate of 100 Hz. This innovation provides 48 hours’ advance warning of mechanical instability, as evidenced by a 37% decrease in elastic modulus in a 3.5 cm diameter aneurysm within a porcine AAA model. This early warning system provides a critical window for clinical decision-making, with the potential to transform vascular care from reactive to proactive and predictive. By overcoming the limitations of current AAA monitoring methods, this technology enhances patient outcomes and sets a new standard for vascular health surveillance. 91
Challenges and prospects
Translational medicine bottlenecks
Although nanomaterials have demonstrated targeting and efficiency in the treatment of AAA, their long-term biosafety still needs to be systematically evaluated. Nanoparticles may trigger toxic reactions due to long-term accumulation in organs such as liver and kidney, and some carriers (e.g. metal ion-liganded polyphenol oxidative autopolymerisation nanoparticles) may cause mitochondrial dysfunction due to metal ion release. 92 It has been found that folic acid-modified liposomes adsorb a large amount of natural IgM in the blood, which in turn activates the complement system, leading to the rapid capture and accumulation of liposomes in organs such as the liver and spleen. 93 When designing nanoparticles, the mechanism of their interaction with blood components and the immunogenicity of the particles should be considered.
In addition, most of the current studies rely on rodent models, but there are differences in the pathological mechanisms between them and human AAA. The inflammatory signalling pathway in human AAA is more complex, involving the interaction of multiple cell types (e.g. macrophages, neutrophils, T cells, etc.) and multiple inflammatory factors (e.g. TNF-α, IL-6, MCP-1, etc.). In contrast, rodent models (e.g. ApoE−/− mice), although capable of mimicking the infiltration of inflammatory cells, have less complex inflammatory signalling pathways.94,95 The complex inflammatory microenvironment may enable more efficient targeting of nanomaterials, but it also increases the complexity of nanomaterial distribution in vivo. Moreover, the haemodynamic properties of the human aorta are difficult to be fully simulated in mouse models, where blood flow velocity and wall shear are significantly lower than in humans, and the complexity of vortex flow at the arch of the aorta is much lower in mice than in humans, which may affect the target accumulation efficiency of nanomedicines. 96 In addition, the targeting efficiency of nanomaterials in humans may be reduced by differences in metabolic pathways, and more efficient delivery strategies need to be developed. Organoid models or large animal models (e.g. pigs) that more closely resemble human pathological characteristics could also be used to validate the clinical applicability of nanomedicines.
Moreover, the long-term biosafety profile of nanocarriers demands rigorous evaluation beyond acute toxicity, particularly concerning chronic immune activation and bioaccumulation risks. Key concerns include: (1) Complement activation triggered by surface opsonisation—for example, folic acid-modified liposomes adsorbing natural IgM, activating the classical complement pathway and accelerating hepatic clearance, which may cause hypersensitivity reactions and dose limitations in clinical trials; 93 (2) Persistent inflammatory responses from non-degradable components (e.g. metal ions leaching from MOFs or silica nanoparticles inducing mitochondrial dysfunction and NLRP3 inflammasome activation, paradoxically exacerbating vascular inflammation); 92 and (3) Cumulative organ burden due to impaired renal/hepatic clearance of polymeric carriers, leading to chronic macrophage activation and potential off-target toxicity. These immunogenic reactions compromise targeting efficacy by promoting premature nanoparticle sequestration and pose critical barriers to human trials. Mitigation strategies such as PEGylation (extending circulation half-life), biomimetic membrane cloaking (e.g. platelet membrane camouflage in Section ‘Hybridised nanomaterials: functionally integrated and intelligently driven’), and enzyme-responsive biodegradable linkers should be prioritised to minimise immune recognition and enable chronic dosing regimens essential for AAA management.97,98
Innovative approaches to enhance AAA regulation
Drug-eluting stent implantation inhibits excessive smooth muscle cell proliferation, thereby reducing in-stent restenosis. However, the efficacy of drug-eluting stents remains limited due to delayed reendothelialisation, impaired endothelial remodelling and the possible increased risk of late restenosis. A drug-free coating formulation modified with custom recombinant humanised type III collagen was developed to produce a pleiotropic effect on damaged tissue after stent implantation. This drug-free coating formulation has anticoagulant, anti-inflammatory, and inhibitory endothelial proliferation properties. It was shown that the drug-free coating favoured the endothelialisation process and induced the conversion of smooth muscle cells to a contractile phenotype. Animal studies have shown that drug-free stents reduced in-stent restenosis and improved neovascular intimal healing in rabbit and porcine models compared to drug-eluting stents. 99
Hemodynamic-optimised nanoparticle delivery remains critical for clinical translation. Under complex human hemodynamics with elevated shear stress, nanoparticles face substantial transluminal penetration barriers. Computational fluid dynamics (CFD) solves the fundamental equations of fluid dynamics (e.g. the Navier-Stokes equations) numerically and is capable of modelling and analysing haemodynamic parameters such as wall shear (WSS), pressure distribution and velocity fields. 100 The geometrical irregularities of the AAA lead to localised pressure concentrations (e.g. top of the tumour pressures are 30%–50% higher than in normal aorta) and in patients with AAA, the CFD can quantify the location of peak pressures and predict the risk of rupture at weak points (e.g. calcified plaque edges). Recent CFD analyses reveal that high-aspect-ratio nanorods exhibit 3.2-fold greater endothelial extravasation efficiency than spherical counterparts under pathological shear conditions, leveraging shear-induced alignment and reduced tumbling forces. To exploit this, shear-responsive nanocarriers incorporating thrombin-sensitive PEG shells or von Willebrand factor-mimetic peptides demonstrate targeted payload release at high-shear sites. Future designs should integrate real-time CFD-predicted shear maps from patient-specific CT angiography to optimise nanoparticle size (ideal 50–100 nm), shape (aspect ratio >8), and surface ligand density for site-specific margination. 101 In addition, eddy currents in the lumen of the aneurysm exacerbate vessel wall vibration and promote fatigue injury, which is significantly associated with the annual rate of AAA. 102
Closed-loop systems integrating treatment, imaging and prognostic monitoring are the future trend. Bismuth nanoparticles have attracted attention due to the high X-ray absorption and acceptable biocompatibility of the element bismuth, showing their potential to be converted into a commercial CT contrast agent, 103 a property that may make them potentially applicable in real-time monitoring of AAA, and bismuth nanoparticles can be intensively investigated in the future as a CT contrast agent for real-time monitoring of morphological changes in aneurysms. Bismuth nanoparticle cores (BiNPs) exemplify this approach: their high X-ray attenuation (5.2×10³ HU·cm³/g) enables microcalcification detection via contrast-enhanced CT, while conjugated MMP-9 inhibitory peptides (KD ~0.8 nM) mitigate ECM degradation. 104 In addition, the combination of photothermal therapy (PTT) and gene therapy (GT) shows great potential in the treatment of AAA. PTT-GT synergistically inhibits inflammation and promotes vascular regeneration, providing non-invasive and reduced side effects. In animal models, the combined application of PTT and gene therapy significantly inhibited the progression of AAA, reduced the release of inflammatory factors, and promoted vascular regeneration. 105 PTT-GT further optimises the imaging technology, and through real-time monitoring of the inflammatory response and vascular regeneration during the treatment process, it can assess the therapeutic effect and provide a basis for the adjustment of the subsequent treatment regimen, which realises a more accurate treatment. The PTT-GT can be used to monitor the inflammatory response and vascular regeneration during the treatment process in real time, which can provide a basis for the adjustment of the subsequent treatment plan and achieve more accurate treatment monitoring.
Nanoparticle geometry critically dictates vascular penetration and bioactivity. Rod-shaped mesoporous silica nanoparticles penetrate aneurysmal endothelia 4.7-fold deeper than spherical equivalents, facilitating MMP modulator delivery to medial VSMCs. After being taken up by activated endothelial cells, rod-shaped particles are retained for a shorter period of time than spherical particles, and are transported to deeper tissues (such as the vascular smooth muscle layer) more quickly. This geometry enhances binding avidity to fragmented elastin by presenting multivalent peptide ligands along the nanorod axis. 81 Experimental evidence shows that the penetration efficiency of 25 nm diameter mesoporous silica nanoparticles (MSNs) in the blood-brain barrier model is 5.38 times that of 50 nm spherical particles, because they are more likely to pass through the paracellular pathway or transcellular transport of endothelial cells. 106
The application of artificial intelligence (AI) can enhance the accuracy and efficiency of clinical decision-making. AI-driven smart sensors enable real-time biomechanical and biochemical feedback during nanomaterial-based AAA therapy, translating continuous wall-stress and inflammatory data into dynamic treatment adjustments. In a 2024 porcine model, ML algorithms coupled to wireless piezoelectric nanosensors provided 48-hour advance warning of mechanical instability, outperforming periodic imaging and allowing earlier, localised nano-drug release that reduced rupture risk by 37%. 107
Gene-editing nanocarriers, such as those based on the CRISPR/Cas system, have achieved significant progress in the safe delivery and long-term modulation of vascular tissues for AAA therapy. However, several challenges remain. Non-viral delivery systems, such as lipid nanoparticles (LNPs), can enhance endothelial targeting and reduce off-target effects through surface modification, but their cytotoxicity and risks of genomic integration require rigorous evaluation. Emerging strategies, including Cas9-EDV (enveloped delivery vehicles), utilise antibody fragments to enable cell-specific editing and have demonstrated successful T cell targeting without off-target events in animal models. 108 Additionally, LNPs encapsulating thermostable Cas9 ribonucleoproteins (RNPs) have achieved editing efficiencies of 37% in the liver and 16% in the lungs, with low immunogenicity. Nevertheless, a study by the Guangzhou Branch of the Chinese Academy of Sciences reported that long-term Cas9 expression in a porcine model induced DNA double-strand breaks, transcriptional dysregulation, and increased mutations in tumour driver genes, indicating potential carcinogenic risks. 109 To enhance safety, a Harvard–MIT team engineered the Cas9 enzyme structure to reduce off-target activity. 110 In summary, gene-editing nanocarriers have demonstrated therapeutic potential in vascular delivery, but their long-term safety must be further validated through vector optimisation (e.g. controllable expression systems) and large-animal model studies.
Conclusion
AAA is a degenerative chronic inflammatory vascular lesion characterised by irreversible dilatation and abnormal remodelling of the abdominal aorta, which is usually asymptomatic but carries a very high mortality rate in case of rupture. The advent of nanomaterials and nanotechnology has facilitated the development of new therapeutic modalities and diagnostic treatments for AAA. This article reviews the use of nanomaterials in clinical diagnosis and treatment, discusses the properties of various nanotherapeutic modalities, and explores their advantages and potential applications. Despite considerable progress in nanomaterials for cardiovascular disease therapy over the past decade, the transition from laboratory research to clinical practice still faces significant challenges, including the lack of specific animal models for aneurysms, poor tissue permeability, regulatory barriers, and biodistribution. The primary challenge confronting nanomaterials at present is the enhancement of their therapeutic efficacy and diagnostic efficiency while maintaining their safety.
Future research must also consider in vivo biodegradability and possible long-term consequences, given the accumulation of nanoparticle drugs for long-term use in vivo and the potential for more durable efficacy. In addition, preclinical studies of nanoparticle drug delivery and aortic disease imaging are currently focused on rodent models. Although the use of nanomaterials in these models has produced encouraging results over the past decade. However, a single model is not sufficient to fully simulate the pathological environment of AAA patients. Therefore, multiple validations and studies in various models are necessary before translating these findings into clinical applications.
Despite remarkable pre-clinical advances, translation of AAA-targeted nanomedicine is gated by four intertwined bottlenecks: murine models inadequately mimic human-scale hemodynamics and the multi-cell inflammatory milieu, causing >80% of nano-formulations to fail in large-animal validation; pathological shear stress at calcified plaques cuts nanoparticle penetration by 72%, demanding high-aspect-ratio carriers or CFD-guided personalisation; systemic dosing triggers metal-ion leakage from MOFs and IgM-mediated complement capture, diverting >60% of payloads to liver/spleen and fuelling chronic toxicity; and the absence of FDA guidance on nanocarrier degradation products, coupled with the lack of scalable GMP routes for complex systems like magnetically guided exosome nanomotors, stalls clinical entry. Overcoming these hurdles will require convergent strategies: porcine AAA or vascular organoid models to recapitulate human pathophysiology, shear-responsive PEGylated/biomimetic nanostructures that evade immune surveillance, real-time AI-integrated sensors to titrate drug release, and regulatory-industry consortia to standardise manufacturing and safety metrics. Only by systematically de-risking these barriers can nanomedicine advance from ‘great potential’ to first-in-human trials and ultimately transform AAA management from passive surveillance to precision, preventive therapy.
Footnotes
Author contributions
All authors contributed significantly to, and are in agreement with, the content of the manuscript. XY, Y., and ZH, Y., did the writing-original draft. WP, Z., JC, Q., did writing-review and editing. WM, Z., JC, Q., did project administration and wrote guidance.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China (82460103 and 82460104), Key Science and Technology Innovation Project of Jiangxi Provincial Health Commission (2024ZD005 and 202210617), Youth Project of the Science and Technology Department of Jiangxi Province(20252BAC200450), Science and Technology Research Project of Jiangxi Provincial Department of Education-Youth Project (GJJ2400114) and The Second Affiliated Hospital of Nanchang University Funding Program (2023efyA02).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
No new data was generated.
