Abstract
Background:
Polycaprolactone (PCL) is a highly recognized synthetic polymer for its biocompatibility, ease of fabrication and mechanical strength in bone tissue engineering. Its applications have extended broadly, including regeneration of oral and maxillofacial lost tissues. Its usefulness has brought attention of researchers to regenerate periodontal lost tissues, including alveolar bone, periodontal ligament and cementum. The aim of this systematic review was to obtain an updated analysis of the contribution of PCL-based scaffolds in the alveolar bone regeneration process.
Methods:
This review adheres to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines for systematic reviews. A computerized search of the PubMed, EBSCO, Scielo and Web of Science databases was performed, restricting literature search to published studies in English or Spanish between January 2002 and March 2023. Database search returned 248 studies which were screened based on title, author names and publication dates.
Results:
Data from 17 studies were reviewed and tabulated. All studies combined PCL with other biomaterials (such as Alginate, hydroxyapatite, bioactive glass, poly (lactic-co-glycolic acid)), growth factors (BMP-2, rhCEMP1), and/or mesenchymal stromal cells (adipose-derived, bone marrow, periodontal ligament or gingiva mesenchymal stromal cells). PCL scaffolds showed higher cell viability and osteoinductive potential when combined with bioactive agents. Complementary, its degradation rates were affected by the addition or exposure to specific substances, such as: Dopamine, Cerium Oxide, PLGA and hydrogen peroxide.
Conclusions:
PCL is an effective biomaterial for alveolar bone regeneration in periodontally affected teeth. It could be part of a new generation of biomaterials with improved regenerative potential.
Introduction
Periodontitis is a multi-factorial long-standing disease that affects the supporting tissues of the teeth. According to the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions it is classified by a staging and grading system. Its more severe forms (stages III/IV) are greatly prevalent, with an affected population range between 10.8% and 19% worldwide.
1
Periodontitis leads to alveolar bone loss, which is normally categorized in two patterns: horizontal and vertical. This last type corresponds to an irregular type of bone resorption that is known to be particularly harmful to the prognosis of the affected tooth.
2
Intrabony defects are a type of bone lesions of vertical or angular nature, where the alveolar crest adopts a coronal position with respect to the most apical part of the periodontal pocket.
2
This entity usually has different types of configurations depending on the amount of remaining bony walls, which will (among other factors) guide the clinical treatment of choice for its resolution.
2
It has been shown that in intrabony defects there is a tendency for deep probing depths to remain after non-surgical periodontal therapy and, therefore, these are sites with a propensity for progression of periodontitis and consequent tooth loss.
2
It has been reported that sites with residual probing depths of 6 mm or more have a 10 times greater risk of loss compared to sites with probing depths of 3 mm or less. Additionally, sites with grade II or III furcation compromise present an additional 5–13 times higher risk of suffering the same consequence compared to molars without furcation compromise.2–4 Matuliene et al. analyzed tooth survival and risk factors associated with tooth loss in 172 patients treated for periodontitis. The follow-up consisted of a mean time of 11.2 years. They described a much higher odds ratio for tooth loss when residual periodontal pocket depths (PPD) were 5 mm or higher, reaching an odds ratio of 37.9 and 64.2 at site and tooth level respectively when PPD was ⩾7 millimeters.
5
Therefore, bony defects with a vertical component are often considered to require surgical intervention beyond cause-related periodontal therapy, in order to decrease the probing depth, increase the attachment gain at the expense of minimal gingival recession and reduce the possibility of further periodontal breakdown. Today the surgical approach of preference considers minimally invasive accesses and adjunctive use of biomaterials for regenerative purposes.
5
Guided tissue regeneration (GTR) is a therapeutic principle and a surgical technique introduced in 1982 that came to revolutionize surgical periodontal treatment. Unlike other treatment strategies whose purpose is to achieve periodontal healing through the development of a long epithelial attachment, the goal of GTR is to regenerate periodontal ligament, cementum and alveolar bone
Methods
Protocol and registration
The development of this study was based on the PRISMA guidelines. The corresponding registration in PROSPERO, ID CRD42022339366.
Acceptability standards
Inclusion and exclusion standards
For the conduction of this research, we selected contemporary studies in which guided bone regeneration was accomplished with the use of scaffolds with a macrostructure made of PCL, either alone or in combination with other constituents, such as: stem cells, molecules and/or biopolymers. The inclusion standards were as follows:
Articles written in English or Spanish language.
Scaffold with a structure based upon PCL for regeneration of alveolar bone lost due to periodontitis.
Available information about biocompatibility, mineralization potential, Alkaline Phosphatase activity and osteogenic expression of genes Osteonectin, Osteopontin, Runx-2 and/or collagen type I.
Study design: randomized controlled studies.
Case reports, editor letters, reviews and studies without explicit ethical committee approval were excluded from our study. Reference period of research was between years 2002 and 2023.
Information sources
In order to identify the most up-to-date research regarding the application of PCL-based scaffolds in periodontal regeneration we used the following databases: PubMed, EBSCO, Scielo and Web of Science. No strings were set up in respect of the constituents added to the PCL scaffold. The following keywords were used to conduct our research: “Polycaprolactone scaffold” OR “PCL scaffold” AND “Alveolar bone regeneration”; “Polycaprolactone scaffold” AND “Periodontal regeneration”; “Polycaprolactone scaffold” AND “intrabony defect” OR “intraosseous defect”; “Polycaprolactone scaffold” AND “Periodontal disease”; “Polycaprolactone scaffold” AND “Guided tissue regeneration” OR “Periodontal regeneration” NOT “dental implant.” Time limit in each Database was set up to the year 2002.
Research selection and recording methods
Study election was assessed by two reviewers (F.A, F.T) separately. After screening by title, abstract and ultimately by full text reading, qualified articles were chosen following inclusion and exclusion requirements, imported and organized in a specific data portfolio in the software Zotero®. Three researchers (F.A, F.T, C.K) contributed in the data processing stage. The following information was retrieved from each study: Design of the study, in vitro and/or in vivo analysis and components that integrated the PCL scaffold. Three separate spreadsheets were design, in which data regarding: cell proliferation and viability assays, mineralization potential assays (described as Alizarin Red staining assay and Alkaline Phosphatase Activity) and RT-PCR for gene expression of osteogenic markers (Osteonectin (ON), Osteocalcin (OCN), Runx-2, Alkaline Phosphatase (ALP) and collagen type I (col-I)) from every included study was collected. The primary outcome was referred to these dimensions. The second outcome was referred to information available on degradation rates of the PCL-based scaffolds, both descriptive and quantitative data, among the selected studies.
The Flow chart used for the selection of the studies is shown in Figure 1.

Flow chart of selected studies (based on PRISMA Guidelines).
Risk of bias assessment
Sixteen studies were conducted in vitro, assessing the properties of the above-mentioned PCL scaffolds. Quality assessment was provided by the QUIN tool to assess the risk of bias of in vitro studies. 10 The QUIN tool includes 12 points with scoring and grading options. For each item, the lowest score was =0 (Not specified) and the highest score was =2 (Adequately Specified). After the evaluation, the lowest record was equal to 5, the highest record was equal to 15, and on average the quality of the articles was evaluated to be equal to 9.93 (Table 1). Overall, the osteo-inductive ability of PCL-based scaffolds to provide alveolar bone regeneration was analyzed with unequivocal methods. Operator details, randomization, outcome assessor details and blinding got, in general, the worst score among the selected studies.
Quality assessment of included studies based on The QUIN tool for assessing risk of bias of in vitro studies.

For the studies that were conducted in vivo, quality assessment was provided by the Newcastle Ottawa Scale. 11 The lowest record was 5, the highest was 9, and on average the quality of the articles was evaluated to be equal to 6.7 (Table 2). Definitions of control and comparability were, in general, the most poorly described.
Quality assessment of in vivo included studies based on Newcastle Ottawa scale.

: star assigned; -: star not assigned.
Results
Study selection and characteristics
The process of article selection is put on view in Figure 1. In the initial search, a total of 252 records were detected from the following databases: Pubmed, Web of Science, EBSCO and Scielo. A total of 99 records were recorded after removal of duplicates. Then records were screened by title and abstract and 32 studies were selected for full article evaluation. After this stage, 17 studies were finally selected for this systematic review. After assessing the quality of the selected studies using The QUIN tool for in vitro studies and Newcastle-Ottawa for studies with an in vivo experimental phase, they were submitted to data collection process. The 17 selected studies were randomized controlled studies with and in vitro and/or in vivo experimental design. In all studies PCL scaffolds were combined with other components, such as: Simvastatin, 18 Magnesium Phosphate, 26 Magnesium Oxide, 19 Cerium Oxide nanoparticles, 22 Strontium-doped nanohydroxyapatite, 14 Poly-lactic-co-glycolic-acid, 20 Jagged-1 protein, 12 zinc oxide, 25 adipose-derived mesenchymal stem cells, 15 bioglass and Copper-doped bioglass, 24 nanohydroxyapatite15,23,heparin-dopamine/heparin-dopamine and Bone Morphogenic Protein-2, 21 polydopamine, 16 calcium phosphate 27 or recombinant human cementum protein I. 13 One study evaluated osteogenic potential with two different manufacturing processes: coaxial electrospinning and single nozzle electrospinning. 19 Eleven studies were conducted in vivo. These studies used the following experimental animal design: 30 cranial defects in rats13,22,12 femur defects in New Zealand white rabbits, 23 70 periodontal defects in rats,16,17,25,26 subcutaneous implantation in rats18,27,10 periodontal defects in sheep 28 and 18 periodontal defects in mongrel dogs. 15
Biocompatibility
Cell proliferation and viability was evaluated in 16 from 17 studies. Four of them used the MTT (3-[4,5-dimethylthiazol-2-yl]-2,5 diphenyl tetrazolium bromide) assay with human periodontal ligament cells,12,13 mouse fibroblasts (cell line L929),
12
human bone osteosarcoma epithelial cells (U2O2)
14
and adipose-derived stem cells (ADSC).
15
Four studies evaluated cell viability with Live/Dead assay, using the following cell sources: mouse fibroblast cell line (L929)
12
and human periodontal ligament cells.16–18 Seven studies evaluated cell proliferation on scaffolds using Cell Counting Kit-8 on human periodontal ligament cells17–22 and rat bone marrow stromal cells (rBMSCs).
23
One study applied the MTS assay to evaluate viability on alveolar bone marrow-derived mesenchymal stem cells (aBMSCs).
24
Two studies used Alamar Blue colorimetric assay: one on human periodontal ligament stem cells
25
and the other on human bone marrow mesenchymal stem cells.
26
The studies evaluating biocompatibility with MTT assay showed no significant difference in cell viability between the groups, considering control as tissue culture plate (TCP). The study that compared PCL and PCL with hydroxyapatite (HA) showed a slight improvement on cell viability with PCL/HA scaffold, associated with increased roughness of its surface.
12
In the same study, cell viability of PCL and PCL/HA membranes were not altered when immobilized Jagged1 protein was added, showing that this osteogenic inducer does not alter cell viability.
12
A multilayered scaffold made of PCL demonstrated that the addition of Strontium (Sr) and nanohydroxyapatite (nHA) improved its cell viability.
14
The only statistically significant difference was at day three, when the absorbance values of PCL/20Sr-nHA scaffold was significantly higher (
Results of individual studies: MTT assay, Live/Dead assay, Cell counting Kit – 8 assay, Alamar blue assay and MTS assay.
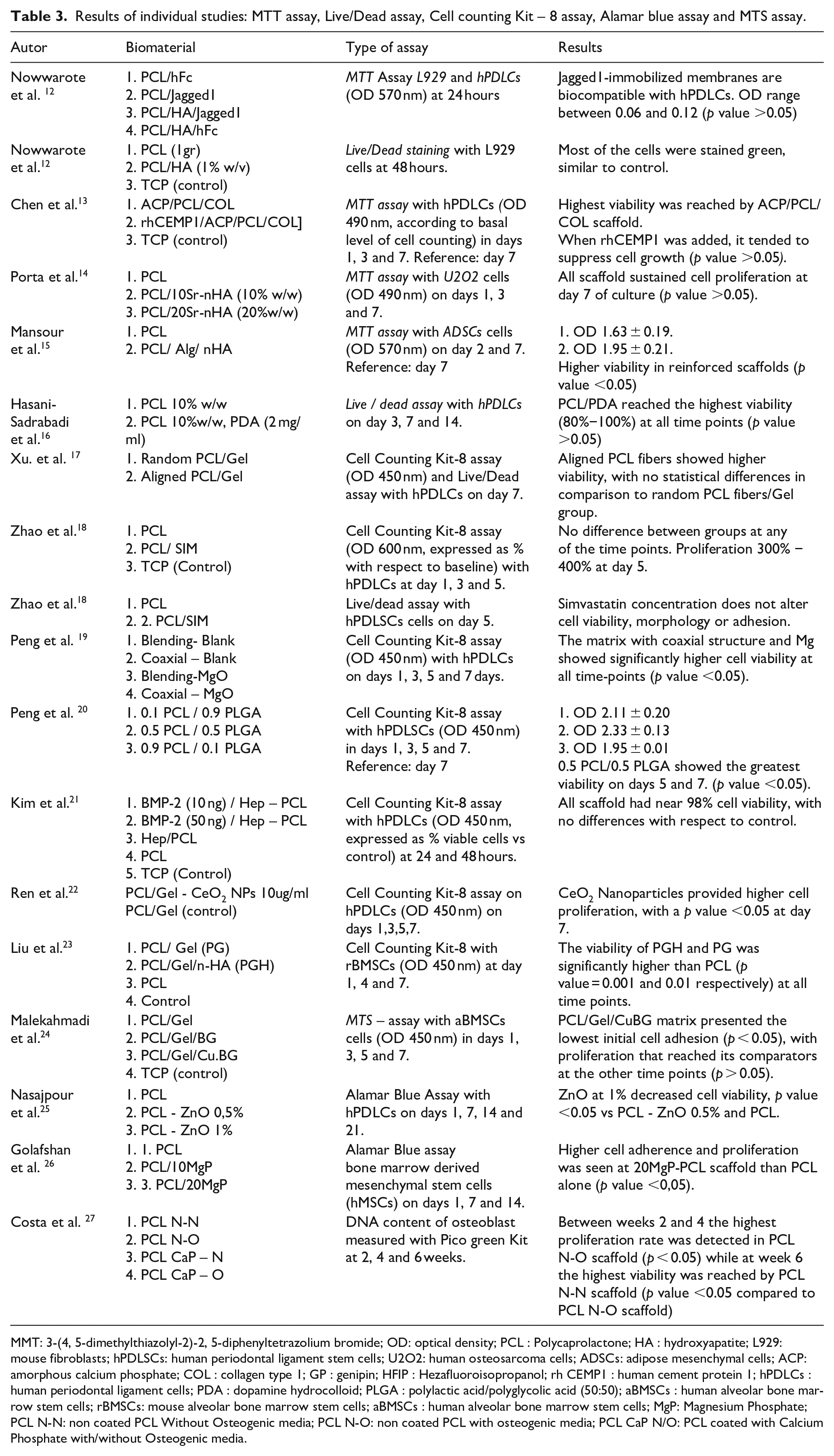
MMT: 3-(4, 5-dimethylthiazolyl-2)-2, 5-diphenyltetrazolium bromide; OD: optical density; PCL : Polycaprolactone; HA : hydroxyapatite; L929: mouse fibroblasts; hPDLSCs: human periodontal ligament stem cells; U2O2: human osteosarcoma cells; ADSCs: adipose mesenchymal cells; ACP: amorphous calcium phosphate; COL : collagen type 1; GP : genipin; HFIP : Hezafluoroisopropanol; rh CEMP1 : human cement protein 1; hPDLCs : human periodontal ligament cells; PDA : dopamine hydrocolloid; PLGA : polylactic acid/polyglycolic acid (50:50); aBMSCs : human alveolar bone marrow stem cells; rBMSCs: mouse alveolar bone marrow stem cells; aBMSCs : human alveolar bone marrow stem cells; MgP: Magnesium Phosphate; PCL N-N: non coated PCL Without Osteogenic media; PCL N-O: non coated PCL with osteogenic media; PCL CaP N/O: PCL coated with Calcium Phosphate with/without Osteogenic media.
Osteogenic potential. Calcium deposits formation and alkaline phosphatase activity
Osteoinductive potential of PCL scaffolds was analyzed in 12 studies. All of them performed either Alizarin Red assay, Alkaline Phosphatase activity assay or both. Only one article
25
evaluated the percentage of mineralized area/total well area in a PCL/Zinc Oxide scaffold using Xylenol Orange dye. The addition of Jagged-1 to a PCL/HA scaffold provided greater formation of mineralized deposits and higher Alkaline Phosphatase activity.
12
The incorporation of Cerium Oxide nanoparticles,
22
Simvastatin,
18
Magnesium Oxide,
19
Bone Morphogenetic Protein-2,
21
Magnesium Phosphate
26
and Calcium Phosphate
27
into the PCL scaffold showed improved osteoinductive capacities, expressed as higher absorbance values in calcium deposits formation and ALP activity. The study that included Bone Morphogenetic Protein-2 into the PCL scaffold (BMP-2/Hep-PCL) added nogging as a BMP-2 suppressor and proved that high values of ALP activity and calcium deposits were related to the BMP-2 released by the PCL scaffold itself and consequent osteogenic differentiation of periodontal ligament cells.
21
The incorporation of Zinc Oxide raised the levels of ALP activity in PDLCs but differences between experimental group and plain PCL did not reach statistical significance.
25
Regarding PCL scaffolds combined with other polymers without bioactive molecules, the study from Peng et al. showed that a 50:50 proportion of PCL/PLGA scaffold provided the best osteoinductive potential with statistical significance when compared with 0.1PCL/0.9PLGA and 0.9PCL/0.1PLGA scaffolds.
20
When analyzing a biphasic scaffold made by two different manufacturing techniques: PCL solution electrospinning for the periodontal compartment and PCL melt electrospinning for the bone compartment, the cell sources that generated the greatest number of mineralized deposits and higher ALP activity were periodontal ligament cells and bone marrow stem cells (with no statistical differences between them) followed by gingival cells (
Results of individual studies: Osteogenic potential in ALP activity assay, Alizarin Red S assay, calcium assay; Xylenol orange assay.

PCL: polycaprolactone; HA: hydroxyapatite; hPDLCs: human periodontal ligament cells; U2OS: human osteosarcoma cells; Sr-nHA: Sr-doped nano hydroxyapatite; ALP: alkaline phosphatase activity; PDL: periodontal ligament; PLGA: Poly(D,L-lactic-glycolic); PDLCs: periodontal ligament cells; hPDLCs: human periodontal ligament cells; BMP-2: bone morphogenic protein 2; SIM: simvastatin; GC: gingival cell; Bm-MSC: bone marrow-derived mesenchymal cells; MgP: Magnesium Phosphate.
Expression of osteogenic gene markers: Osteonectin (ON), osteocalcin (OCN), Runx-2, alkaline phosphatase (ALP) and collagen type I (Col-I)
A total of 11 studies analyzed the expression of osteogenic markers by real-time polymerase chain reaction. When PCL scaffolds where blended with bioactive molecules, such as: Polydopamine,
16
BMP-2,
21
nanohydroxyapatite,
23
Simvastatin,
18
Zinc Oxide
25
or Magnesium Oxide
19
the mRNA expression levels were considerably higher, reaching statistical differences. The addition of rhCEMP to a PCL scaffold combined with amorphous calcium phosphate and type I collagen abruptly downregulated the mRNA expression of OCN with a
Expression of osteogenic markers, quantified by Real Time Polymerase Chain Reaction (rt-qPCR).

PCL: Polycaprolactone; HES1: Hes Family BHLH Transcription Factor 1; HEY1: Hes Related Family BHLH Transcription Factor With YRPW Motif 1; OCN: Osteocalcin; OPN: Osteopontin; OG: Osteogenic; Runx-2: RUNX Family Transcription Factor 2; Col1: Collagen Type 1; BMP-2: Bone Morphogenetic Protein Type 2; BSP: Bone Sialoprotein.
In vivo osteogenic potential. Bone regeneration experiments in animal models. Micro-CT analysis, H&E staining and Masson staining
From all the included studies, 11 reported outcomes on in vivo experimental models.
One study 15 evaluated the regenerative potential of a PCL scaffold enhanced with hydroxyapatite in class II furcation defects in mongrel dogs. Seven studies16–18,22,25–27 analyzed the osteoinductive effect of PCL scaffolds on rat models. Four of them created periodontal defects in rats.16,17,25,26 Another two PCL-based scaffolds were subcutaneously implanted.18,27 Two study groups inserted the scaffold in a rat calvaria defect.19,22 One study 28 evaluated the regenerative effect of a biphasic scaffold made from a combination of melt and solution PCL electrospinning in periodontal defects in sheep. PCL based scaffold with 0.5% concentration of Zinc Oxide were inserted in the femur of 12 rabbits. 23 The time of the included studies ranged between 4 and 10 weeks, with 8 weeks being the most frequent time point of analysis. All studies reported higher bone formation in experimental groups in comparison with empty controls. When bioactive molecules were added, such as: Polydopamine, 16 n-hydroxyapatite/Heparin coating 23 or Cerium Oxide nanoparticles 22 and Simvastatin 18 the osteogenic potential was enhanced. In the study from Vaquette et al. the addition of cell sheets did not provide more bone formation compared to PCL scaffold alone. 28 On the contrary, a study that created class II furcation defects in 18 dogs showed a higher bone formation rate when PCL/Alginate/nano-hydroxyapatite scaffolds were pre-seeded with adipose derived stem cells and then implanted into the periodontal defect. 15 A summary of all included in vivo studies can be seen in Table 6.
Results of individual in vivo studies: Micro-CT analysis, H&E and Masson trichrome staining.

PCL: polycaprolactone; nHAP: nano hydroxyapatite; Alg: alginate; ADSCS: adipose mesenchymal cells; H&E: hematoxylin-eosin; PDA: dopamine hydrocolloid; PGH: PCL/Gel/n-HA; HB: Hybrid bi-layer; CEJ: amelocementary line; PG: PCL/Gel; SIM: simvastatin; GC: gingival cell; Bm-MSC: bone marrow-derived mesenchymal cells; PDLCs: periodontal ligament cells; OBs: osteoblasts; CS: Cell Sheets; BM: Basal Medium; OM: Osteogenic Medium; TG-GEL: Transglutaminase/Hydrogel.
Degradative properties of PCL scaffolds
Only four studies16,17,20,22 reported outcomes on PCL scaffold degradative properties. The inclusion of bioactive molecules, such as Dopamine, tended to increase the degradation rate. Additionally, the exposure of PCL/PDA scaffolds to PBS (Phosphate-buffer saline) and hydrogen peroxide up to 50 days accelerated its degradation by 20% and 40% respectively. 30 The presence of PLGA significantly accelerated mass loss of the PCL/PLGA scaffolds. Complementary, the addition of this polymer tended to decrease the pH and in consequence speed up degradation. 20 The addition of Cerium Oxide nanoparticles to a PCL/Gel membrane promoted the loss of mass. This could suggest that this component increases the degradation rate of the PCL membrane. 22 According to the study from Xu. et al, the arrangement of PCL fibers tends to increase mass loss when they are aligned (degradation of 21.4 ± 3.3% the first day vs 16.5 ± 5.5% with random fibers) although difference did not reach statistical significance. 17
Discussion
Periodontal tissues get damaged by the establishment of a chronic immunoinflammatory disease named periodontitis.
29
In its severe forms, periodontitis will cause destruction of the periodontal ligament attachment and alveolar bone which, if left untreated, will ultimately lead to tooth loss.
2
To prevent this ultimate catastrophic scenery from happening, different regenerative strategies have been developed that include the use of biomaterials to guide tissue regeneration (periodontal ligament, cementum and bone)
Conclusions
Our systematic review confirms the validity and potential of PCL as a biomaterial for alveolar bone regeneration in periodontally affected teeth. It is worth mentioning that although several of the articles included in this review included data regarding periodontal ligament and cementum regeneration, the focus of this review was on the potential of PCL to regenerate the bony compartment of periodontal tissues affected by periodontitis. Considering all the analyzed data, the combination of PCL with more hydrophilic polymers and/or molecules renders greater cell adherence and proliferation, improves its degradation rates and provides higher osteogenic potential both at in vivo and in vitro experimental settings. Even with the variety of published articles related to the design of PCL-based scaffolds for guided bone regeneration, there are no reports of clinical applications to date. Given the proven versatility and efficiency of PCL stated in this review, we encourage research groups to move forward with the validation of this biomaterial in clinical settings.
Footnotes
Acknowledgements
We would like to thank Valentina Veloso (MSc, PhD) for her assistance and guidance in this research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Contributorship
Conceptualization, C.K.; methodology, validation, investigation, F.A/F. T; risk of bias analysis R.O; writing – original preliminary form preparation, C.K; writing—review and editing, C.K, R.O.
Guarantor
CK.
