Abstract
Objective:
To investigate the Pinus halepensis extracts and determine its healing and antibacterial effects, and to evaluate the treatment of skin burns.
Methods:
Aqueous and ethanolic extracts and topical based on Aleppo pine plant extracts were prepared. Thirty male and female Wistar rats were used to study the cutaneous toxicity of extracts from the bark of P. halepensis. The extracts’ healing potential for burn wounds were also assessed by evaluating the clinical and macroscopic aspects of the wounds. The antibacterial activity of crude extracts of P. halepensis as well as its wound healing abilities was verified in this investigation.
Results:
In animals with acute dermal toxicity, there were no signs of treatment-related toxicity or death. The extracts of these plants could be transformed into phytomedicines for the treatment of infected wounds. The results demonstrated that formulated ointments are successful in treating second-degree burns in rats and may be suitable for the short-term therapeutic treatment of second-degree burns.
Conclusion:
This study successfully answered our problem, regarding the efficacy of our extract for treating second-degree burns in rats. Further studies are needed to confirm these results by identifying the molecules responsible for these activities and examining their mechanism of action.
Introduction
Burns are traumatic pathologies responsible for significant morbidity and mortality. They are defined as a partial or total destruction of the skin covering or the underlying tissues by a thermal, electrical or chemical agent or by ionizing radiation.1–3
Given the importance of the skin as a barrier structure protecting the body from external shocks, particularly infectious ones, post-burn sequelae are one of the most problematic disorders in the medical world.4,5 Even though the survival rate of patients with severe burns has improved over the past few decades, infection remains the main cause of death,6,7 because of the heavy care burden,8,9 and the often limited use of many “conventional” products because of their variable effectiveness, their possible adverse effects, their unavailability and their price, which is considered high in developing countries. In addition, burns are characterized by opportunistic infections as the skin breaks down. Therefore, the treatment of burns must be accompanied by antibiotics in order to avoid microbial infection in the burned parts.
The solution to these many issues is the use of natural products; these products, from traditional medicine, have long proven their effectiveness for the treatment of burns and other human diseases, and have very interesting advantages in terms of availability, safety, and low cost.10–12
According to the WHO, nearly 80% of the populations of developing countries in Africa use traditional medicine. 13 Many plants have a therapeutic potential that would allow them to play a beneficial role in terms of very important preventive action for human and animal health.14,15 It is also recognized that plants are an important source of bioactive molecules such as terpenoids, flavonoids, tannins, and phenolic compounds, which are able to improve the healing process of burns. Herbal remedies also have some advantages including accessibility, fewer side effects and low cost. 16
In the present study, we focus on the Moroccan medicinal plant Pinus halepensis (P. halepensis) belonging to the Pinaceae family. P. halepensis is very well known as the wild Pin d’Alep (Aleppo pine) plant, native to the Mediterranean region. Traditionally it is used as a coagulant in the artisanal production of sheep and goat’s cheese, and to help heal burns.
An extensive range of ethnomedicinal surveys has reported the utilization of P. halepensis in traditional medicine to combat various health issues such as respiratory problems (including bronchitis, pneumonia, respiratory infections, and colds), inflammation, wounds, urinary problems, gastric and intestinal ulcers, prostate infection, and infertility. Moreover, it is renowned for its antiseptic properties and for stimulating the adrenal glands.17–20
However, to our knowledge, the antibacterial activities of P. halepensis bark have not been investigated. The aim of this study is to determine the healing and antibacterial effects and evaluate the treatment of skin burns with extracts of P. halepensis.
Materials and methods
Plant
The plant material consists of the bark of P. halepensis collected in its natural habitat around the Khemisset province in January 2019. This species was identified by botanist Professor Hamid Abdelhalim Khamar and a specimen (No. RAB93519) was deposited in the herbarium of the botany department at the Scientific Institute of Rabat. After drying at room temperature and protected from sunlight, the bark was finely ground using a grinder to obtain a fine powder. The powder was then sieved through sieves of different particle sizes, to recover a very fine and homogeneous powder with a particle size of less than 125 μm (Figure 1).

The plant and the bark of P. halepensis were collected from the Khemisset province in January 2019.
Preparation of extracts and topicals based on plant extracts
Preparation of aqueous and ethanolic extracts
The total aqueous extract (AEPH) and the ethanolic extract (EEPH) were prepared in the same way. The powder (250 g) of the bark of P. halepensis was macerated (by exhaustion times 4) in 1 L of distilled water or absolute ethanol under magnetic stirring for 24 h, at room temperature and at l shelter from light. The extracts were filtered on Wathmann paper then concentrated under reduced pressure on a rotavapor at 50°C and the residue obtained was freeze-dried in order to completely eliminate traces of solvent. The extracts were stored at +4°C.
Preparation of topicals
In the present work, the plant extracts were applied as a 20% ointment. The extracts were moistened with inert excipients (Table 1).
Ingredient for 70 g of ointment.

The ointments thus prepared were stored in sterile bottles in the refrigerator at +4°C.
Study animals
The study on cutaneous toxicity and healing potential of P. halepensis extracts involved a total of 60 Wistar rats, 30 male and female rats for the cutaneous toxicity study and 30 male rats for the healing potential study. The rats weighed between 180 and 300 g for the cutaneous toxicity study and between 180 and 250 g for the healing potential study and were obtained from the central animal facility of the Faculty of Sciences of Kenitra. The female rats were non-pregnant and nulliparous, and all animals had healthy, intact skin. The rats were transferred to the central animal facility of the Faculty of Medicine and Pharmacy in Rabat 15 days prior to the experiment to allow for acclimatization. The rats were individually housed in cages under controlled environmental conditions, with a temperature of 25 ± 1°C, humidity between 60% and 70%, and alternating 12 h of light and 12 h of darkness. The rats were fed conventional food and had access to tap water continuously through bottles, which were regularly disinfected with bleach. The cages and litter were also changed daily to maintain cleanliness and prevent infection.
The research was carried out under the supervision of the Laboratory of Pharmacology and Toxicology and the Central Animal Facility of the Faculty of Medicine and Pharmacy of Rabat, following ethical approval from the Mohammed V University of Rabat. The study adhered to the recommended guidelines outlined in the “Guide to the Care and Use of Laboratory Animals” issued by the National Academy of Sciences and published by the National Institutes of Health. The experimenters conducted the study in sterile conditions and took measures to minimize the number of animals needed for the experiments and their suffering.
Antibacterial activity
In order to evaluate the antimicrobial potential, the study tested the activity of the sample against five bacterial strains: Staphylococcus aureus Collection of Institut Pasteur (CIP) 483, Bacillus subtilis CIP 5262, Escherichia coli CIP 53126, Pseudomonas aeruginosa CIP 82118, and Salmonella enterica CIP 8039. The minimum inhibitory concentration (MIC) was determined using a modified resazurin microtiter-plate assay, as previously reported by Sarker et al. 21 Any observable color changes from purple to pink or colorless were recorded as a positive result, and the lowest concentration at which a color change occurred was considered the MIC value.
Acute dermal toxicity
The acute dermal toxicity of aqueous and ethanolic extracts of P. halepensis bark was assessed in rats using a limit test protocol 402 of the Organisation for Economic Co-operation and Development (OECD, 1987) at a dosage of 5000 mg/kg body weight (BW) following a preliminary test at 2000 mg/kg of (PC). The acute skin toxicity limit test involved 30 Wistar rats, consisting of an equal number of males and females. A day before the test, an area of the dorsal region of the animals’ trunk equivalent to or greater than 10% of the total body surface was shaved using an electric stretcher, avoiding any skin lesions. The rats were then fasted and placed individually in cages, randomly divided into three groups of 10 rats each, comprising of an equal number of males and females (n = 10).
The control group was administered the vehicle (water), and the test groups were administered the aqueous and ethanolic extracts. A pre-weighed dose was moistened with water and gently massaged onto the shaved skin area to facilitate product penetration, which was then covered with gauze and held in place with a bandage for 24 h. Afterward, the dressing and gauze were removed, and the skin was cleaned with lukewarm water, and the rats were returned to their individual cages.
Following the application of the extracts, the rats were observed every 30 min for 10 h on the first day and daily for 14 days. Throughout this observation period, the weight of each animal was recorded once a week, and behavioral manifestations, signs of local and systemic toxic symptoms, and the number of deaths were documented daily.
Experimental model for the study of the healing of cutaneous burns
The study protocol
A deep second degree burn, affecting an area less than 10% of the body surface, was performed using the technique described by Walker and Mason 22 with some modifications.
In our burn model, the metal cage used in the Walker and Mason study was replaced by a Plexiglas device containing a recess in the center. This device is a plate measuring 30 cm long, 20 cm wide and 1 cm thick.
In our study, the size of the chosen burn area was 3.14 cm2. This surface has been materialized on the Plexiglas plate by a central circular recess 2 cm in diameter. The implementation of the device will enable the targeted induction of burns in a specific area of the skin that passes through the recess, allowing for precise control over the extent of the burn and ensuring uniformity in the burn area in animal models. (No reference provided, but feel free to add one if available).
The burn wounds were produced after depilation of the backs of the rats with an electric razor and anesthesia with ketamine dosed at 50 mg/mL by intraperitoneal injection at the rate of 90 mg/kg of body weight.
The animal, still under anesthesia, was placed on its back on the Plexiglas plate. The legs are held in place by rubber straps and the skin on the back has been pulled through the hollow circle. The plate carrying the rat was immersed for 10 s in a water bath at 90°C. so that only the skin protruding under the plate was immersed.
The burnt skin was carefully dried and the rats were randomly divided into seven groups containing six rats each as follows:
• Control group: untreated rats or group of control rats;
• TVH group: vehicle-treated rats or group of vehicle-control rats;
• SSD group (=SDA): rats treated with 1% silver sulfadiazine (SSD) or reference group of rats
• PHA group: rats treated with the ointment containing 20% of the aqueous extract of the bark of P. halepensis;
• PHE group: rats treated with the ointment containing 20% of the ethanolic extract of the bark of P. halepensis.
Wound treatment was performed immediately after burn induction and once daily until day 18. Approximately 0.5 g of each treatment was evenly applied to the wound to cover the entire burned area. The burned animals did not show any mobility disorders or disturbances in food or drink intake, they were put in individual cages.
Assessing the healing potential of burn wounds
Clinical and macroscopic evaluation of wounds
The clinical evaluation of the animals was carried out by monitoring their mortalities daily and their body weights every 3 days. The clinical evolution of the wounds was carried out daily in the morning by the macroscopic examination of the lesions throughout the healing period of the burns. Gross examination of wounds is based on observation of the appearance of changes in the color, odor and appearance of the wounds, the formation of edema, bullae or crusts, bleeding, swelling, and secretion.
Calculate wound shrinkage rate
Healing was assessed by calculating the rate of wound retraction. For this, the surfaces of the wounds were measured at D0, D3, D6, D9, D12, D15 and D18 days after induction of the burn. The burn site was photographed using a Canon SX710 HS digital camera (20.3 megapixels; zoom = 1). The device was mounted on a support ensuring a constant distance of 15 cm between the lens of the device and the back of the animal and the lighting conditions were respected. In parallel, direct manual tracing of wound surfaces on transparencies was carried out. The photos and the transparencies were analyzed by image processing software (Image J software) to calculate the surfaces and determine the shrinkage rate.
The shrinkage rate was calculated by the following formula:
Retraction of the wound on day Dx = [(size at D0 − size at Dx) / size at D0] ×100 (Senthil Kumar et al. 10 ).
Histopathological evaluation
The histological evaluation was carried out at the end of the experiment on D18 after sacrifice of all the animals under ethical conditions. The biopsies were performed by taking a sample from the scar using a scalpel. The samples were then immersed in a 10% aqueous solution of buffered formalin to ensure their fixation. The fixed specimens were sent to the pathological anatomy laboratory for routine histological processing which consists of paraffin embedding or embedding, 4 µm sectioning by a microtome and making of a slide before hemalun-eosin staining and Masson’s green trichrome.
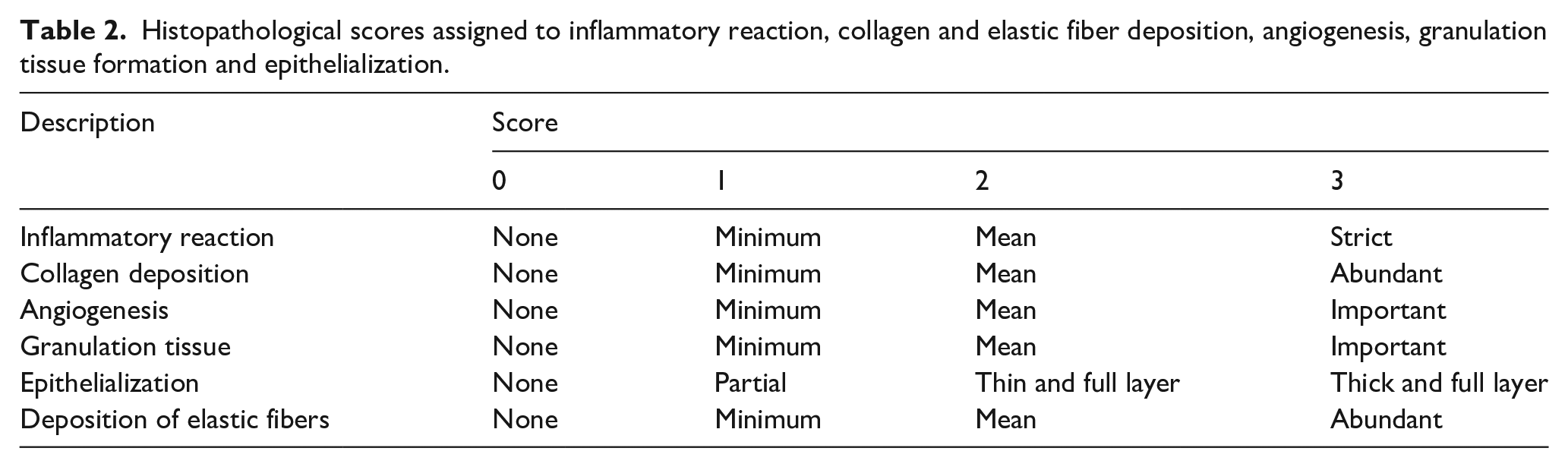
Wound healing potential was assessed histologically by determining the degree of inflammatory reaction, collagen and elastic fiber deposition, angiogenesis, granulation tissue formation and epithelialization. A score of 0–3 was assigned to each parameter studied (Table 2).23,24 Masson’s trichrome green staining and orcein staining were used for qualitative estimation of collagen deposition and elastic fibers respectively.
Histopathological scores assigned to inflammatory reaction, collagen and elastic fiber deposition, angiogenesis, granulation tissue formation and epithelialization.
Results and discussion
Antibacterial activity
In our study, the antibacterial activity of two extracts from the bark of P. halepensis was studied against five bacterial species. The results (Table 3) show that the ethanolic extract has antibacterial activities against S. aureus CIP 483, B. subtilus CIP 5262 at a similar minimum inhibitory concentration (MIC) equal to 0.25 mg/mL, as well as E. coli CIP 53126 and S. entirica CIP 8039 and P. aeroginosa CIP 82118 at a similar MIC equal to 0.5 mg/mL for all bacterial species. The aqueous extract also revealed antibacterial activity against the five bacterial species at similar MICs, equal to 1 mg/mL but lower than those of the ethanolic extract.
Evaluation of the antimicrobial activity of P. halepensis (PH) aqueous (EA) and ethanolic (EE) extracts and a standard drug against bacteria strains.

EEPH: ethanolic extract of Pinus halepensis; AEPH: aqueous extract of Pinus halepensis.
Skin toxicity
No indications of systemic toxicity were observed within the 48-h period following the removal of the dressing, and no alterations were detected in the skin, eyes, mucous membranes, or behavior during the 14-day monitoring period.
In addition, the present study did not reveal any mortality following acute dermal exposure of rats to a dose of 5000 mg/kg of aqueous and ethanolic extracts of the roots of P. halepensis, which shows a tolerance of these extracts in rats and therefore an LD50 greater than 5000 mg/kg dermal.
At the end of this experiment, according to line 402 of the OCDE guidelines (OECD, 1987), the aqueous and ethanolic extracts of the roots of P. halepensis can be considered as non-toxic by the dermal route. However, the study of the sub-chronic and/or chronic cutaneous toxicity of these extracts would be essential to secure their long-term use (Figure 2).
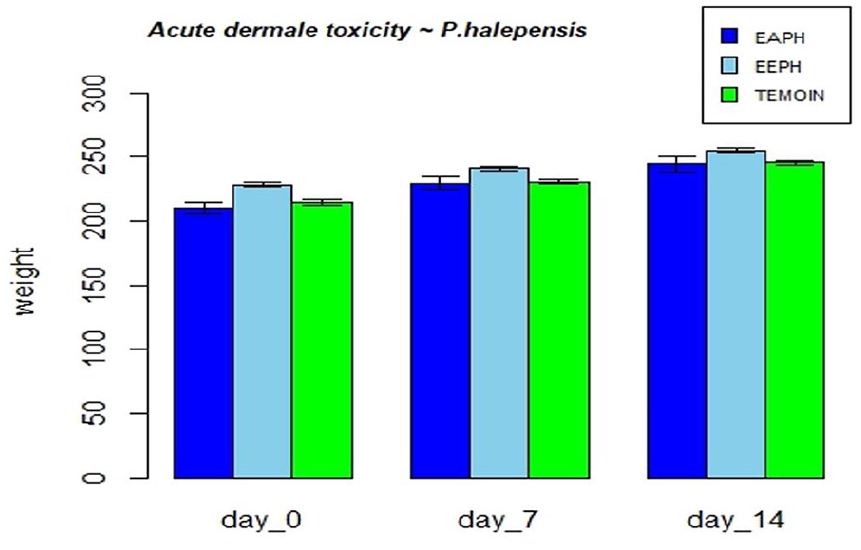
Evolution of body weight of animals in acute dermal toxicity with P. halepensis extracts treatment.
Effect on the evolution of body weight
The mean body weight of rats in the VAS groups was not statistically different (p < 0.05) from that in the control group. Body weight gain during the experimental period (D0 to D18) was 2.5, 3, 4, and 4.75 g, respectively, for the Control, Control VH, SSD, and EEPH groups. Only the weight of the rats having received EAPH increased from 247.75 g on day 0 to 257 g on day 18, representing a body weight gain of 10.75 g. Figure 3 represents the evolution of the average body weights of the rats receiving the various treatments every 3 days.
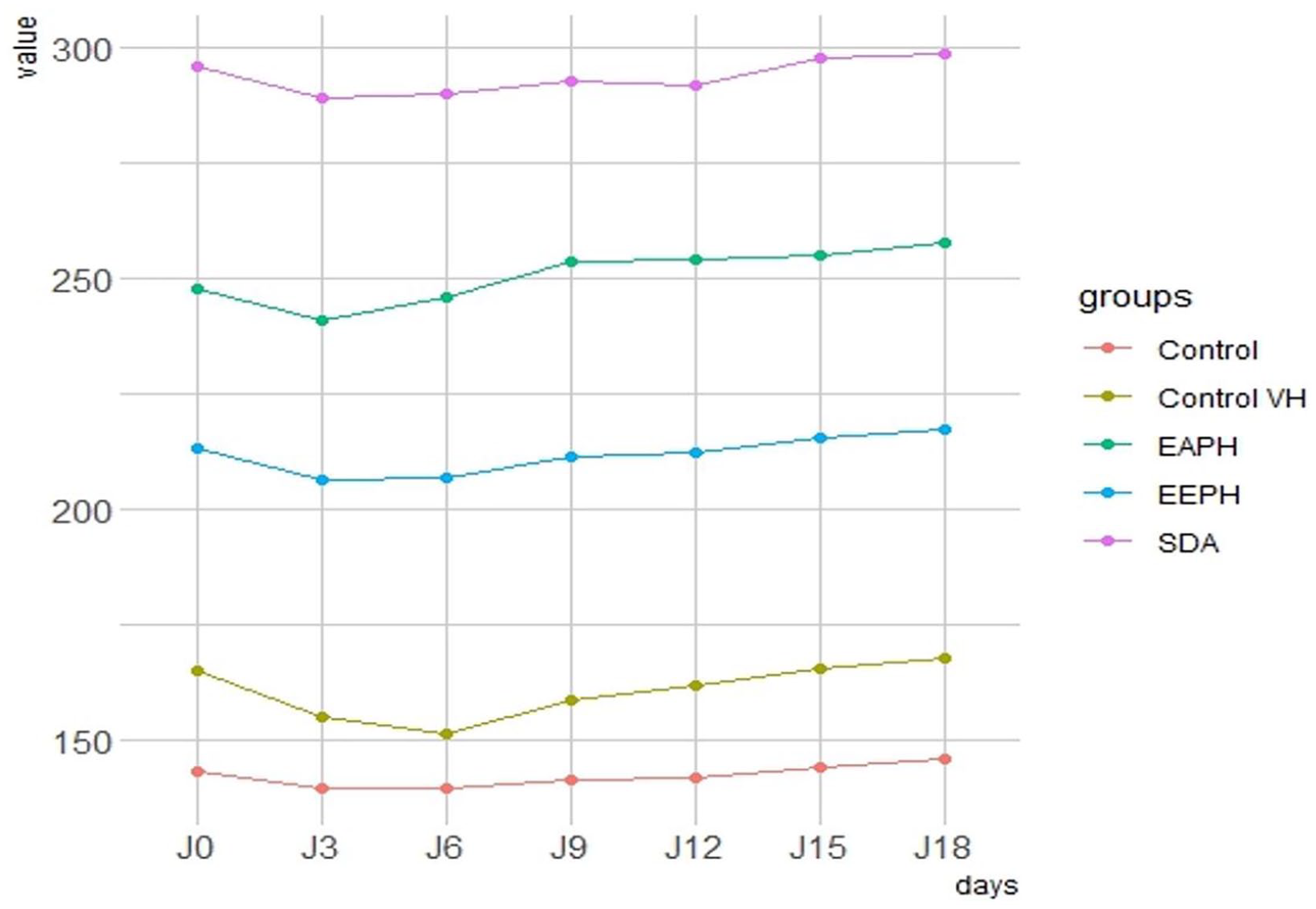
Evolution of body weights of animals treated with P. halepensis extract, SDA, Control VH, and Control in burn study.
Macroscopic analysis of wound healing
Topical use of Pinus halepensis compounded into an ointment base on wound healing in rats resulted in a considerably (P < 0.05) greater rate of wound healing in rats in the current investigation. The wound area after P. halepensis ointment administration was indicated in Figure 4. Wistar rats treated within 12–18 days had a superior healing pattern with full wound closure (Figure 4), whereas the control group needed more than18 days.
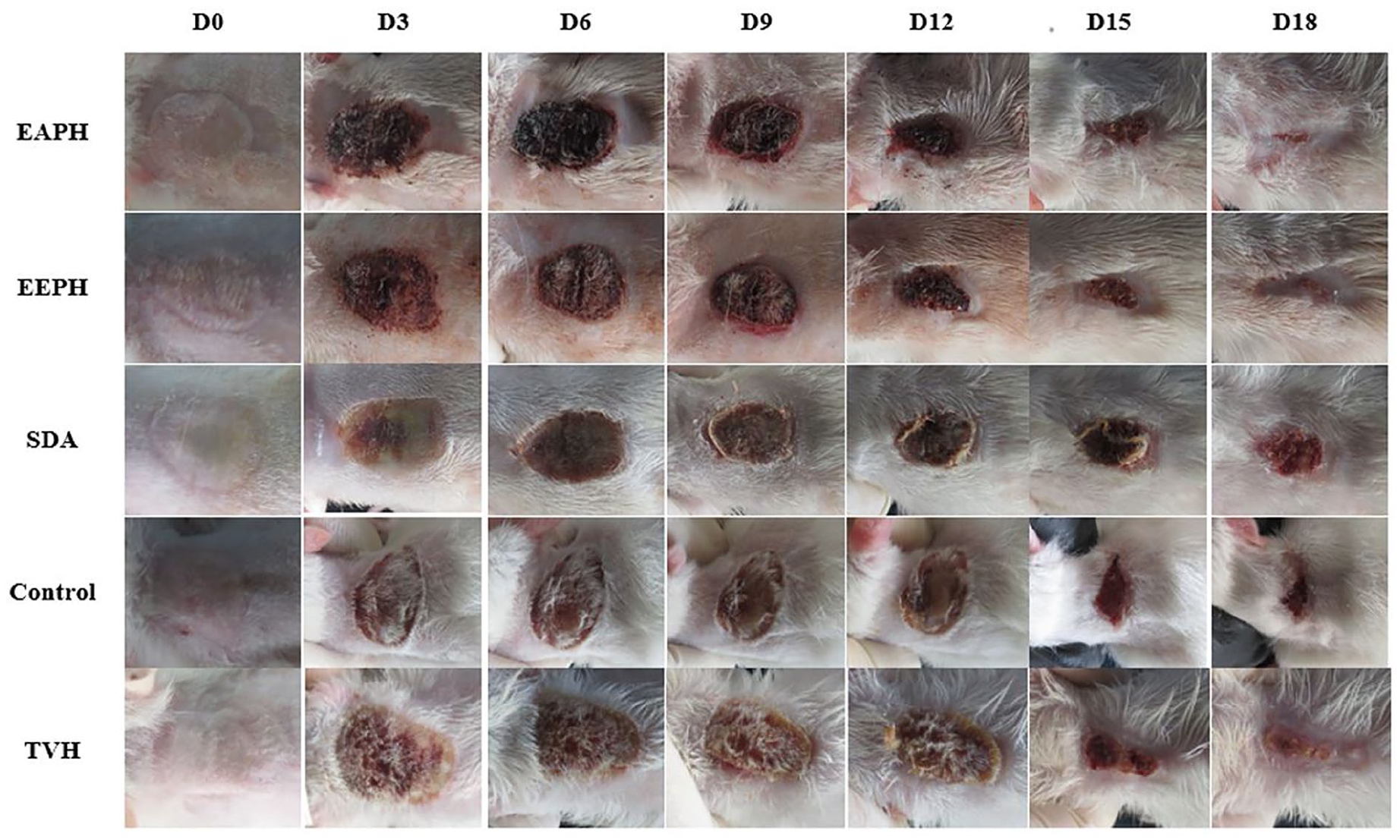
Macroscopic observation of burn wounds on rats after days 1, 3, 6, 9, 12, 15, and 18 of treatment with AEPH, EEPH, SDA (standard drug), Control VH and Control (no-treated group).
Analysis of wound retraction rate
Figures 5 to 7 show the differences in wound area assessed at days 6, 12, and 18 post-burn in the three treatment groups compared to the two control groups. Eighteen days after the burn, the groups treated with the EEPH and AEPH extract had achieved more than 75% healing. The group treated with EEPH achieved a healing rate of 32.5% at day 6 and 66% after 12 days, which was not significantly different (p > 0.001) from the group treated with the extract of AEPH which showed healing rates of 27% and 62% on days 6 and 12, respectively.
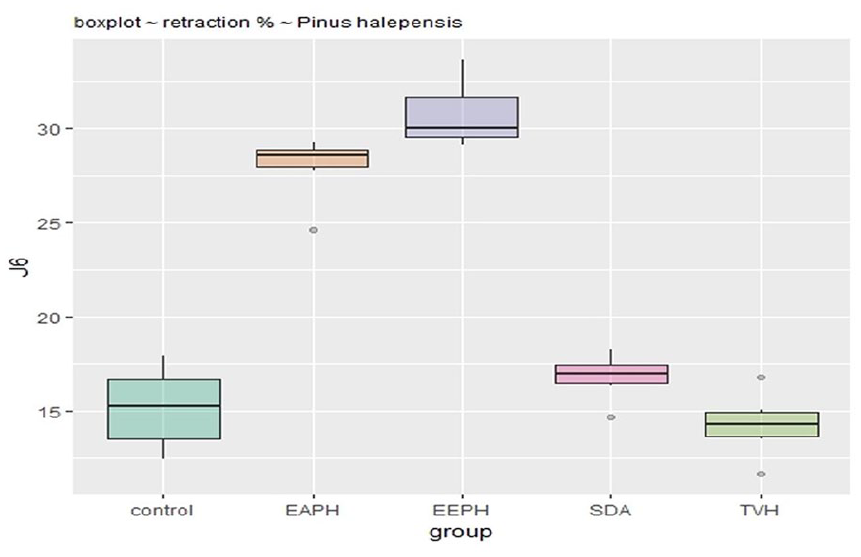
Retraction rate in groups treated with P. halepensis after 6 days.
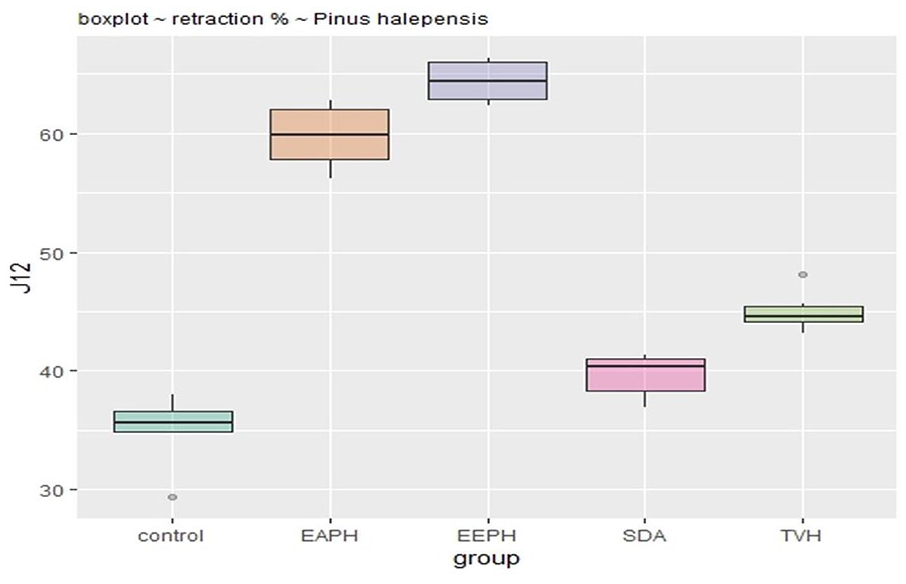
Retraction rate in groups treated with P. halepensis after 12 days.
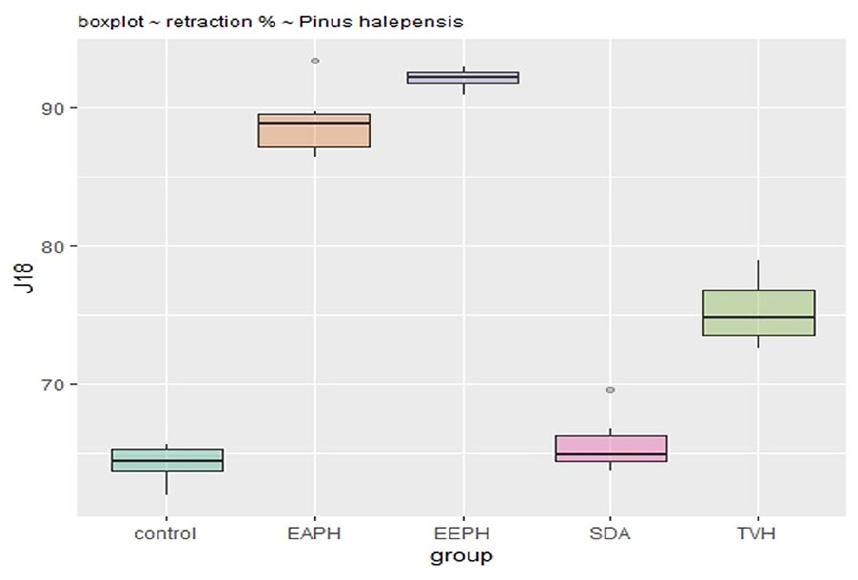
Retraction rate in groups treated with P. halepensis after 18 days.
The percentage of healing in the SSD-treated group was significantly lower (P < 0.001) than in the extract-treated group, at 66.4% on day 18.
The percentage of wound contraction of the ethanolic extracts of P. halepensis (92%) and the aqueous extract (89.5%) at 18 days is significantly higher (P < 0.001) than in the untreated group (65.5%) and vehicle-treated groups (66.5%).
Histological analysis of wound tissues
On the 18th day following the initiation of burn induction, a histological assessment of the wound sites was conducted, employing three different staining techniques. Figures 8 to 10 present histological cross-sections of the wound scars observed through an optical microscope. After 18 days of treatment, it was observed that the control, control VH, and SSD groups exhibited non-reepithelialized epidermis. In the dermal layers, ulceration was evident, accompanied by the presence of necrotic tissue debris intertwined with a variety of inflammatory cells, resulting in congestion and edema (as depicted in Figure 8).
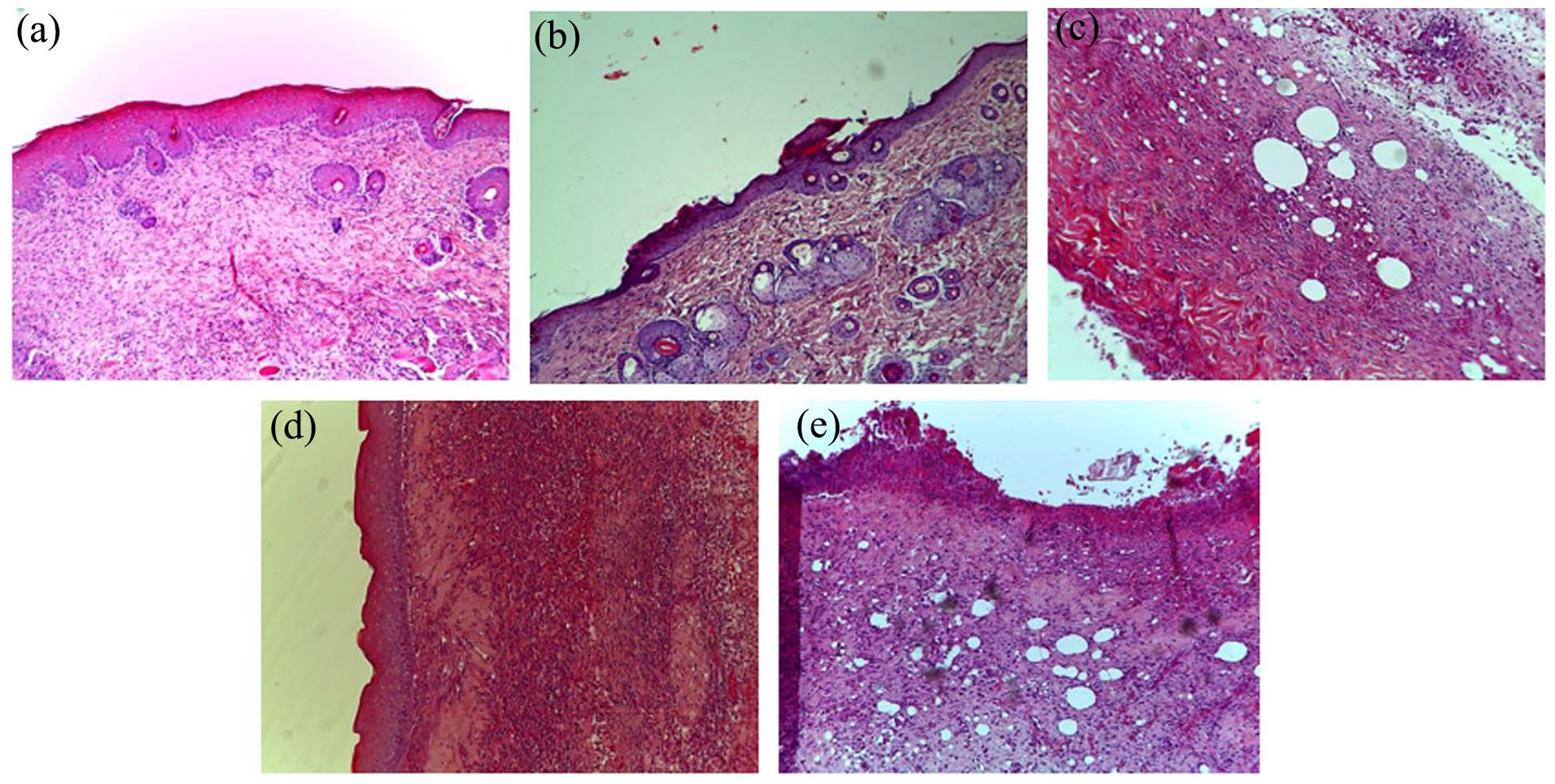
Histopathological change examination of coloration with hematoxylin and eosin of the skin of control and treated groups of rats: (a) AEPH, (b) EEPH, (c) Control, (d) TVH control, and (e) SSD.
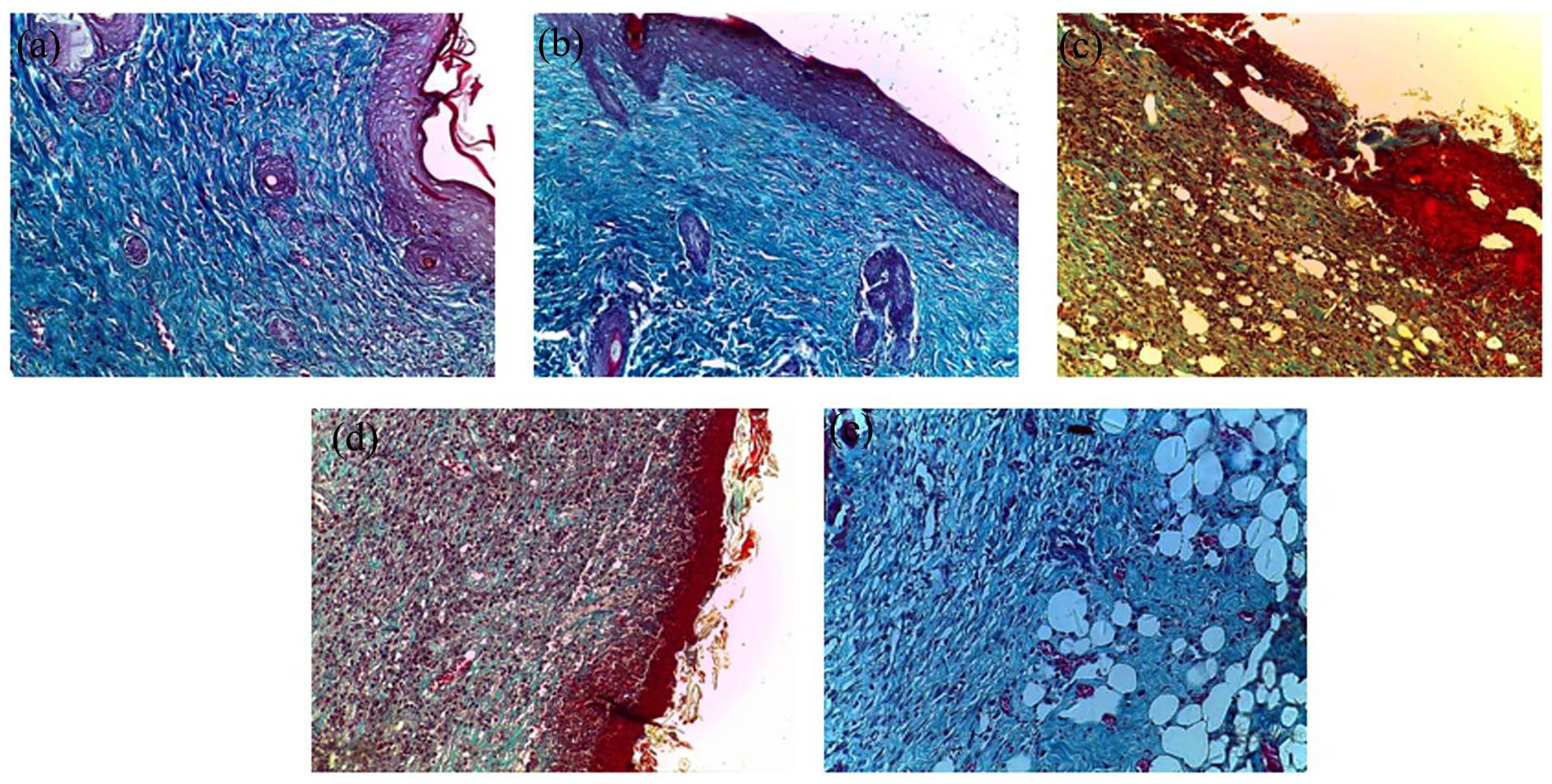
Histopathological change examination of coloration with green trichrome from Masson of the skin of control and treated groups of rats: (a) AEPH, (b) EEPH, (c) Control, (d) TVH control, and (e) SSD.
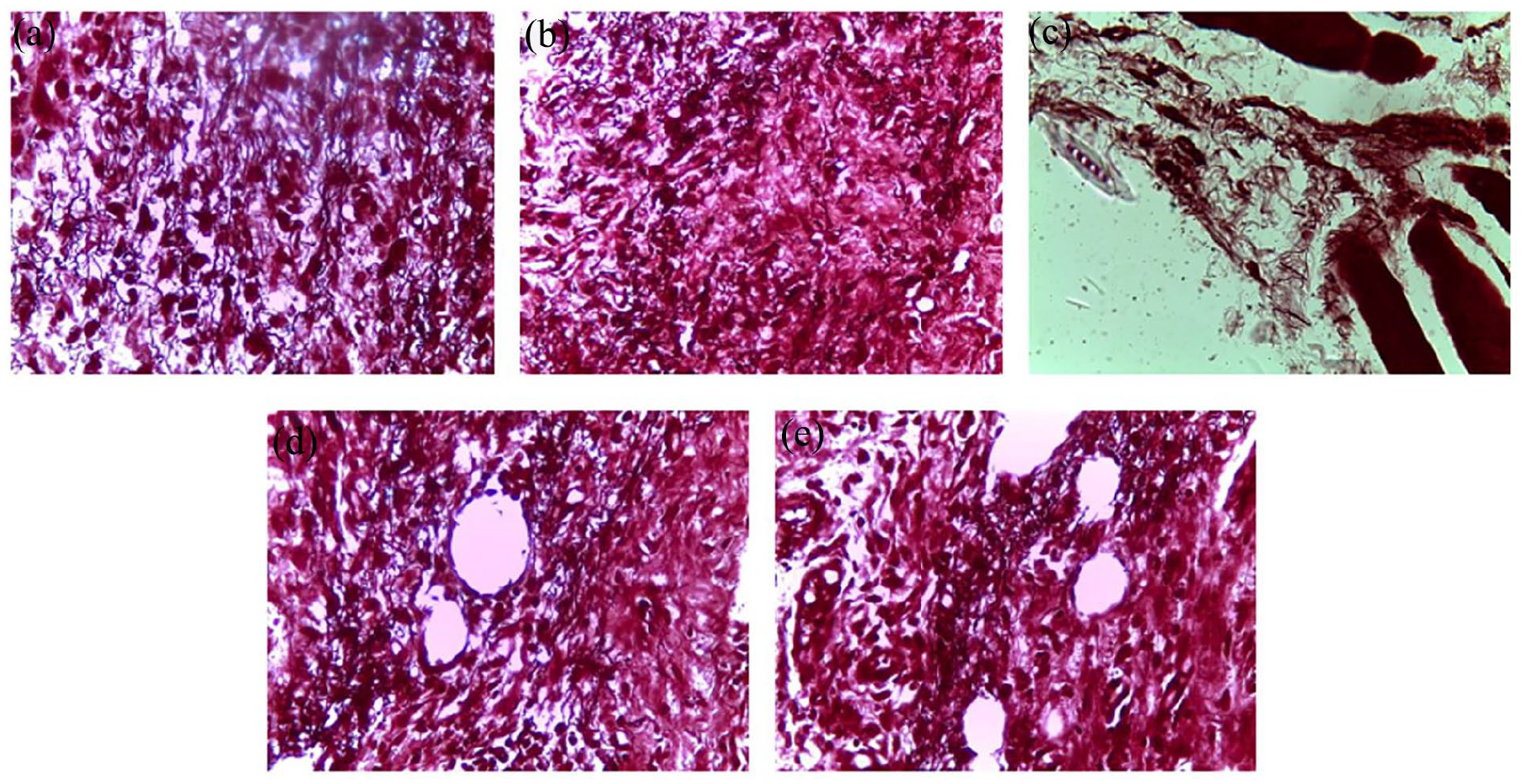
Histopathological change examination of coloration with orcein of the skin of control and treated groups of rats: (a) AEPH, (b) EEPH, (c) Control, (d) TVH control, and (e) SSD.
Treatment with P. halepensis extracts yielded notable outcomes, including substantial epithelialization, a reduction in inflammatory cell presence, and the absence of necrotic tissue remnants. Moreover, it spurred the generation of a modest but discernible amount of extracellular matrix and facilitated the emergence of fresh blood vessels and hair follicles, as exemplified in Figure 8.
To assess collagen fiber deposition and organization, we utilized Masson’s Trichrome Vert staining for histological analysis, as depicted in Figure 9. Results indicated that the control, control VH, and SSD groups exhibited sparse and disorganized collagen fibers. Concurrently, epidermal ulceration persisted alongside a polymorphic inflammatory infiltrate in the dermal layers, accompanied by edema, suggestive of an incomplete healing process (Figure 9).
In contrast, the groups treated with P. halepensis extracts exhibited remarkable enhancements in wound healing. This was demonstrated by a higher density and superior organization of collagen fibers, evident in our analysis of collagen quantity and quality (Figure 9).
We also scrutinized the deposition of elastic fibers by employing orcein staining on histological sections. Under the optical microscope, these elastic fibers presented as undulating bundles, occasionally interconnected, displaying a reddish-brown hue against a beige backdrop (Figure 10). Our findings underscored the abundance of elastic fibers in the histological sections of the P. halepensis extract-treated groups, a feature absent in the other groups.
Histological analysis of wound biopsies was also performed based on a scoring system by studying collagen and elastic fiber deposition, inflammatory response, appearance of granulation tissue, angiogenesis and epithelialization (Figures 8–13).
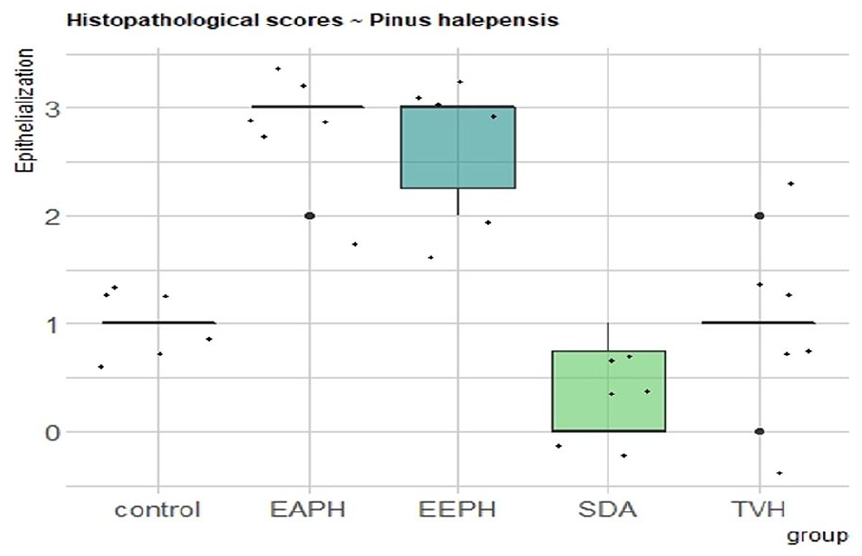
Histopathological scores for epithelialization.
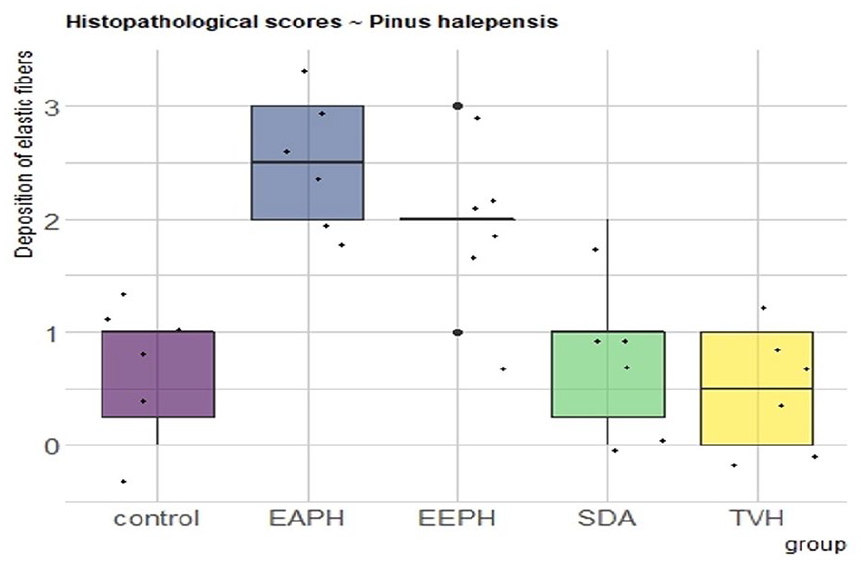
Histopathological scores for elastic fibers.
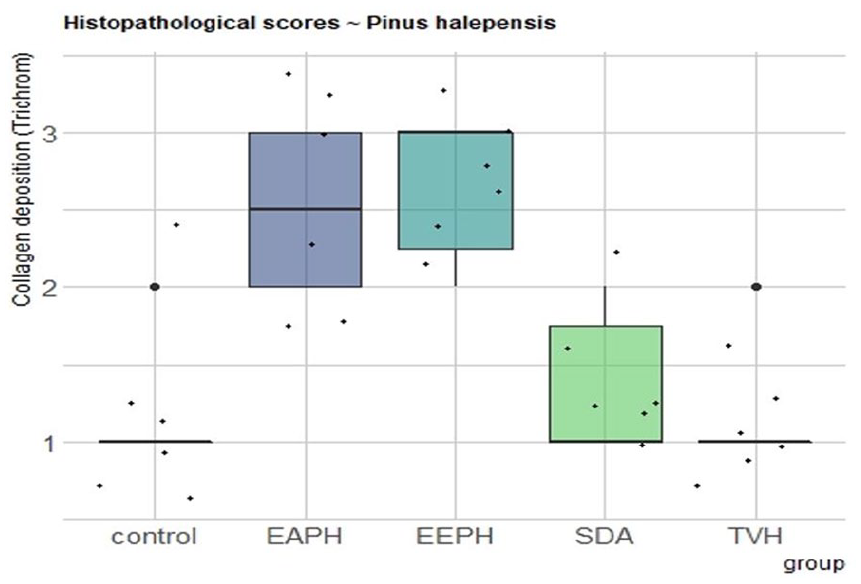
Histopathological scores for collagen.
Histopathological scores for epithelialization
The results show that there is no significant difference in the re-epithelialization score between the control and TVH control groups (p > 0.05), while there is a significant difference when comparing them to SDA therapy (p > 0.05). The highest re-epithelialization scores were found for EAPH and EEPH (Figure 11), and they have comparable significance (p < 0.01). The treatment groups (EEPH and EAPH) had higher re-epithelialization observations than the control (Control and TVH) and SSD groups (p < 0.05). Both the quantity and quality of collagen were increased with P. halepensis extracts. Herbal medicine groups had improved inflammation and angiogenesis processes.
Histopathological scores for elastic fibers
Elastic fiber deposition was highly controlled over the 18 days (Figure 12), and comparable patterns were detected across Control, TVH Control, and SDA, with little significant difference (p > 0.05). A low elastic fiber score was observed in scars treated with SDA, and in the control groups. During day 18, significantly more elastic fibers were found inside dermal scar tissue in the treatment groups that received EEPH and AEPH for wound healing after a skin burn (P < 0.05).
Histopathological scores for collagen
The proportion of collagen bundles in the microscopic fields was used to assess collagen synthesis in the skin. Collagen analysis revealed no significant difference between P. halepensis extracts (EEPH and AEPH), characterized by better collagen synthesis, while there was a small difference in score between the control and the TVH control, having shown a low rate of synthesis (Figure 13).
Histopathological scores for inflammatory cells
In all groups, the histopathological picture is almost identical. On day 8, the number of inflammatory cells was higher in the wounds of the control, TVH control and SDA groups compared to the groups treated with the creams containing 20% P. halepensis extracts (Figure 14).
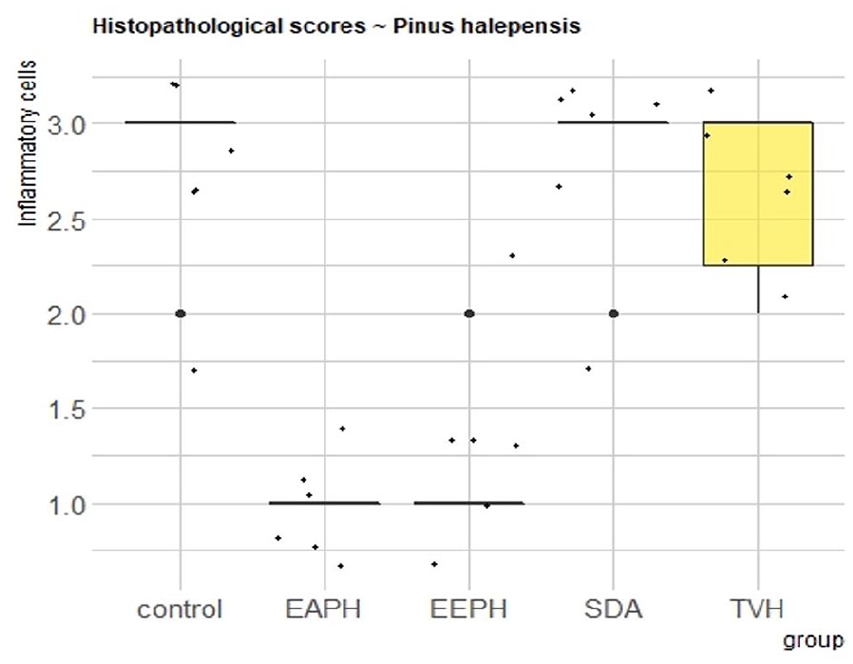
Histopathological scores for inflammatory cells.
Histopathological scores for angiogenesis
The best aspect of all groups is the moderate creation of new blood vessels (angiogenesis), while the control and control TVH groups have modest angiogenesis (Figure 15).
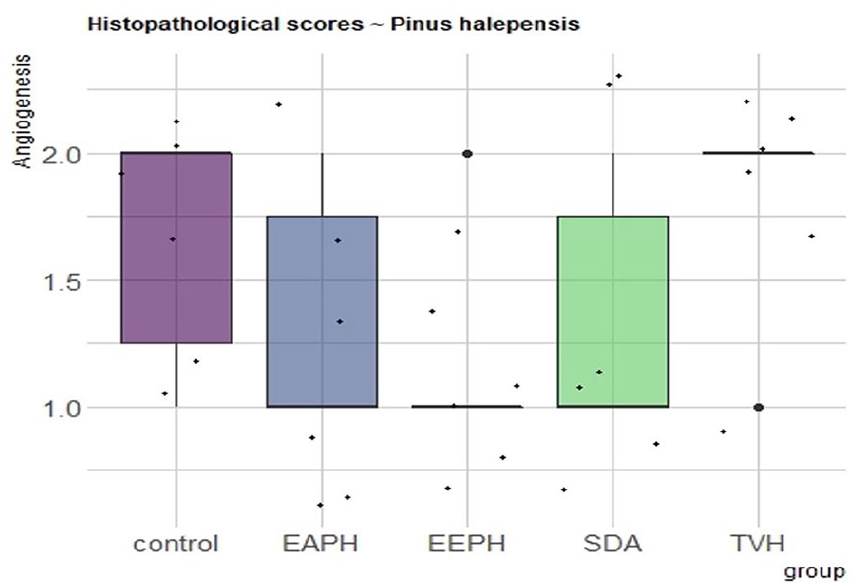
Histopathological scores for angiogenesis.
Histopathological scores for granulation tissue
The volume and distribution of granulation tissue over the treatment group (Figure 16) was significantly lower in the P. halepensis ointment group (p 0.01) than in the control group (p > 0.05). The best wound healing results are shown in EEPH and AEPH (P. halepensis treatment), which have high collagen density with good organization, complete and mature epithelium, low inflammatory cell count and angiogenesis, respectively.
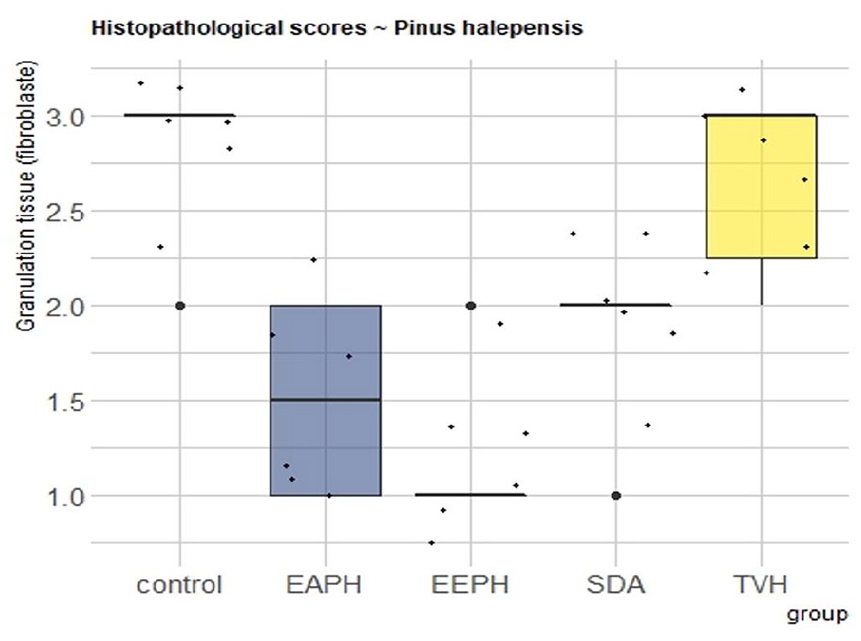
Histopathological scores for granulation tissue.
Discussion
Burns and Thomson are particularly vulnerable to complications.25,26 Complications can disrupt the healing process in various ways. An infection is the most likely consequence. The comparison between the standard drugs (Chloramphenicol silver sulfadiazine) and the topical drugs (P. halepensis extract ointment) revealed that the topical drugs were highly active and similar to Sulfadiazine against gram-negative and gram-positive bacteria. It has no major side effects.27,28 The findings of the current study indicate that the ethanolic extract obtained from the bark of P. halepensis possesses strong inhibitory effects against multi-drug resistant strains of both gram-positive and gram-negative bacteria. Other studies29,30 examined the antibacterial activity of methanol and ethanol extracts of P. halepensis seeds, barks and cones against S. aureus and E. coli. Although an antimicrobial study on pomegranate reveals the antibacterial process, the active ingredient inside pomegranate causes toxicity in bacteria by changing and interacting with enzymes and substrates.31,32 In addition, Salim et al. evaluated the antibacterial activity of extracts from seeds, bark, and cones of Palestinian Aleppo pine in their study. They conducted a phytochemical analysis to identify the compounds present in these extracts and subsequently tested their efficacy against strains of Staphylococcus aureus and Escherichia coli. The results demonstrated a significant inhibitory activity of the bark and cone extracts against these gram-positive and gram-negative bacteria. 33
The use of silver sulfadiazine as a positive control (SDA group) in this study produced the worst results in terms of collagen creation. Adverse effects of silver sulfadiazine that are clearly demonstrated in this experiment include retardation of wound contracture and delayed and incomplete epithelialization, which are also reported in a study by Muller et al. 34 The risk of delayed healing is increased when the inflammatory process is prolonged and severe. Skin layer turnover is one of the most significant indicators of burn healing during treatment.35–37 Therapy with P. halepensis extracts revealed regenerative benefits in the dermis layers, according to our histological evaluation.
Several studies have recently identified natural compounds, such as those derived from medicinal plants, for the development of new wound healing drugs. In this context, our study attempted to evaluate the healing of burns using an ointment based on a well-known Moroccan medicinal plant for the treatment of skin burns. Natural herbal materials are used in Asian and Indian medicine as they have advantages over manufactured drugs. The inclusion of many active ingredients in herbal medicines has the potential to broaden the therapeutic spectrum and improve treatment outcomes. 38 However, no research on the effectiveness of P. halepensis ointment against burns had previously been done, according to the literature. Our research focused on this. The impact of P. halepensis extract on rats’ burns was found to be remarkable.
In research conducted by Akhoondinasab et al., 39 the aloe vera group had a significantly shorter healing time for grade 2 wounds than the silver sulfadiazine group. Since contraction plays an important role in second-degree wounds, especially in the loose skin of rats, this impact was most noticeable with second-degree burns. In our study, the rate of contraction was higher in the EEPH and AEPH groups than in the SDA and control groups. Moreover, epithelialization is an essential part of wound healing and determines its success. Re-epithelialization is necessary for a wound to be considered healed. Histological data showed that all treated groups had a higher rate of epithelialization than the control group.
Inflammation has three functions: eliminating damaged cells, vasodilating inflammatory cells and extravasating inflammatory cells. In our investigation, P. halepensis ointment had a significant effect on collagen and inflammatory cells. It was comparable to earlier findings, which showed that topical application of avocado and jojoba oil to rats increased collagen production and reduced the number of inflammatory cells during wound healing.40–42
Inflammation can also contribute to the production and degeneration of skin cells. Therefore burns, which are prone to severe inflammation, should be treated with anti-inflammatory and antioxidant drugs to reduce the risk of delayed healing. 16 As demonstrated by our previous work, 16 P. halepensis has a variety of active compounds, each of which may function as an antibacterial, anti-inflammatory, and powerful antioxidant.
Collagen deposition in burnt skin was higher in wounds from the EEPH group. Indeed, epithelialization was greater in wounds treated with extracts of P. halepensis. In other treatment results, Kotian et al. 43 revealed that all treated groups had better collagen deposition at the wound site than the control group. The organization of collagen in the treated groups was ordered and intertwined horizontally. In contrast, the collagen fibers of the control group were obliquely and loosely arranged. Angiogenesis is one of the most studied processes in a burn wound. 44
In this study, excellent elastic fibers production and maximum angiogenesis were associated with an effective burn healing process. Collagen production was significantly stimulated when the dosage of P. halepensis extract was increased to 20% ointments. Compared with the control and normalization of silver sulfadiazine, this result showed that the ointment with 20% P. halepensis extract could establish an excellent burn healing process in rats. Based on these results, the use of a 20% AEPH and EEPH ointment to promote an ideal burn wound healing process likely provides a synergistic mechanism.
Crucially, the extracts did not cause the rats any discomfort or pain during the brief duration of the wound treatment, as they showed no symptoms of restlessness, scratching or biting the site of the wound when the extracts were administered.
Conclusion
The antibacterial activity of crude (aqueous, EA, and ethanolic, EE) extracts of P. halepensis (PH), as well as their wound-healing properties, were verified in this investigation. In animals with acute dermal toxicity, there were no signs of treatment-related toxicity or death. The extracts of these plants could be transformed into phytomedicines for the treatment of infected wounds. The results demonstrated that AEPH and EEPH ointments are successful in treating second-degree burns in rats and may be suitable for the short-term therapeutic treatment of second-degree burns.
Footnotes
Data availability
The data used to support the findings of this study are included within the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2024R33), Riyadh, Saudi Arabia.
Ethical disclosures
The authors declare that no experiments were performed on humans for this study.
Confidentiality of data
The authors declare that they have no patient data used in this study and declare they have followed the protocols of their work center on the publication of data.
