Abstract
Background
This study evaluated the surface morphology, chemical composition and adhesiveness of lithium disilicate glass ceramic after acid etching with hydrofluoric acid or phosphoric acid.
Methods
Lithium disilicate glass ceramic specimens polished by 600-grit silicon carbide paper were subjected to one or a combination of these surface treatments: airborne particle abrasion with 50-μm alumina (AA), etching with 5% hydrofluoric acid (HF) or 36% phosphoric acid (Phos), and application of silane coupling agent (Si). Stainless steel rods of 3.6-mm diameter and 2.0-mm height were cemented onto treated ceramic surfaces with a self-adhesive resin cement (Clearfil SA Cement). Shear bond strengths between ceramic and cement were measured after 24-hour storage in 37°C distilled water.
Results
SEM images of AA revealed the formation of conventional microretentive grooves, but acid etching with HF or Phos produced a porous surface. Bond strengths of AA+HF+Si (28.1 ± 6.0 MPa), AA+Phos+Si (17.5 ± 4.1 MPa) and HF+Si (21.0 ± 3.0 MPa) were significantly greater than those of non-pretreated controls with Si (9.7 ± 3.7 MPa) and without Si (4.1 ± 2.4 MPa) (p<0.05). In addition, HF etching alone (26.2 ± 7.5 MPa) had significantly higher bond strength than AA alone (11.5 ± 4.0 MPa) (p<0.05). AA+HF, AA+Phos and HF showed cohesive failures.
Conclusions
Etching with HF or Phos yielded higher bond strength between lithium disilicate glass ceramic and self-adhesive resin cement without microcrack formation.
Introduction
For today's dental patients, aesthetic appearance matters to them as much as functionality. All-ceramic restorations, in the form of inlays, onlays, veneers, crowns or fixed partial dentures, have ubiquitously pervaded the dental field because they are metal-free and offer high aesthetic appeal (1). To meet different clinical situations, different types of dental ceramics of varying chemical compositions have become commercially available, such as leucite, lithium disilicate, alumina, magnesia and zirconia (2–3–4–5). Heat-pressed or computer-aided design and computer-aided manufacturing (CAD/CAM) fabricated lithium disilicate glass ceramics are preferred over other types of dental ceramics because of these superior characteristics: excellent mechanical properties, accuracy of fit and optical properties that mimic those of natural teeth (6–7–8–9–10).
Adhesively bonded lithium disilicate glass ceramic restorations have a higher load-bearing capability than those conventionally cemented (9, 11). This is because adhesive resin cements improve their bond strength and fracture toughness (12, 13). Before adhesive bonding, it is important that the ceramic substrate be applied with an optimum surface pretreatment method as a prerequisite to good bonding (14). After pretreatment, varying degrees of microporosity or irregularity are formed on a ceramic surface according to its microstructure and composition (15). The topography of pretreated ceramic surfaces significantly influences the formation and maintenance of mechanical and chemical bonds (15). Therefore to ensure good bonding, manufacturers of ceramic systems frequently specify both the luting cement and the recommended surface pretreatment for the ceramic substrate (16).
Surface pretreatment can be achieved by chemical modification (such as silane coating and acid etching) (17, 18) and/or mechanical methods (such as airborne particle abrasion with alumina) (16, 19–20–21). Airborne particle abrasion with alumina has been proven to be effective in ceramic surface roughening and is generally used in the dental field. However, it is difficult to regulate the operating air pressure or adjust the abrasive blasting direction. Consequently, excessive airborne particle abrasion results in microcrack formation on and chipping of ceramic surfaces. Chemical treatment by acid etching is a comparatively less aggressive and safer method to change the microstructure of ceramic surfaces. Hydrofluoric acid etching is a method known to be effective in achieving strong durable bonds with glass ceramics. Hydrofluoric acid attacks the silica phase and yields a topographic pattern which favors resin bonding (14, 22, 23). On the other hand, hydrofluoric acid is classified as a hazardous substance. When hydrofluoric acid diffuses into cells, it can kill them by disrupting their metabolism and cause severe burns (24). Hence, hydrofluoric acid should not be used for intraoral procedures, despite its efficacy in yielding good bond strength (24–25–26).
Both airborne particle abrasion and hydrofluoric acid etching have inherent flaws that render them unsuitable for clinical use. While a microrough surface is desirable, aggressive pretreatment may result in strong and deep degradation of the ceramic and weaken the ceramic itself. Fractures in ceramics can be initiated by a microcrack formation in the crystalline phase, which in turn has probably been weakened by the pretreatment process. An alternative that reduces surface degradation is hydrofluoric acid etching with 1.23% acidulated phosphate fluoride gel (23, 27). Although this method can produce the characteristic rough surface similar to that yielded by hydrofluoric acid etching on feldspathic porcelain, it produces a less prominent etching pattern (28–29–30). To achieve utmost bond strength without material weakness, a clinically safe and effective pretreatment on lithium disilicate glass ceramics remains to be realized.
Phosphoric acid etching has emerged as a popular intraoral surface pretreatment method for tooth substrates and restorative materials (31). Like etching by hydrofluoric acid and acidulated phosphate fluoride, phosphoric acid etching is able to produce clean, high-energy and microrough surfaces intraorally. Microporosity created by phosphoric acid etching also provides micromechanical retention, but might be weaker than that rendered by hydrofluoric acid and acidulated phosphate fluoride. To circumvent the use of caustic agents such as hydrofluoric acid and acidulated phosphate fluoride, phosphoric acid seems like a good alternative etchant for ceramic surfaces.
To obtain clinically safer and optimum surface treatment methods, the purpose of this study was to evaluate the effects of 2 different types of acid etching on the morphology and characteristics of lithium disilicate grass ceramic, and the bond strength of lithium disilicate glass ceramic luted with a resin cement.
Methods
Lithium disilicate glass ceramic specimens
The lithium disilicate glass ceramic material used in this study was IPS e.max Press (Ivoclar Vivadent, Schaan, Principality of Liechtenstein). It had the following chemical composition in % mass: SiO2 57.0-80.0, Li2O 11.0-19.0, K2O 0.0-13.0, P2O5 0.0-11.0, ZrO2 0.0-8.0, ZrO 0.0-8.0, other oxides 0.0-10.0 and coloring oxides 0.0-8.0 (Tab. I). A total of 120 lithium disilicate glass ceramic specimens were prepared, each measuring 6 mm in diameter and 3 mm in thickness. The surface of each specimen was polished with 600-grit silicon carbide abrasive paper (Struers A/S, Rodovre, Denmark) under water irrigation until standardized surface roughness was achieved. Each finished specimen was embedded in a clear acrylic resin cylinder.
Materials used in this study

Surface treatment methods
Embedded specimens were randomly divided into 3 groups (Tab. II). The first 2 groups were etched with either phosphoric acid (Phos) (n = 40) or hydrofluoric acid (HF) (n = 40). The third group comprised the non-acid-etched controls (Control) (n = 40). Each group was subdivided into 4 groups: airborne particle abrasion (AA) and silane treatment (Si), airborne particle abrasion only, silane treatment only, or no surface treatment (Tab. II). Where applicable for a particular group of specimens, surface treatments on the bonding surface were carried out in this order: airborne particle abrasion, then acid etching, followed by silane treatment.
Experimental conditions for each group

AA = airborne particle abrasion; Phos = phosphoric acid etching; HF = hydrofluoric acid etching; Si = silane treatment.
- indicates without that treatment.
Using a grit blaster (Hi-Blaster III; Shofu, Kyoto, Japan) with the nozzle positioned 5 mm from the lithium disilicate glass ceramic surface, airborne particle abrasion using 50-μm aluminum oxide particles (Al2O3; Perlablast® Micro; Bego, Bremen, Germany) was carried out at a pressure of 2.8 MPa for 10 seconds. After grit blasting, specimen surfaces were ultrasonically cleaned for 2 minutes followed by drying with an air stream.
Acid etching was carried out with either 36% phosphoric acid (K-etchant Gel; Kuraray Noritake Dental Inc., Tokyo, Japan) or 5% hydrofluoric acid (IPS Ceramic Etching Gel; Ivoclar Vivadent). Phos was applied for 30 seconds or HF for 20 seconds on the specimen surface, followed by AA. Etched surfaces were thoroughly rinsed with water to remove etchant, then gently blown dry using an air syringe.
After AA and/or acid etching surface treatment(s) where applied to each group, a silane coupling agent (Clearfil Ceramic Primer; Kuraray Noritake Dental Inc.) was applied in a thin layer to the entire specimen surface using a disposable sponge.
Bonding procedure
After a gentle blow-dry with oil-free air, a stainless steel rod (3.6-mm diameter, 2.0-mm height) was cemented onto the treated ceramic surface using an auto-mixed, dual-cure, self-adhesive resin cement (Clearfil SA Cement; Kuraray Noritake Dental Inc.). Cementation was carried out using a Teflon mold jig, where a 10-N load was applied and maintained for 3 minutes. Bonded specimens were light-cured for 20 seconds each from the 2 opposite sides.
Shear bond strength test
All bonded specimens were immersed in distilled water (37°C ± 2°C) for 24 hours before the shear strength test. Each specimen was placed in a shear test fixture (Fig. 1), and shear bond strength was measured using a universal testing machine (Autograph AG-X; Shimadzu, Kyoto, Japan). Up to a maximum of 490 N, an external shear force was transmitted by a flat, blunt, 1-mm-broad shearing edge at a crosshead speed of 0.5 mm/min, at a 90-degree angle to the direction of the load and at the back of the loading plate. Stress at failure was automatically calculated and recorded as the shear bond strength via the accessory software.
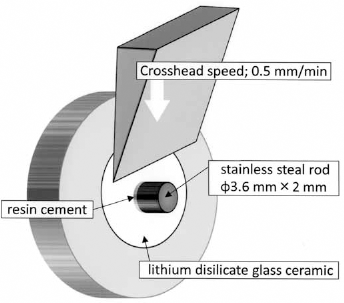
Schematic picture of shear bond strength test.
After debonding, the fractured surfaces of ceramic specimens were examined under a light microscope. Failure caused by shear fracture was classified into 1 of the following 3 types: (i) adhesive failure between resin cement and ceramic, (ii) cohesive failure within resin cement or (iii) mixed-mode failure (adhesive-cohesive).
Shear bond strength data for each group of lithium disilicate glass ceramic specimens were statistically compared through 1-way analysis of variance (ANOVA) and Scheffé post hoc test within 5% error limits (p<0.05) using IBM SPSS Statistics (SPSS Inc., Chicago, IL, USA).
Morphological and chemical analyses
To detect and observe morphological and chemical composition changes in the pretreated surfaces, scanning electron microscopy (SEM), X-ray powder diffraction (XRD) and energy-dispersive X-ray spectroscopy (EDXS) were carried out.
SEM analysis (DS-720; Topcon Corp., Tokyo, Japan) was carried after the specimen was dried in a desiccator for 24 hours and then gold-coated using an ion sputter (JFC-1100E; JEOL, Tokyo, Japan). XRD analysis (CuKα1 radiation, λ = 1.5406 Å; RINT-2500; Rigaku, Tokyo, Japan) was carried out under these operating conditions: 40 kV acceleration, 200 mA current and scanning rate of 0.02° per second for 2θ/θ scan. EDXS analysis (Voyager III M3100; NORAN Instruments Inc., Middleton, WI, USA) was carried out to analyze the element distributions on the pretreated surfaces.
Results
Shear bond strength
Shear bond strengths of HF-treated groups – AA+HF+Si (28.1 ± 6.0 MPa), AA+HF (27.7 ± 6.2 MPa), HF+Si (21.0 ± 3.0 MPa) and HF (26.2 ± 7.6 MPa) – were significantly higher than those of control+Si (9.7 ± 3.7 MPa) or the control (4.1 ± 2.4 MPa) (p<0.05) (Tab. III). There were no incidences of adhesive failure in the HF-treated groups (Tab. III). Interestingly, HF+Si showed a lower shear bond strength than treatment by HF alone. This tendency was not observed for Phos and AA pretreatment groups.
Mean shear bond strengths and failure modes of the pretreatment groups
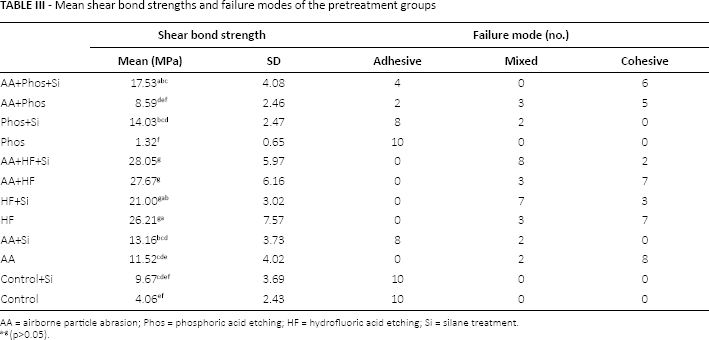
AA = airborne particle abrasion; Phos = phosphoric acid etching; HF = hydrofluoric acid etching; Si = silane treatment.
(p>0.05).
Phos treatment alone exhibited the lowest shear bond strength (1.3 ± 0.7 MPa) among all of the pretreatment groups (Tab. III). All specimens treated by Phos only exhibited adhesive failure, as was the case for the control group with and without silane treatment (Tab. III). In Phos-treated groups, silane treatment (Phos+Si) – which exhibited 14.0 ± 2.5 MPa – was needed to increase the bond strength to lithium disilicate glass ceramic.
AA treatment had to be combined with both Phos (AA+Phos; 8.6 ± 2.5 MPa) and silane treatment (AA+Si; 13.2 ± 3.7 MPa) to achieve a significant increase in shear bond strength over AA only (11.5 ± 4.0 MPa) (p<0.05).
Morphological changes
SEM-acquired secondary electron images revealed morphological changes according to surface pretreatment method. Compared with the non-pretreated control (Fig. 2A), Phos treatment did not result in significant visible change to the ceramic surface (Fig. 2B), but HF produced a porous surface (Fig. 2C). AA caused the formation of microretentive grooves (Fig. 2D), and AA combined with acid etching produced deeper microretentive grooves (Fig. 2E, F). Figure 2E shows that when AA was followed by Phos treatment, a slightly porous structure was produced.

Secondary electron images of lithium disilicate glass ceramic surfaces according to surface pretreatment (magnification ×3,000 and ×10,000): (
The backscattered electron image of non-pretreated control (Fig. 3A) shows both high and low contrast areas on the ceramic surface. With Phos treatment only (Fig. 3B), the high-contrast area became slightly decreased when compared with the control (Fig. 3A). As for the air-abraded surface (Fig. 3D), it showed a similar ratio of contrasts to the control (Fig. 3A). For HF only (Fig. 3C) and AA followed by HF (Fig. 3F), mainly the high-contrast area was shown. AA followed by Phos (Fig. 3E) caused the low-contrast area to increase when compared with AA only (Fig. 3D).
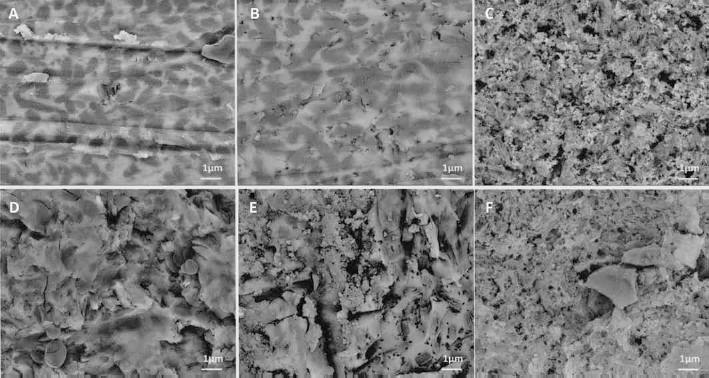
Backscattered electron images of lithium disilicate glass ceramic surfaces according to surface pretreatment (magnification ×10,000): (
Chemical composition changes
The ceramic surface was mainly composed of Li2Si2O5, Li2SiO3 and Li3PO4 (Fig. 4). When compared with the non-pretreated polished surface, there were no differences in chemical composition after acid etching with either HF or Phos (Fig. 5).
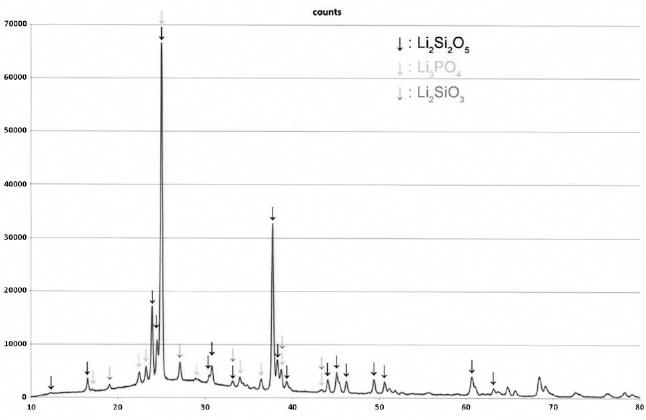
X-ray powder diffraction (XRD) analysis of lithium disilicate glass ceramic surface.
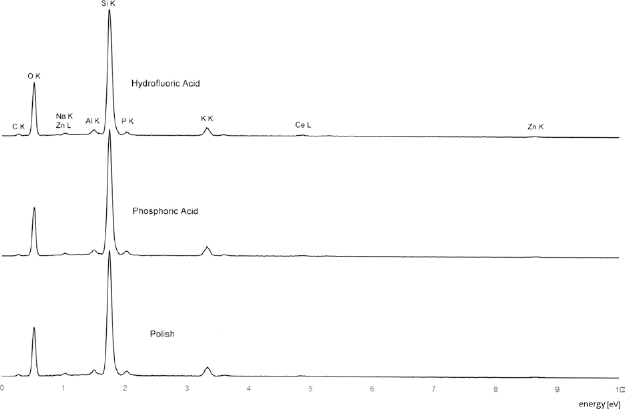
Energy-dispersive X-ray spectroscopy (EDXS) analysis of pretreated lithium disilicate glass ceramic surface.
Discussion
Clinical performance of ceramic restorations relies heavily on the adhesion of the resin cements to restorative materials and tooth substrates. To improve the bond strength of ceramic surfaces to resin cements, methods such as roughening with airborne particle abrasion or acid etching have been developed and advocated (23). Actual bond strength between a ceramic material and resin cement is a very complex issue because it involves myriad factors, such as microstructure of ceramic surface, viscosity of resin cement and mechanical properties of ceramic material. Different surface pretreatment methods may also produce different topographic changes on different types of ceramic materials, hence yielding different bond strength results. Although an aggressive pretreatment on ceramic surfaces produces the highest bond strength, a deep and strong microroughness formed by airborne particle abrasion or hydrofluoric acid etching may result in surface degradation and yield compromised ceramics with microcracks. Therefore, a safer ceramic pretreatment method needs to be developed to achieve utmost bond strength without material weakness.
To enhance resin–ceramic bonding in the present study, a silane coupling agent was applied on the lithium disilicate glass ceramic surface so that the reaction with the latter's silica ingredient would produce chemical covalent bonds (14, 21). Results showed that silane treatment provided higher bond strength than the control group without surface pretreatment. However, all specimens treated with silane coupling agent alone showed adhesive failure.
In the present study, airborne particle abrasion produced a microstructure which promoted mechanical retention to the lithium disilicate glass ceramic surface. The AA group saw an increase in bond strength and a higher incidence of mixed-mode failures when compared with the control group without surface pretreatment. A combined treatment regime of air abrasion and silane coupling agent application led to higher bond strength than with each treatment alone. However, the resultant bond strength was not an outcome of synergism. A higher incidence of adhesive failures was seen in the AA+Si group when compared with the AA group.
Acidic etchants attack the glassy phase of ceramic materials and expose silica oxides, yielding topographic changes that increase micromechanical retention and improve chemical bonding with silane coupling agents and resin-based luting cements (14). Surface topographies produced after etching with phosphoric or hydrofluoric acid were markedly different from that produced after air abrasion. SEM results revealed that hydrofluoric and phosphoric acid etching created different etching patterns. Hydrofluoric acid readily reacted with lithium disilicate glass ceramic and dissolved a significant amount of surface material, producing a porous surface. However, etching with hydrofluoric acid alone failed to create the microcrater-like appearance which was characteristic of air-abraded surfaces. On the other hand, phosphoric acid etching only modified the surface superficially to produce a minimally roughened surface.
Hydrofluoric acid is an aggressive etching agent on silica-based ceramic surfaces. It selectively dissolves the glassy or crystalline components of these ceramics and produces an irregular porous surface (14). Porous surfaces increase the surface area and thus the penetration of resins into microretentive spaces formed on the etched ceramic surfaces (32). The vastly different surface topographies thus accounted for the markedly higher bond strength exhibited by hydrofluoric acid etching alone as compared with phosphoric acid alone. The fluctuation of low- and high-contrast areas detected in this study indicated that these contrast areas should be silicone and lithium oxide areas, respectively. Hence, the low-contrast area increased with acid etching because silicone oxide is easier to dissolve than lithium oxide.
Although chemical changes after acid etching were not detected through XRD and EDXS analyses, hydrofluoric acid etching pronouncedly increased the bond strength of lithium disilicate glass ceramic, independent of any combined surface treatment(s) with silane coupling agent and/or airborne particle abrasion. No adhesive failures occurred in any of the 4 HF-treated groups. However, silane treatment after hydrofluoric acid etching decreased the shear bond strength and resulted in predominantly mixed-mode failures. When hydrofluoric acid etching was used in combination with air abrasion and silane treatment, mixed-mode failure was also the predominant failure mode, although bond strength yielded by the AA+HF+Si group was not statistically different from that of the HF group. Hydrofluoric acid etching produced insoluble silica-fluoride salts which could remain as by-products on the ceramic surface and interfere with resin-ceramic bonding (33). These fluorine-containing by-products deposited on the ceramic surface prevented the silane coupling agent and/or luting resin to fully penetrate the porous ceramic surface created by hydrofluoric acid etching. They also weakened the binding effect between alkoxy group in silane coupling agent and the 10-methacryloxydecyl dihydrogen phosphate (10-MDP) adhesive monomer of the luting cement used in this study, resulting in lower bond strength.
Interestingly, silane coupling agent exerted vastly different effects on phosphoric acid-etched and hydrofluoric acid-etched surfaces. Compared with the AA+Phos group, a synergistic outcome was apparently realized in the Phos+Si group with a significant increase in bond strength and a decrease in adhesive failures. Phosphoric acid etching did not produce any phosphorus-containing compounds to be deposited on the surface, or these by-products did not have a complex structure and could be easily washed off from the phosphoric acid-etched surface. Therefore, unlike the case of hydrofluoric acid etching, the binding between silane alkoxy group and the 10-MDP adhesive monomer was not hindered.
Hydrofluoric acid is a strong, corrosive and a highly toxic chemical. Hence, it was an encouraging finding that phosphoric acid could be a potential etchant which increases the bond strength of lithium disilicate glass ceramics. Phosphoric acid etching is also a good alternative to airborne particle abrasion which poses a high risk of producing ceramic frameworks with edge chipping and microcracks. When phosphoric acid etching is combined with silane treatment to improve resin–ceramic bonding, only a thin coating of silane coupling agent needs to be applied on the ceramic surface.
Conclusions
Within the limitations of the present study, the following conclusions were drawn:
Etching with hydrofluoric acid or phosphoric acid resulted in higher bond strength between lithium disilicate glass ceramic and self-adhesive resin cement.
Although hydrofluoric acid etching produced more reliable and consistent bond strength results than phosphoric acid, phosphoric acid is a less hazardous substance which produced good bond strength results when etching was accompanied with silane treatment.
Footnotes
Financial support: This work was partially supported by a Grant-in-Aid for Scientific Research (KAKENHI), grant number 15K11159, from the Japan Society for the Promotion of Science (JSPS).
Conflict of interest: None of the authors has any financial interest related to this study to disclose.
