Abstract
Guided tissue regeneration/guided bone regeneration membranes (GTR/GBR) are widely used to repair damaged bone, manufactured using various materials, including synthetic polymers, natural polymers, metals, and inorganic compounds. Our previous study showed that nano-attapulgite (nano-ATP) composite membranes could significantly increase bone regeneration. Here, the porous nano-ATP-incorporated CH membranes were fabricated via the technique of freeze gelation (FG). The property and thermal stability of membranes were tested by scanning electron microscopy (SEM), Fourier-transform infrared spectroscopy (FTIR), and thermogravimetric analysis (TGA). Cell proliferation and morphology on the membranes were also studied. In vivo Micro-CT and histological analyses were used to evaluate new bone formation in rat cranium defect models. The results indicated that with increasing ATP content, the water absorption, porosity, swelling rate, and degradation rate of chitosan membranes significantly decreased; conversely, the thermal weight loss and mechanical strength exhibited a marked increase. Culture of mouse mesenchymal cells (D1) showed that ATP-incorporated membranes increased cell proliferation and matrix deposition. Furthermore, the results from Micro-CT and histological staining demonstrated that ATP-incorporated membranes could increase new bone formation. These results suggest that this novel nano-ATP incorporated membrane has great potential for bone tissue engineering applications and may lead to effective bone regeneration and repair.

Introduction
Guided tissue regeneration/guided bone regeneration membranes (GTR/GBR) are variety extensively used in bone tissue engineering (BTE).1,2 An excellent GTR/GBR membrane provides a favorable environment for bone regeneration, prevents connective tissue over-ingrowth, and increases bone tissue formation in defect sites.3,4 Thus, the research community has been working hard to fabricate the ideal GTR membrane with excellent biocompatibility, cell-occlusion properties, biodegradability, osteoconductive capability, and space-making ability.4–6
Chitosan (CH) is a biopolymer polysaccharide that possesses beneficial biomaterial properties, including biocompatibility, biodegradability, non-toxicity, and bacteriostatic effect.7,8 Hence, CH and its derivatives are widely used in biotechnology and biomedicine, spanning areas such as wastewater treatment, textile and food industry, agriculture, antibacterial and antiviral activities, drug delivery systems, wound dressings, dental materials, and tissue engineering. For example, various CH-based self-healing hydrogel dressings have been prepared in recent years to cope with increasingly complex wound conditions. CH and its derivative-based nanomaterials for the delivery of drugs, therapeutic gene transfer, delivery of vaccine. It is widely used in wound care, particularly to stop bleeding, promote wound healing, and provide antimicrobial properties.9–11 However, the primary limitation of CH is its relatively low mechanical properties and reduced bioactivities, which do not meet repairments of bone tissue engineering applications.12–14 Recently, incorporating natural or synthetic materials as composites with CH enhances mechanical properties and bioactivities, have been developed for this problem. The inorganic materials, including Hydroxyapatite (HA) and beta-tricalcium phosphate (β-TCP), have been incorporated in CH and processed enhanced mechanical properties and bone regeneration in vitro. The polymers, such as poly(vinyl alcohol; PVA), 15 poly-ɛ-caprolactone (PCL), 16 collagen, 17 silk fibroin, 18 mixed with CH efficiently reinforces its mechanical properties, and improves its bioactivities of the membrane.
Nano-Attapulgite (Nano-ATP) is a hydrous magnesium-rich aluminosilicate mineral with a layered chain structure. It is a kind of natural nano-clay with excellent adsorption performance, colloidal performance, carrier performance, and reinforcement performance due to its porosity, large specific surface area, high viscosity, and absorption ability.19,20 Based on the characterization of ATP, our previous studies revealed that ATP can promote the expression of osteoblast-related marker genes in mesenchymal stem cells (MSCs) under non-osteogenic induction conditions, with a positive correlation to ATP concentration. 21 Additionally, ATP not only enhanced cell proliferation but also significantly improved bone tissue regeneration and repair. By utilizing 3D bioprinting technology, we developed a Gelatin/Sodium Alginate composite hydrogel, which markedly increased the survival rate of printed cells, enhanced the mechanical strength of the hydrogel, and accelerated bone regeneration. 22 Furthermore, the ATP/hydroxyapatite (HA) composite material fabricated via 3D bioprinting exhibited synergistic effects in promoting vascularization and bone tissue regeneration. The combination of nano-ATP and HA as a bone repair material holds great clinical potential for significantly improving the efficacy of bone regeneration and functional reconstruction. 23 Moreover, we demonstrated that ATP could effectively load drugs, with the controlled release of these drugs synergistically enhancing vascularization and bone regeneration and repair. 24
The most common techniques, such as freezing, salt leaching, and electrospinning, have been used to fabricate CH membranes.25–27 However, these techniques have shown drawbacks, including the difficulty controlling the pore size, low interconnectivity, and uniform pores.28,29 For instance, salt-leaching only creates limited and tiny pores in CH membranes. 30 PCL/CH membranes fabricated by electrospinning produce a small pore size which significantly restrains cell proliferation and tissue ingrowth. The technique of freeze gelation (FG) was also reported to offer a more convenient, time- and energy-efficient method to fabricating porous membranes compared to freeze-drying and enables an easily operated scale-up process.31,32 Qasim et al. 33 have fabricated the porous membranes of CH with and without hydroxyapatite (HA) via FG with two different solvents systems, acetic acid (Aca) or ascorbic acid (Asa). The results showed that HA incorporated membranes supported cell proliferation, increased the matrix deposition for a long term, and indicated their potential for GTR applications in bone lesions and regeneration.
In this work, our purpose is that the porous ATP-incorporated membranes fabricated by FG could improve bone regeneration. For this purpose, we tested the membranes incorporated with different ratios of CH/ATP by evaluating their chemical, mechanical, and biological properties. Afterward, the biological effect of fabricated membranes on bone-forming capability is evaluated in the rat cranium defects model. It was expected that these novel membranes produce suitable properties as biomaterials for bone regeneration.
Materials and methods
Characterizations of ATP
Preparation of ATP: 12 g of ATP was slowly added to a 0.2 M hydrochloric acid (HCl) solution and stirred for 30 min. The mixture was then centrifuged at 4500 rpm/min for 10 min, followed by washing with deionized water 3 times, 5 min/time. The product was dried naturally, ground, and sieved through a 200-mesh screen. The obtained ATP was dispersed in 100 ml of anhydrous ethanol and ultrasonicated for 10 min (to dissociate the ATP nanobundles). After natural drying, the sample was ground again and sieved (200-mesh). Finally, it was stored at room temperature for further use.
The surface morphology of ATP was observed using Scanning Electron Microscopy (SEM, J Hitachi S3400 N, Japan). The samples were fixed on copper stubs and sputter-coated with an Au-Pd film. A high-resolution transmission electron microscope (TEM, HITACHI-7000FA, Japan) was used to take the images of ATP nanoparticles after the sample was dispersed in anhydrous ethanol by sonication and dropped onto a micro grid. SEM (J Hitachi S3400 N, Japan) was performed to analyze the composition of ATP.
Preparation of CH/ATP membranes
The CH/ATP membranes were fabricated using FG. 33 Briefly, CH (190–310 kDa molecular weight, Deacetylation degree > 90%, Aladdin, China) was dissolved in 0.2 M acetic acid (Aca; Life Scientific, China) solution to create 3% w/v solutions and stirred for 12 h until a homogeneous solution was formed. Further, nano-ATP (150–500 nm, Lanzhou Institute of Chemical Physics, Chinese Academy of Sciences) powder was added into the CH solution to form a viscous polymer homogeneous solution and then stirred for 12 h at room temperature. Membrane compositions (CH: ATP Ratio): 100:0 (3wt% CH, 0wt% ATP), 50:50 (3wt% CH, 3wt% ATP), 60:40 (3wt% CH, 2wt% ATP), 70:30 (3wt% CH, 1.29wt% ATP), respectively. The homogeneous solution was cast into a 9.0 cm petri dish before storing at 4°C for 3 h and then at −20°C for 12 h. A solution of 3 M Sodium hydroxide (Aladdin. China) in 100% ethanol (Aladdin. China) in a ratio of 1:1 (v/v) was pre-cooled to −20°C and used to submerge frozen membranes for 12 h. Finally, membranes were washed with PBS (0.02 M, pH = 7.4) to remove the remaining neutralizing solution and dehydrated with multiple ethanol washes (70%, 80%, 90%, 95%, and 100%), 15 min each. Afterward, the samples were immersed in a solution made of glycerol (Aladdin. China) and distilled water in the ratio of 1:10 for 15 min, air-dried, and stored at room temperature. The fabricated four different membranes were named CH:ATP (100:0), CH:ATP (70:30), CH:ATP (60:40), and CH:ATP (50:50).
Surface characterization of CH/ATP membranes
The morphology of the different CH/ATP membranes was observed by Scanning Electron Microscopy (SEM, JSM-6701; JEOL, Tokyo, Japan). The Energy Dispersive X-ray Spectroscopy (EDX) was used for the element of the membranes.
Physical and chemical characterization of CH/ATP membranes
In vitro degradation test
The membranes (
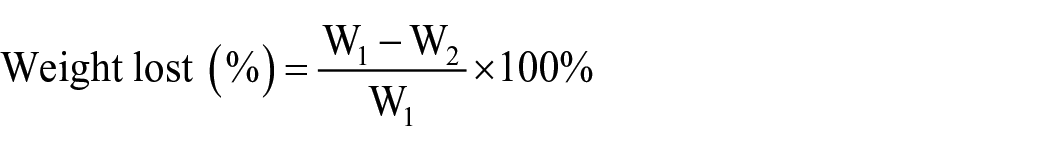
Where W1 is the initial dry mass of samples, W2 is the dry mass of the membranes after immersing in degradation media.
Swell ratio analysis
The different membranes were cut into 20 mm × 20 mm × 3.0 mm (width × length × thickness) were dried and weighed before storing in PBS (0.2 M, pH = 7.4) at 37°C to allow any swelling to occur. At set time intervals (0, 1, 2, 4, 8, 12, 24, 48, and 72 h), samples were removed from PBS and any excess water on the surface was removed gently with filter paper before weighing samples. The swelling ratio was obtained with the following equation:

Where W1 is the weight of the soaked membrane and W2 is the weight of the dry membrane.
Porosity measurement
The membranes (

Where M1 is the initial weight of ethanol in the bottle, M2 is the weight of ethanol-containing membranes, and M3 is the weight of ethanol after removal of the membranes.
Contact angle measurement
The contact angle analysis (FTA125, First Ten Angstroms, USA) was performed to assess the hydrophilicity of the membranes. The membranes were first chopped into 2 cm2 square pieces (
Thermogravimetric analysis (TGA)
Thermogravimetric analysis (TGA) of the membranes was investigated using a NETZSCH STA 409C/3/F in nitrogen condition. The temperature was gradually increased from 25 to 1000°C at a heating rate of 10°C/min. The curve was drawn according to residual weight (%) versus temperature (°C).
Fourier transform infrared spectroscopy (FTIR)
Fourier Transform Infrared Spectroscopy (FTIR, AVATAR 360, NICOLET, USA) was employed to identify the functional groups of the membranes. The samples were fixed to the ATR crystal. Testing was conducted under the following conditions: a resolution of 4 cm−1, 64 scans, and the frequency region range was 400–4000 cm−1. Three samples were examined three times.
Mechanical testing of membranes
The tensile strength testing was carried out at room temperature with MTS 4204 universal testing machine (MTS system (China) Co., LTD, China) with a 10 KN load cell. The membranes were cut into 10 mm × 60 mm × 3 mm (width × length × thickness) blocks. After the membranes were inserted between two clamps exposing a surface area of 40 mm2, the upper clamp was raised at a crosshead speed of 5 mm/min until the breaking point.
In vitro bioactivity test
For the bioactivity, the different membranes were cut into 20 mm × 20 mm × 3.0 mm (width × length × thickness), and immersed in 2 ml of simulated body fluid (1 × SBF, pH = 7.4) solution in 24 well-plate and incubated at 37°C, 5% CO2, and 95% relative humidity for 7, and 14 days with the SBF changed every 2 days. Then the SBF solution was taken away from 24 well-plate at the desired time and washed with DI water three times, then the membranes were freeze-dried (Freezezone 4.5; Labconco Corp., Kansas City, MO, USA). Finally, the dried samples were observed and analyzed using SEM and EDX.
Cell culture
D1 cells (mouse multipotent mesenchymal precursor cell line, ATCCCRL-12424, ATCC, USA) were cultured in DMEM/F12 (D/F12) complete medium (Gibco, USA) supplemented with 10% fetal calf serum (FCS) and 1% penicillin/streptomycin and 5% CO2 at 37°C. The sterilized membranes were soaked in the D/F12 medium overnight. A total of 1.2 × 106 cells per 20 µl of D/F12 complete culture medium were seeded on the surface of membranes for 30 min before adding 1 ml of culture medium and continuously incubated at 37°C with 5% CO2 for 1, 3, and 7 days. The culture medium was changed twice a week.
Scanning electron microscopy (SEM)
The cells-seeded membranes were cultured until day 7, then the membranes were rinsed in PBS (pH = 7.4, 0.01 M;
Cell proliferation evaluation
The cell proliferation was evaluated using a CCK-8 assay according to the instructions. On day 1, 3, and 7, the membranes were washed three times with PBS (0.2 M, pH = 7.4) and transferred to a 96-well plate. Then, the D/F12 medium containing 10% volume of CCK-8 was added to every well and then incubated for 4 h. Finally, optical density (OD) at 450 nm wavelength was evaluated using an ELISA reader (Bio-Rad Model 550; Bio-Rad, Berkeley, CA, USA).
In vivo cranial defect model and membranes implantation
All animal experiments were approved by China’s Animal Research Authority and Ethics Committee of the Chan-gzhou Second Hospital. 48 Wistar rats were selected to establish a rat cranium defect model. The rats were randomly divided into five groups as follows: Control group, CH:ATP (100:0) group, CH:ATP (70:30) group, CH:ATP (60:40) group, and CH:ATP (50:50) group. Rats were anesthetized with 3% pentobarbital sodium by intraperitoneal injection of 0.3 ml/100 g. The skin and soft tissue were carefully dissected. Two circular defects (5 mm in diameter) were created using trephine at both sides of the cranium of each rat. Afterward, the different materials (5 mm in diameter, 3 mm in thickness) were implanted in the different groups. Ultimately, muscles and skin were sutured layer by layer, and animals were recovered from the anesthesia after giving 2 mg/kg carprofen postoperative for analgesia. Penicillin (400,000 U) was given through an intramuscular route every 24 h for 7 days. At 4, 8, and 12 weeks postoperative, rats were sacrificed by overdose of pentobarbital sodium anesthetized.
Micro-CT evaluation
The samples (cranium/material complexes) in different groups were fixed in 10% formaldehyde at room temperature for 72 h and scanned with Micro-CT (GE Healthcare, USA) for 4, 8, and 12 weeks post-surgery. After 3D reconstruction, the images were visualized and analyzed using the built-in software.
Histological staining
After Micro-CT evaluation, the samples were decalcified in 10% EDTA-Na2 (pH = 7.0) solution for 6 weeks at room temperature and subsequently dehydrated, embedded in paraffin, and sectioned at a 5 µm thickness. Hematoxylin and eosin (H&E), and Masson’s trichrome (MT) Stain Kit were performed to assess new bone formation at bone defect areas.
Statistical analyses
All data were expressed as means ± standard deviations (SD). SPSS 13.0 statistical software (SPSS, USA) was used to analyze all data. One-way analysis of variance (ANOVA) with an LSD post hoc test was performed to compare the differences between each group.
Results
Characterization of ATP
The characterization of the nona-ATP particles was observed by SEM and TEM. Figure 1(a) and (b) showed the ATP acicula-like nanorods with a 200–500 nm length and diameter of approximately 30 nm. EDX results showed that the main elemental compostion (mass%) of ATP was C (5.28%), O (12.76%), Mg (9.28%), Si (49.47%), Al (8.77%), Ca (2.62%), K (1.5%), and Fe (9.63%; Figure 1(c)).

SEM, TEM micrographs, and composition of the ATP. (a) SEM image. (b) TEM image. (c) The results of the EDX spectroscopy analysis of ATP.
Morphology of the CH/ATP membranes
SEM analysis of the surface morphology of the CH/ATP membrane is shown in Figure 2, It can be seen from Figure 2 that CH membranes were dense surface structures, without any porosity in their surface (Figure 2(a) and (b)). Conversely, ATP incorporated into CH membrane, seldom porosity was observed on their surface. Specifically, the uniformly larger pores can be seen in CH/ATP (70:30) membranes (indicated by arrows; Figure 2(d) and (e)). Meanwhile, with the increase of ATP, a large number of nano-ATP crystals are distributed on the surface of the CH/ATP membrane (Figure 2(g)–(k)). EDX results showed that strong signals were detected for Si, Mg, and Al elements, indicating the presence of ATP in the CH/ATP membranes (Figure 2(f), (i), and (l)).

SEM micrographs of the surface topography of the various membranes. (a and b) CH membranes. (d and e) CH/ATP (70:30) membranes. (g and h) CH/ATP (60:40) membranes. (j and k) CH/ATP (50:50) membranes. (c, f, i, and l) The results of the EDX spectroscopy analysis of the CH/ATP membranes, (c; 100:0), (f; 70:30), (i; 60:40; U), (l; 50:50) membranes.
Figure 3a1–a3 show representative optical images of CH/ATP membrane. The cross-section morphology of CH and CH/ATP membranes was observed by scanning electron microscopy (SEM), as shown in Figure 3b1–e3. The SEM images illustrated that the membranes displayed three layers construct, including a relatively dense surface layer and a loose internal layer. CH membranes exhibited an uneven porous structure with uniformly distributed 200–300 µm diameter pores (Figure 3b1–b3). However, there is an obvious difference between the cross-section morphology of CH and CH/ATP membranes. When ATP was incorporated into CH, the sized pores decreased compared to CH membranes, and the pore dimensions were mainly in the range of 100–200 µm (Figure 3c1–c3). In contrast, with the increase of ATP incorporation, the morphology of CH/ATP membranes was clearly altered. It was found that the uneven porous structure had completely disappeared, displaying the lamellar structure with smaller uniformly distributed sized pores (Figure 3d1–e3). These results suggested that ATP incorporation can affect the porosity and morphology of CH membranes.

Characterization of cross-sectioned CH/ATP membrane. (a1–a3) Optical images of CH/ATP (50:50) membrane. (b1–b3) SEM images of CH/ATP (100:0) membranes. (c1–c3) SEM images of CH/ATP (70:30) membranes. (d1–d3) SEM images of CH/ATP (60:40) membranes. (e1–e3) SEM images of CH/ATP (50:50) membranes.
Physical and chemical properties of the CH/ATP membranes
Contact angle measurements
As well as known, the water contact angle is higher for rougher surfaces than for smoother ones. Therefore, the contact angle of CH and CH/ATP membranes was measured using the contact angle analysis. It can be found from Figure 4(a) that the contact angle of CH and CH/ATP membranes was considerably changed. There are lower contact angles of CH/ATP membranes than that of CH membranes, which means increasing hydrophilicity and increasing rougher surfaces of CH membranes when ATP is incorporated.

Characterization of CH and CH/ATP membranes. (a) The contact angles. (b) Porosity. (c) The weight loss. (d) Swell ratio. (e) TGA curves of CH/ATP membranes. (f) Elastic modulus of CH/ATP membranes. (g) FTIR spectra of CH/ATP membranes.
Porosity
The porosity of CH and CH membranes incorporating ATP with different concentrations is shown in Figure 4(b). It can be noticed that the porosity of the CH membranes was 62.5%, while that of the CH:ATP (70:30), CH:ATP (60:40), and CH:ATP (50:50) membranes were 71.4%, 35.1%, and 30.6%, respectively. A significant decrease in the porosity was observed as the concentration of ATP increased, particularly in CH:ATP (60:40) and CH:ATP (50:50) membranes, significant differences were observed between the CH membrane and CH/ATP membrane. It could be noticed that the decrease in the porosity of the CH/ATP membrane was attributed to the morphology of the membranes was altered when the concentration of ATP increased.
The degradation
Membranes were immersed in PBS (0.1 m, pH = 7.4) containing Egg Hen lysozyme for 10 weeks. The degradation rates of the membranes are shown in Figure 4(c). By 1 week, the loss of CH/ATP (50:50) membranes weighed 38.34% of their original mass. However, the other membranes loss weighed 56.23% of its original mass. Even after 10 weeks of immersion, CH/ATP (50:50) membranes loss weighed 63.0% of its original mass. Conversely, the other membranes’ loss weighed 80% of its original mass (Figure 4(c)), indicating that ATP incorporation decreased the degradability of the CH membranes.
Swelling percentage
Figure 4(d) shows the swelling percentage of the different membranes in PBS solution with incubation time. The CH:ATP (100:0) membrane exhibited the highest swelling ratio (up to 80%) compared with CH:ATP (70:30; up to 70%–75%), CH:ATP (60:40; up to 70%–65%), and CH:ATP (50:50) membranes (up to 60%–65%), indicating CH membranes incorporating ATP exhibited less swelling percentage as compared to CH membrane. Moreover, the swelling percentage of CH:ATP membranes decreased with increasing ATP concentration. The decrease in swelling of incorporating ATP membranes was attributed to their lower porosity. The results were the same as the porosity results.
Thermogravimetric analysis (TGA)
Figure 4(e) exhibits TGA curves of CH and CH/ATP membranes obtained by the thermogravimetric analysis. It can be observed from Figure 4(d) that the thermal decomposition of CH and CH/ATP membranes undergoes two stages. First, no significant degradation occurred before 250°C for all membranes; weight loss of 5% around 350°C, which could be attributed to moisture within the material. The major decomposition of the CH and CH/ATP membranes ranged from 350°C to 450°C, and the weight loss had a significant difference for membranes, which was attributed to dope ATP decrease thermal degradation of membranes (Figure 4(e)), indicating that ATP incorporation increases the thermal stability of the CH membranes.
Mechanical characterization
Mechanical properties of the CH and CH/ATP membranes are shown in Figure 4(f). The elastic modulus was 0.9 (±0.23) MPa for CH membranes at room temperature (25°C). However, when ATP was incorporated into the membranes, the elastic modulus was significantly higher than that of the CH membranes. The highest increase was observed for CH/ATP (50:50) membranes 2.8 (±0.18) MPa compared to CH/ATP (60:40) membranes 2.6 (±0.16) MPa and CH/ATP (70:30) membranes 1.8 (±0.46) MPa. The overall trend shows a significant increase in elastic modulus with increasing ATP concentration.
Fourier transform infrared (FTIR) spectroscopy
Figure 4(g) shows the FTIR spectrum of CH and CH/ATP membranes. It was observed that CH had absorption bands at 3435 and 2945 cm−1 as the stretch of O–H and C–H. The absorption bands at 1634 cm−1 and 1050 cm−1 for C=O of the CH (Figure 4(g)). A typical characteristic absorption band can be observed between 1130 cm−1 and 800 cm−1, which peaks representing the Si-O-Si stretching and the Si-O stretching of the CH:ATP (70:30), CH:ATP (60:40), and CH:ATP (50:50) membranes (Figure 4(g)), indicating ATP successfully incorporated into CH membranes.
In vitro bioactivity investigation
Figure 5 shows the SEM image of the surface morphology of the CH and CH/ATP membranes after 7 days of immersion in SBF. The formed crystals were found on the surface of all membranes. However, their morphology was not the same. In the CH/ATP (100:0) membranes, only a smaller apatite-liking crystals were formed (Figure 5(a)). The spherical crystals were present on the surface of the CH/ATP (60:40; Figure 5(c)) and (50:50; Figure 5(d)) membranes. In contrast, the apatite-liking crystals were formed on the surface of CH/ATP (70:30) membranes (Figure 5(b)). Figure 5(e) shows the chemical composition of the apatite-liking crystals on the CH/ATP (70:30) membranes. Strong signals were detected for Ca and P elements, indicating the presence of Hydroxyapatite (HA) in the membranes (Figure 5(e)). The results showed that 30% ATP incorporated into CH significantly increased the bioactivity of the membranes, indicating that this property is beneficial for bone formation.

SEM images of the different membranes after simulated body fluid (SBF) incubation for 7 days. (a) CH:ATP (100:0) membranes. (b) CH:ATP (70:30) membranes. (c) CH:ATP (60:40) membranes. (d) CH:ATP (50:50) membranes. (e) The results of the EDX spectroscopy analysis of the CH:ATP (70:30) membranes.
Cell viability
D1 cells were seeded and cultured on different membranes for 1, 3, and 7 days, respectively. SEM and CCK-8 were employed to evaluate the cell adhesion and proliferation on the membranes. Figure 6(a) shows the SEM images of D1 cells on the membranes after being cultured for 7 days. A large amount of cell adhesion was observed on the surface of CH/ATP (70:30) membranes. Conversely, a small number of cells were found on the other membranes. CCK-8 results showed the cell proliferation did not indicate a statistically significant difference on day 1. However, the cell growth on the CH/ATP (70:30) membranes increased in comparison with the other membranes (

Cells grown on the different membranes. (a) SEM image of cells on the surface of the different membranes at 7 days. (b) the cell proliferation was assessed using CCK-8. Data show the mean ± standard deviation (
Micro-CT scan analysis
To further verify the effect of CH/ATP membrane on bone formation, the different CH and CH/ATP membranes were implanted into rat cranium defects for 4, 8, and 12 weeks, respectively. The reconstructed 3D micro-CT images of the cranium are shown in Figure 7(a). At 4 weeks post-surgery, no new bone formation was detected in CH membranes groups. In contrast, a small amount of new bone was found within the bone defect region in the CH/ATP groups. The increased new bones were observed within the bone defect region in the CH/ATP (70:30) groups at 8 weeks postoperatively (Figure 7(a)). After 12 weeks of surgery, a large amount of new bone was seen in the CH/ATP (70:30) group, followed by that in the CH/ATP (60:40) group, compared to that in the CH/ATP (50:50) and CH membranes group. Consistent with the Micro-CT scanning results. Quantification of newly formed bone shown in Figure 7(b) and (c). The Bone mineral density (BMD; around 0.19 mg/cm3) and bone volume/tissue volume ratio (BV/TV; around 12.84%) were higher in the CH/ATP (70:30) groups than those of CH (around 0.072 mg/cm3 and 6.62%), CH/ATP (60:40; around 0.17 mg/cm3 and 10.90%), and CH/ATP (50:50; around 0.14 mg/cm3 and 9.80%) groups at 4 weeks post-surgery. At 8 weeks, the BMD (around 0.23 mg/cm3) and BV/TV (around 18.04%) was increased in the CH/ATP (70:30) group than that of CH (around 0.087 mg/cm3 and 10.44%), CH/ATP (60:40; around 0.20 mg/cm3 and 16.84%), and CH/ATP (50:50; around 0.17 mg/cm3 and 12.92%) groups (
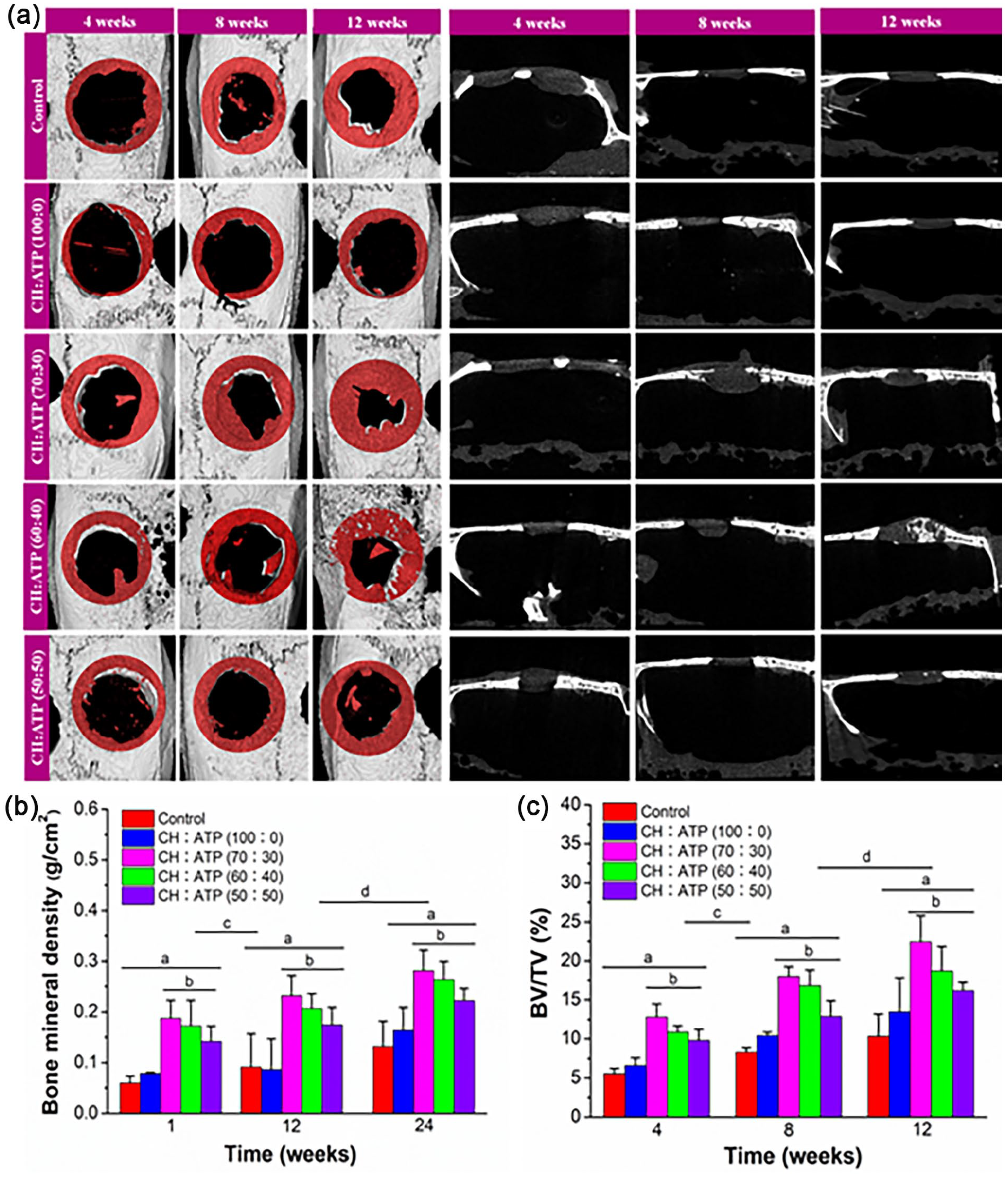
In vivo evaluation of new bone formation in the cranium defects that implanted the membrane at 4–12 weeks after post-surgery. (a) Micro-CT scanned images. (b) Bone mineral density (BMD). (c) Bone volume/Tissue volume ratio (BV/TV), Data show the mean ± standard deviation (
Histological assessment
H&E and Masson staining of bone defect sites post-implantation was carried out to assess the regenerative bone profiles (Figure 8). Consistent with micro-CT scanning results, only a tiny amount of new bone tissue was observed adjacent to the bone-biomaterial interface in the CH: ATP (100:0) group at week 12 (Figure 8(a) and (b)), which consisted of mature and immature collagen deposition indicated by Masson staining (Figure 8(c)). Conversely, at 12 weeks post-surgery, it can be found from Figure 7(a) and (b), CH:ATP (70:30) group displayed a larger number of new bones in the core of membranes. Meanwhile, mature bone tissues were present in the membranes of the CH:ATP (70:30) group (Figure 8(c)). However, CH : ATP (60:40; Figure 8(a) and (b)) and CH:ATP (50:50; Figure 8(a) and (b)) groups exhibited emergence of several independent bone islands regenerated bone tissue was creeping from old bone and almost bridged at the central of the defect. Masson staining showed that only smaller mature bone tissues were present in the core of membranes (Figure 8(c)). The results indicated that the porous nano-ATP-incorporated CH membranes exhibited greater potential as a functional biomaterial for bone defects. Furthermore, incorporating 30% ATP in the CH membranes also significantly promotes new bone formation.
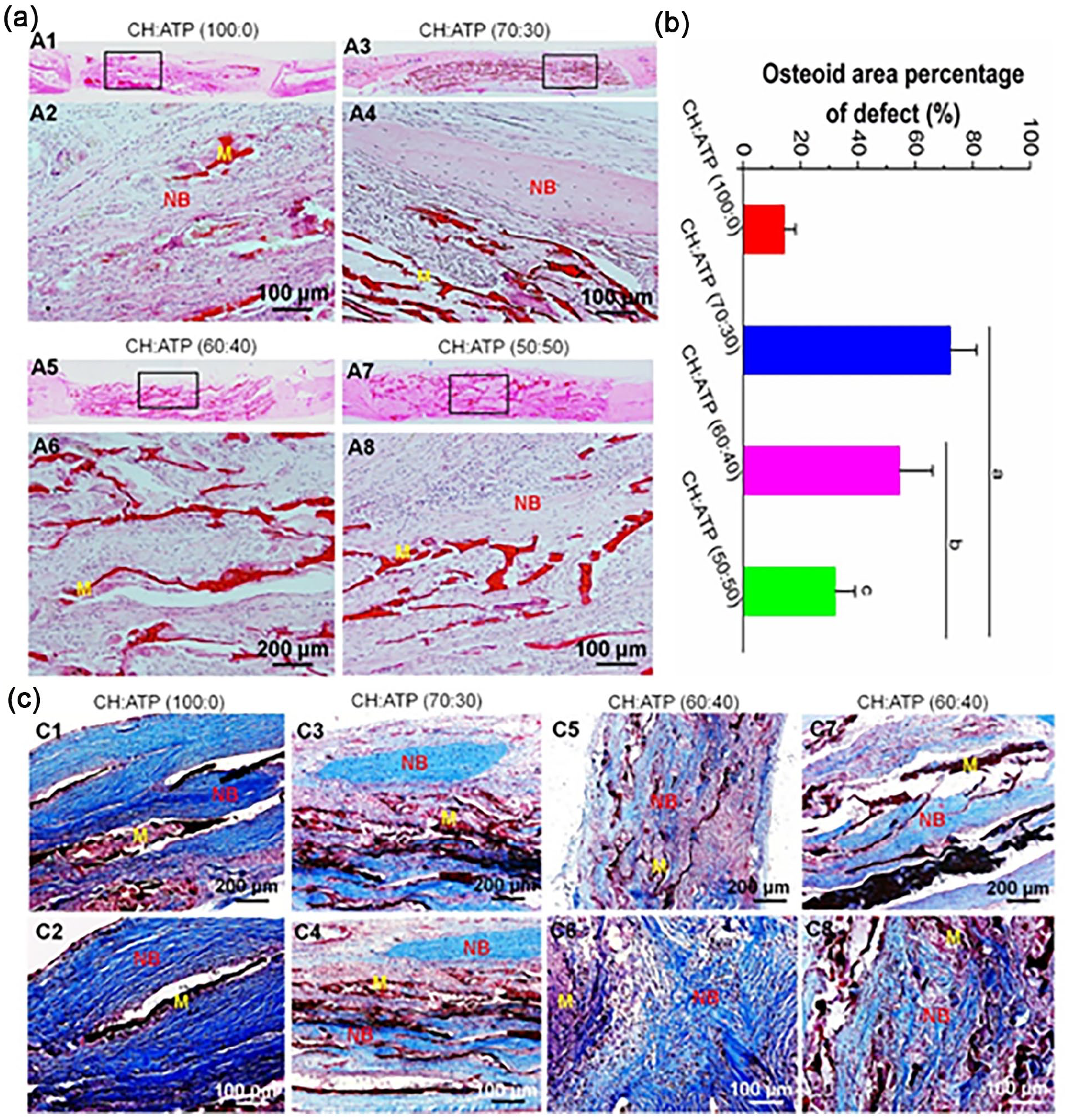
Histological analysis of new bone regeneration in the cranium defects that implanted the membrane at 12 weeks post-surgery. (a) H&E-stained images. (b) Show the quantitative data from (a). Data display the mean relative values calculated from three independent experiments (mean ± SD). a
Discussion
CH is therefore commonly utilized in membrane matrices for bone healing. Meanwhile, CH-based membranes as bone biomaterials have been demonstrated by numerous studies.35–37 As well investigated, ideal GBR membranes as bone membranes should have the following advantages: (1) porous structures for increasing cell adhesion, proliferation, migration, and differentiation; (2) sufficient mechanical properties; (3) controllable degradation rate; (4) excellent biocompatibility, and osteoconductive.38–40 Compared with traditional CH as the material of bone membranes, CH membranes mixed with other natural or synthetic materials improved mechanical properties and increased biocompatibility and osteoconductive.41,42 In recent studies, porous membranes for GBR were fabricated with nano-clay and CH, which improved mechanical properties and promoted cell adhesion, proliferation, and differentiation.43,44 Therefore, incorporating other materials as ideal GBR membranes is a promising strategy in bone tissue engineering applications.
In this work, membranes were manufactured by incorporating CH-ATP using freeze gelation (FG). SEM results showed that the tri-layered structure of the cross-sectional surface could be seen. The outer layer of the membrane displayed a relatively dense structure, and the middle layer showed the honeycomb structure in the membrane of CHI and CH/ATP with 30% ATP. However, with ATP increased, the membrane displayed the lamellar structure with smaller uniformly distributed sized pores in CH/ATP membrane with 40% and 50% ATP. The relationship between the microstructure alter and the increase of ATP is not well understood.
Biomaterials dedicated to tissue engineering must be characterized by low density and high porosity, which should be at least 90%. 23 Multiple pores with interconnecting channels provide nutrient and oxygen delivery and enable CO2 and metabolite removal. Such porosity is also needed for the new tissue formation in three dimensions as well as angiogenesis and neovascularization processes. The porosity is an essential factor for GTR membranes as it affects the progression of osteogenesis. 45 Small pore sizes (<100 µm) reduce cell migration and restrict nutrient delivery and waste removal. Additionally, it limits the amount of new tissue regeneration. 46 It has been reported that the minimum recommended pore size for a membrane is 100 µm to achieve adequate vascularization of the tissue/organ being repaired or regenerated. 47 However, other studies have reported increased cell migration and tissue formation with pores around 300 µm. These results show the suitable pore size variation from 100 to 400 µm. 48 Our study showed that the pore size of the CH/ATP membrane with 30% ATP ranged from 300 to 400 µm as compared to that of the CH membrane. However, the pore size of the CH/ATP membrane with 40% and 50% ATP ranged from 30 to 80 µm, which was relatively lower than the CH membrane and the CH/ATP membrane with 30% ATP. This may be due to the incorporation of nano-ATP within the CH network structure, resulting in the closing of pores, therefore, lowering the porosity.
Membrane biodegradability is also a vital factor affecting tissue regeneration. 49 The degradation rate of the membrane should meet the growth rate of the formation of the new tissue. 50 In other words, the rapid degradation rate led to the lack of mechanical support during the new tissue forming, whereas the slow degradation rate affects new tissue regeneration. Hence, the appropriate degradation rate is essential for the biomaterials in BTE. Several studies demonstrated that nano-clay particles, including montmorillonite natural clay, laponite, and kaolin incorporation into other materials, not only increased the mechanical strength and shape fidelity but also enhanced cell adhesion and promoted osteogenic differentiation of mesenchymal stem cells.51–53 Our work indicated that the membrane’s weight loss was significantly decreased compared to the CH membrane, with the incorporated ATP increased. The primary reason why ATP mixed with CH incorporation altered the membrane structure is that reduced porosity and slowed the hydrolysis-driven degradation rate.
Swelling ratio is another parameter important for tissue engineering membranes, as the increased swell ratio of membrane implanted in vivo could lead to the loosening and dislocation of the implant. Because CH has high hydrophilicity, it is easy to lose and dislocate as bone materials. Therefore, it is necessary to reduce the swelling ratio of CH membrane. Bhowmick et al. 54 fabricated biomimetic organically modified montmorillonite clay (OMMT) supported CH/hydroxyapatite-zinc oxide nanocomposites. The composite showed a decreased swelling capacity with increasing hydrophobic-modified clay. In this study, we found that blending CH with ATP showed less swelling ratio as compared to CH membrane. Moreover, the swelling ratio decreased with increasing ATP concentration.
The mechanical strength of the membrane plays a crucial role in BTE. However, the mechanical properties of CH membranes are inferior to those of normal bone. Therefore, their applications are restricted owing to their weak mechanical strength. Now, this disadvantage has been overcome by mixing the CH membranes with other synthetic or natural polymers efficiently reinforces its mechanical properties and improves its bioactivities. Fang et al. 55 fabricated the CH/tri-calcium phosphate bio-composite bone cement. The addition of tri-calcium phosphate can enhance the mechanical properties of CH membrane. Uswatta et al. 56 fabricated porous injectable spherical membranes using CH biopolymer, sodium tripolyphosphate (TPP) and nano-hydroxyapatite (nHA). The results showed that the mechanical properties of the membrane were significantly improved compared with the membrane without TPP and nHA addition. However, due to HA alone is a brittle material, composites prepared from HA and CH showed unideal mechanical strength. To settle this problem, Kar et al. 57 fabricated a composite by adding montmorillonite (MMT)-type clay into HA-CH composite can enhance mechanical properties. Clay has a layered silicate structure and hence the incorporation of montmorillonite (MMT)-type clay into a composite. Here, we found that the mechanical strength of the CH/ATP membranes was significantly increased compared with that of CH membranes. The increased mechanical strength of the membrane with an increase in ATP content is probably due to the electrostatic interactions at hydrogen bonds between CH and ATP, enhancing its mechanical strength.
Post-SBF immersion SEM analysis revealed the formation of crystals on the surface of all membranes. However, their morphology varied significantly. Smaller apatite-like crystals were observed on the CH/ATP (100:0) and CH/ATP (60:40) membranes, whereas spherical crystals predominated on the CH/ATP (60:40) and (50:50) membranes. This morphological difference can be primarily attributed to variations in the release levels of magnesium (Mg²⁺) and silicon (Si⁴⁺) ions, which increased with higher ATP content. Additionally, changes in the pH of the surrounding medium may have influenced the biomineralization activity of the membranes, consistent with previous reports.58,59 Nevertheless, this needs further validation through in-vivo studies in the future.
An ideal bone tissue engineering membrane should provide a suitable micro-environment for cell adhesion, proliferation, differentiation, and new tissue formation.60,61 Extensive studies have been used CH to produce membranes for bone tissue engineering, but the use of CH as membrane is limited by having reduced bioactivities. 62 Li et al. 63 prepared CH -hyaluronic acid (CH-HA) membranes incorporating nano-pearl powder (NPP), the results showed that the mechanical properties of membranes were increased with NPP content, whereas the osteogenic characteristics were significantly promoted by upregulating the expression of Col αI, OCN, OPN, and Runx2 genes, due to nano-pearl’s bioactive characteristics and favorable mechanical properties. Tamburaci et al. 64 applied diatomite and polyhedral oligomeric silsesquioxanes (POSS) as bioactive agents to fabricate CH (CS)/Na-carboxymethylcellulose (Na-CMC) polymer membranes. The results showed that both inorganic reinforcements not only increased the mechanical strength, but also enhanced the surface mineralization in osteogenic medium. Gurucharan et al. developed a novel CH-hydroxyapatite (CSHA) composite scaffold through an ultrasonication-assisted synthesis route, wherein nano-hydroxyapatite (nHAp) particles were homogenously incorporated into a carboxymethyl CH (CMC) matrix within aqueous medium, followed by lyophilization to achieve three-dimensional porous architecture. Remarkably, the engineered CSHA scaffold demonstrated significant bioinductive properties by markedly upregulating two critical biomarkers in human dental pulp stem cells (hDPSCs): vascular endothelial growth factor (VEGF) and dentine sialophosphoprotein (DSPP). This dual enhancement of VEGF and odontogenic DSPP gene expressions suggests synergistic potential for dentin-pulp complex regeneration. Furthermore, the scaffold exhibited superior mineralization capacity, as evidenced by intensified Alizarin Red staining and distinct apatite crystal formation observed via SEM-EDX after 21-day immersion in simulated body fluid. 58 In our study, it was found that a larger number of cells adhered and proliferated on the CH/ATP (70:30) membranes. However, a seldom number of cells were observed on the other membranes. The main reason is ATP has strong adsorption properties and can spontaneously adsorb bioactive molecules from body fluids and other body fluids to the material, which could build up a benign microenvironment for adherent cells. Therefore, our results indicated that incorporating the appropriate ATP into the CH membrane exhibited good cytocompatibility and promoted the proliferation of MSCs.
Numerous in vitro and in vivo studies have been conducted using natural and synthetic materials to fabricate bone membranes for bone regeneration.65,66 However, some CH-based membranes lacked of bioactivities and osteoconductivity. Here, In vivo, the processes of bone regeneration were assessed by Micro-CT, and results showed that the fibrous tissue was filled with the defect in the control group, even after 12 weeks. By contrast, in week 8 and 12 after surgery, the defects in the CH/ATP (70:30) membranes group were mainly filled up with large amounts of newly formed bone. Significantly more dense bones were seen in the defect, especially in the CH/ATP (70:30) membranes group. On the other hand, H&E and Masson staining confirmed the new bone formation in all the groups. In this case, there was no bone tissue found in the control group. After 4 weeks, some bony tissue was observed in CH/ATP (70:30) groups. However, only small amounts of bony tissue were observed in the control group after 8 weeks. Contrarily, a higher quantity of new bone formation was observed in CH/ATP (70:30) group. After 12 weeks of post-surgery, the regenerated mature bone that had migrated into the membranes was found in CH/ATP (70:30) groups. These results indicated that CH/ATP (70:30) membranes had the best effect on promoting bone regeneration than any other group. This was probably because ATP can increase cell growth and differentiation. Our previous studues22,67 demonstrated that ATP significantly promotes MSCs differentiation into osteoblasts in vitro and enhance new bone regeneration in vivo. Recent advances in scaffold design have demonstrated significant potential for enhancing bone regeneration. Liu et al. 68 developed a nacre-mimetic cerium-doped nano-hydroxyapatite/CH layered composite scaffold (CeHA/CS), which markedly accelerated new bone formation by suppressing bone resorption activity while promoting osteogenesis. Similarly, Boda et al. 69 utilized electrospinning technology to fabricate a novel aerogel scaffold incorporating polyvinyl alcohol (PVA)/CH hybrid nanofibers and tricalcium phosphate. This scaffold facilitated sustained release of calcium (Ca), phosphorus (P), and silicon (Si), thereby enhancing bone regeneration and new bone formation. In another study, Zhai et al. 70 designed a poly (4-acryloylmorpholine) clay-based nanocomposite hydrogel capable of releasing magnesium (Mg²⁺) and silicon (Si⁴⁺) ions. This system exhibited excellent osteogenic differentiation potential in primary rat osteoblasts (ROBs) and effectively promoted new bone formation upon implantation. In our study, it was collectively demonstrated that the CH membrane mixed with appropriate ATP exhibited an excellent mechanical properties and bone regeneration effect.
Conclusions
In this work, we have successfully fabricated the porous ATP-incorporated ATP/CH membranes by freeze gelation. The increased incorporation of ATP led to a change in the CH membrane structure. The uneven, porous structure completely disappeared, displaying the lamellar structure with smaller uniformly distributed sized pores. The CH/ATP (70:30) membranes exhibited a better proliferation of BMSCs than other ATP/CH membranes and CH membranes. In vivo, the CH/ATP (70:30) membranes exhibited significantly more new bone formation than treatment with the other ATP/CH membranes or CH membranes. Therefore, a conclusion can be drawn that a novel ATP-incorporated membrane is a promising candidate for bone tissue engineering applications.
Footnotes
Acknowledgements
We would like to thank Ai qin Wang for providing us with ATP materials and Guan Je Bao for contributing to mechanical tests.
Author contributions
Conceptualization, Hongbin Zhao.; methodology, Yiping Wen.; software, Dong Zhou.; validation, Chun Liu, and Jiayi Ma.; formal analysis, Siyu Wu and Jun Liu.; investigation, Yiping Wen.; resources, Ting Dai.; data curation, Fang Shao; writing—original draft preparation, Yiping Wen; writing—review and editing, Hongbin Zhao.; visualization, Xiaoyu Wu.; supervision, Dong Zhou.; project administration, Hongbin Zhao.; funding acquisition, Hongbin Zhao. All authors have read and agreed to the published version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funding from the Key Project of Science, and Technology of Jiangsu Province (Grant BE2018644) and Changzhou Young Talents Training Project (CZQM2020070).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
