Abstract
Background:
Melanin can be used in biomedical nanomaterials, but its solubility in water and bioavailability are low.
Aim:
Melanin nanoparticles were prepared and then PEG-natural melanin nanoparticles (NMNP-PEG) were obtained with good performance and optimize their (water solubility, dispersion stability, chelating metal ions, photothermal stability, drug delivery, and biocompatibility), therefore improve the water solubility of melanin and broaden its application scope in biology, medicine, food, and other fields.
Methods:
MFAK (melanin from apricot kernels) and NMNP-PEG were prepared and characterized using ultraviolet-visible spectrophotometry (UV-Vis), high-performance liquid chromatography (HPLC), Fourier-transform infrared spectroscopy (FTIR), 1H nuclear magnetic resonance (NMR), and electron microscopy. The chelation rate of metal ions, photothermal effect, doxorubicin loading, and cytotoxicity (MCF-7 cells) were examined.
Results:
UV-Vis, HPLC, FTIR, and NMR indicated that NMNPs contained melanin. NMNPs could be successfully modified using PEG. Under physiological pH conditions (pH 7.4), the metal ion chelation rate of NMNP-PEG increased with time and peaked at 12 h. The photothermal assay showed a temperature enhancement of 26.3°C with 1 mg/mL NMNP-PEG, compared with 1.9℃ with water. The NMNP-PEGs had a typical peak for doxorubicin in the FTIR spectrum, and the peak intensity was proportional to the drug loading. The release of doxorubicin in an acidic buffer was 40.8% at 24 h, almost threefold that in a neutral buffer (11.9%). There was no obvious cytotoxicity from NMNP-PEG.
Conclusion:
NMNP-PEG displays good stability, high metal ion chelation ability, efficient photothermal conversion potential, drug-retaining capability, sustained controlled drug release, and biocompatibility. This study provides a theoretical basis for NMNP-PEG applications in medicine (targeting specific sites to diagnose and treat diseases), food (extending the shelf life of food), and biology (as metal ion chelating agents to remove heavy metals from wastewater).
Introduction
Melanin has excellent physical and chemical properties and can bind metals, drugs, and other chemicals.1,2 Melanin can form highly cross-linked heteropolymers bound to matrix scaffolding melanoproteins through covalent links. 3 The antioxidant ability of melanin is directly associated with its degree of polymerization, 4 and suboptimal polymerization can lead to a pro-oxidant form of melanin involved in melanoma development and retinal degeneration. 5 Melanin is also involved in innate defense against bacteria, fungi, and neurodegenerative diseases.6,7
In general, melanin is not stably dispersed in water, restricting the analyses and experiments on uniformly distributed systems, 8 but nanotechnology can be used to improve the water dispersion of melanin. 9 Indeed, nanoparticles can increase the solubility and bioavailability of natural products. 10 At the same time, new characteristics, such as surface properties and side effects, can be generated.
In addition to covalent cross-linking, non-covalent interactions during the supramolecular assembly of melanin are critical in determining the nanomaterial properties. 11 In addition, other polymers can be used with melanin for various technological applications. One of those polymers is polyethylene glycol (PEG), which increases the bioavailability and stability of nanoparticles in vivo.12,13 Melanin- and PEG-based nanomaterials can be used for photodynamic therapy, 14 photothermal therapy (PPT), 15 drug delivery systems, 16 photoacoustic tomography, 13 and magnetic resonance imaging (MRI). 17 Drug delivery is a promising application of melanin-based nanotechnology, especially to increase potency while minimizing the systemic side effects of chemotherapeutic agents.16,18–20
This study aimed to prepare PEG-natural melanin nanoparticles (NMNP-PEG) and optimize their water solubility, dispersion stability, chelating metal ions, photothermal stability, drug delivery, and biocompatibility. The natural melanin from apricot kernels (MFAK) was isolated and characterized in this study.
Methods
Preparation and purification of MFAK
MFAK was extracted by the alkali-solution method and acid-isolation assisted by ultrasound, as previously described.21,22 Apricot kernels (collected from wild apricot trees in Taiyuan, Shanxi Province, China) were dried and ground into a powder. Crude melanin was suspended in 2 M HCl by incubating the solution in a water bath at 100°C for 2 h and centrifuging it at 10,000 rpm for 10 min. Ethanol was used to wash away the hemicellulose from the crude melanin until the supernatant was colorless. The melanin was dissolved in NaOH and precipitated by adding acid until the supernatant was colorless (repeated several times). Finally, the pure melanin was washed with deionized water three times and vacuum-dried at 70℃.
Preparation of the NMNPs
The NMNPs were prepared using an ultrasonic method according to the different solubilities of melanin in various solvents, based on previous studies.9,23,24 Method 1: MFAK was dissolved in 10% ethylene glycol, adjusting the pH to 7.0 using 1 M HCl, with sonication (200 W, 60 kHz) for 30 min. Method 2: MFAK was dissolved in 10% dimethyl sulfoxide (DMSO), adjusting the pH to 7.0 with 1 M HCl, with sonication (200 W, 60 kHz) for 30 min. Method 3: MFAK was dissolved in NaOH (0.1 mol/L), adjusting the pH to 7.0 with 1 M HCl, with sonication (200 W, 60 kHz) for 30 min. Method 4: The nanoparticles prepared using Method 3 were treated by an ultrasonic cell crusher with a power of 200 W for 10 min, adjusting the pH to 7.0 with 1 M HCl and centrifuging the mixture; the pellet was rinsed thrice with deionized water and dried.
Surface modification of the NMNPs with mPEG2000-NH2
PEG chains were introduced to the NMNPs to enhance their water solubility. Surface-modified NMNPs were prepared by mixing NMNPs with mPEG2000-NH2 (Aladdin Chemicals Co., Ltd., Shanghai, China) at a mass ratio of 1:2 (pH 9.0).12,25
Ultraviolet-visible spectrophotometry (UV-Vis)
The optical properties of MFAK were investigated using a Lambda 25 UV-Vis spectrophotometer (Perkin-Elmer Life Sciences, Waltham, MA, USA).
High-performance liquid chromatography (HPLC)
Crude and purified MFAKs were dissolved in 0.5 M NaOH before HPLC analysis. The analysis was performed on a Waters e2695 HPLC system (Waters, Milford, MA, USA) with a diode-array detection system. HPLC detection of MFAK was achieved on a Waters Symmetry C18 column (4.6 × 150 mm, 5 μm). Acetic acid 1% in methanol constituted the mobile phase, used for isocratic elution for 80 min. 26 Each elution was balanced for 15 min (liquid phase condition: flow rate: 0.2 mL/min; injection volume: 20 μL; detection wavelength: 280 nm; column temperature: 25°C).
Fourier transform infrared spectroscopy (FTIR)
Crude and purified MFAKs were mixed with dry KBr (200 mg), made into tablets, and scanned at 4000–400 cm−1.
1H nuclear magnetic resonance (NMR)
DMSO-d6 was used to dissolve the purified MFAK. The NMR conditions were temperature of 299.0 K, delay time of 1.000 s, spectral width of 8223.685 kHz, 16 sampling repetitions, observation frequency of 400.10 kHz, and matrix size of 65,536.
Morphology analysis
The purified MFAK was sputter-coated with gold and observed directly using scanning electron microscopy (SEM) (SU8010, Hitachi, Tokyo, Japan).
NMNP characterization
The diameters of the NMNPs were determined by dynamic light scattering (DLS) (Zeta Plus, Malvern Panalytical Ltd., Malvern, UK). FTIR and 1H NMR spectra of the NMNP/NMNP-PEG powder were determined using the above methods. The morphologies of the NMNPs and NMNP-PEGs were observed by transmission electron microscopy (TEM) (Hitachi, Tokyo, Japan).
Metal ion chelation
For metal binding reactions, 4 mL of FeCl3 (10 mg/mL) was added to 1 mL of NMNP-PEGs (40 mg/mL), and the pH of the solution was adjusted to 7.4 immediately. The reaction was carried out in the dark for 0.5, 1, 2, 4, 8, 12, 24, and 36 h. 27 NMNP-PEGs bound to Fe(III) were separated by centrifugation. The amounts of unbound ions in the supernatants were calculated using a UV-Vis spectrophotometer. The binding properties of the NMNP-PEGs to CuSO4 and ZnCl2 were determined using the same method.
Photothermal effect of NMNP-PEG
NMNP-PEGs in aqueous solutions (0.25, 0.5, and 1 mg/mL) were placed into a quartz cuvette and exposed to a near-infrared (NIR: 808 nm) laser at 3.6 W/cm2 for 700 s. 15
Drug loading and stimulus-responsive drug release by NMNP-PEGs
Doxorubicin (DOX) (Aladdin Chemicals Co., Ltd., Shanghai, China) was loaded onto the NMNP-PEGs by mixing 1 mL NMNP-PEG with 2 mL of varying concentrations of DOX under magnetic stirring. After 12 h, NMNP-PEG/DOXs were collected using centrifugation at 15,000 rpm for 10 min and washed three times with deionized water. The drug-loading capacity of the NMNP-PEGs was measured by UV-Vis (wavelength: 490 nm). Next, 0.1 mL sample solution was centrifuged at 15,000 rpm for 10 min to remove the NMNP-PEG/DOXs. The supernatant was analyzed by HPLC to measure the unloaded DOX. The drug loading in the NMNP-PEGs was calculated by subtracting the drug released from the amount initially loaded.
The NMNP-PEG/DOXs (15 mL) were packaged in a dialysis tube (MWCO = 2.0 kDa) and placed in 40 mL of phosphate-buffered saline (PBS: pH 7.4)). pH was changed from 7.4 to 5.0 for pH-responsive release. H2O2 was added to the PBS at a final concentration of 20 mM. At different points, 1 mL of solution was sampled (1 mL of PBS was added in order to maintain volume) to measure the concentration of the released drug by HPLC.
Cell culture and cytotoxicity assay
The breast cancer cell line MCF-7 was obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA). The cells were cultured in DMEM medium at 37℃, 5% CO2. The DMEM medium contained 10% fetal bovine serum, 100 units/mL penicillin, and 100 mg/mL streptomycin. The cytotoxicity of the NMNP-PEGs was analyzed on MCF-7 cells seeded into a 96-well plate (104 cells/well). NMNP-PEGs (0.78–400 μg/mL) were added and incubated for 48 h. A CCK-8 assay was performed to determine cell viability.
Statistical analysis
Descriptive statistics were used. Continuous data were presented as means ± standard deviations. Data analysis was performed using OriginPro Portable 8.5 (OriginLab, Northampton, MA, USA).
Results
Characterization of MFAK and NMNP
The characteristics of MFAK and NMNP are presented as Supplemental Material.
Surface modification of the NMNPs
The NMNPs prepared by this method retained the key functional groups of MFAK. mPEG2000-NH2 was used to improve water solubility, biomodification, and biocompatibility (Figure 1). In addition, the surface of the NMNPs was modified effectively with mPEG2000-NH2. The peaks of mPEG on mNMNP-PEG were at 2880 cm−1 (alkyl C-H stretching) and 1110 cm−1 (C-O-C stretching), demonstrating that the NMNPs were successfully modified (Figure 1).
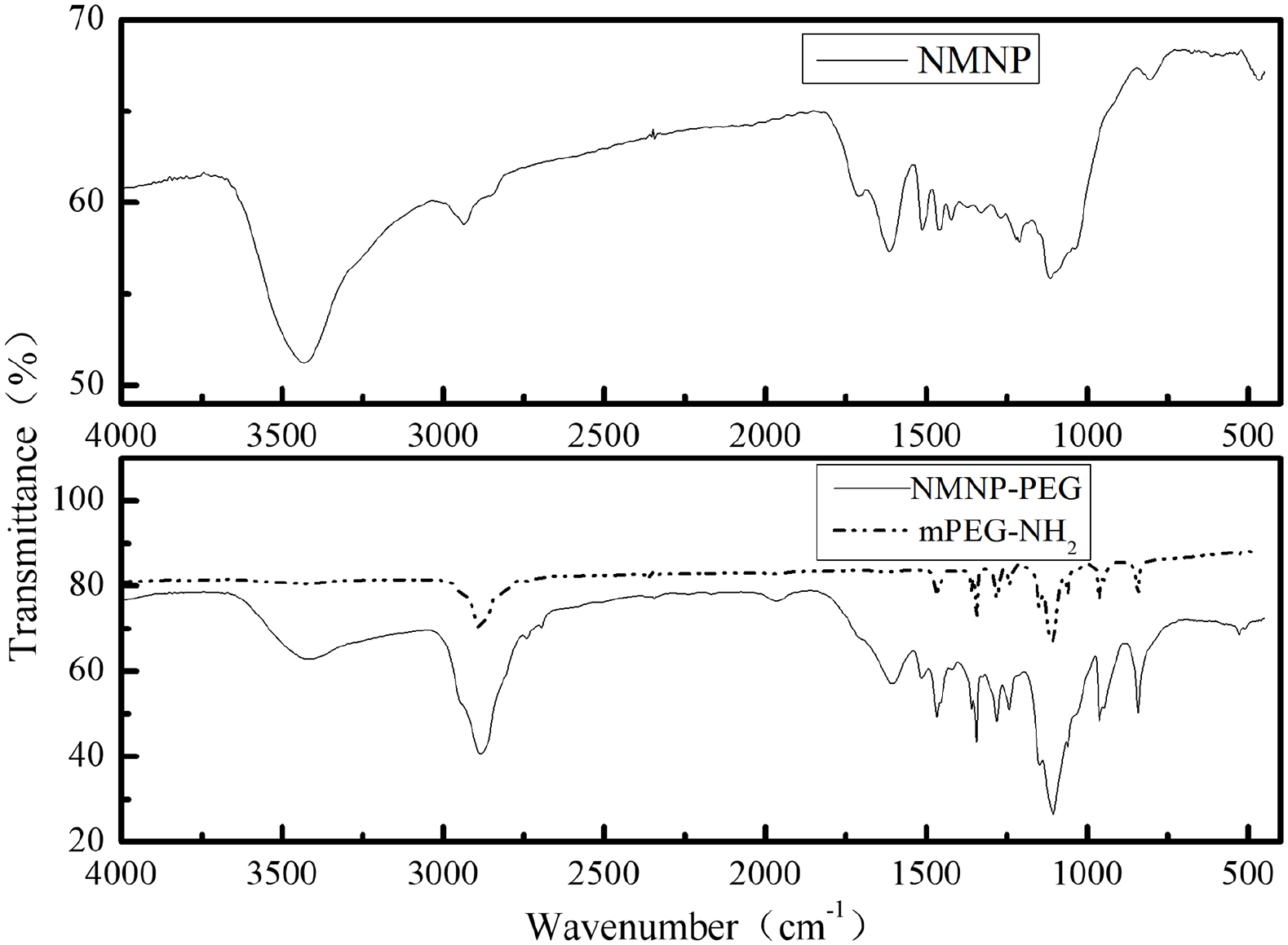
Fourier-transform infrared (FTIR) spectrum of mPEG2000-NH2 and NMNP-polyethylene glycol (PEG).
TEM
The NMNPs prepared using Method 4 (Figure 2(A(b))) showed a smaller size, narrower size distribution, and better dispersibility than the NMNPs obtained using Method 3 (Figure 2(A(a))). Therefore, Method 4 was the optimal method for preparing NMNPs. The particle size determined by TEM was 5–15 nm. After the NMNPs prepared by Method 4 were modified, the NMNP-PEGs were 15–20 nm in diameter (Figure 2(B)) and indicated excellent dispersion stability.

Functional characterization of the natural melanin nanoparticles (NMNPs): (A) transmission electron microscopy (TEM) images of NMNP: (a) NMNP prepared by Method 3 and (b) NMNP prepared by Method 4, (B) TEM images of NMNP-polyethylene glycol (PEG), (C) metal ion chelation rate of melanin from the apricot kernel (MFAK) and NMNP-PEGs, and (D) photothermal heating curves of NMNP-PEG with different concentrations irradiated by NIR laser at a power density of 3.6 W/cm2.
Metal-ion chelation
It can be seen from Figure 2(C) that under physiological pH conditions (pH 7.4), the metal ion chelation rate of the NMNP-PEGs increased with time and peaked at 12 h. The maximum chelating amounts of NMNP-PEGs to Fe(III), Cu(II), and Zn(II) were 2.99, 0.75, and 0.33 mmol/g, respectively. The maximum chelation rates of Fe(III), Cu(II), and Zn(II) were 48.51%, 10.10%, and 4.54%, respectively. Compared with the chelation rate of MFAK, the chelation rates increased by 19.30%, 3.61%, and 2.28%, respectively.
In vitro photothermal effect of the NMNP-PEG
The temperature of the NMNP-PEG solution increased with irradiation time, as shown in Figure 2(D). The final temperature was enhanced by 26.3℃ in the solution with 1 mg/mL NMNP-PEGs, compared with 1.9℃ for water alone. Hence, NMNP-PEGs had a photothermal conversion efficiency of 28.6%.
Drug-loading and stability of NMNP-PEG/drug
Compared with NMNP-PEGs, a typical DOX peak was detected at 490 nm (Figure 3(a)). The peak intensity increased with the drug loading amount. The drug loading was the highest at a 61.5% DOX-weight ratio. A small amount of DOX (about 11.9%) was released from the NMNP-PEGs after a 24 h incubation in PBS (Figure 3(b)), which meant that the NMNP-PEGs could retain a drug load under physiological conditions.

Doxorubicin loading of natural melanin nanoparticles (NMNP)-polyethylene glycol (PEG): (a) UV-visible spectra of NMNP-PEG with different loadings of doxorubicin, (b) pH and ROS-responsive drug release from NMNP-PEG/doxorubicin, and (c) The cytotoxicity of NMNP-PEG on MCF-7 cells.
Drug release study
DOX release could be triggered from NMNP-PEGs under acidic conditions (pH 5.0) (Figure 3(b)). The cumulative release of DOX in an acidic environment was 40.8% at 24 h, almost threefold the release using a neutral buffer (11.9%). Moreover, under oxidative stress, DOX release was increased compared with the neutral buffer.
Toxicity of NMNP-PEG
NMNP-PEG showed no significant cytotoxicity in the experimental concentration range (Figure 3(c)).
Discussion
The physicochemical characterization of MFAK revealed no obvious absorption peak, but its optical density was negatively correlated with the increase of wavelength, as supported by the literature. 28 The characteristic absorption peak at 260–280 nm was in line with melanin isolated from plants and animals and with synthetic melanin. 28 The absorbance plotted against wavelength suggested that the extraction yielded typical melanin.21,29 Furthermore, the FITR spectra were similar to the literature, 30 with OH and NH groups at 3500–3400 cm−1,31 aliphatic C-H stretches (such as CH2 and CH3) at 2930 cm−1,32 aliphatic C = C stretches at 1453 cm−1,33 aromatic C = C stretches and COO stretches at 1650–1600 cm−1,34 phenolic COH bends and phenolic NH stretches at 1400–1380 cm−1,35 C-O stretching of polysaccharides at 1047 cm−1,27 aromatic ring substitution at 800–600 cm−1,33 and aliphatic groups and glycosidic linkages at 2930 and 1047 cm−1. In addition, as could be expected, the purity of the purified MFAK was higher than its crude counterparts.
The HPLC chromatograms of MFAK showed a profile with a single signal, as previously reported.33,36 The chromatogram profile of crude/purified MFAK showed a main single symmetrical elution peak at 7.126 and 7.293 min. The peak for MFAK was similar to the peak of melanin synthesized by the oxidation of tyrosine, indicating that MFAK might have a similar structure to standard melanin. 33 On the other hand, MFAK showed that the main compound was eluted in the range of 7.126–10.0 min, and a minor chromatographic peak was eluted at approximately 9 min. The results showed that MFAK might contain more than one melanin species or derivatives.
The 1H-NMR results showed it was hard to perform quantitative analysis and showed an estimated spin-spin coupling resulting from intramolecular disorders of MFAK. The peak at 6.0–7.5 ppm suggested the presence of macromolecules. 26 The signals at 6.0–7.5 ppm indicated aromatic hydrogens or aromatic heterocycles, which hinted at the complicated chemical environment around aromatic hydrogens. 37 The signal peak in the range of 3.2–4.2 ppm was caused by CH2 or CH3 groups connected with a nitrogen or an oxygen atom. 26 In the aliphatic region, signals within the range of 0.8–1.0 ppm could be assigned to CH3 groups of alkyl fragments caused by residual proteins. Signals at 2.0 ppm belonged to methylene or ester groups, indicating the presence of carboxylate structures. Therefore, the FTIR and 1H-NMR spectra indicated that the functional groups of MFAK/NMNP included aromatic rings containing potential functional groups such as hydroxyl (OH), carboxyl (COOH), methyl (CH3), and amine (NH).
The analysis of the NMNPs revealed similar results to MFAK, indicating that the properties of melanin were retained, but the 1H-NMR showed that compared with MFAK, NMNP had only the disappearance of the peak at 6.0–7.5 ppm, which was likely due to the degradation of the macromolecules into smaller molecules. Then, to improve water-solubility, biomodification, and biocompatibility, mPEG2000-NH2 was used because the NH group can react with the dihydroxyindole/indolequinone in melanin. 38 The nanoparticles prepared using Method 4 had a size of 15–20 nm and excellent dispersion stability.
Functionally, the NMNP-PEGs could chelate Fe(III), Cu(II), and Zn(II), suggesting a potential for use in patients with metal poisoning or for loading ions for imaging.23,31,33 In addition, NMNP-PEGs could be loaded with doxorubicin, as previously shown.2,17,39 The NMNP-PEGs could gradually release the doxorubicin with time, which could be modulated by pH and reactive oxygen species (ROS). The NMNP-PEGs are rich in phenyl, NH, and OH groups on their surface, endowing them with the ability to load multiple drugs by π-π stacking and/or hydrogen bonding interactions. 12 The drug release triggered by an acidic environment was probably attributed to the protonation of NH on the NMNP-PEGs and/or on the DOX molecules. 16 DOX was continuously released after 20 mM H2O2 was added into the neutral buffer, and the cumulative release amount of DOX was 21.92% after 24 h of H2O2 treatment. H2O2 oxidized the polyphenol groups abundant on the NMNP-PEGs, thus destroying the hydrogen bonding between NMNP-PEG and drugs. 40 The microenvironment of tumors is mildly acidic, and the highly dysregulated metabolic state leads to oxidative stress. Under such stress, tumors are associated with an increased generation of ROS.12,40 Conditions such as acid and high ROS could promote the targeted release of NMNP-PEGs anticancer drugs. Contrasting with complex drug delivery systems, NMNP-PEGs could load a variety of anticancer drugs without any additional modifications and respond to different stimuli to trigger the release of the loaded drug. Furthermore, the results indicated an excellent photothermal ability of NMNP-PEG, suggesting its possible use in targeted photothermal therapy, as suggested by previous studies. 12 Before in vivo studies could be conducted, the cytotoxicity of NMNP-PEGs was confirmed in MCF-7 cells for 48 h and using concentrations ranging from 0.195 to 100 μg/mL. The results suggested no significant cytotoxicity, as supported by previous studies.41,42
This study has limitations. It mainly focused on the physicochemical properties of the NMNP-PEGs, and their exact eventual use for therapy must be demonstrated in vitro and in vivo. In addition, four different synthesis methods were tested, but further optimization is still necessary, as well as the eventual adaptation of the final method to an industrial scale.
Conclusions
The results showed that NMNP-PEGs have good stability, high metal ion chelation ability, efficient photothermal conversion potential, drug-retaining capability, sustained controlled drug release, and biocompatibility. This study provides a highly promising biomaterial for widespread biomedical applications. This study provides a theoretical basis for NMNP-PEG applications in medicine (targeting specific sites to diagnose and treat diseases), food (extending the shelf life of food), and biology (as metal ion chelating agents to remove heavy metals from wastewater).
Supplemental Material
sj-docx-1-jbf-10.1177_22808000221124418 – Supplemental material for Improving the solubility of melanin nanoparticles from apricot kernels is a potent drug delivery system
Supplemental material, sj-docx-1-jbf-10.1177_22808000221124418 for Improving the solubility of melanin nanoparticles from apricot kernels is a potent drug delivery system by Li Gao, Liu Yang, Lixiao Guo, Haibin Wang, Yinghu Zhao, Jun Xie and Nan Shi in Journal of Applied Biomaterials & Functional Materials
Footnotes
Authors’ contributions
Li Gao and Liu Yang carried out the studies, participated in collecting data, and drafted the manuscript. Lixiao Guo and Haibin Wang performed the statistical analysis and participated in its design. Yinghu Zhao, Jun Xie and Nan Shi participated in acquisition, analysis, or interpretation of data and draft the manuscript. All authors read and approved the final manuscript.
Data Availability
The datasets used and/or analyzed during the current study are included within the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Research Project Supported by Shanxi Scholarship Council of China (2020-118); Shanxi key laboratory of pharmaceutical biotechnology, Shanxi Institute of Biology (KF202002); Undergraduate Education and Teaching Reform project by North University of China (ZJ2020077); the Key Research and Development Program of Shanxi Province (201803D221013-4 and 201903D221033).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
