Abstract
Regeneration and reconstruction of segmental bone defects (SBD) is a clinical challenge in maxillofacial surgery and orthopedics. The present study evaluated efficacy of guided bone-regeneration (GBR) of rat femoral SBD using osteoconductive equine-bone (EB) and beta-tricalcium phosphate (beta-TCP) grafts, either with or without platelet-derived growth-factor (PDGF). Following ethical-approval, 50 male Wistar-Albino rats (aged ~12–15 months and weighing ~450–500 g) were included. A 5 mm femoral critical-size SBD was created and animals were divided into five groups depending on the graft material used for GBR (EB, EB + PDGF, Autograft, beta-TCP, beta-TCP + PDGF; n = 10/group). Following 12-weeks of healing, animals were sacrificed and femur specimens were analyzed through qualitative histology and quantitative histomorphometry. There was new bone bridging femoral SBD in all groups and qualitatively, better bone formation was seen in autograft and EB + PDGF groups. Histomorphometric bone-area (BA %) was significantly high in autograft group, followed by EB + PDGF, beta-TCP + PDGF, EB, and beta-TCP groups. Addition of PDGF to EB and beta-TCP during GBR resulted in significantly higher BA%. After 12-weeks of healing, EB + PDGF for GBR of rat femoral segmental defects resulted in new bone formation similar to that of autograft. Based on this study, GBR with EB and adjunct PDGF could be a potential clinical alternative for reconstruction and regeneration of segmental bone defects.
Introduction
Anatomic and functional reconstruction of post-traumatic and pathological segmental bone defects (SBD) is a major challenge in maxillofacial surgery, owing to its complex three-dimensional anatomy. 1 Despite availability of several bone substitutes, SBD reconstruction is constrained by the need for large volume, structural integrity, and adequate density of graft material. 2 In addition to the popularly used vascularized and non-vascularized block autografts, several methods of maxillofacial SBD reconstruction have been reported, including distraction osteogenesis and tissue engineering techniques.1,3 –5 Although the gold-standard bone autografts are osteoinductive, osteoconductive, and osteogenic, they are associated with significant donor site morbidity and are only scarcely available from specific anatomic sites. 6 In order to mitigate disadvantages of autografts and to fulfil clinical demands of osseous reconstruction, bone substitutes from human cadaveric (allografts), animal (xenografts), and synthetic (alloplasts) sources have been developed.7,8 However, these bone substitutes retain only the osteoconductive mineral microstructure, as they undergo deproteinization (allografts and xenografts) and sterilization to reduce risk of disease transmission.7,9
Majority of clinical cases reporting about particulate bone substitutes for maxillofacial bone grafting involve treatment of small and medium, periodontal, and dentoalveolar defects.4,6,10 –14 Although tissue engineering techniques such as use of osteoconductive scaffolds with or without adjunct factors or stem cells, and guided bone regeneration (GBR) have been reported for the treatment of maxillofacial bone defects, they are not used for SBD reconstruction.15 –18 GBR involves placement of particulate osteoconductive graft material within an osseous defect and covered by a resorbable or non-resorbable barrier membrane. 8 While GBR progresses through the same steps of bone healing (hematoma, granulation tissue, callus formation, and remodeling), the addition of mineralized scaffold surrounded by a barrier membrane enhances migration of cellular components and growth factors, prevents fibrous tissue invasion and promotes early bone formation and mineralization.19,20 Several clinical and pre-clinical studies have reported successful bone growth with GBR using different osteoconductive materials and resorbable collagen membrane (RCM).8,11,19 –25 Considering the above facts, it is indeed alluring to hypothesize that GBR could be used for reconstruction and regeneration of SBDs, which are otherwise treatable only with large quantities of autografts.5,18 GBR as a tissue engineering strategy could facilitate the use of a promising triad of osteoconductive scaffolds, stem cells, and growth factors for bone regeneration. As a result of which issues such as surgical morbidity and lengthy hospitalization, usually associated with segmental defect reconstruction, could be potentially eliminated.6,18
Studies have reported better bone regeneration with adjunctive use of bone morphogenic proteins (BMP), platelet rich plasma (PRP), platelet-derived growth factor (PDGF) and mesenchymal stem cells (MSCs).12,21,23,26 –34 A common denominator among them all is the presence of growth factors (GFs) such as BMPs belonging to transforming growth factor-β (TGF-β) superfamily and PDGF. 22 Synthesized by platelets, osteocytes, and monocyte-macrophage cells, PDGF promotes chemotaxis and proliferation of osteoblasts during bone healing.10,21 PDGF is also capable of enhancing bone regeneration in patients with compromised healing due to systemic comorbidities such as smoking, osteoporosis, and diabetes. 35 Recombinant human PDGF-BB isoform (rhPDGF-BB) is approved by U.S. Food and Drug Administration (FDA) for clinical dentoalveolar and peri-implant bone regeneration.12,13,31,35 Based on recent studies reported in the literature, PDGF is capable of augmenting periodontal soft and hard tissue regeneration, and extraction socket bone healing, in addition to being capable of enhancing MSCs to accelerate orofacial bone regeneration.16,17,36 Nevertheless, none of these studies report a role for PDGF in segmental bone defect regeneration in the maxillofacial region. Considering the above paucity in scientific evidence, it would indeed be novel to evaluate the efficacy of adjunct PDGF on GBR of maxillofacial SBD.
In order to test bone regeneration in SBD, standardized translational animal models with critical size segmental defects (CSSD) are imperative.9,37,38 Studies have confirmed the reliability of rat models for reporting qualitative and quantitative histological assessment of bone healing and regeneration, in both membranous (calvarium) and endochondral (femur) bone defects.8,23,25,33,34,37,39,40 Although the effectiveness of GBR for treating SBD and the adjunctive role of PDGF have not been studied extensively, based on an earlier in-vivo radiographic study, the present authors reported new bone formation in rat femoral SBD after 2–4 weeks of GBR with alloplastic bone substitute and PDGF. 41 This study envisaged assessment of long term bone healing after GBR of segmental defects along with adjunct PDGF. The objective of the present histomorphometric study was to evaluate GBR of rat femoral segmental defects grafted with equine bone and beta-tricalcium phosphate, either with or without PDGF, after 12-weeks of healing.
Materials and methods
Ethical approval, sample size, randomization, and grouping of study animals
After ethical approval from College of Dentistry Research Center, King Saud University, Riyadh, Saudi Arabia (CDRC approval numbers #PR 0041 and #PR 0043), fifty skeletally-mature male Wistar-Albino rats (aged ~12–15 months; weighing ~450–500 g) were included in the study. Animals were acclimatized and housed (4–5 animals per rodent cage) under veterinary supervision in a controlled laboratory environment (temperature 22°C–24°C ; humidity 45%–55%; 12-hourly light/dark cycles; ad libitum access to food and water). All procedures were performed in compliance with NIH guidelines for care and use of laboratory animals (NIH Publication #85-23 Rev.1985). 8 Following subcutaneous implantation of digital identifier micro-chip (Pet-ID Microchips Ltd., England), animals were randomly divided into one of five groups as described in Table 1.
Grouping of the study animals according to bone graft material used.

CSSD: critical size segmental defect; GBR: guided bone regeneration; PDGF: platelet derived growth factor; RCM: resorbable collagen membrane; TCP: tricalcium phosphate.
General anesthesia and surgical procedure
According to “ASTM Standard Guide for Preclinical in vivo Evaluation in Critical Sized Segmental Bone Defects (F2721-09),” a CSSD cannot heal without intervention and a 5 mm mid-diaphyseal femoral defect has been reported as a CSSD in rats.37,38 A similar defect was surgically created under general anesthesia (GA) in the right femur of all animals in this study. Following intraperitoneal injection of ketamine hydrochloride (60–100 mg/kg body weight) (HIKMA Pharmaceuticals, Amman, Jordan) and xylazine hydrochloride (5–16 mg/kg body weight) (Laboratorios Calier S.A., Barcelona, Spain), GA was ascertained by absence of corneal and righting reflexes. 42 The animal was positioned on its left lateral side and skin over right femur was shaved and disinfected. The femur was exposed through a skin incision and dissection through subcutaneous, fascial and muscular tissue. A 4-hole mini plate (1.0 mm thickness and 25 mm length) (KLS Martin, Tuttlingen, Germany) with graded span (7 mm) was fixed to the femoral diaphysis using 4 bicortical screws (1.5 mm diameter × 7 mm length) (KLS Martin, Tuttlingen, Germany), and a 5 mm CSSD was completed using a rotary cutting hand-piece under coolant irrigation. The CSSD was circumferentially covered with a RCM, filled with suitable biomaterial based on study group and secured in place by 6–0 resorbable sutures (Vicryl-Polyglactin 910; ETHICON Inc., Somerville, NJ, USA). The surgical wound was closed in layers using resorbable sutures. All animals were monitored for vital signs, hemorrhage and post-operative infection. Parenteral antibiotic (Amoxicillin hydrochloride 30 mg/kg body weight; Betamox, Norbrook Laboratories, UK) and analgesic (Metamizole 60 mg/kg body weight; Analgivet, Vemedim- Pharma, Cantho City, Vietnam) were administered once before surgery and after 6, 24, and 48 h (Figure 1).
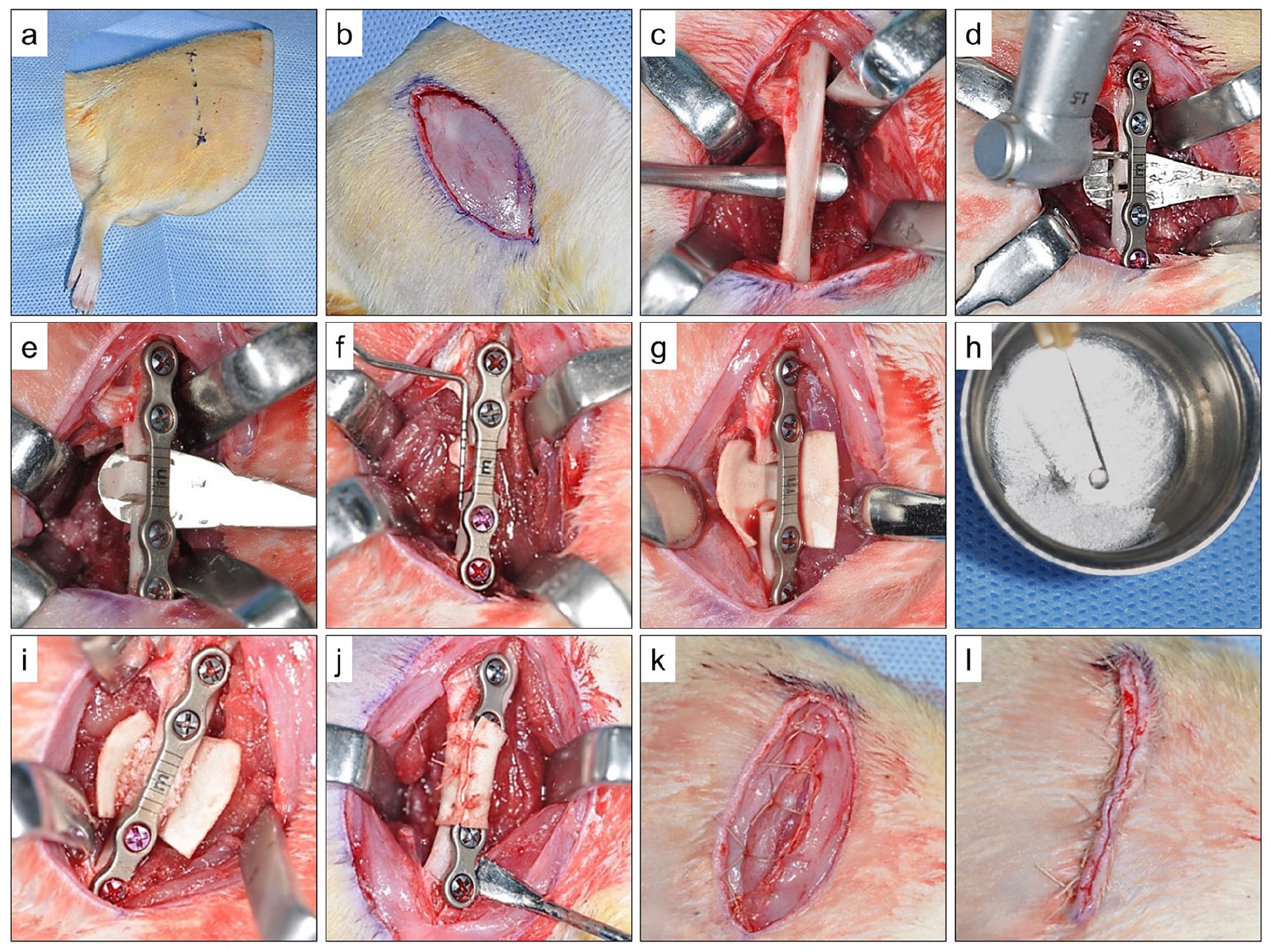
Representative images showing the surgical steps: (a) surgical site preparation and marking of skin incision, (b) completed skin incision, (c) exposure of the right femur bone after dissection of soft tissues, (d) Mini-plate fixation on anterolateral aspect of femur and segmental defect marking with aid of gradations on mini-plate span, (e) completed femoral segmental defect, (f) confirmation of segmental defect length (5 mm) with a measuring probe, (g) resorbable collagen membrane (RCM) placed around the defect, (h) soaking osteoconductive bone substitute material in PDGF (depending on study group), (i) placement of bone substitute material within the defect (depending on study group), (j) RCM sutured around the defect, (k) closure of sub-cutaneous tissue layers, and (l) Closure of skin wound. (Reproduced in part with permission from previously published studies of the authors: (1) Binsalah et al., 43 (2) Badwelan et al. 41 )
Study animal sacrifice and preparation for histology
After 12-weeks of healing, study animals were euthanized by 20% potassium chloride injection and right femoral bone was excised along with the regenerated CSSD. Following fixation in 10% neutral-buffered formalin for 72 h, mini-plates were removed from the femoral specimens and subjected to decalcification in EDTA (0.5 M; pH 7.5) for 3 weeks. Using an automated rotary microtome (Leica RM2255, Leica Biosystems Inc., Richmond, IL, USA), 4 µm thick histological sections were prepared from the decalcified, paraffin embedded specimens, at an orientation paralleling the long axis of femur and stained with hematoxylin and eosin (HE) and Masson’s Trichrome (MT). The HE and MT stained sections/slides were scanned in a “Digital Pathology Slide Scanner” (Aperio CS2, Leica Biosystems Inc., Richmond, IL, USA) and imaged using a “Pathology Slide Viewing Software” (Aperio ImageScope – ver. 12.3.2.8013, Leica Biosystems Inc., Richmond, IL, USA). Qualitative histological assessment focused on bone regeneration within CSSD in addition to presence or absence of residual bone graft/substitute, fibrous tissue ingrowth, inflammation, foreign tissue reaction and connective tissue bridging the defect. New bone within CSSD was characterized based on its unorganized lamellar pattern of woven bone, increased cellularity with irregularly arranged osteocytes, neovascularization and matrix staining intensely with hematoxylin than eosin. 8 Similarly, based on MT staining, new bone or osteoid was qualitatively identified by its bluish-green color and mature bone by red color. 15
Histomorphometric analysis
A rectangular region of interest (ROI), measuring 4500 pixels × 2500 pixels at 96 DPI was captured from the “Pathology Slide Viewing Software,” at 8× magnification and exported to an image processing software (GNU Image Manipulation Program – GIMP Ver. 2.10.8 Freeware). The ROI image was viewed using ImageJ Software (Image processing and analysis in java freeware – Ver. 1.4; National Institutes of Health, Bethesda, MD, USA) for histometric measurement of new bone area (BA%). Areas of new bone formation within the ROI were identified using “Threshold Color” control panel of the software, and selected areas were measured in pixels. The BA% for each histological section was calculated by dividing area of newly formed bone (in pixels) by total area of ROI (in pixels). Figure 2 describes in detail the steps in histometric assessment.
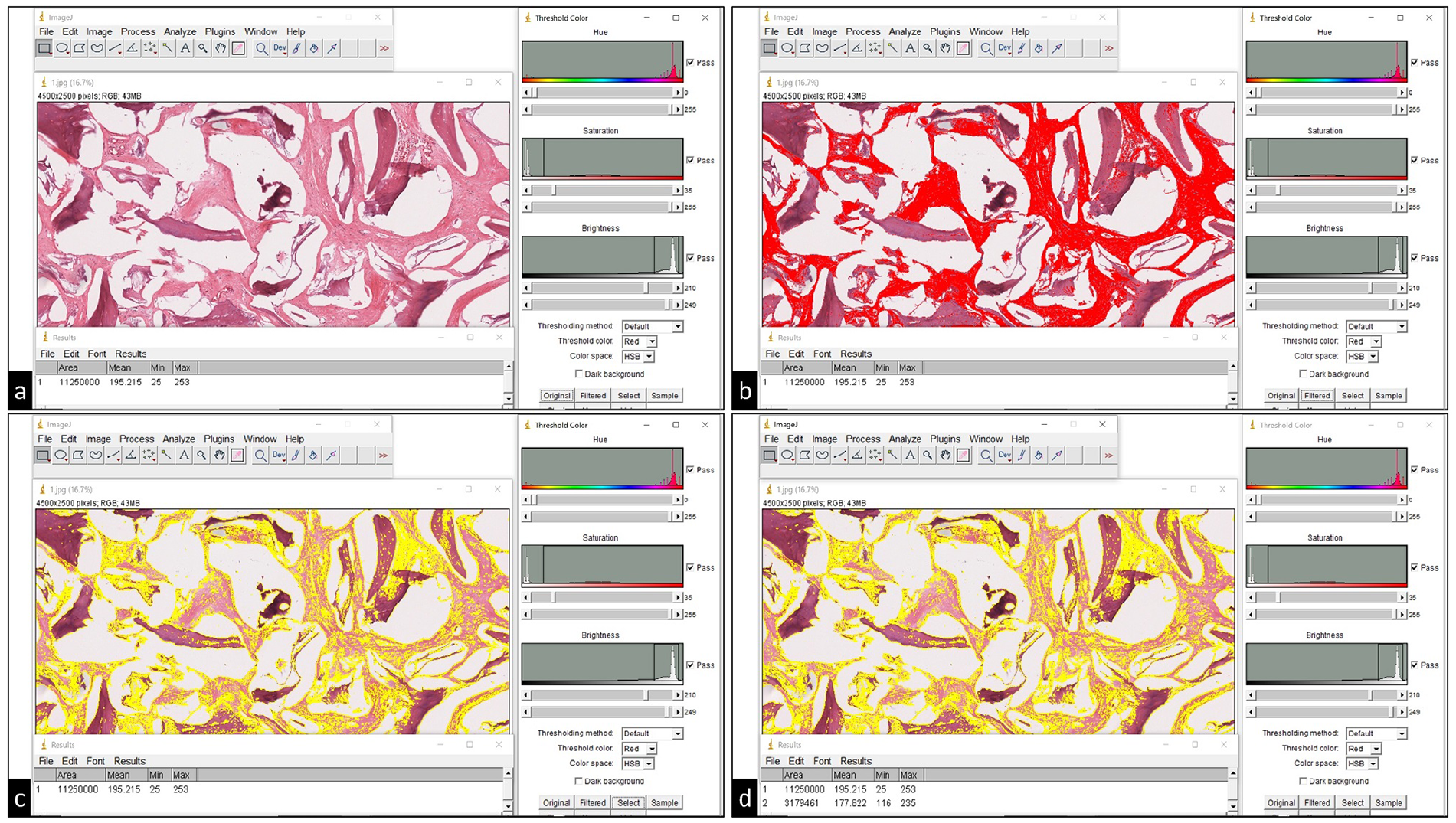
Representative images of screenshots obtained during histomorphometric analysis of bone area (BA %) within the region of interest (ROI): (a) ROI image (top left) is opened in ImageJ software and its area is measured in pixels (bottom left), (b) newly formed bone within the defect ROI is selected (red color) based on predefined ranges for hue (0–255), saturation (35–255) and brightness (210–249), using the “Threshold Color” control panel (right side of image), (c) Newly formed bone area within the ROI is digitally identified (yellow color) using “Select” button on the “Threshold Color” control panel, and (d) Total area of the ROI and area of newly formed bone are measured in pixels (bottom left) to calculate the BA%.
Statistical analysis
Descriptive statistics for BA% in each group was calculated based on cumulative data obtained from individual histological sections of each femoral specimen. Mean histometric BA% was compared between the groups using one-way ANOVA with Tukey HSD (honest significant difference) post-hoc test. All statistical analyses were performed using SPSS software (IBM SPSS Statistics Version 20, Armonk, NY, USA), assuming a 95% significance level (p < 0.05).
Results
All study animals recovered without any post-operative complications and remained healthy until they were sacrificed after 12-weeks of healing. There was no incidence of plate bending, fracture, exposure or surgical site infection.
Qualitative histological evaluation
Light microscopic evaluation of HE and MT stained sections revealed new bone formation bridging the defect in all study groups. Histological images at differing magnifications (original magnification ×2, ×10 and ×40) for the different study groups are described in Figures 3 and 4. Except for AB (autologous bone) group, grafted bone particles (equine bone and beta-TCP) were discernible in the histologic sections in all other groups. In the absence of adjunct PDGF, new bone formation in EB (equine bone) and beta-TCP groups was primarily observed close to the edges of the defects only. On the contrary new bone was observed even in the defect center in EB + PDGF and beta-TCP + PDGF groups. Under HE staining, new bone appeared intensely eosinophilic with a woven lamellar pattern, abundant osteocytes in the matrix and evidence of neovascularization. This was reaffirmed by the interspersed areas of bluish-green and red colored MT staining of immature osteoid and woven bone within the defect. While equine bone appeared similar to acellular bone matrix (eosinophilic in HE / dark red in MT), beta-TCP particles were mildly eosinophilic (HE) or light red (MT) and amorphous in appearance. Minimal fibrous tissue invasion, predominantly extending from the periosteal fibrous layer, was observed in central regions of the defect in EB and beta-TCP groups. Although remodeling of new bone, evidenced by an organized lamellar pattern, was not observed in any of the groups, early remodeling was observed in certain areas within the defect in AB group. There was no histological evidence of foreign body reaction or inflammatory infiltrate in any group (Figures 3 and 4).
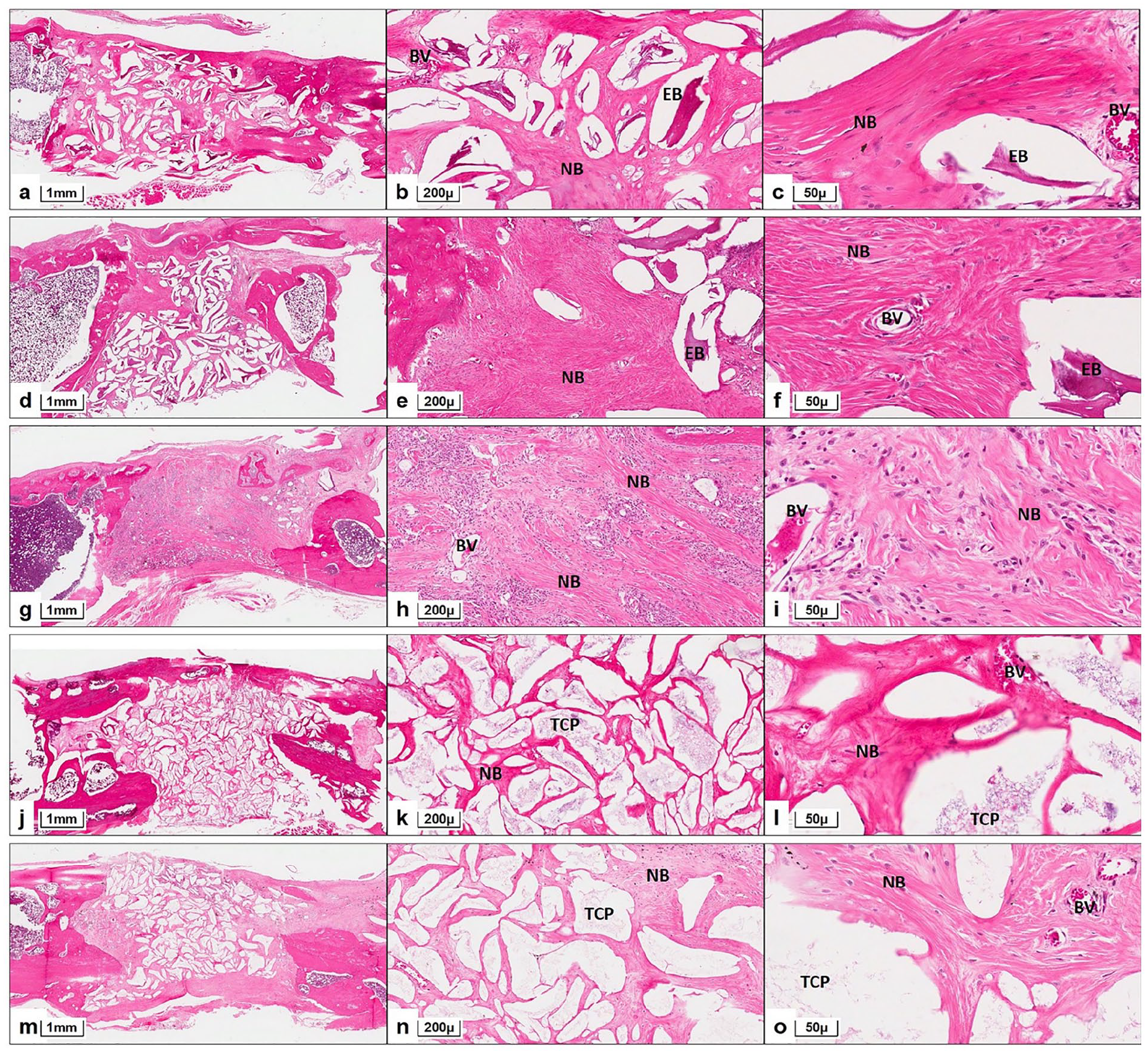
Representative histological images of rat femur segmental defect along with bone grafting in different study groups, after 12-weeks of healing. Equine bone (EB) group: (a) Defect is bridged by newly formed bone (NB) with interspersed EB particles (HE ×2), (b) NB in center of defect is evidenced by presence of irregularly arranged lamellae and osteocytes, along with blood vessels (BV) and is seen surrounding acellular EB particles. Fibrous tissue invasion within defect is minimal (HE ×10), (c) Woven lamellar pattern of NB with abundant osteocytes and acellular EB particles are seen clearly (HE ×40). EB + PDGF group: (d) NB with interspersed EB particles is bridging the defect and is greater near defect edges (HE ×2), (e) Dense NB with woven lamellar pattern, abundant osteocytes and neovascularization is seen close to defect edges. Acellular EB particles are surrounded by NB and fibrous tissue invasion is minimal (HE ×10), (f) Difference between NB with neovascularization (BV) and grafted EB are clearly observed (HE ×40). Autologous bone (AB) group: (g) Entire defect is bridged by a layer of NB with evident bone marrow spaces. Periosteal fibrous tissue layers are observed superior and inferior to bone bridging the defect. (HE ×2), (h) Center of defect shows several islands of NB with neovascularization (BV) and varying degrees of cellularity indicating progressive bone regeneration. (HE ×10), (i) NB shows areas of mineralization and organized lamellar pattern suggestive of early remodeling (HE ×40). Beta-TCP group: (j) NB interspersed with large quantities of TCP particles is seen filling entire defect. Periosteal fibrous tissue layer is seen superior to bone bridging the defect and minimal fibrous tissue is seen extending into middle of the defect. (HE ×2), (k) Continuous strands of NB, without any clear lamellar pattern or neovascularization, are seen around amorphous eosinophilic TCP (HE ×10), (l) Woven lamellar pattern with neovascularization (BV) is observed in NB surrounding TCP. Areas of TCP resorption are indicative of continuing new bone formation (HE ×40). Beta-TCP + PDGF group: (m) Thick bridge of NB with TCP particles is seen filling the defect and large amounts observed close to defect edges (HE ×2), (n) NB with abundant osteocytes and woven lamellar pattern is observed around scanty amorphous eosinophilic TCP particles. NB is observed as continuous strands surrounding TCP (HE ×10), (o) NB with neovascularization (BV) is observed and resorption of TCP is more pronounced as islands of remnant TCP are scanty (HE ×40).

Representative histological images of rat femur segmental defect along with bone grafting in different study groups, after 12-weeks of healing, stained with Masson’s Trichrome (MT). Equine bone (EB) group: (a) (MT ×2), (b) (MT ×10), (c) (MT ×40). EB + PDGF group: (d) (MT ×2), (e) (MT x10), (f) (MT x40). Autologous bone (AB) group: (g) (MT ×2), (h) (MT ×10), (i) (MT ×40). Beta-TCP group: (j) (MT ×2), (k) (MT ×10), (l) (MT ×40). Beta-TCP + PDGF group: (m) (MT ×2), (n) (MT ×10), (o) (MT ×40). The above histological images reaffirm the findings outlined in Figure 3. The low magnification images (×2) show new bone and residual bone graft bridging the defect, along with defect edges showing the native bone (dark red color). At higher magnifications (×10 and ×40) newly formed bone (NB) is seen interspersed with areas of immature osteoid (green color) and mature woven bone (red color). Residual equine bone particles (EB) are seen resembling acellular lamellar bone matrix (dark red color) and residual TCP particles are seen as amorphous mineral matrix. Blood vessels (BV) indicative of neovascularization during bone healing can be appreciated at highest magnification (×40).

Bar graph with standard deviation bars representing the mean values of histomorphometric bone area (%) within femoral segmental defects in different study groups after 12-weeks of healing. Statistical comparisons (p-value) are also shown.
Quantitative histomorphometric evaluation
Bone area (BA%) measured through histomorphometric evaluation, showed new bone formation in all groups after 12-weeks of healing. Greatest mean BA% was observed in AB group (62.012 ± 1.269) followed by EB + PDGF (40.837 ± 3.313), beta-TCP + PDGF (38.504 ± 2.294), EB (30.619 ± 1.691), and beta-TCP (28.536 ± 1.851) groups (Figure 5). One-way ANOVA revealed statistically significant differences in mean BA% between the different groups (F = 289.32, p < 0.001). Based on Tukey HSD post-hoc test, AB group had significantly higher mean BA% in comparison to all other groups (p < 0.01). Addition of adjunct PDGF to both EB and beta-TCP resulted in greater new bone formation evidenced by significantly greater mean BA% in EB + PDGF and beta-TCP + PDGF groups (p < 0.05). Interestingly, the difference in mean BA% between EB + PDGF and beta-TCP + PDGF groups were not statistically significant. Similarly, difference in mean BA% between EB and beta-TCP groups were not statistically significant.
Discussion
The earliest documented technique of managing maxillofacial segmental continuity defects is using reconstruction plates. 44 While maintaining anatomic continuity, they resulted in soft-tissue invasion into the defect and made dental rehabilitation difficult. Functional loading often lead to plate bending and fracture, loosening of screws and plate exposure with scarring and infection. Although reconstruction plates are still used clinically to stabilize segmental defects, grafting the defect helps avoid previously mentioned complications. 44 In the present study, femoral CSSD were reconstructed through GBR using different bone substitutes and mini-plate fixation mimicking reconstruction plate in clinical scenario. Bending or fracture of mini-plates was not observed in any of the study groups and histological findings revealed no soft tissue invasion within the defects, after 12-weeks of healing. On the contrary, plate bending has been reported in non-grafted rat femoral segmental defects, stabilized only by mini-plates. 43 The above evidences justify the role of mini-plate fixation and GBR for structural stability and unhindered bone formation within femoral CSSD.
Very few studies in the literature have evaluated bone regeneration and healing in rat femoral defects. While Brown et al. 26 and Harada et al., 34 respectively reported the use of BMP and MSC impregnated scaffolds for bone regeneration, Poser et al. 37 proposed a model for standardized CSSD creation and rigid immobilization using custom-fabricated PEEK plates. Based on femoral defects grafted with beta-TCP and hydroxyapatite (HA), Eftekhari et al. 39 reported better bone formation after 45 days of healing. Although similar findings were observed after 12-weeks with all bone substitutes used in the present study (equine bone and beta-TCP), the addition of PDGF resulted in significantly higher bone formation (Figures 3, 4 and 5). Particulate autologous bone used for GBR of femoral CSSD resulted in greatest histometric BA%, when it was used as a positive control in this study (Figures 3, 4 and 5). There was no issue of donor site morbidity, as bone removed during surgical creation of CSSD was used as autograft. While this is not clinically feasible during bone resection (due to pathology/trauma), specialized aspirators to harvest autograft during osseous surgery have been reported. 2 In addition to being proven as the best alternative, the current results also indicate a promising role for particulate autograft bone either alone or in combination with osteoconductive scaffolds for GBR of segmental defects, owing to its osteoinductivity. 2
Several clinical and experimental studies have reported the biocompatibility of equine bone and its ease of availability in large quantities, which were comparable to that of the more commonly used bovine bone.6,13,21 Moreover, equine bone has been shown to induce angiogenesis and osteoblastic differentiation, thereby enabling early new bone formation, sustained remodeling, and mineralization. 6 Addition of PDGF to equine bone in the present study, resulted in new bone with mineralization similar to that of autologous bone by 12-weeks (Figures 3 and 4). These findings are in coherence with a clinical study by De Angelis and Scivetti, 13 who used equine bone with PDGF for dentoalveolar ridge augmentation and observed large amounts of new bone with remodeling, after a period of 3 months. Furthermore, the qualitative and quantitative histological outcomes of GBR with equine bone and PDGF observed in this study, are similar to that of experimental studies which used xenograft with PDGF for bone regeneration, in different animal models.21,32
Alloplastic beta-TCP has been used an osteoconductive scaffold for regeneration of small to medium bone defects in orthopedic and dental surgery, and are capable of indirect osteoinductivity through entrapment of growth factors and osteoprogenitor cells.6,11,39,45 Since beta-TCP resorbs faster than osteoconductive allografts and xenografts, it enables early bone formation within the grafted site and mineralization through hastened release of calcium and phosphorus ions.8,46 The ability of beta-TCP to enhance GBR and peri-implant bone healing have been reported by several experimental studies.8,23,24,32,39,47 Using beta-TCP in rat femoral defects, Eftekhari et al. reported new bone formation within 4-weeks and significantly greater bone formation than with hydroxyapatite by 8-weeks. 39 While similar results were observed in the present study too, addition of PDGF to beta-TCP resulted in even better qualitative and quantitative bone formation, than with beta-TCP alone (Figures 3, 4 and 5). These findings are in accordance with the study of Choo et al., 31 who reported enhanced peri-implant bone formation with beta-TCP and PDGF.
Although GBR of femoral CSSD with equine bone resulted in greater new bone formation than with beta-TCP, the present study showed better overall quantitative and qualitative histological results with adjunct PDGF (Figures 3, 4 and 5). Healing bone tissue expresses BMP and PDGF for mesenchymal cell differentiation and proliferation, and the addition of these factors could potentially render osteoconductive graft materials as osteoinductive.6,22 PDGF is released from the alpha-granules of platelets in response to injury and is chemically characterized as a dimer derived from PDGF-A/B polypeptide chains.28,29 Interestingly, PRP which is considered to release PDGF through platelet lysis, availed no clinical benefit towards osseous healing and regeneration. 14 Consequently, a recombinant isoform of PDGF (rhPDGF-BB) was synthesized and found to be potent in exerting chemotactic and osteoproliferative actions through cell surface tyrosine kinase receptors. 22 While several studies have reported beneficial effects on bone healing when combining rhPDGF-BB with osteoconductive scaffolds,10,12,21,30 –33 the present study evaluated the adjunct role of rhPDGF-BB along with equine bone and beta-TCP for GBR of segmental defects (femoral CSSD), which has not been reported earlier. Although, BMPs and MSCs have been reported as adjuncts to GBR, they are associated with disadvantages, such as uncontrolled bone formation seen with BMPs and difficulty in cell culture and isolation of MSCs.23,26,27 In the absence of similar demerits as observed in this study, it is indeed alluring to assume PDGF as a feasible and ideal adjunct to GBR, even for segmental defect regeneration.
The outcomes of the present study were based on standardized methodology including animal model selection, uniformity of femoral CSSD, use of RCM for all GBR procedures and follow-up evaluation protocol.8,38 The present histomorphometric study is one among few that has comprehensively evaluated GBR with osteoconductive scaffolds and adjunct PDGF, specifically for femoral segmental defects in a translational animal model. A limitation of the present study was the use of only healthy animals as against those with comorbidities affecting bone healing, such as diabetes mellitus and osteoporosis. Similarly, absence of foreign body reaction to the grafted biomaterials was reported only on the basis of clinical parameters and absence of inflammatory cellular infiltrate, and not based on immunohistochemical analysis for macrophage activation. These limitations should be considered prior to clinical extrapolation of the present results and can be a potential avenue for future studies.
Conclusion
Based on the results of the present study, it could be concluded that PDGF used as an adjunct with osteoconductive bone substitutes for GBR of femoral segmental defects in a rat model, results in enhanced quantitative and qualitative new bone formation. Although the combination of equine bone with PDGF for GBR of femoral CSSD, after 12-weeks of healing, resulted in new bone which was quantitatively lesser than that of defects treated with autograft bone, it was qualitatively similar. Nevertheless, equine bone with PDGF resulted in the greatest amount of new bone formation than the other substitutes. In view of the associated morbidity and availability of autografts, GBR with PDGF and substitutes like equine bone could be an alternative to conventional reconstruction techniques for segmental defects. Similarly, the current findings indicate the need for further clinical research on tissue engineering of segmental bone defects through GBR.
Supplemental Material
sj-jpg-1-jbf-10.1177_2280800020987405 – Supplemental material for Guided bone regeneration with osteoconductive grafts and PDGF: A tissue engineering option for segmental bone defect reconstruction
Supplemental material, sj-jpg-1-jbf-10.1177_2280800020987405 for Guided bone regeneration with osteoconductive grafts and PDGF: A tissue engineering option for segmental bone defect reconstruction by Mohammed Alkindi, Sundar Ramalingam, Osama Alghamdi, Omar Mohamed Alomran, Mohammed Awadh Binsalah and Mohammed Badwelan in Journal of Applied Biomaterials & Functional Materials
Supplemental Material
sj-jpg-2-jbf-10.1177_2280800020987405 – Supplemental material for Guided bone regeneration with osteoconductive grafts and PDGF: A tissue engineering option for segmental bone defect reconstruction
Supplemental material, sj-jpg-2-jbf-10.1177_2280800020987405 for Guided bone regeneration with osteoconductive grafts and PDGF: A tissue engineering option for segmental bone defect reconstruction by Mohammed Alkindi, Sundar Ramalingam, Osama Alghamdi, Omar Mohamed Alomran, Mohammed Awadh Binsalah and Mohammed Badwelan in Journal of Applied Biomaterials & Functional Materials
Supplemental Material
sj-jpg-3-jbf-10.1177_2280800020987405 – Supplemental material for Guided bone regeneration with osteoconductive grafts and PDGF: A tissue engineering option for segmental bone defect reconstruction
Supplemental material, sj-jpg-3-jbf-10.1177_2280800020987405 for Guided bone regeneration with osteoconductive grafts and PDGF: A tissue engineering option for segmental bone defect reconstruction by Mohammed Alkindi, Sundar Ramalingam, Osama Alghamdi, Omar Mohamed Alomran, Mohammed Awadh Binsalah and Mohammed Badwelan in Journal of Applied Biomaterials & Functional Materials
Supplemental Material
sj-jpg-4-jbf-10.1177_2280800020987405 – Supplemental material for Guided bone regeneration with osteoconductive grafts and PDGF: A tissue engineering option for segmental bone defect reconstruction
Supplemental material, sj-jpg-4-jbf-10.1177_2280800020987405 for Guided bone regeneration with osteoconductive grafts and PDGF: A tissue engineering option for segmental bone defect reconstruction by Mohammed Alkindi, Sundar Ramalingam, Osama Alghamdi, Omar Mohamed Alomran, Mohammed Awadh Binsalah and Mohammed Badwelan in Journal of Applied Biomaterials & Functional Materials
Footnotes
Acknowledgements
The authors extend their appreciation to the Deanship of Scientific Research at King Saud University for funding this work through research group no (RG-1440-075).
Author’s contribution
MA – Conceptualization, formal analysis, funding acquisition, writing – reviewing and editing; SR – Conceptualization, formal analysis, investigation, resources, writing – original draft, writing – reviewing and editing; OA – Formal analysis, project administration, resources, writing – reviewing and editing; OMA – Data curation, investigation, writing – original draft, writing – reviewing and editing; MAB and MB – Data curation, investigation, writing – reviewing and editing.
The authors are grateful to Ms. Jeannette Gonzales-Quinsay, Lab Technologist, Oral Histopathology Laboratory, Dental University Hospital, King Saud University, College of Dentistry, for her assistance in preparation and processing of histopathology specimens.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Deanship of Scientific Research, King Saud University: Research group no. RG-1440-075.
Ethical approval
Ethical committee at the College of Dentistry Research Center (CDRC), King Saud University, Riyadh, Saudi Arabia (Approval numbers #PR 0041 and #PR 0043).
Supplemental material
The reviews of this paper are available via the supplemental material section.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
