Abstract
The goal of this study is to understand the ability of a newly developed barrier membrane to enhance bone tissue regeneration. Here in this study we present the in vitro characterization of the barrier membrane made from type I collagen and crosslinked by oligomeric proanthocyanidins (OPCs). The effects of the membrane (P-C film) on cell cycle, proliferation, alkaline phosphatase activity, and mineralization were evaluated using the human osteoblast cell line MG-63, while the barrier ability was examined using MG-63 cells, as well as the human skin fibroblast cell line WS-1. The pore size is one of the factors that plays a key role in tissue regeneration, therefore, we evaluated the pore size of the membrane using a capillary flow porometer. Our results showed that the mean pore size of the P-C film was approximately 7–9 µm, the size known to inhibit cell migration across the membrane. The P-C film also demonstrated excellent cell viability and good biocompatibility, since the cell number increased with time, with MG-63 cells proliferating faster on the P-C film than in the cell culture flask. Furthermore, the P-C film promoted osteoblast differentiation, resulting in higher alkaline phosphatase activity and mineralization. Therefore, our results suggest that this P-C film has a great potential to be used in guided bone regeneration during periodontal regeneration and bone tissue engineering.
Introduction
In a century characterized by a large aging population, periodontal diseases are more frequently encountered. The global burden of healthcare directly attributed to oral diseases, such as periodontal diseases, caries, and tooth loss, is estimated at US$298 billion annually. 1 Following the removal of microbial plaque and calculus, guided bone regeneration (GBR) plays an important role in rebuilding the alveolar ridge for dental implant placement. GBR is also commonly used after tooth extractions, since crestal bone resorption is unavoidable. 2 GBR is a surgical technique that involves the use of barrier membranes to maintain a space over the bone defect. The purpose of barrier membranes, either resorbable or non-resorbable, is to block the ingrowth of soft tissues and provide a time window for the regeneration of hard tissues. 3
GBR was first described in a dog model by Nyman and Karring. 4 Several follow up studies by his group and others introduced the idea of using polytetrafluoroethylene (PTFE) membranes.5–8 PTFE is mechanically rigid, making it suitable for maintaining a space for GBR 9 ; however, it is non-resorbable and requires surgical removal. Titanium mesh is another popular non-resorbable material utilized in GBR with good space retention, especially for large defects,10,11 though its large porosity causes fibrous ingrowth, making it less favorable. 12 For these reasons, resorbable membranes composed of aliphatic polyesters have been investigated.13,14 Various resorbable membranes made with polyesters are currently commercially available and are completely degradable within 4–48 weeks 15 ; however, these membranes have been reported to elicit inflammatory responses at implant sites.16,17
Collagen-based membranes have received special attention, since collagen is a naturally-occurring and the most abundant protein in the body. Collagen membranes are resorbed in the oral cavity by collagenases, enzymes produced by bacteria, macrophages, and polymorphonuclear leukocytes18,19; however, the fast degradation rate of natural collagen makes it a potential drawback. Meanwhile, crosslinking can delay collagen resorption in vivo, therefore, crosslinked collagen membranes have been widely explored. For example, non-crosslinked collagen membranes have been reported to degrade faster than glutaraldehyde- and ribose-crosslinked collagen membranes, as measured on day 28 in a rat calvarial defect model. 20 Furthermore, the ideal membrane should degrade at the same rate as the regeneration of new tissues.
Oligomeric proanthocyanidins (OPCs) are a class of polyphenols found in plants. OPCs are primarily known for their antioxidant activity and provide protection through the direct scavenging of free radicals.21–23 OPCs form strong hydrogen bonds with soluble collagen, converting it into a non-degradable form. 24 Currently, there are numerous crosslinking agents, such as formaldehyde, glutaraldehyde, hexamethylenediiscyanate, and diphenylphosphoryl azide, which are commonly used to produce commercial non-resorbable collagen membranes. 25 Glutaraldehyde is the most widely used crosslinking agent of collagen for biomedical applications26,27; however, it is also cytotoxic and exposure to glutaraldehyde leads to apoptosis. 28 On the other hand, OPCs are non-toxic and are reported to have antibacterial, antiviral, anti-inflammatory, and anticarcinogenic properties, making them good candidates as alternative crosslinking agents.29–32
Here in this study, we utilized collagen and OPCs, extracted from grape seed extract and used as a natural cross-linker, to develop a barrier membrane suitable for stimulated bone regeneration. The in vitro properties, such as surface morphology, pore size, and biocompatibility, of the OPC crosslinked collagen membranes were evaluated. Fibroblast and osteoblast cell lines were chosen to study the biocompatibility and cell migration across this newly developed barrier membrane. Finally, cell proliferation and osteoblast differentiation were assessed to demonstrate its suitability for use in orthopedic applications.
Materials and methods
Materials
Type I collagen from bovine skin, molecular weight around 300,000, was obtained from Devro Pty Limited (Bathurst, Australia). OPCs extracted from grape seeds were purchased from Gino Biotechnology Ltd (Taipei, Taiwan). Cell culture supplies, such as minimum essential medium (MEM), fetal bovine serum (FBS), penicillin-streptomycin-amphotericin B, L-glutamine, sodium pyruvate, and non-essential amino acids, were purchased from Gibco (Thermo Fisher, Waltham, MA). All other analytical grade chemicals used in this study were purchased from Sigma Aldrich (St. Louis, MO) or J.T. Baker (Phillipsburg, NJ, USA) unless specified otherwise.
Preparation of collagen membrane
The bone regeneration membrane was prepared from type I collagen crosslinked by OPCs. Briefly, collagen (1 g) was mixed into 25 mL of phosphate buffered saline (PBS) and stirred for 4 h at 4°C. Next, isopropanol (1 g) was mixed with 25 mL of PBS and added to a well-mixed collagen solution (final concentration of collagen solution was 20 mg/mL). According to our previous study, the optimized condition to prepare the OPCs crosslinked collagen membrane has been investigated and applied. 33 Collagen/OPC solution was prepared by mixing 4.5 mL 20 mg/mL collagen solution with 0.5 mL 10% OPC solution (in 5% citric acid); the final concentration of OPCs in the mixture was 1%. Collagen/OPC solution was poured into a 4 cm diameter mold and dried overnight in a 37°C incubator. Next, the membrane (P-C film) was further washed with deionized water and dried before further use. For in vitro studies, the P-C film was sterilized using 75% ethanol and 30 min of UV light.
Pore size measurement
The pore size of the P-C film was measured using a nitrogen gas adsorption technique. Briefly, 3 × 3 cm2 P-C film was soaked in a Gal Wick solution (with a surface tension of 15.9 dyn/cm). The P-C film, measured under wet and dry conditions, was placed on the sample holder of a capillary flow porometer (CFP-1100AX; Porous Materials Inc., Ithaca, NY). The pore size was determined using nitrogen gas (gas flow of 0–5 psi). The pore size distribution was calculated based on the Brunauer-Emmett-Teller (BET) theory.
Cell culture
Human osteosarcoma (MG-63) and fibroblast (WS-1) cell lines were purchased from the American Type Culture Collection (ATCC, Manassas, VA). Cells were cultured in MEM containing 10% FBS, 100 IU/mL penicillin, and 100 mg/mL streptomycin in a humidified incubator at 37°C and 5% CO2.
Membrane cell migration
The cell permeability of the P-C film was examined using a 13 mm Swinnex Filter Holder (Merck Millipore, Darmstadt, Germany). The P-C film was placed into the filter holder and seeded with 5 × 104 cell/cm2 of MG-63 or WS-1 cells. Cells were cultured for 1, 3, and 7 days and stained with Hoechst 33258. Stained P-C films were imaged using a fluorescent microscope (50i, Nikon, Japan).
Biocompatibility
Biocompatibility of P-C films was evaluated using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) assay (Sigma, USA). Briefly, the P-C film was placed into the wells of a 24 well-plate and seeded with 5 × 104 cells/cm2 cells in 200 µL MEM for 10 min to allow the cells attach to the film. The medium was later added to bring the total volume to 1 mL and cells were cultured for 1, 2, and 3 days. The MMT assay was performed according to the manufacturer’s instructions. The absorbance at 570 nm, with the reference absorbance at 630 nm, was determined using a microplate reader (Sunrise remote F039300, Tecan, Austria). Cells seeded in the wells without the film (a standard culture plate surface) were used as controls. The optical density (OD) was normalized to the control on day 1 and presented as relative cell viability.
Cell cycle analysis
To study the cytocompatibility of the P-C film, the cell cycle progression of MG-63 cells was determined. Briefly, MG-63 cells were cultured on the P-C film (5 × 104 cell/cm2, 24 well-plates) for 1, 2, and 3 days; cells plated in the wells without the film were used as controls. Next, the cells were harvested, fixed in ice-cold 70% ethanol for 15 min, centrifuged at 500 RCF, resuspended in PBS, and stained with 0.5 mL 50 µg/mL propidium iodide in PBS (containing 100 mg/mL RNase and 0.05% Triton X-100) for 40 min at 37°C in the dark. Cell cycle analysis was performed using a flow cytometer (BD FACS Calibur™ Flow Cytometer; Beckman Coulter, Brea, CA, USA).
Cell morphology
The morphology of MG-63 cells cultured on the P-C film was investigated using scanning electron microscopy (SEM). Briefly, cells cultured on the P-C film for 1, 3, and 7 days, were fixed with 2.5% glutaraldehyde in cacodylate buffer (0.1 M sodium cacodylate-HCl pH 7.4), the surfaces were coated with a 100 Å layer of Ag-Pt, and samples were visualized by SEM (S-3000H, Hitachi, Tokyo, Japan).
Cell proliferation
The proliferation of MG-63 cells cultured on P-C films was evaluated by DNA quantification. Cells plated on P-C films in 24-well plates at a density of 5 × 104 cell/cm2 were cultured for 1, 3, 7, and 14 days. Next, cells were harvested and DNA was extracted using a DNA extraction kit (Cell DNA Mini Kit; Geneaid, Taiwan). DNA concentration was measured using a UV-Vis spectrophotometer (SP8001; Metertech, Taiwan) at a wavelength of 260 nm.
ALP activity and mineralization
Osteogenic differentiation of MG-63 cells was evaluated using the alkaline phosphatase (ALP) activity assay and mineralization. Cells were seeded at a density of 5 × 104 cell/cm2 on P-C films in 24 well-plates with regular medium. On days 1, 3, 7, and 14, MG-63 cells were collected, lysed using 0.1% Triton X-100, and ultrasonicated for 5 min. ALP activity was detected using the hydrolysis of 4-nitrophenyl phosphate into 4-nitrophenol method (the amount of 4-nitrophenol is directly proportional to the ALP activity). The formation of 4-nitrophenol was measured at 405 nm absorbance using a microplate reader (Tecan, Sunrise remote F039300, Männedorf, Switzerland). Mineralization (calcium precipitation) was detected using the von Kossa staining. This method is based on silver ions in the stain replacing calcium ions and precipitating after the reaction. Cells were cultured as described above, fixed on days 7 and 14 using 10% formaldehyde for 30 min, and then rinsed twice with deionized water (DI water). Next, cells were stained with 5% AgNO3, exposed to UV light for 1 h, washed with deionized water, and then incubated with 5% Na2SO4 for 2 min. Finally, cells were washed with deionized 2–3 times and imaged using an optical microscope (Nikon, Ti-U, Japan).
Statistical analysis
The Student’s t-test was used to calculate statistical significance. All data are expressed as the mean ± standard deviation (n = 5). A p-value <0.05 was considered as statistically significant.
Results
Surface morphology and pore size measurement
First, we characterized the surface morphology of the P-C film and its pore size. The microstructure was evaluated by SEM (Figure 1(a) and (b)), and the macrostructure of the film was visualized using an optical microscope (Figure 1(c)). We observed that the film was dried evenly, maintaining the circular shape of a Petri dish. SEM showed that collagen fibers were arranged in networks, with pores visible between the fibers. The pore size of the P-C film was measured using a capillary flow porometer. Our results demonstrated that the pore size of the film ranged from 3.3 to 19.6 µm, with the average diameter of 7–9 µm (Figure 1(d)).

Characterization of P-C film surface morphology: (a) SEM at 500× magnification, (b) 1000× magnification, (c) optical microscope, and (d) the pore size distribution of the P-C film.
Cell migration
The purpose of the P-C film is to be used as a barrier membrane for guided bone regeneration, therefore, next, we examined its cell permeability using MG-63 and WS1 cells. For that purpose, the P-C film was placed in a Swinnex filter holder, cells were seeded on top, and cultured for 1, 3, and 7 days (MG-63 cells – Figure 2, WS-1 cells – Figure 3). At the end of the specified incubation period, the film was stained with Hoechst 33258 to observe cells on both sides of the film. On day 1, MG-63 cells were detected on the top side of the film; WS-1 cells were also observed. On days 3 and 7, more stained cells from both cell lines were present on the top side of the film; however, no cells were observed on the bottom side of the film at any time point, and in both MG-63 and WS-1 cell lines. The well spreading cellular morphologies showed that the prepared materials were well biocompatible. It has been reported the morphological behavior of cells correlated to the biocompatibility of material.34,35
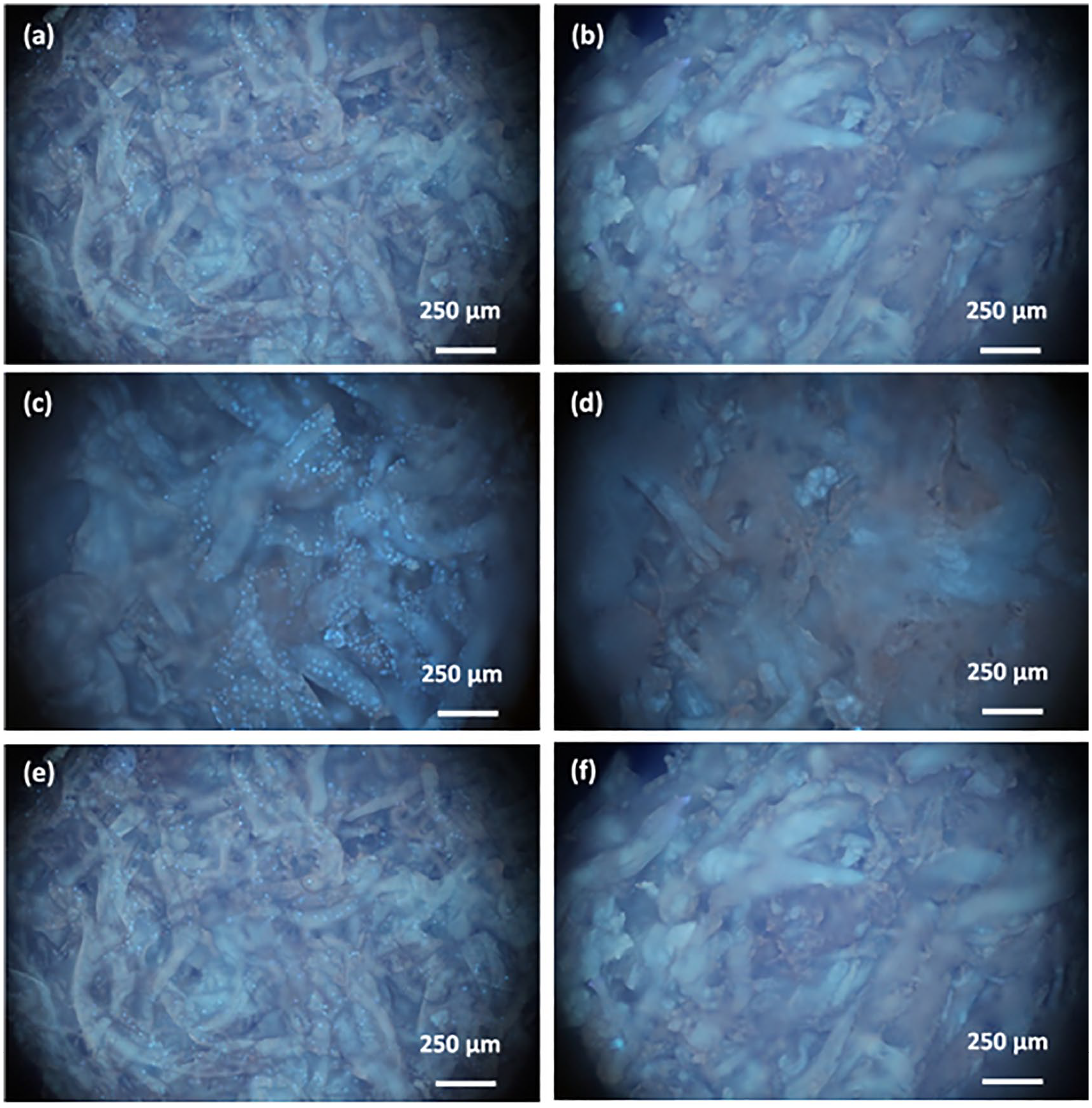
Evaluation of cell migration. Fluorescent microscopy images of MG-63 cells seeded on the P-C film for 1 (a, b), 3 (c, d), and 7 (e, f) days. Images of the top (left column) and the bottom (right column) of the film.

Evaluation of cell migration. Fluorescent microscopy images of WS-1 cells seeded on the P-C film for 1 (a, b), 3 (c, d), and 7 (e, f) days. Images of the top (left column) and the bottom (right column) of the film.
Biocompatibility
To assess viability, MG-63 cells were directly seeded onto the surface of the P-C film, incubated for 1–3 days, and then evaluated using the MTT assay (Figure 4). The cell viability of MG-63 cells on P-C film was higher on day 1 compared to the control; however, there were no statistically significant differences between the groups on days 2 and 3.

The cell viability of MG-63 cells seeded on the P-C film was measured using the MTT assay. Cells cultured in the wells without the film were used as controls.
Cell cycle analysis
To evaluate the cell cycle distribution, MG-63 cells were seeded on the P-C film for 1, 2, and 3 days, and then analyzed using flow cytometry. Figure 5 shows the cell cycle distribution and the summary of cell cycle phases. No apparent sub-G1 phase was detected at any time point. The distribution of G0/G1, S, and G2/M phases of cells seeded on the P-C film for 2 and 3 days was not significantly different from the control. At the same time, a significantly higher percentage of cells was found at the S and G2/M phases in the P-C film on day 1 compared to the control.

The effect of P-C film on MG-63 cell cycle distribution in vitro. Cell cycle analysis was performed on cells cultured on the P-C film for 1, 2, and 3 days; cells cultured in the wells without the film were used as controls.
Cell morphology
The cell morphology of MG-63 seeded on the P-C film was observed by SEM (Figure 6). Cells cultured on the surface of P-C film appeared to spread evenly on the surface of the film over time, starting on day 1, and an increasing number of cells could be seen on the film by days 3 and 7.

SEM micrographs showing the morphology of MG-63 cells cultured on the P-C film for 1 (a, b), 3 (c, d), and 7 (e, f) days; 500× (a, c, e) and 1000× (b, d, f) magnification. Red arrows indicate cells growing on the material.
Cell proliferation
To evaluate the proliferation of MG-63 cells on P-C films, a DNA quantification assay was performed (Figure 7). Compared to the control, the DNA concentration of the cells on the P-C film was higher only on day 3, while there were more cells in the control group for all other time points. After cell attachment, cells appeared to proliferate rapidly, as demonstrated by the increase in DNA concentration in both groups over time; however, the cell number started to decrease on day 7, with the trend continuing on day 14.

The proliferation of MG-63 cells on P-C film was determined using the DNA quantification; cells cultured in the wells without the film were used as controls.
ALP activity and mineralization
To examine the effect of P-C film on MG-63 osteogenic differentiation, ALP activity and mineralization ability were evaluated. During the 14-day period, ALP activity (Figure 8(a)) of both groups followed the same trend: first, the levels increased with time (days 3 and 7), but then started to decrease on day 14. The ALP levels in the cells plated on the P-C film were higher compared to the control at all time points except day 7; statistical significance was only observed on day 14. The presence of mineralized nodules was evaluated using von Kossa staining (Figure 8(b)). On day 7, no silver deposits, indicating the presence of calcium, were observed in both groups; however, stained mineral nodules were visible on day 14 in the wells containing the P-C film.

(a) The ALP activity assay of MG-63 cells cultured on P-C film was performed on days 1, 3, 7 and 14; cells cultured without the P-C film were used as controls. *p < 0.05 versus control and (b) Von Kossa staining of MG-63 cells was performed on days 7 and 14. Red circle indicates stained mineral nodules.
Discussion
Barrier membranes for GBR are commonly used for treating periodontal defects or other conditions. Here we successfully prepared a barrier membrane as a P-C film by crosslinking collagen using proanthocyanidins. The ideal barrier membrane should prevent the migration of cells into the underlying structures, while supporting new bone formation. Type I collagen is the most abundant protein in bone, playing a key function as a structural component. 36 At the same time, collagen has been a popular biomaterial because it promotes cell adhesion and proliferation of mesenchymal stem cells. 37 Unlike other more commonly used cross-linking agents, which are toxic, proanthocyanidins are naturally occurring cross-linkers that benefit collagen synthesis in dentin biomodification. 38 Here we investigated the P-C film in vitro using both MG-63 and WS-1 cells to evaluate its potential for use as a barrier membrane.
Physical properties are important for the success of a barrier membrane: the membranes are supposed to promote cell attachment of certain cell types, as well as to block the migration of other cell types. In our previous publication, we have demonstrated that the mechanical strength of the crosslinked collagen membranes using different concentrations of OPCs ranged from 30 to 60 kPa. 33 For the optimized concentration of 10% OPCs as used in this study, the water content reached 70%. Nevertheless, mechanical properties are responsible for cell adhesion, proliferation, and differentiation behavior. And the arrangements of cell over time correlate to mechanical performances of the materials.39–41 The effect of surface roughness is a crucial factor in cell-surface crosstalk. 42 The microscale topography and porosity of the P-C film, as evaluated by SEM, showed strands of collagen fibers arranged around the pores. Our results demonstrated that MG-63 and WS-1 cells were able to attach and proliferate on the surface of the P-C film at the levels similar to the controls. The majority of human cells range in size between 2 and 120 µm. 43 For example, the size of osteocytes is 10–20 µm, while the sizes of fibroblasts and epithelial cells are 10–15 and 8–21 µm, respectively.44–46 Even though the average pore size of the P-C film membrane was in the range of 7–9 µm, the size that should have allowed the cells to move through it, the fluorescent staining of both MG-63 and WS-1 cells showed that the cells did not migrate to the other size of the film.
Collagen, either in resorbable or crosslinked forms, has been a popular material of choice for GBR. Here in this study, we verified that the P-C film is biocompatible and suitable for application with osteoblast-like cells. Higher cell viability on the P-C film compared to the control on day 1 suggested that this material is not only biocompatible, but could also promote the growth of MG-63 cells. The absence of the P-C film cytotoxicity was further confirmed by cell cycle analysis and, since no sub-G1 phase was identified, our results indicated that this material would not lead to apoptosis. SEM images of MG-63 cells seeded on the P-C film also confirmed the initial attachment, as well as cell proliferation, since a large number of cells was observed on the film surface. These results confirm the observations by Behring et al., 47 who reviewed 17 original studies and concluded that collagen membranes allow fibroblasts and bone-forming osteoblasts to attach to the surfaces, regardless of the source or treatment of collagen.
Upon initial cell attachment, osteoblasts undergo proliferation, differentiation, and mineralization to generate new bone. During the proliferation study, we observed a rapid growth in the beginning of the experiment to reach confluence, followed by a reduction of cell numbers by day 14. Our results suggested that this P-C film promoted the proliferation of osteoblasts, since cells cultured on the P-C film had reached the peak growth first. Similar to cell proliferation, the ALP activity of the cells plated on the P-C film was also higher than that of the control (except on day 7), suggesting that the film promoted and increased the proliferation and differentiation cycle of MG-63 cells. Calcium deposition, evaluated by von Kossa staining, was only detected in cells cultured on the P-C film, indicating that this material stimulated differentiation, leading to the mineralization of MG-63 cells. Collagen coating, either applied on two or three dimensions, has been reported to promote both proliferation and osteogenic differentiation of mesenchymal stem cells or osteoblast-like cells.37,48,49 Our results demonstrated that this P-C film is osteoconductive and osteogenic and, therefore, is well-suited for guided bone regeneration. Our future experiments will focus on characterization of this biomaterial in vivo, using animal models of periodontal regeneration and peri-implant responses.
Conclusion
The development of biomaterials for guided bone regeneration (GBR) is a challenge from the engineering and biological perspectives. Even though a number of advancements has been made over the past 30 years, collagen is frequently used because of its biological properties and flexibility for modifications. Here we developed a crosslinked collagen membrane using proanthocyanidins as the crosslinking agents. With a pore size of 7–9 µm, this P-C film appears to successfully block the migration of WS-1 and MG-63 cells across the membrane. Furthermore, the attachment and initial growth of cells on the P-C film demonstrated biocompatibility, as well as the absence of toxicity. The proliferation, differentiation, and mineralization of MG-63 cells cultured on this film were accelerated. Therefore, this P-C film could be a good candidate as a barrier membrane for GBR.
Footnotes
Acknowledgements
The first and second authors contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful for the financial support by the Ministry of Science and Technology of Taiwan (MOST 104-2622-E-027-027-CC3; MOST 104-2221-E-027-061, MOST 105-2221-E-027-028, MOST 106-2221-E-027-034, MOST 107-2221-E-027-014), and the Development and Reform Commission of Jilin Province, under Grant No. 2017C059-5.
