Abstract
Background:
Biocompatible materials are used for treatment of blood circulatory system diseases, especially abdominal aortic aneurysms. The most popular and often used are knitted and polymer vascular patches. The aim of this study was to optimize the manufacturing process of implantable materials, ensuring antibacterial activity useful for treating abdominal aorta aneurysms.
Methods:
The vascular patch was manufactured from Trevira® yarn. The parameters of the intermediate product and vascular patch were tested according to standard procedures.
Results:
The vascular patch, manufactured from microsilver-containing yarn, with crimps on the surface of the patch, has been found useful for treatment of abdominal aorta aneurysms. Introducing crimps on the surface of the patch resulted in reduction of water permeability and enabled cutting of the graft at various angles without fraying at the cut ends of the biomaterial. The final vascular patch was marked by a gradual release of silver within 48 hours.
Conclusions:
On the basis of the performed test, it has been demonstrated that an implantable material for the treatment of abdominal aorta aneurysms was obtained, and that it can be considered as an alternative for currently used vascular patches. The final vascular patch was marked by a gradual release of silver during the first period of incubation. The antibacterial properties of the final product were confirmed by observation of a significant reduction in the number of Staphylococcus aureus and Klebsiella pneumoniae bacterial colonies.
Keywords
Introduction
One of the fields in modern material engineering is the search for new materials useful for manufacturing advanced biomaterials, including biomaterials used for the treatment of blood circulatory system diseases. The fact that these types of illness are the cause of death of 7 million patients worldwide each year should be emphasized. One of these lethal blood circulatory system diseases is abdominal aortic aneurysm. First records regarding wrapping abdominal aortic aneurysms date back to the 1940s. 1 Poppe wrapped the frontal–lateral surface of the aneurysm using cellophane; this strengthened the aneurysm’s wall and inhibited its expansion. 2
The development of science, in both material engineering (structure, type, and construction of the chosen material) and treatment methods of abdominal aorta aneurysms, allows for the choice of the most appropriate treatment method for each patient (personalized medicine). Among the new methods of treatment currently being used, there are methods involving implantation of bifurcated and fenestrated stent-grafts.3–6 An important feature in this case is the construction of a stent with the properties required for reducing the risk of aneurysm rupture. 6 Other methods might be considered as the synergy between right choice of material, treatment method, and tissue engineering. Using materials that had been implanted with appropriate cells prior to the surgery influenced the compatibility of the material and, as a consequence, reduced the risk of complications during and after surgery.7–11 Nevertheless, resection surgeries of aneurysms or aneurysm ‘wrapping’ procedures are still carried out. The methods mentioned are performed to protect patients from rupture of the aneurysm, especially in the case of high-risk patients, where there is a high mortality rate. At present, vascular prostheses and patches are used as a straight piece of knitwear of the required dimensions. The most popular are materials manufactured out of polyester fibers (Dacron®) or polytetrafluoroethylene. The biocompatibility of polytetrafluoroethylene and Dacron has been proved in many in vivo and clinical tests.12–17 Apart from biologically stable materials (practically non-degraded in an organism), biodegradable materials are increasingly being used. The best known are: polylactic acid and its copolymers, poly-ε-caprolactone, poly(glycolic acid), alginates, or chitin derivatives.18–24 However, in the case of these polymers, there is no data showing their beneficial effect in manufacturing vascular grafts or long-term evaluation after surgery. Analysis of currently used vascular grafts shows that the patches are manufactured with the use of two different textile technologies: weaving and knitting. Application of weaving technology produces a product with very low water permeability. In the first period after implantation (1–3 months), this feature seems to have an advantage; however, during healing-in or overgrowing with tissue it has a clear disadvantage (>3 months). Products manufactured using a knitting technique might reproduce the features of natural blood vessels.25–27 They are characterized by sufficient strength, excellent elasticity, and no fraying of the ends. The high biocompatibility of products manufactured using a knitting technique led to an excellent healing-in and good clinical results in long-term observation. However, the safety of the materials used was threatened by bacterial infection of the implanted vascular prostheses. 28 The most commonly used solution is based on the application of materials modified with classic antibiotics, 29 which is unfavorable from the point of view of bacterial resistance or nano- or microsilver modifications. 30 There is no vascular patch on the market intended for wrapping coronary and abdominal arteries. Until now, the desired effect has been obtained by cutting a vascular prosthesis into smaller fragments and sewing them together to create a graft of the correct diameter. On the market there are available: knitted products (Vascutec Inc., Jotec, Atrium, InterGard, B. Braun) and polymer products from Cormatrix and Synovis.
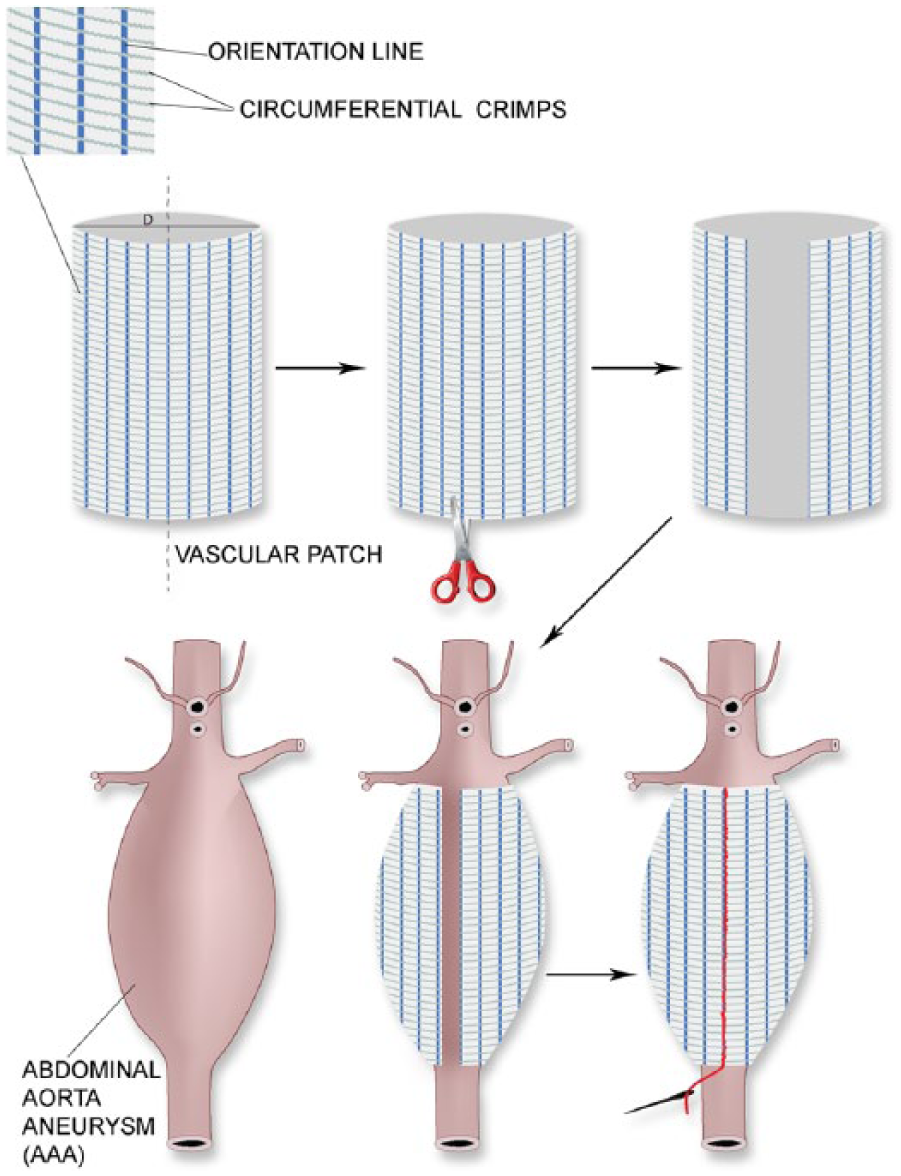
The aim of this study was to optimize the manufacturing process of implantable materials useful for the treatment of abdominal aortic aneurysm. The proposed materials will be used in the wrapping technique of aorta aneurysm treatment. Figure 1 illustrates how the proposed solution would be applied.
Wrapping abdominal aorta aneurysm using the proposed vascular patch.
In the case of the proposed non-sealed vascular patch, the crucial points were: the features of the material and the construction parameters of the device, strictly related to the medical procedure.
The proposed vascular patch has to possess a microfibrous structure appropriate for tissue overgrowth, adequate elasticity, and optimal dimensions and size, which will be retained unchanged during the healing-in process. Moreover, in the case of this implant, an important issue will be the possibility of surgically adjusting the graft. The adjustment could be performed by cutting the knitted fabric at different angles and directions, thereby avoiding fraying of the cut ends of the graft, which could lead to inflammation processes.
Materials and methods
Manufacture of the vascular patch
The trade product Trevira® (Germany) was used to manufacture vascular patches. Trevira® is a polyester yarn modified with microsilver, ensuring antibacterial properties. The characteristics of the yarn were tested previously 31 and the obtained results allowed the use of this material to manufacture a final patch, which was formed using a warp knitting machine, giving an intermediate product for the manufacture of vascular patches. Comparison studies of physical and chemical properties of three different commercially available yarns used for manufacturing textile implants have demonstrated that the crucial properties for medical application are chemical purity and good mechanical properties of the applied materials.
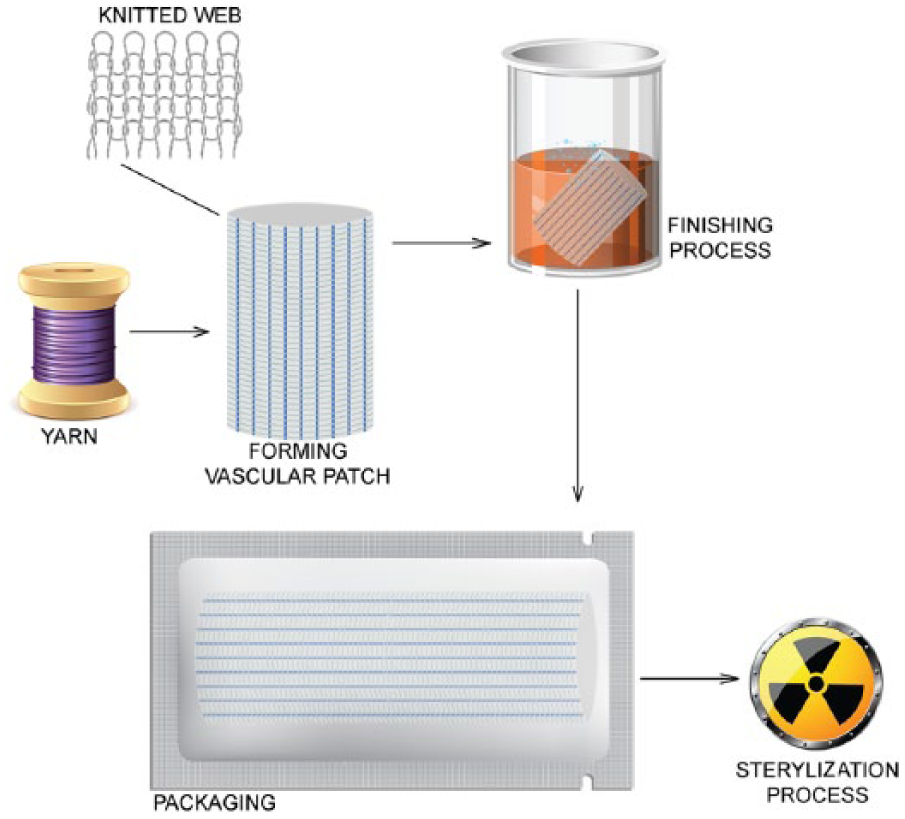
In the next step, cylindrical knitted fabric was modified by the incorporation of circumferential crimps according to their recommended manufacturing technology. 32 The last manufacturing step involved removing solid contamination in a thermal stabilization process. After drying the knitted fabric, standard organoleptic control and packing measures were taken. The final stage of the production process of the vascular patches was radiation sterilization with a 25 kGy dose. The production process for the vascular patches is outlined in Figure 2.
Vascular patch manufacturing process.
Methods of testing the vascular patch
Chemical structure of the material
The chemical structure was analyzed using Fourier transform infrared spectroscopy (FTIR). The tests were carried out using an FTIR spectrometer equipped with an attenuated total reflection (ATR) accessory module, at a wavelength range from 600 cm−1 to 4000 cm−1. An FTIR NIR JASCO V-57 PC spectrometer with an Ulbricht sphere was used.
Thermal properties and crystallinity degree
Tests of thermal properties and crystallinity degree were performed in accordance with the requirements of PN-EN ISO 10993-18:2006 and ISO 10993-19 norms, using a differential scanning calorimetry ‘Diamond’ Perkin-Elmer device under the following parameters: atmosphere, nitrogen, 20 cm3/min; standard Al dish; heating rate: 20 °C/min; cooling rate: 20 °C/min, temperature range: 20–280 °C; weight of the sample: 5–7 mg. The instrument was equipped with an Intracooler II cooler and ‘Paris’ firmware managing software, the software being used for both managing the device and analysis of the results. The analyses were performed with the following method: heating I (temperature from 20 °C to 260 °C), cooling (temperature from 260 °C to −20 °C); heating II (temperature from 20 °C to 260 °C). Determination of transformation temperature was based on maximum and minimum peaks. Heat of transformation was determined by integration of peak. The measurements conditions were adjusted to the composition of the sample (polyethylene terephthalate).
Microscopic structure
Microscopic structure was analyzed using a Nova NanoSEM 230 scanning electron microscope manufactured by FEI (field emission gun) with an X-ray EDS Apollo 40 SDD microanalyzer manufactured by EDAX and an optical microscopic (Delta Optical Genetic Pro).
Water vapor permeability
The gravimetric modified cup method based on GB/T1037-88 was used to determine the water vapor transmission of yarns.
Density of the knitted fabric
The density of the knitted fabric was determined from a count of meshes per 1 cm2 under optical microscope examination, using a Delta optical microscope equipped with Smart Analysis 2.0 software.
Contact angle
The contact angle was determined using KRUSS, DSA 100.
Chemical parameters
Chemical parameters were determined based on tests applied in accordance with appropriate requirements for biomaterials: PN-EN ISO 3071 for establishing the pH, PN-P 04896 for permanganate oxidization ability; PN-P 04990 for ultraviolet absorbance at a wavelength 230 and 245 nm−1; PN-P 04781-03 for determination of Cl− ions; PN-P 04781–04 for determination of SO42− ions; PN-P 04992 for determination of NH4+ ions; PN-P 04607 for determination of compounds soluble in isopropyl alcohol. In all cases, water extracts from tested materials were used.
Release of silver ions
To determine silver ion release from the vascular patches, extracts from the knitted fabric were obtained. For this purpose, approximately 20 g of the knitted fabric products was immersed in 10 ml of extraction solution. The extraction solution used was phosphate buffered saline (pH, 7.4) and a 0.45% solution of HNO3. The extraction was conducted under dynamic conditions, at 37 ± 1 °C, in accordance with PN-EN ISO 10993-12. The silver concentration in the extracts was established according to PN-EN ISO 15586.
Silver content
Silver content was analyzed according to the standard procedure by using an atomic absorption spectroscopy method (Thermo Scientific iCE 3500) recommended for the determination of metal ion concentration in a polymer matrix.
Antibacterial activity tests
To evaluate the antibacterial properties of vascular patches, antibacterial activity tests were performed in accordance with PN-EN ISO 20743 for the Staphylococcus aureus ATCC 6538 and Klebsiella pneumoniae ATCC 4352 bacterial strains. Suspensions with densities from 1.0 × 105 to 3.0 × 105 CFU/ml of both bacteria species were prepared. The test samples were infected with 0.2 ml of the suspension for each sample. Directly after infecting all tested samples (samples containing antibacterial additives and control samples), 20 ml of a neutralizer was added. The samples were cultivated in trypticase soy agar (TSA) growth medium at dilutions from 100 to 10−4 in order to determine the initial quantity of bacteria per sample. Six test samples (three control samples and three test samples) were incubated at a temperature of 37±2 °C for 24 h. After incubation, 20 ml of neutralizer was added again and the samples were stirred. Next, the samples were cultivated in TSA growth medium at dilutions from 100 to 10−4 in order to determine the final quantity of bacteria per sample. The calculations were made for each sample separately, according to the formulas included in PN-EN ISO: 20743. The tested samples were also directly assessed for antibacterial activity in accordance with AATCC test method 147-1998.
Results and discussion
In the first stage of the study, the physical properties of cylindrical knitted fabric formed on a warp knitting machine (intermediate product), and of vascular patches (final product; the result of incorporating circumferential crimps in an intermediate product, finishing treatment with thermal stabilization, and subsequent radiation sterilization) were determined. From the obtained data and a microscopic examination of the structure (Figure 3), it can be stated that the process of adding crimps and the finishing treatment influence two parameters: water vapor permeability (for intermediate product, 2000 ml/min/cm2; for vascular patch, 1500 ml/min/cm2) and density of the knitted fabric (intermediate product, 295.7 meshes/cm2; vascular patch = 1552.6 meshes/cm2). The observed changes may be due to the micro- and macro-structure changes (Figure 3) resulting from the mechanical and thermal treatment of the intermediate product, causing a reduction in the distance between yarns in the loops of knitting materials, which finally resulted in an increase in the density and a decrease in water vapor permeability. Additionally, the contact angle data confirmed this observation. In the case of the intermediate product, the structure did not allow a drop of water to permeate (hydrophilic surface), while for the vascular patch, the contact angle was 144° (indicating a hydrophobic surface).

Micrographs: (A) intermediate product; (B) vascular patch; (C) a drop in the crimp surface of the vascular patch; (D) no drop during the test of the intermediate product.
The formation of the crimps (‘protrusions’ giving the final corrugated structure) on the surface of the cylindrical knitted fabric (intermediate product) reduced the value of the parameter related to water permeability. This is very important for preventing blood outflow in the case of a rupture in the aneurysm wall. Full sealing of the vascular patch takes place during the process of preparing the graft for implantation (the preclotting procedure).
Additionally, it has been found that the incorporation of circumferential crimps in the intermediate product, the thermal stabilization, and the radiation sterilization did not affect the properties involved in fixing suture pull-out strength and the stability of the predicted dimensions of the implant.
The most important feature of a good biomaterial is its safety, so it is necessary to test for the presence of residues from the production process as well as to conduct an analysis of the compounds contaminating the starting material. Figure 4 presents the chemical properties of the intermediate product and the final vascular patch. Studies of all these chemical parameters are necessary for the registration of biomaterials. The results for the vascular patch (final product) have improved compared with the results for the intermediate product (cylindrical knitted fabric formed on a warp knitting machine). A preliminary analysis, comparing the properties of different commercially available yarns, as well as the final products, has already been presented. 31
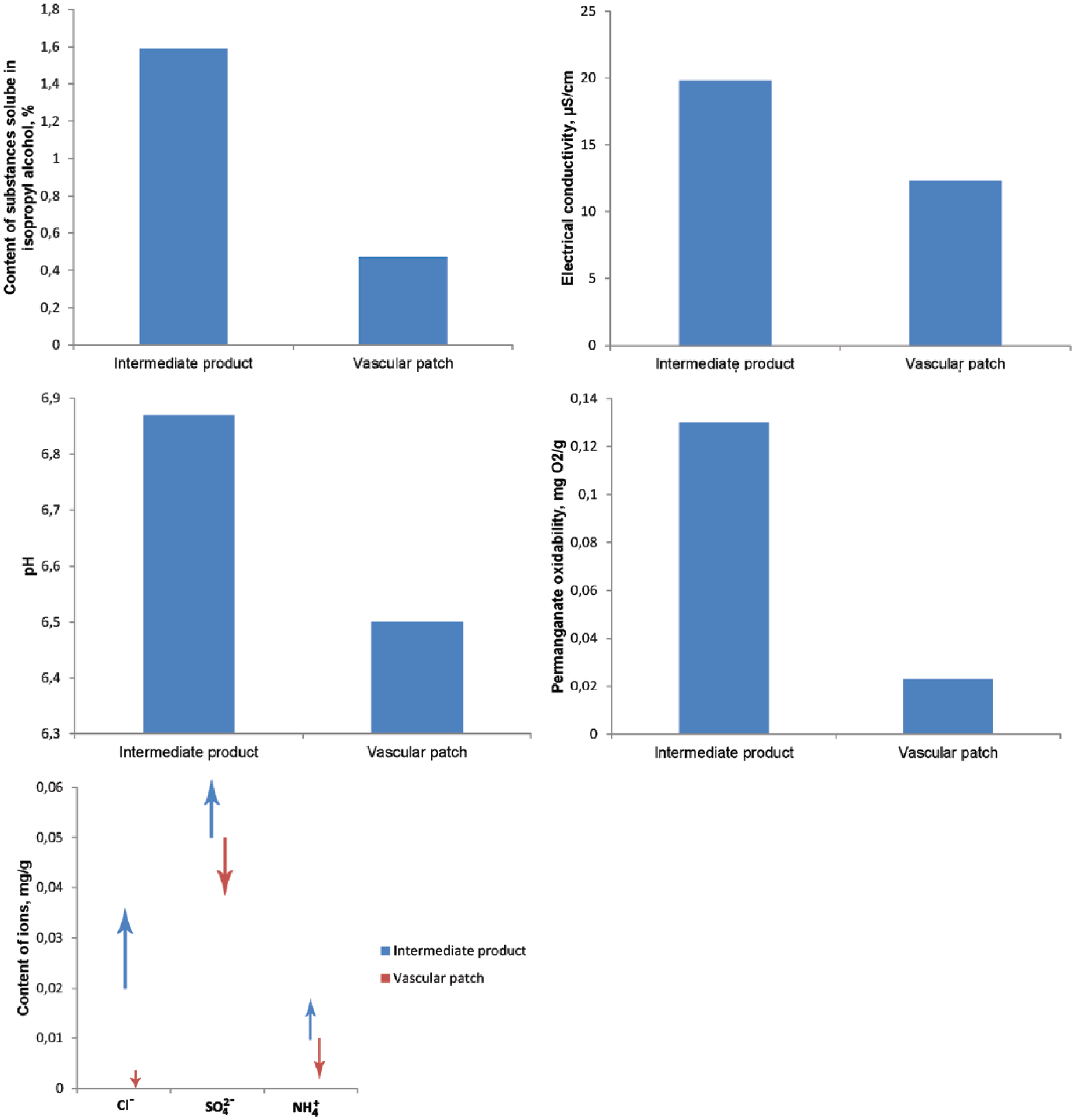
Chemical parameters of the intermediate product and the vascular patch.
This analysis of the presented results indicates that the process of finishing treatment with thermal stabilization of material with added circumferential crimps in the intermediate product reduced the content of Cl−, SO42−, and NH4+ and led to a reduction of electric conductivity from 19.8 µS/cm to 12.3 µS/cm. At the same time, the content of substances soluble in isopropyl alcohol was significantly reduced from 1.59% in the intermediate product to 0.47%, a decrease of 70%. Moreover, the susceptibility to oxidation of the final product was considerably reduced compared with the intermediate product; this is important for the durability and safety of the vascular patch.
FTIR analysis was used to determine whether using thermal stabilization in the finishing treatment affected the chemical structure of the vascular patch (final product). The results were then compared with data obtained for the intermediate product (cylindrical knitted fabric formed on a warp knitting machine).
Analysis of the recorded FTIR spectra showed that the finishing treatment of the knitted fabric and the sterilization process did not influence the chemical structure of the polymers. Both spectrograms (Figure 5) showed bands with a similar intensity, which is characteristic for polyester. There were distinctive bands for the following wavenumbers:
3428 cm−1, reflecting the oscillation of binding of –OH groups, corresponding to terminal groups of polyester;
2968 cm−1, related to the oscillation of methylene groups presented in the polyethylene terephthalate macromolecule;
1716 cm−1, reflecting strong valence oscillation of the C=O group;
1017 cm−1, 871 cm−1, and 722 cm−1, resulting from oscillation of aromatic bonds.
The obtained results were practically the same as characteristic oscillation bands for polyester (for example, Dacron® polyester yarn). On the basis of the performed FTIR tests it was found that the presence of silver in the tested material did not change the spectral characteristic of the material.
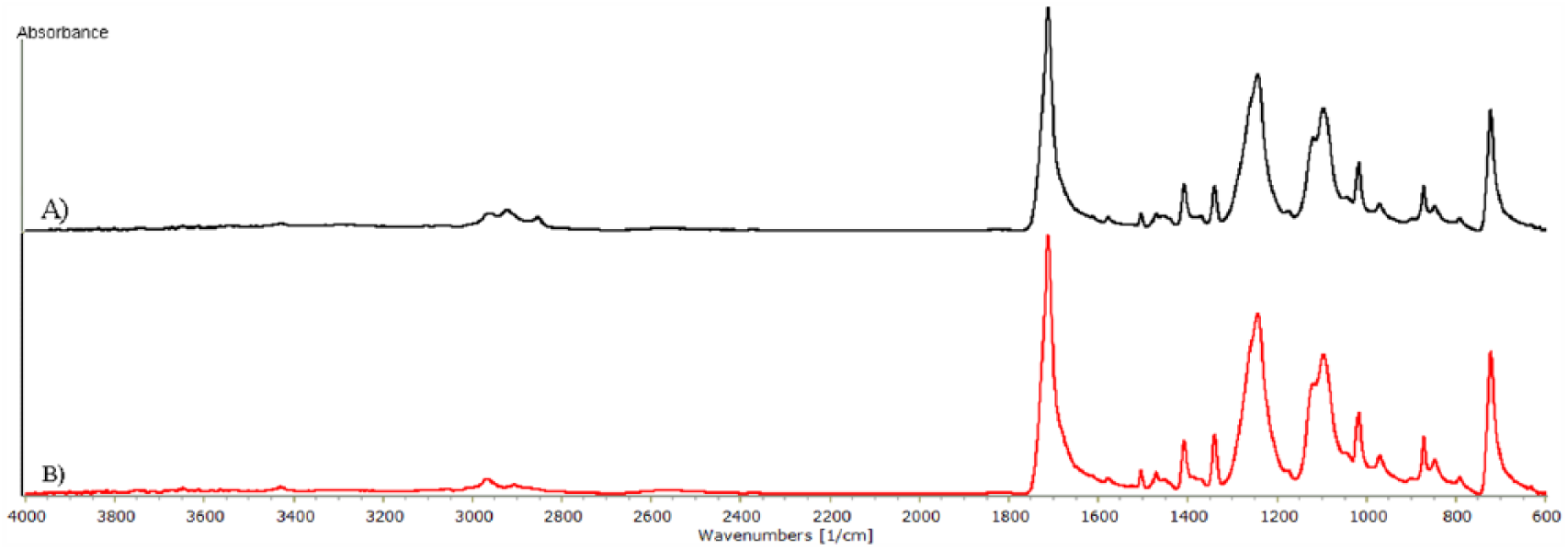
FTIR-ATR spectra of: (A) intermediate product; (B) vascular patch.
An important parameter from the point of view of implantable materials is the supramolecular structure of the materials used. It is widely recognized that long-term use of implantable polymer materials in a biological environment promotes their corrosion. Finally, the material might be defragmented and the fragments of the implant might be released into the surrounding tissues, causing irritation of the tissue or an inflammatory reaction. The release of crystalline fragments from the implant is particularly dangerous, as they are less prone to degradation. This can lead to long-lasting inflammatory processes and thus also increases the probability of neoplasia. Long-term observations have shown that, in the case of fragmentation of crystalline materials, there has been a spread of cytotoxity.33–35
To determine this parameter, differential scanning calorimetry analysis of the knitted fabric (intermediate product) and the final vascular patch were made. The tests were carried out in the following setting: heating I, cooling, and heating II. Based on the differential scanning calorimetry studies, the impact of the finishing process was tested. In this case, it was important to determine the impact of the finishing treatment on the crystallinity degree of the final vascular patch. The polymer melting enthalpy was determined as the result of this analysis through extrapolation of data of the polymer with increased crystallinity.
It was found that the crystallinity degree amounted to 8.7% in the case of the knitted fabric (intermediate product) used for the manufacture of vascular patches; this value is appropriate and safe for implantable material. A slightly larger crystallinity degree, 9.0%, of the vascular patch (final product) might be caused either by measurement error or by the presence of bleaching substances used in the manufacturing process. The analysis proved that the final treatment during manufacture does not significantly influence the crystallinity degree. In both cases, it is near 9%. The level of this parameter is comparable to the crystallinity degree of widely used polyester yarns in implants, whose values fall between 6% and 14%. The analysis of scanning electron micrographs and energy dispersive X-ray spectrographs of the knitted fabric subjected to finishing treatment and of the vascular patch showed that both wet and thermal treatments do not change the structure of the product (Figure 6).
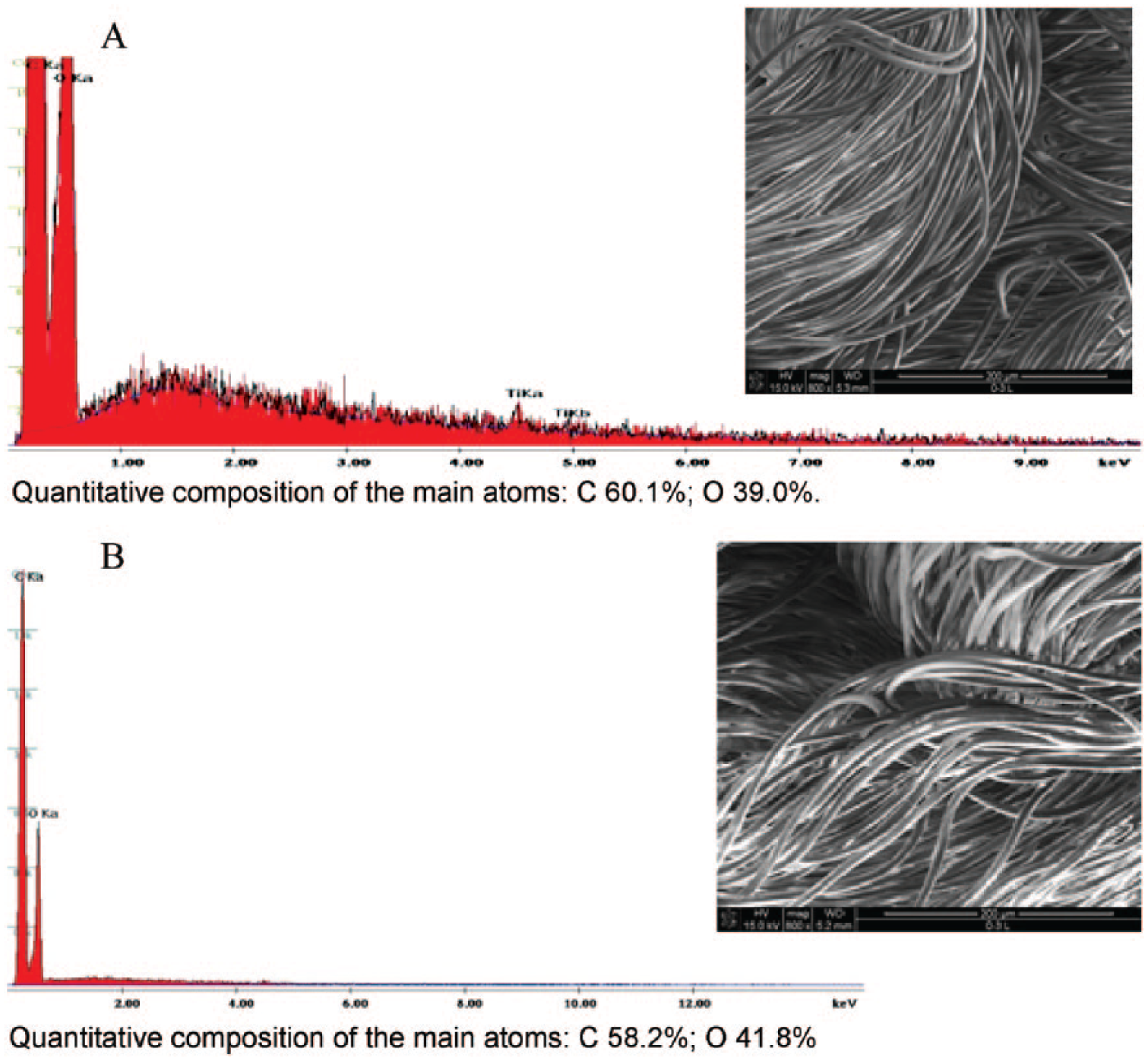
Scanning electron microscopy and energy dispersive X-ray spectroscopy of: (A) knitted fabric (intermediate product) used for manufacturing vascular patch; (B) final vascular patch.
What came as a surprise was the fact that scanning electron micrographs and energy dispersive X-ray spectrographs show no microsilver (active substance) present in the starting material (trade product Trevira®) or the final product. This may be attributed to the fact that the microsilver was not deposited on the outer surface of the fiber, but rather incorporated in the matrix of the fiber material. This claim is made because the atomic absorption spectroscopy results showed that the content of the microsilver was 194 ppm for the knitted fabric (intermediate product) and 134 ppm for the vascular patch (final product).
To ensure that microsilver was not removed during the manufacture of the vascular patch, an analysis of its release from the final product was performed.
Long-term tests of microsilver release from the final vascular patch in phosphate buffered saline showed its increased growth over time. Almost 80% of the active substance (microsilver) was released in the first 50 hours (Figure 7a), and the remaining microsilver was released rather slowly during the following 336 hours (Figure 7b). The microsilver being released in such a way may have a beneficial effect on the correct interaction between the implant and natural tissues.
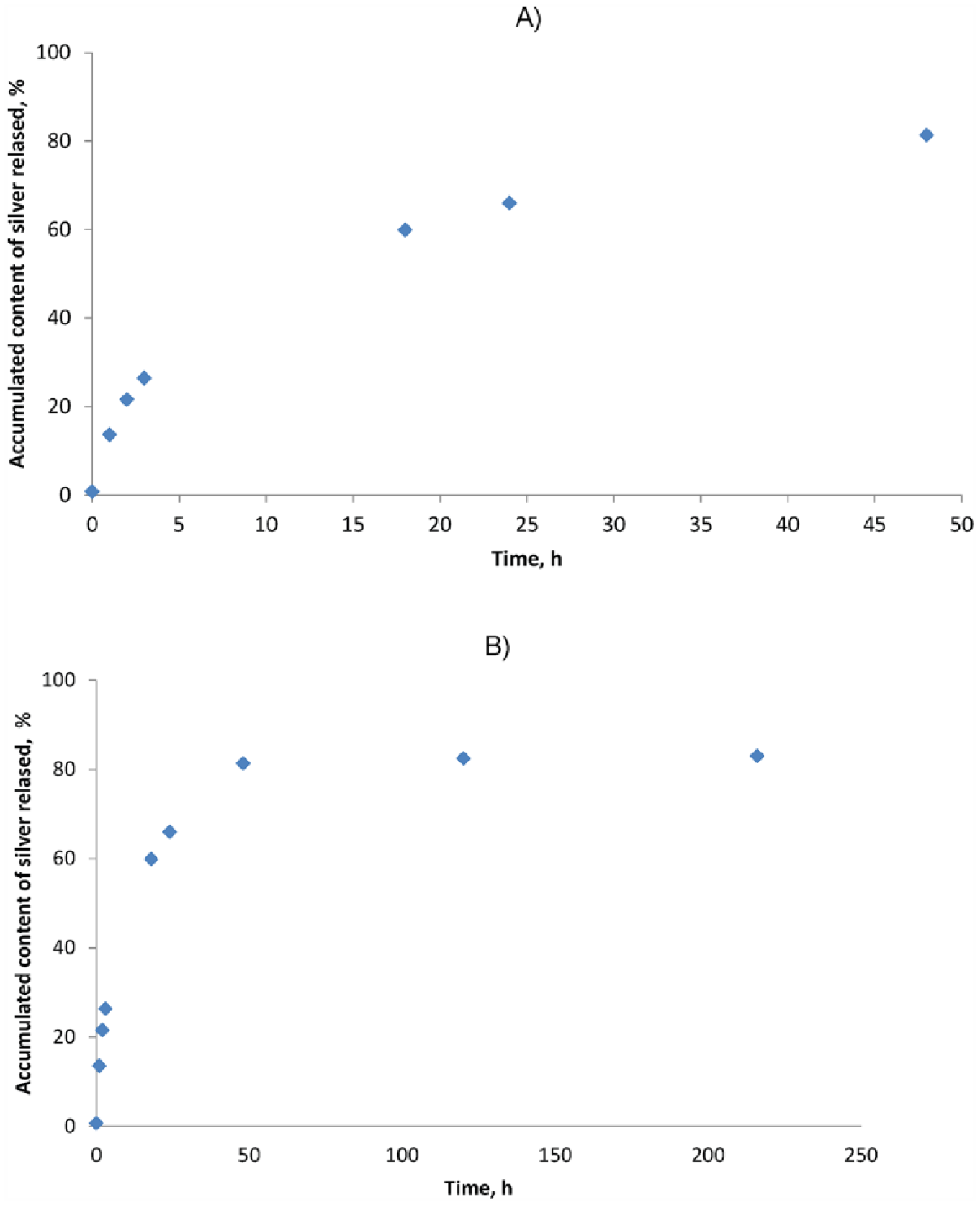
Percentage dependency of released silver as a function of incubation time: (A) 48 hours, (B) 336 hours.
The obtained profile of release of the active substance (microsilver) into the buffered saline solution (phosphate buffered saline) can be considered appropriate from the point of view of microbial infection occurring in the first hours after surgery. A systematic release of 80% of the microsilver from the fiber material during 50 h takes place. From analytic calculations it was found that the amount of silver released during 24 h amounted to 120 µg/l. Assuming that the vascular patch would have the following dimensions: diameter 50 mm and length 200 mm, this means that 360 µg of silver would be released from the patch. Thus, for a patient with a weight of 70 kg, the amount of microsilver released would be 5.14 µg/kg of bodyweight.
The calculated amount of released microsilver, owing to its antibacterial activity, indicated that this amount should protect the field of surgical operation from bacterial infections. To verify this assumption, inhibitory growth tests of microbes were performed.
Quality tests of antibacterial activity did not show growth inhibition zones for either Staphylococcus aureus or Klebsiella pneumoniae. However, the quantity tests showed a reduction of bacterial colonies by 93.64% in the case of Staphylococcus aureus and 97.31% for Klebsiella pneumoniae.
Conclusions
The tests carried out during the research enabled development of a vascular patch for the treatment of abdominal aorta aneurysm by wrapping the affected vessel. The material can be used in cases where there is no possibility of using other treatment methods. The process of modifying the surface of the knitted fabric by adding circumferential crimps enabled the water permeability of the implantable material to be reduced. The applied technology of vascular patch production allowed the possibility of adjusting the implant to a specific case (the possibility of cutting the patch at various angles). Additionally, the introduction of orienting lines enabled easier application and monitoring after surgery and also allowed for adjustment of the diameter of the implant according to individual requirements. The reduction of Staphylococcus aureus and Klebsiella pneumoniae colonies and a simultaneous retention of the assumed release profile of the active substance will undoubtedly reduce the risk of post-operative complications related to microbial infections. It has also been shown that proper finishing treatment is required to ensure proper purity of the final product. On the basis of the performed test, it has been demonstrated that an implantable material for the treatment of abdominal aorta aneurysm was obtained. The material can be considered as an alternative for currently used vascular patches.
Footnotes
Declaration of conflicting interest
None declared.
Funding
This work was supported by the National Centre for Research and Development (grant number NCBiR 04612/C.ZR-10-6/2010).
