Abstract
Aim:
The aim was to compare the repair bond strength and surface topography of lithium disilicate ceramics (LDC) and hybrid resin ceramics (HRC) using different surface conditioning treatments [low level laser therapy (LLLT), photodynamic therapy (PDT), hydrofluoric acid (HF) with silane and air abrasion (AA) and silane].
Material and method:
Sixty specimens each of LDC and HRC were used. Discs were prepared for each group (6 × 2 mm), conditioned using different regimes. Specimens in group 1 and 5 were laser irradiated using Er,Cr:YSGG (ECYL), group 2 and 6 were conditioned using methylene blue photosensitizer (PDT), group 3 and 7 surface was treated with hydrofluoric acid and silane (HFA-S), group 4 and 8 conditioned with Al2O3 air abrasion and silane (AA-S). A Porcelain Repair Kit was used according to manufacturer recommendation in all samples. Peak universal bond adhesive was rubbed on ceramic surface and then bonded with composite resin. For shear bond strength testing the specimens were placed in a universal testing machine. A stereomicroscope at 40x magnification was used to analyse failure pattern. Five specimens in each group after surface treatment were evaluated for surface changes and topography using scanning electron microscopy. The mean repair bond strength was calculated using ANOVA and Tukey’s post hoc test at a significance level of (
Result:
The highest repair bond strength was observed in group 3 (LDC) (20.57 ± 3.58 MPa) (HFA-S), whereas, the lowest score was displayed in Group 2 (LDC) using methylene blue photosensitizer (MBPS) (12.18 ± 1.08 MPa). Similarly, in HRC the highest repair SBS was presented in group 8 (AA-S) (20.52 ± 2.51 MPa) and the lowest SBS values were exhibited by PDT treated group 6 (13.22 ± 0.62 MPa).
Conclusion:
A combination of mechanical and chemical surface treatments should be used in order to achieve adequate repair bond strength between resin composites and ceramic interface of LDC and HRC.
Introduction
Dentistry has greatly evolved in the past few decades with the advent of modern materials and novel techniques. The need for these materials and techniques appeared due to the development of contemporary aesthetic materials for improved facial appearance and dental aesthetics. 1 The use of all-ceramic indirect restorations has now become more popular among dentists, to overcome the relatively inferior aesthetic outcomes of metallic restorations. 2
Computer-aided Designing and Computer-aided Manufacturing (CAD/CAM) Technology made industrial-grade prefabricated blocks of ceramic materials available for use as the modern standard of care for permanent restorations, such as inlays, onlays, veneers, crowns and bridges. These materials have numerous advantages over conventional materials, such as: uniform quality, better physical and mechanical properties, reproducibility of restorations, and reduced costs and duration of fabrication. 3 There is a variety of ceramic blocks available for milling different types of dental restorations through CAD/CAM technique. These include feldspathic glass ceramics, leucite-reinforced glass ceramics, lithium disilicate glass ceramics, aluminum-oxide and yttrium tetragonal zirconia polycrystals, composite resins and nano-hybrid ceramics.3,4 Their mechanical properties vary according to their chemical nature and that also influences their bonding to various luting agents.4,5
However, the quick delivery and superior aesthetics of such ceramic restorations comes with its own challenges, compared to their versatile metallic alternatives. The inherent brittle nature of the ceramic materials makes them prone to fracture during clinical use, mostly due to parafunction and trauma.6,7 Replacing the fractured ceramic restoration is however not feasible, because it costs more and is less conservative of the natural tooth structure.8,9 Therefore, a direct repair of such restorations has to be considered as a more viable option. The repair procedure involves surface treatment of the fractured ceramic interface to create roughness through mechanical means to increase surface area, such as air abrasion (AA); laser irradiation2,10–13 or chemical means to increase the wettability for bonding, by acid etching with hydrofluoric acid (HFA) and bonding with Silane coupling agents.2,14,15 The treated surface is then ready to receive an adhesive composite resin material to restore the defect.
The various materials and techniques used for the surface conditioning of different types of ceramic materials to optimize the adhesion of the additional resin materials onto the substrate, requires thorough consideration of the chemical nature of both interfaces in order to achieve a durable outcome. 16 In the authors knowledge from indexed literature, limited evidence is available on surface treatment of Lava ultimate hybrid resin ceramics (HRC) with the purpose of repair by composite resin. It is hypothesized that lithium disilicate ceramics (LDC) and HRC surface treated with photodynamic therapy (PDT) and laser using Er,Cr:YSGG (ECYL) will exhibit comparable repair bond strength to conventional surface treated regimes (HFA and AA) with silane. Therefore, the aim of this in-vitro study was to compare the repair bond strength and surface topography of LDC and HRC with different surface treatments including low level laser therapy (LLLT), PDT, HFA and AA.
Material and method
Specimen preparation
The existing study followed CRIS (Checklist for reporting In-vitro studies) guidelines and was conducted at Specialist Dental Research Centre and was completed in 1 month duration. Forty specimens of LDC (IPS Emax Press; Ivoclar/Vivadent, Schaan, Liechtenstein) and HRC (3M™ Lava™ Ultimate CAD/CAM Restorative for CEREC) in the form of discs were prepared from prefabricated blocks. Prepared discs had a diameter of 6mm and thickness of 2 mm. All the discs were disinfected using 96% isopropanol for 180 s and air dried. Based on different surface conditioning regimes, LDC and HRC prepared discs were randomly allocated into eight groups (
Specimens in groups 1 to 4 and groups 5 to 8 belonged to LDC and HRC materials respectively. Specimens discs from group 1 and 5 were conditioned using Er,Cr:YSGG (ECYL) wavelength 2790 nm (Millennium; Biolase Technology, Inc., San Clemente, CA, USA) at 30 Hz frequency and power 3.75 W, pulse rate of 140 µs with a 300 µm sapphire tip, MZ8 in a non-contact position perpendicular to conditioning surface in a circular motion for a duration of 120 s. Air water ratio was maintained at 65% to 35%.
Samples from group 2 and 6 were surface treated with PDT using methylene blue photosensitizer (MBPS). MBPS was used at a concentration of 100 mg/L (Qualigens Fine Chemicals, Mumbai, India) and was activated by means of a diode laser (ADV Laser, Picasso, Italy) at power 1.5 W and wavelength 810 nm. The fibre optic tip diameter of 150 µm was placed perpendicular to the surface and was moved over the ceramic surface for a duration of 30 s.
Sample discs from group 3 and 7 were conditioned using conventional 9.6% HFA gel (Ivoclar, Vivadent, Germany) for 60 s, rinsed with strong jet of air water spray for 10 s and air dried.
Specimens from group 4 and 8 were surface treated with aluminium trioxide (Al2O3) silicate particles (CoJet system; 3M ESPE, St. Paul, MN) air abrasion (AA) for 10 s at 2 bar pressure from a distance of 10 mm using an intraoral sandblaster. After AA all specimens were washed under running water and air dried (Table 1).
Means and SD for repair bond strength values among study groups using ANOVA and Tukey multiple comparisons test.
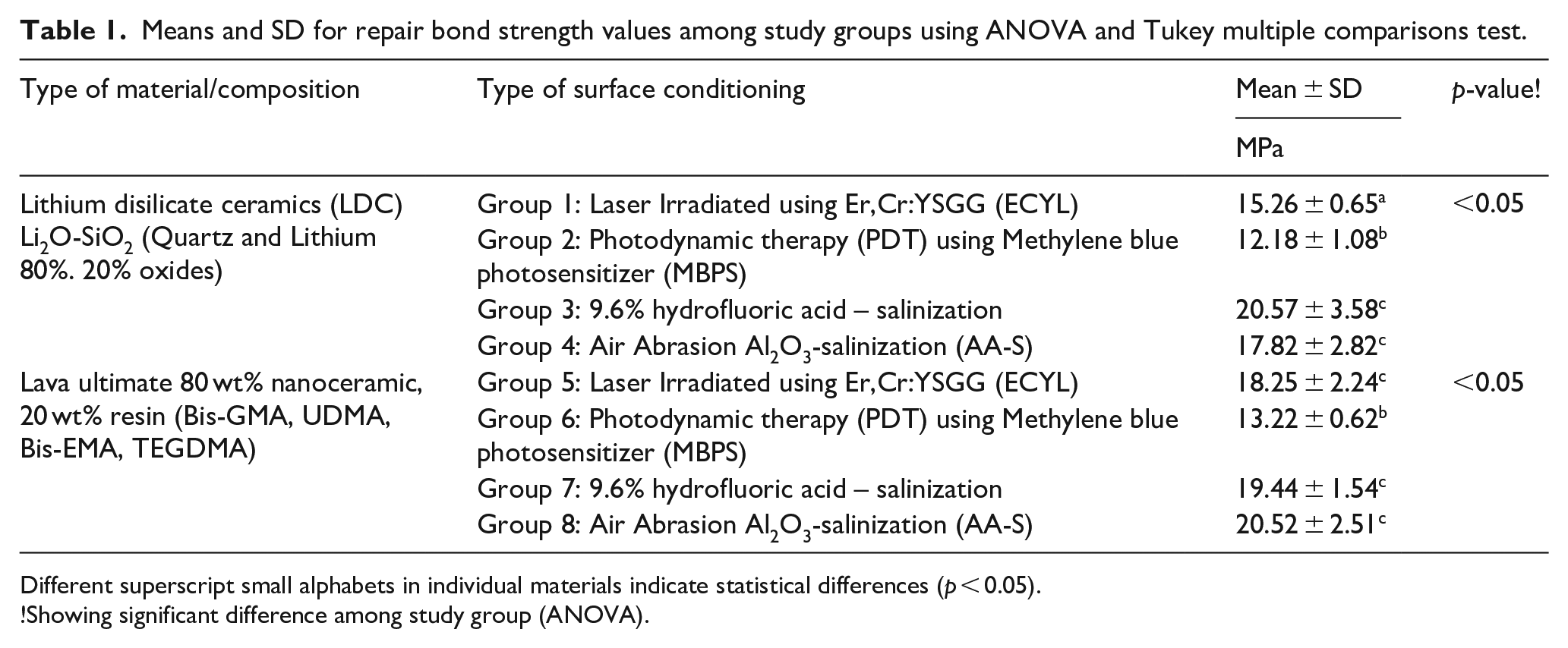
Different superscript small alphabets in individual materials indicate statistical differences (
!Showing significant difference among study group (ANOVA).
Application of repair resin composite
After performing conditioning protocols, samples from groups 3, 4 and 7, 8 received silane application (Monobond Plus ceramic primer Ivoclar vivadent) for 60 s and air dried. Specimens from group 1, 2, 5 and 6 did not receive a coating of silane coupling agent. A Porcelain Repair Kit (Ultradent Products Inc., South Jordan, UT, USA) was used according to manufacturer recommendation in all samples. Peak universal bond adhesive (Ultradent Products Inc., South Jordan, UT, USA) was applied on ceramic surface with a single stroke of brush and light cured for 15 s (Bluephase G2, Ivoclar, Vivadent). Repaired surface was bonded with a composite (Clearfil Majesty Esthetic; Kuraray, Medical, Tokyo, Japan) incrementally (2 mm), using a Teflon mold and cured for 40 s. Bonded specimens were stored in artificial saliva at 37°C for 24 h following placement in thermocycler (Mini Opticon BioRad, USA) with a dwell time of 30 s between 5°C to 60°C.
Shear bond strength testing (SBS) and failure mode analysis (FMA)
A universal testing machine (Lloyds, LF, plus, Ametek Inc, Great Britain, UK) was used to determine the SBS (ASTM D4501-01(2014) of the ceramic disc structure at a crosshead speed of 1 mL/min and 2.5 KN force. The samples were assembled and fixed in a jig and force was applied perpendicular to the bonded surface until repair failure. SBS was measured in Megapascal (MPa). Fractured discs were examined under stereomicroscope (SZX-ILLB100, Olympus Optical Co. Ltd., Tokyo, Japan) by a single investigator at 40× magnification. Failure pattern was classified into adhesive, cohesive and admixed types.
Scanning electron microscopy
Qualitative assessment of surface topography and characteristics for five specimens in each group was performed with SEM. Specimen surfaces were wiped with ethanol and dried. Sputter coating (6 nm thickness) of the specimens with gold powder (JFC – 1100 fine coat ion sputter) at 40 mA for 250 s was performed and specimens were secured in the chamber of Scanning Electron Microscope (JEOL JSM-6360 LV, Tokyo, Japan) with a double-sided carbon tape. SEM images were captured (10 kV) at multiple magnifications (×500, ×1000, ×1500, ×2000 to ×5000) with wide field mode for visual interpretation.
Statistical analysis
Normality of the data was assessed with Kolmogorov-Smirnov and Shapiro-Wilk test. Descriptive statistics that is, Means (MPa values) were compared using one-way analysis of variance (ANOVA) and Tukey’s post hoc test was done for multiple comparison using statistical program for social science (SPSS version 21, Inc., Chicago, US) at a significance level of
Results
Adhesive bond strength
Table 1 presents mean and standard deviation (SD) of repair SBS in MPa. The highest repair bond strength in LDC was observed in group 3 (HFA-S) (20.57 ± 3.58 MPa). Whereas, the lowest bond strength was displayed in group 2 PDT (12.18 ± 1.08 MPa). Similarly, in HRC the highest repair bond strength was found in group 8 (AA-S) (20.52 ± 2.51 MPa) and the minimum repair bond values were presented by group 6 PDT specimens (13.22 ± 0.62 MPa).
In LDC repair bond strength of group 3 (20.57 ± 3.58 MPa), group 4 (17.82 ± 2.82 MPa) were found to be comparable (
Moreover, based on conditioning regimes, group 2 of LDC (12.18 ± 1.08 MPa) and group 6 of HRC (13.22 ± 0.62 MPa) treated with PDT exhibited the lowest SBS. In addition, group 3 and group 7 surface treated with HFA-S, group 4 and group 8 surface conditioned with AA-S and group 5 surface treated with ECYL were found to be comparable (

Bar graph demonstrating repair bond strength values among different experimental groups.
Among failure types, specimens in groups 1, 2 and 6 showed 60%, 70% and 70% of was adhesive failures respectively (Table 2). Among groups 3, 4, 7 and 8, adhesive failures ranged from 10% to 20%. Among specimens of groups 4, 7, 5 and 3, cohesive failures were 50%, 60%, 70% and 80%, respectively. Surprisingly, specimens in group 8 showed 90% of admixed failures. In addition, groups 4 and 5 exhibited 30%of admixed failures and samples in groups 1, 6 and 7 showed 20% of admixed failures respectively (Table 2). Failure modes are presented in Figure 2(a) to (c).
Number of samples for modes of failure among different experimental groups.

AA-S, abrasion; Al2O3, salinization; ECYL, Er,Cr:YSGG laser; HF, hydrofluoric acid; LDC, lithium disilicate; LU, lava ultimate; PDT, photodynamic therapy; S, silane.

SEM micrographs of lithium disilicate samples within different study groups. (a) (ECYL-30 Hz, 3.75 W) – Lased surface showing patches of locally sintered ceramic due to matrix heating, reorganization and agglomerated LDC crystals (arrow). Shows both micro and macro-irregularities. The irregularities are irregular and non-uniformly spread. (b) (PDPDT-Methylene blue-Diode laser treated) – Increase heat from laser in the presence of methylene blue cause washout of silica matrix around LDC needle like crystals. Resulting in uniform irregularities on the treated surface without the patchy sintered matrix. Porosities are mostly regular and small, with few large size craters (arrow). (c) (HFA treated 9.6%, 60 s) Showing classic etched surface with needle like LDC crystals. Acid dissolution of silica matrix forming craters, shelves and empty areas around crystals. Some showed crevices and sulci (arrows) resulting form silica matrix erosion.
Surface topography
Different surface treatment showed variable effect on material surface. Lithium disilicate specimens treated with ECYL at 30 Hz and 3.75 W power showed patches of locally sintered ceramic due to matrix heating causing it to reorganize and exhibit agglomerated LD crystals (Figure 3(a)). It showed irregularly distributed micro and macro-irregularities. Among PDPDT treated LDC samples with Methylene blue and Diode laser, a washout of silica matrix around LDC needle like crystals was seen due to the heat from laser in the presence of methylene blue. Resulting in uniform irregularities on the treated surface without the patchy sintered matrix. Porosities were mostly regular and small, with few large size craters (Figure 3(b)). Figure 3(c) presents HFA treated (9.6%, 60 s) LDC samples with classic etched surface with needle like crystals. Acid dissolution of silica matrix formed craters, shelves and empty areas around crystals. Some specimens showed crevices from acid erosion of silica matrix.
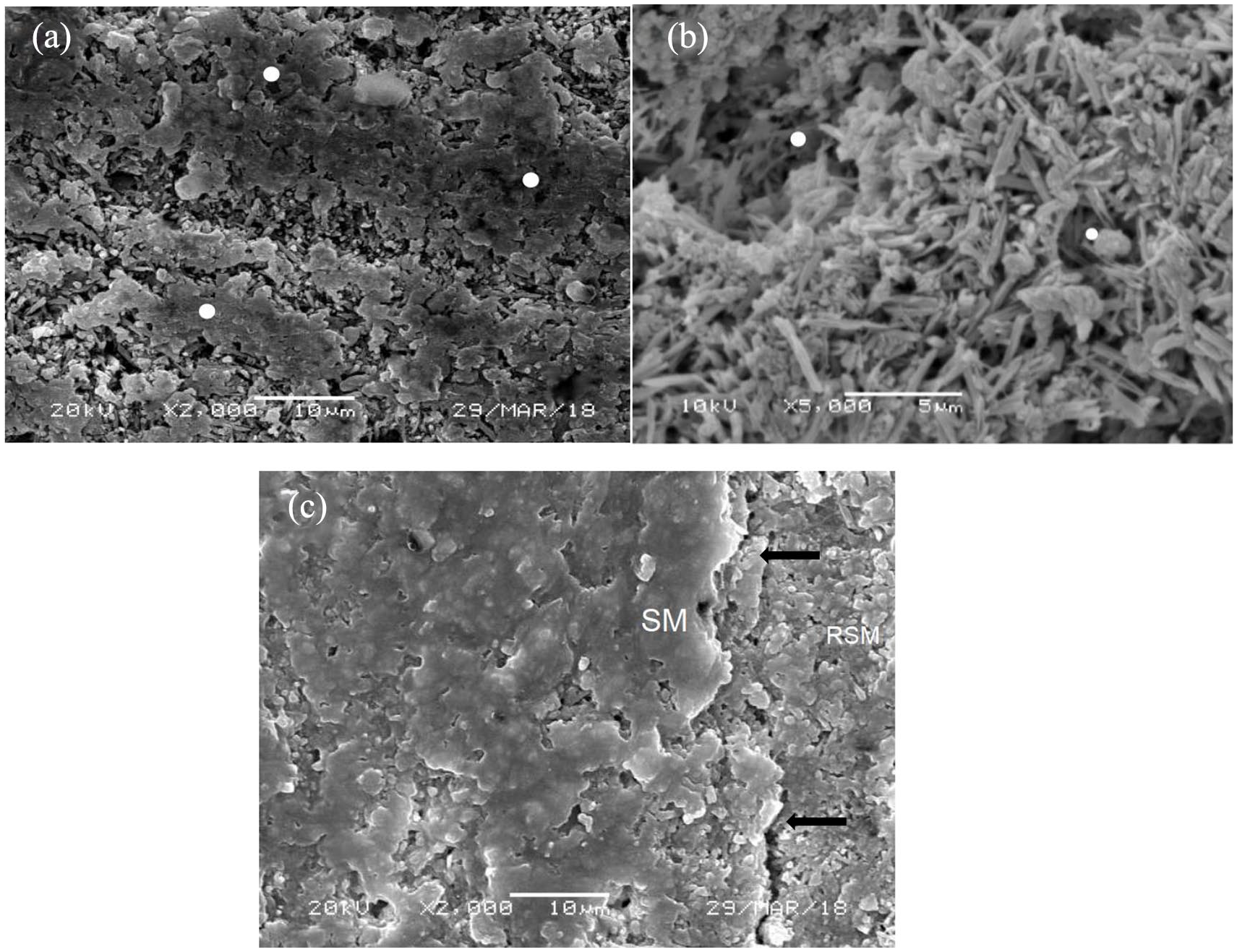
SEM micrographs of Hybrid Resin ceramic (HRC) samples within different study groups. (a) (ECYL-30 Hz, 3.75 W) – Lased surface showing multiple zones of locally sintered HRC (arrows) due to resin matrix heating, reorganization and fusion (arrow). Shows both micro and macro-irregularities. The irregularities are irregular in the form of micr-crust. (b) (PDPDT-Methylene blue-Diode laser treated) – Shows predominant erosion of resin matrix around ceramic crystals. A consistently irregular surface with a high energy but evenly distributed micro rough topography. Dissipation of heat from diode in MB distributes heat through surface, showing lack of fused matrix. (c) (HFA treated 9.6%, 60 s) – Uniformly distributed irregular surface showing resin matrix and ceramic crystals (arrows). Acid erosion causes equal dissolution of matrix around filler particles and allows for consistently similar micro surface topography.
HRC samples surface treated with ECYL showed multiple zones of locally sintered material due to resin matrix heating and fusion. This fusion was higher than LDC samples resulting in both micro and macro-irregularities. The surface irregularities were also present in the form of micro-crust (Figure 4(a)). Figure 4(b) displays PDPDT (Methylene blue-Diode laser) treated specimens, with a predominant erosion of resin matrix. A consistently irregular surface with evenly distributed micro-rough topography was observed. Dissipation of heat from diode in MB distributed heat through surface, showing lack of fused matrix. Specimens conditioned with HFA (Figure 4(c)), exhibited uniformly distributed irregular surface showing resin matrix and ceramic structure. Acid erosion caused equal dissolution of matrix around filler particles consistently similar micro surface topography in samples. In addition, specimens with aluminium trioxide (Al2O3) silicate particles showed consistent moderate porosity and surface with hybrid surface structure

SEM micrographs of Hybrid Resin ceramic (HRC) samples within different study groups. (a) (ECYL-30 Hz, 3.75 W) – Lased surface showing multiple zones of locally sintered HRC (white spots) due to resin matrix heating, reorganization, and fusion. The irregularities are irregular in the form of micro-crust (White arrows). (b) (PDPDT-Methylene Blue-Diode laser treated) – Shows predominant erosion of resin matrix. A consistently irregular surface with evenly distributed micro rough topography (arrows showing similar ceramic crystal exposure and micro-rough topography). Dissipation of heat from diode in MB distributes heat through surface, showing lack of fused matrix (shown with white spot). (c) (HFA treated 9.6%, 60 s) – Uniformly distributed irregular surface showing a combination of resin matrix and ceramic crystals (arrows). Acid erosion causes equal dissolution of matrix around filler particles and allows for consistently similar micro surface topography.
Discussion
The current study was based on the supposition that LDC and HRC surface treated with PDT and ECYL will exhibit comparable repair bond strength to conventional surface treatment regimes, HFA and AA with silane. The hypothesis was partly accepted as ECYL exhibited comparable repair bond strength to HRC. Whereas LDC conditioned with PDT and ECYL unveiled significantly lower repair bond strength to conventional surface treated regimes (HFA and AA) with silane.
Surface conditioning of ceramic is essential to improve the adhesion of repair composite to the ceramic surface. Bond eminence and resilience indicates clinical success of repaired ceramics. 17 In the present study, effectiveness of repair bond performance was assessed through SBS testing. The method is found to be consistent, displays results quantitatively, is cost-effective and can be used for comparative analysis between different experimental groups.18,19
LDC and HRC conditioned by PDT using MBPS displayed the lowest bond strength (12.18 ± 1.08 MPa) (13.22 ± 0.62 MPa) respectively (Table 2). Possible explanation to this outcome is the cationic and hydrophilic nature of methylene blue (MB) which when activated under low level laser therapy (LLLT) results in water sorption deteriorating the bond performance. Moreover, in the existing study MB was used at a high concentration of 100 mg/L. A study by Wainwright et al., 20 asserts that high concentration of MB is directly proportional to water sorption, corroborating to the results of present study.
ECYL conditioning of LDC demonstrated low repair bond strength (15.26 ± 0.65 MPa). However, ECYL surface treatment of HRC revealed comparable bond strength to conventional regimes (18.25 ± 2.24 MPa) (Table 2). ECYL works at a wavelength of 2.78 µm and it is estimated that ECYL is able to remove glass phase and improve micromechanical retention with increase irregularity in crystalline structure of HRC henceforth, improving repair bond strength. 8 Furthermore, HRC consists of 80% filler by weight, to which evidence suggests that greater the filler content, better the adhesive bond values. 21 These finding were found to be in line with the work by Hatipoglu and Barutcigil. 22 Furthermore, poor repair bond values in LDC conditioned with ECYL can be due to heat induction during laser irradiation causing damage to the superficial layer which gives weak attachment to underlying surface and composite resin resulting in repair bond failure. 23 Our findings are similar to the work by Akyil et al. 24
The highest repair bond strength in case of LDC was observed in HFA-S (20.57 ± 3.58 MPa) treated specimens (Table 2). A probable reason to this outcome is the hexafluoro-silicates that are formed as a result of the chemical reaction between the etchant and the glass matrix, which not only provides rough surface for micromechanical retention, but also increase surface energy for the silane coupling agent.24–26 These findings were comparable with the repair bond values in group 4 (AA-S) and also validate the findings in the study by Colares et al. 15 and Ataol and Ergun. 27
The highest repair bond strength in case of HRC was observed in AA-S (20.52 ± 2.51 MPa), followed by HFA-S (19.44 ± 1.54 MPa) specimens (Table 2). A probable description to these conclusions is that aluminium trioxide particle (Al2O3) abrades and roughens the ceramic surface under defined pressure. This indirectly favours undercut formation facilitating silane agents into these grooves improving repair bond strength. 3
In regards to failure mode analysis, adhesive failure was predominant in group 1, 2 and 6. Samples in these groups had low repair bond values which correlates to adhesive failure. 28 Moreover, this type of failure mode is also related to type of substrate, method of SBS testing and mode of conditioning method.10–12 In addition, debonded samples in group 3, 4, 5, 7 and 8 exhibited cohesive and admixed failure types. Available evidence suggests better adhesion is associated with high repair bond values and this method of failure. 29 Moreover, it can be suggested that silane might have played a contributing role in the observed cohesive failures among groups 3, 4 and 7.
Lack of atomic force microscopy (AFM) of treated surfaces of HRC and LDC, along with dispersive spectroscopy of debonded samples, marks one of the possible limitations of the study. Use of photosensitizers at variable concentration with different irradiation protocols needs to be investigated in different clinical settings. Use of thermocycling and aging of specimens along with use of artificial saliva for specimen storage may replicate oral settings and provide better and reliable repair bond strength indicators and information for the clinicians. Therefore, further studies are recommended in this regard.
Conclusion
Mechanical and chemical surface treatments should be used in order to achieve adequate repair bond strength between resin composites and ceramic interface. HFA-S along with AA-S showed predictable bonding outcomes as compared to PDT for both LDC and HRC. Moreover, laser conditioning of HRC using ECYL can be used as a potential alternate to conventional conditioning regimes.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful to the Researchers supporting project at King Saud University for funding through Researchers supporting project No. (RSP-2019-44).
