Abstract
Alternative calcium phosphates are strong candidates as tooth-whitening to mitigate the adverse effects of hydrogen peroxide (HP). This study examines changes in the color and chemical structure of human enamel induced by a biomimetic calcium phosphate-based bleaching ceramic derived from chicken eggshell, known as the experimental remineralizing Substance (ERS). Forty human molars were assigned to four groups: G1: storage group; G2: treated with 35% HP; G3: treated with ERSs; and G4: treated with HP followed by ers. CIE color coordinates (
Introduction
Dental bleaching is a cosmetic procedure that addresses external and/or internal dental dyschromia. It consists of any process aimed at lightening the color of teeth. Bleaching methods are either physical (mechanical and optical) or chemical. The most used chemical approach for external bleaching of vital teeth involves the application of hydrogen peroxide (HP) or carbamide peroxide (CP). However, these agents are associated with limitations and risks, including tooth hypersensitivity and demineralization.
The whitening mechanism of HP is based on a redox reaction involving the molecular moieties of organic pigments and the action of free radicals. These radicals break molecular chains, leading to a loss of pigment coloration. 1 Free radicals can also affect the organic content by breaking polypeptide chains and degrading amino acids that are components of organic substances.1–3 Simultaneously, HP affects the inorganic content of enamel, including mineral apatite dissolution and the loss of elements such as oxygen (O), sodium (Na), magnesium (Mg), calcium (Ca), and zinc (Zn).4–7 HP use also leads to a decrease in phosphate content, an increase in carbonate groups,4,8,9 changes in the crystallinity index, and alterations in the Ca/P ratio, ultimately resulting in changes to the chemical composition of enamel.10,11
It is worth noting that these changes are directly influenced by treatment conditions, including peroxide concentration, pH, type of activation (e.g. heat, laser radiation, or UV light), temperature, exposure time, the number of applications, and interactions with transition metals, among other factors.12–15
Chemical and morphological changes in teeth caused by bleaching can be partially repaired through natural processes, such as exposure to saliva, and the application of agents including calcium, fluorides, potassium nitrate, amorphous calcium phosphate (ACP), casein phosphopeptide (CPP), calcium silicate, synthetic hydroxyapatite (HA), and synthetic or chicken-eggshell-derived nano-hydroxyapatite (nHA).10,13,15–19 However, despite their beneficial effects, these chemical agents cannot fully restore the original properties of dental tissues.
In response, alternative biomimetic substances based on calcium phosphates are being studied as bleaching agents to overcome the adverse effects of conventional peroxide-based bleaching treatments on enamel. Synthetic calcium phosphates, including hydroxyapatite, zinc carboapatite, and tricalcium phosphate, have demonstrated their ability to induce certain changes in enamel color parameters (
Previous research led to the development of a biomimetic-modified remineralizing agent (MRA) composed of two distinct components. The first component consisted of a mixture of acidic phosphate phases purified from eggshells, while the second was a pH-restoring remineralizing agent.
21
This MRA was evaluated to assess the influence of osmotic pressure and the mass proportions between the two main phosphate components—90%–10% and 50%–50%—referred to as MRA 91 and MRA 55, respectively. The study demonstrated that the exerted osmotic pressure had no significant effect on remineralization, while MRA 91 produced the most substantial color changes in
The main features of ERS include the complete absence of oxidative components—entirely distinct from conventional whitening materials—and its biomimetic composition. Therefore, this study aims to evaluate color and chemical changes in human enamel following whitening treatment with ERS. The null hypothesis proposes that ERS would not alter the color or chemical composition of enamel.
Materials and methods
The Ethics Committee of the Faculty of Dentistry (CIE-028-15), Universidad Nacional de Colombia, Bogotá campus, approved this project.
Material preparation
The Experimental Remineralizing Substance (ERS) is composed of two distinct components. The first is a powder consisting primarily of acidic solid phosphates, applied to wet enamel as an aqueous paste. The second is a liquid solution that restores the natural neutral pH of enamel while supplying additional calcium ions.
Sample size calculation and tooth selection
A non-probabilistic sampling method was used. The sample size was calculated based on a two-sided test with a 5% alpha level and 83% power, allocating 10 teeth per group. Forty extracted molars donated by individuals aged 18–28 were selected for the study after signing the informed consent form. All teeth were thoroughly examined. Those teeth presenting cracks, structural anomalies, fractures, pulp necrosis, cavities, prior whitening, or any restorative treatments were excluded. The selected samples were subjected to the cleaning, disinfection, and conservation protocol established by the Teeth Biobank Guidelines from Universidad Nacional de Colombia. 23
Sample treatments
A 5 × 5 mm area on the buccal surface of the middle coronal third of each tooth was marked using a one-fourth round bur. The four vertices of this square were designated as the zone for observations and measurements. Afterward, the teeth were randomly assigned to four groups (
Methodology and sample characterization
Color analysis by spectrophotometer
After the specimens were rinsed with distilled water and dried with absorbent paper, initial and final color measurements were recorded on the mid-vestibular region of each sample using a calibrated spectrophotometer (Vita Easyshade Advance 4.0, VITA Zahnfabrik, Bad Säckingen, Germany). The operator recorded the results by documenting the instrumental color coordinates: lightness,
In addition, the Whiteness Index for Dentistry (
Perceptibility (PT) and acceptability (AT) by human observers were determined by comparing the results to threshold values established in previous studies.
24
For
Attenuated total reflectance–Fourier transform infrared (ATR-FTIR) spectroscopy
Enamel powder was collected from the treated surfaces using a medium-grit aluminum oxide sandpaper strip (Microdont® TDN 0020/13). For ATR-FTIR analysis, infrared spectra were acquired using a Nicolet iS10 FT/IR spectrometer (Thermo Fisher Scientific®). Spectra were recorded in absorbance mode over the 600–4000 cm−1 range, with 124 scans at a resolution of 1 cm−1. Curve fitting was performed, and integrated areas were measured using curve-fitting software (PeakFit v4.11, Systat Software Inc.). 25
The relative amounts of phosphate, carbonate, and organic content in the enamel samples were calculated by analyzing the peak areas of the absorption bands associated with phosphate, carbonate, and amide groups, as identified in the infrared spectra. 26 Overlapping peaks within the ranges of 900–1200 cm⁻¹ (ν₁–ν₃ PO₄ molecular vibrations, representing mineral phosphate groups), 800–850 cm⁻¹ (carbonate groups), and 1300–1750 cm⁻¹ (carbonate type B substitution and amide groups in the hydroxyapatite structure, Ca10(PO₄)₆(OH)2, where phosphate sites are referred to as position B and hydroxyl sites as position A) were resolved using a second derivative methodology and fitted to a mixed Gaussian–Lorentzian function. This function is referred to as “rat” (ratio) in the Results section.
Based on the ATR-FTIR spectral analysis, the following compositional parameters were calculated to describe enamel mineral properties:
X-ray diffraction
X-ray diffraction (XRD) patterns were obtained using an X’Pert Pro MPD® diffractometer (PANalytical) with CuKα radiation (λ = 1.5406 nm), generated at 40 mA and 45 kV. Scans were conducted over a range of 5–60° (2θ) with an angular step size of 0.0042° and a counting time of 5.08 s per step. The average crystallite size (

Where
Statistical analysis
All color variables—Δ
Results
Color analysis by spectrophotometry
Table 1 presents the results for color coordinates (
Analysis of

For ∆
In terms of the perceptibility (PT) and acceptability (AT) thresholds for
Regarding ∆
Attenuated total reflectance–Fourier transform infrared (ATR-FTIR) spectroscopy
Table 2 presents the most relevant results of the ATR-FTIR statistical analyses—including one-way ANOVA, Shapiro–Wilk, and Kruskal–Wallis tests—for each variable.
Results of ANOVA, Shapiro Wilks, and Kruskal-Wallis test for the most significant variables, including carbonates, phosphates, amides, and others (Degmin, MinCO32−, and Crystallinity Index) across groups.

Phosphates
Rat1095 (the ratio of the 1095 cm⁻¹ peak to the 900–1200 cm⁻¹ region; (
Carbonates
Rat878 (CO3²⁻ ν2 band, type A substitution at OH⁻ sites;
The a1413 (CO3²⁻, type B substitution at PO4 ν3;
Amides
The a1690 band (Amide I;
The degree of mineralization (Degmin;
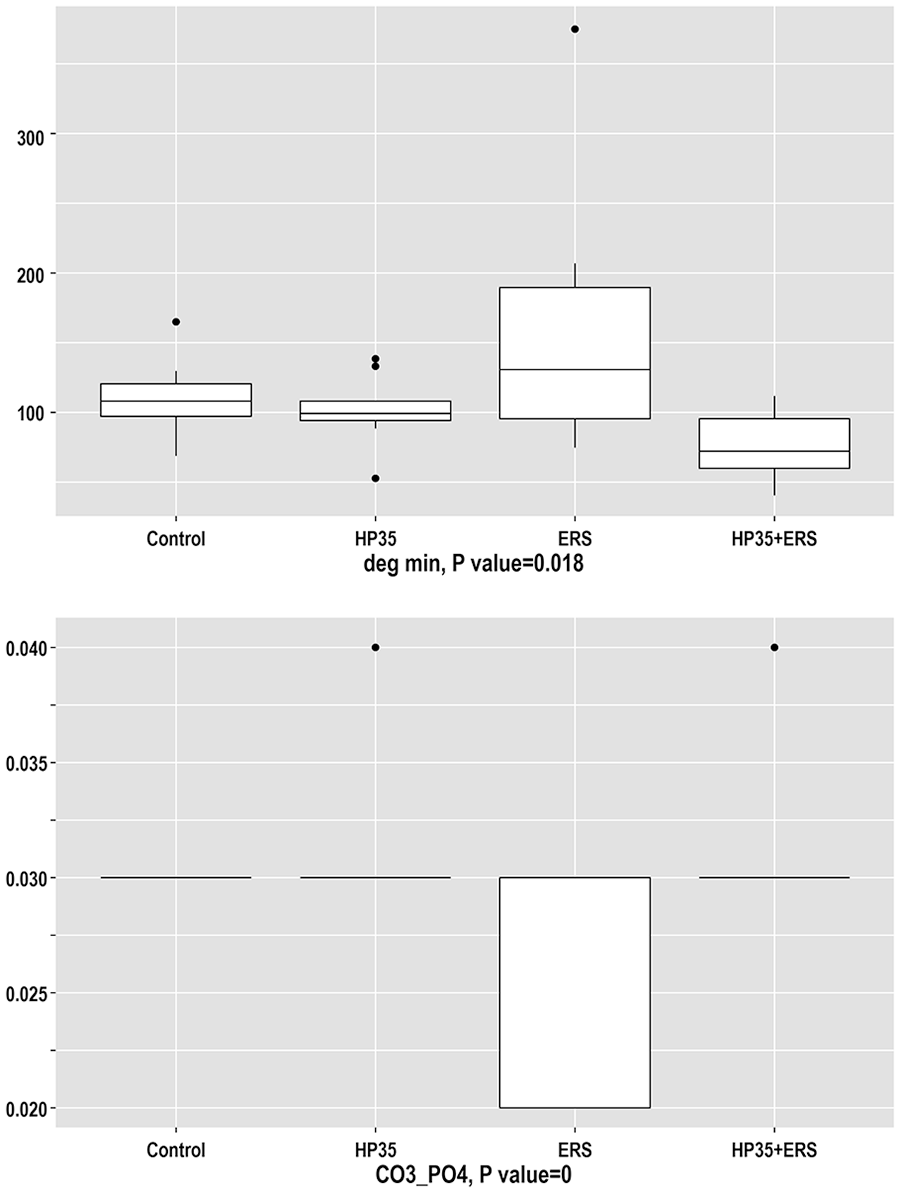
Results of the analysis of Degmin and MinCO32− variables obtained through ATR-FTIR spectroscopy.
The relative mineral carbonate content (MinCO3²⁻;
The crystallinity index (CIIR;
X-ray diffraction analysis
The XRD patterns for the four experimental groups showed diffraction peaks corresponding to hydroxyapatite (HAp) crystals, as identified by JCPDS card no. 9-432 (Figure 2). The average crystallite size of the enamel powder samples was calculated from the 002 diffraction peak, which corresponds to the c-axis reflection of HAp crystals.

X-ray diffraction patterns for the four experimental groups (G1: control, G2: HP 35%, G3: ERS, G4: HP 35% followed by ERS). Miller indices for hydroxyapatite crystal are indicated. Crystallite size measurements were performed for the (002) reflection along the c-axis of HAp crystals.
The results revealed that crystallite size (i.e. crystalline domains) in the experimental groups decreased compared to the G1 (without treatment) (
Crystallite size (

Crystallite size (nm) results by group. The
Discussion
This study evaluated color and chemical changes in human enamel using FTIR and XRD after the application of ERS. The null hypothesis was rejected, as ERS had a significant effect on color, and the observed chemical changes—though subtle—reflected meaningful variations within the same crystalline components (calcium phosphates).
This study used teeth extracted from individuals aged 18–28 years. These teeth exhibited high permeability and a greater concentration of mineral elements—such as calcium (Ca) and phosphorus (P)—in the vestibular face and superficial enamel layer. Additionally, the high ion exchange rate of these teeth is noteworthy, as it reflects active mineral exchange processes. 35 This methodological approach enabled the standardization of specific parameters and the detection of structural and chemical changes in enamel, despite its high intra- and interindividual variability.
Regarding the color results, samples treated with HP did not show significant changes in ∆
The results may be influenced by factors such as reduced enamel thickness on the vestibular side, deproteinization, and a decrease in mineral content (calcium and phosphates). 4
On the other hand, G3 (ERS) exhibited a significant decrease in lightness (
A decrease in yellowness (
Tooth whitening is ultimately achieved by balancing the contributions of the yellow–blue and red–green axes, which can be influenced by colorless, nanoscale crystals that scatter light—similar to how water is transparent, yet the clouds it forms appear white. Hence, in the absence of organic pigments or chromophore ions, the new layers formed by ERS will appear white, regardless of any underlying discoloration in the substrate.
G4 (HP + ERS) showed a significantly diminished Δ
The Δ
These results suggest that ERS is the most effective treatment for altering enamel color, followed by HP. However, the combined treatment (G4, HP + ERS) was less effective than ERS alone.
Although the increase in ΔWI
FTIR spectroscopy is a destructive technique that enables the identification of functional groups and chemical compounds, as well as the analysis of biomimetic materials and their interactions with enamel. 36 The observed peaks facilitate the detection of carbonate and phosphate groups at various sites within the apatite (Ap) unit cells. The typical composition of a chemically synthesized apatite cell in an inert, pure atmosphere is Ca10(PO4)6(OH)2. The calcium positions within the apatite structure can be occupied by various cations, including Ca²⁺, Mg²⁺, Sr²⁺, or even O²⁻. The PO4³⁻ site is known as position B and may be substituted by several anions, primarily carbonate. Position A, associated with the hydroxyl group, can also accommodate anions such as fluoride, chloride, and carbonate. 37 When pure solid apatite (Ap) is exposed to carbon dioxide, CO2 is incorporated exclusively at position A. However, when CO2 is present during Ap formation, it accumulates at both positions A and B. The contents of calcium, as well as sites A and B, can be altered, with several of the 10 calcium atoms potentially replaced by cations such as Mg²⁺ or Sr²⁺. Similarly, substitutions can occur at the phosphate (position B) and hydroxyl (position A) sites. When comparing all the experimental groups to G1, the presence and extent of these substitutions provide insight into the pathways activated by the different treatments.
In the literature, samples treated with HP have shown a decrease in carbonate content.7,11,33,38 The results from this study revealed significant differences in type A carbonate substitution (Rat 878 cm−1) among the treated groups. Specifically, type A carbonate content increased in G2 (HP) and decreased in G3 (ERS) and G4 (HP + ERS) compared to G1 (without treatment). This behavior may be attributed to the additives in 35% HP (Pola Office®), such as high concentrations of viscosity thickeners (73.26%), unspecified catalysts (26.2%), colorants, and a desensitizing agent. 39 These materials, when in contact with the oxidizing agent, generate carbon dioxide, which can be exchanged with hydroxyl groups at position A. Another contributing factor is the inverse correlation between carbonate content and tooth lightness, which may influence crystal size. 40 Increased carbonate content is also associated with greater solubility (in water or saliva) and reduced acid resistance.
ERS is not an oxidizing solid and does not contain organic components. Consequently, no carbonate is generated through the action of an oxidizer on carbon-containing matter, no increase in carbonate content is expected, and there is no anion in the solution capable of displacing the hydroxyl ion. The clinical goal is to achieve remineralization using apatite with the same natural composition found in conservative dentistry.
In G4 (HP + ERS), the treatment begins with the application of HP, followed by thorough rinsing and drying before using ERS. The resulting type A carbonate content in G4 is similar to that of G1 (without treatment) and G3 (ERS). This similarity may be explained by the two-step process applied in G4. In the first step, carbonate content increases, as observed in G2 (HP); however, in the second step, ERS—a remineralizing agent—deposits new layers of apatite formed in the absence of carbonates, thereby reducing the carbonate content at position A.
Likewise, the a1413 band (CO3²⁻ type B substitution at PO4
Instead, in supplying fresh phosphates and cations, G3 and G4 support the reconstitution of mineral content without compromising enamel integrity. In contrast, G2 provides neither calcium nor phosphates; thus, any newly formed apatite likely results from the dissolution of existing enamel apatite and its subsequent redeposition. This redeposition occurs with reduced type B carbonate substitution due to the acidic environment, indicating a process of demineralization.
In that sense, changes in chemical structure may be subject to debate. The presence of apatite both before and after treatment may be misleading. Although the material remained apatite in composition, changes in the calcium-to-phosphorus molar ratio (Ca/P) and alterations at the B-position site indicate that the resulting apatite had different chemical properties. These changes are only achievable through the recent formation of apatite from ions in aqueous solution. In other words, a newly structured apatite was formed from a previously existing, differently structured apatite. Lower carbonate content is then associated with increased resistance to corrosion.
Concerning phosphates, the crystalline apatite difference ratio Rat1095 (signal 1095/900–1200 cm⁻¹) was statistically significant (
Enamel crystallinity results from the combined contribution of its components: bioapatite (the crystalline ceramic phase) and hydrated proteins (in the amorphous phase). Phosphate-related enamel crystallinity may range from high to very low, depending on enamel depth. Reports in the literature regarding phosphate IR peaks are somewhat inconsistent. Some peaks are described as indicators of crystallinity, while others are associated with the “crystalline molecular environment”—a term that remains poorly defined. Other studies suggest that these peaks are not directly linked to crystal concentration.
Moving on to MinCO3²⁻, the carbonate type B substitution ratio (1405 cm⁻¹/mineral phosphate 900–1200 cm⁻¹) provided additional insight into changes in enamel composition.
In the G2 (HP) group, this proportion remained similar to that of G1 (control), as the peroxide primarily targets organic additives and proteins. The resulting carbonate was not incorporated at position B, since this would require access to internal crystal cell positions, which are already protected and inaccessible within the mature apatite structure. For such incorporation to occur, apatite must form at the site.
In the ERS treatment group (G3), the ratio decreased significantly due to the material’s high phosphate content and absence of carbonates. As a result, carbonates were not incorporated at position B. Moreover, the formation of new apatite increases the denominator of the ratio substantially, thereby reducing the overall value and indicating remineralization.
For G4 (HP + ERS), the results suggest a cumulative process in which the first step mirrors that of G2 (HP), while in the second step—ERS application—no carbonates are produced and, as a result, type B carbonate was not increased.
In turn, XRD analysis was used to obtain information on the crystalline structure of bioceramics.
In this study, all groups exhibited diffraction patterns characteristic of a polycrystalline material composed of hydroxyapatite (HAp) crystals. The width of the XRD peaks at half their height was inversely correlated with crystallinity—that is, the degree of perfection in the ordered state of the solid. An ideal, highly ordered crystallite would produce a vertical line with zero peak width. In contrast, greater structural disorder results in broader diffraction peaks. Traditionally, crystallite size is directly associated with crystalline perfection: the larger the crystallite, the higher the degree of order.
In this study, the broadening of the diffraction peak—calculated as the full width at half maximum (FWHM)—corresponding to the 002 reflection (i.e. the
In this case, the crystallite size was significantly larger in G1 (without treatment) compared to the treatment groups (G2–G4), indicating that all treatments reduced the size of apatite particles. Smaller particle size increases light scattering, shifting toward a whiter appearance due to Rayleigh and Mie scattering. In other words, all three experimental groups exhibited a bleaching effect.
Previous studies have shown that the less crystalline and more reactive mineral phase dissolves more rapidly upon acidic treatment of bioapatite (e.g. bones and teeth). Due to acid depletion (HP pH = 2.78), the surrounding fluid becomes supersaturated as it reacts with enamel, leading to the reprecipitation of freshly formed apatite with smaller, semicrystalline particles.
In contrast, the ERS treatment did not involve a low pH environment, instead it provided abundant calcium and phosphate ions, which induced the nucleation of new apatite crystals that grew into small-sized crystallites. These crystalline features closely relate to the compositional variations previously described in the ATR-FTIR analyses.
Finally, regarding clinical relevance, ERS demonstrated significant whitening potential by altering enamel color by reducing crystallite size. These changes—along with increased mineralization and decreased MinCO3²⁻ content—can be associated with improved mechanical properties, including microhardness, compressive strength, and tensile strength, while also reducing susceptibility to wear.
This potential was further supported by the reduction in crystallite size observed across all three treatment groups, which directly correlates with enamel age and indicates that the enamel becomes “rejuvenated.” However, this rejuvenation brings along both the benefits and potential drawbacks of younger enamel. For analogy, while a solid glass rod in one piece is structurally strong, breaking it into smaller fragments reduces the average particle size and compromises its mechanical properties. ERS may also serve as a protective agent against dental erosion. Another notable advantage is that it does not require a protective silicone barrier to prevent hydrogen peroxide-induced burns to the surrounding gingival tissue.
However, a significant drawback of ERS is its prolonged application time, underscoring the need for further research to optimize and accelerate the treatment process.
The limitations of this study include the sample size, the extended application time required for ERS, and, in the case of chemical composition analysis, the use of destructive techniques, which prevented comparison of enamel before and after treatment within the same specimen.
Conclusion
ERS demonstrated a superior color-changing effect compared to HP, as indicated by both CIELAB and CIEDE2000 results. The whitening effects induced by ERS were more perceptible and acceptable than those achieved with HP. ATR-FTIR analysis revealed that, in the ERS-treated group, certain phosphate bands remained unchanged relative to the control group. Additionally, carbonate substitutions at positions A and B were reduced, and the degree of mineralization increased compared to the HP-treated group. XRD analysis showed that all treatments affected enamel crystallite size, resulting in a moderate reduction in mineral crystallinity.
These findings suggest that effective tooth whitening can be achieved without the detrimental oxidative effects associated with hydrogen peroxide treatments.
Footnotes
Acknowledgements
The authors would like to thank the School of Dentistry and the Department of Chemistry at the Universidad Nacional de Colombia, Bogotá campus; the Department of Geology at the Universidad de Oviedo, Spain; Edgar Torres Durán of Fundación Universitaria CIEO, Bogotá, Colombia; and Oscar Emilio Pecho Yataco of the Integrated Regional University of Alto Uruguay and Misiones for their valuable contributions to this work.
Ethical considerations
This study was approved by the Ethics Committee of the School of Dentistry at the Universidad Nacional de Colombia, Bogotá campus, under approval number CIE-028-15.
Consent to participate
All donors received an informational booklet and signed informed consent documents.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Carolina Torres R. received support from the School of Dentistry at the Universidad Nacional de Colombia, Bogotá campus. Pedro Álvarez-Lloret received funding from the European Regional Development Fund (ERDF) under the Next Generation/EU program.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
