Abstract
Background:
Biofilm formation and microbial colonization on the surface of implant devices may cause dental caries and peri-implantitis. Therefore, various surface treatments have been developed to improve the antibacterial activity of titanium implant.
Methods:
Silver-loaded polydopamine coating was formed by immersing pure titanium in dopamine hydrochloride/HCl buffer solution for 24 h in 50 mL silver nitrate solutions with different concentrations for 30 min. Microbial growth inhibition and microbial growth curve analyses for bacterial solutions of
Results:
Silver nanoparticles were uniformly distributed over the whole surface of the polydopamine and silver-coated titanium specimens. The numbers of microbial colonies for both bacteria cultured with surface-modified titanium were significantly lower than those cultured with uncoated titanium. When
Conclusion:
It was confirmed that the coating of polydopamine and silver on the surface of titanium effectively retards the microbial growth, which can cause the formation of biofilm and pathogenesis of gum disease in the mouth.
Introduction
Osseointegration between the bone and an implant, and bacterial aggregation around the implant can have a significant influence on the success or failure of a dental implant. In particular, implant-related infections may lead to serious complications, even if antibiotics are administered regularly and systemically in a clean environment.1–3 Dental implants are constantly exposed to oral bacteria owing to partial contact with the jawbone and gums.
Periodontal diseases occur within the gums including the gingiva, the periodontal ligament, and the alveolar bone. While there are various indirect causes of periodontal disease such as smoking, hormones, poor dentition, diabetes, and family history, the formation of a biofilm, which comprises bacteria and is continuously formed on teeth, is a direct cause. In dental implants, microbial proliferation and biofilm formation can lead to serious infections, resulting in implant failure, systemic disease, pain, and economic loss.4,5
Traditionally, antibiotics have been used to prevent bacterial infections, but antibiotics alone cannot eliminate the bacteria in biofilms, which are a major cause of infection on the implant surface. When the biofilm was formed on the surface of the implant, the biofilm provides a protective barrier, which can shield the bacteria from antibacterial agents including antibiotics.6–8 Moreover, the excessive use of antibiotics not only promotes the proliferation of bacteria with antibiotic resistance but also reduces antibiotic efficacy. Therefore, an effective surface modification with antibacterial function is required to prevent bacterial adhesion and biofilm formation.
Peri-implantitis can even destroy the alveolar bone, depending on the degree of inflammation. Various types of bacteria can affect peri-implantitis or periodontal diseases. Among them,
Polydopamine (PDA), which is a mussel-derived adhesive protein, is created through the self-polymerization of dopamine (3,4-dihydroxyphenylalanine, DOPA). PDA has been extensively researched because it can be applied in various organic and inorganic biomaterials as it is non-toxic and biodegradable and is capable of altering its adhesive properties in the presence of amino acids.16,17 PDA uses threads made of fiber bundles to attach firmly to the surfaces of materials, even in water. Therefore, it can be used as coating material to improve the adhesive force and biocompatibility of biomaterial surfaces.17–21
Many previous studies identified that PDA coatings are useful in a variety of fields, particularly biomedical devices. 22 It was reported that PDA coatings on biomaterials increased the biocompatibility, the resistance of cytotoxicity, and antibacterial activity.23–25 Also, PDA coatings with additional incorporation of the functional material such as hydroxyapatite, chitosan, collagen, and antibiotics on Ti have been developed to enhance the osteointegration, osteogenisis, and antimicrobial activity of dental and bone implants.26–29
Silver has a broad spectrum of antibacterial properties, and has been used for various purposes in medicine for many years. Silver is chemically stable, but silver ions can be released when it is exposed to body fluid, which exhibits antibacterial properties in a wounded area. Silver ions are highly reactive and bind strongly to tissue proteins, causing structural changes of bacterial cell walls and the nuclear envelope. They also have an affinity for hydrogen ions and bind to thiol moieties in microorganisms; as a result, they kill bacteria and other microorganisms through incapacitation.30–33 Thus, much research of nanocomposites, surface modification, and pre-formulation using silver nanoparticles has been carried out to use as the dental and orthopedic materials.34–36 For instance, the excellent antibacterial activity and bio-integration was confirmed through
Materials and methods
Specimen preparation
Pure Ti plates (10 × 10 × 1.5 mm; Kobe Steel Ltd, Japan) were sequentially polished with #400 to #1200 sandpaper. The PDA coating solution (2 g/L) was prepared by completely dissolving dopamine hydrochloride (Sigma-Aldrich, Germany) in a HCl buffer solution (0.01 mol/L, pH = 8.0). Subsequently, PDA was deposited on a Ti specimen by stirring the DOPA solution for 24 h. The self-polymerization of DOPA was visually confirmed by color change of the surface to dark gray. For silver coating, the PDA-coated Ti specimens were immersed in 50 mL silver nitrate solutions (Samchun Pure Chemical Co., Ltd, Korea) with silver nitrate concentrations of 5 g/L (Ag5) and 50 g/L (Ag50) for 30 min, and exposed to ultraviolet radiation (UV). Finally, the specimens were washed with distilled water and dried in a vacuum oven at 25°C.
Surface analysis
The surface morphologies of the Ti specimen coated with PDA and silver were observed by field-emission scanning electron microscopy (FE-SEM; Hitachi S-4800, Japan) and the chemical composition on the surface was confirmed using energy-dispersive X-ray spectroscopy (EDX; Oxford, UK).
Bacterial culture
Each strain was inoculated into the medium which was sterilized at 121°C for 15 min, and cultured at 37°C.
4 mL of each liquid culture was added to a test tube, and the prepared single colony was cultured at 37°C for 16–24 h. An aliquot of the culture medium was stored at −80°C.
McFarland standard turbidity was used as a reference to determine the density of the strain culture media. Each culture medium was adjusted to the turbidity (absorbance 0.09–0.13, density of strain 1 × 107 colony-forming units (CFU)/mL) of the McFarland Turbidity Standard No. 0.5 kit (bioMérieux, France) at 600 nm using a 6131 BioPhotometer (Eppendorf, Germany).
Microbial growth inhibition test by serial dilution assay
Briefly, the specimens were first placed in a 24-well plate, and 100 μL each of the
Microbial growth curve analysis
Growth curve analysis was carried out by measuring the absorbance at 600 nm to determine whether the PDA and silver coating on the surface of Ti affected the growth rate of the microorganisms. The strain culture medium was adjusted to an optical density of 0.02 at 600 nm (OD600). 5 mL turbidity-adjusted strain culture medium was inoculated into a test tube and cultured with specimen at 37°C. Finally, the culture medium was transferred to a 96-well plate every 3 h, and the OD600 value was measured using an enzyme-linked immunosorbent assay (ELISA) reader (EMax, Molecular Device). Figure 1 shows the schematic processing for the preparation of specimen and antibacterial tests.
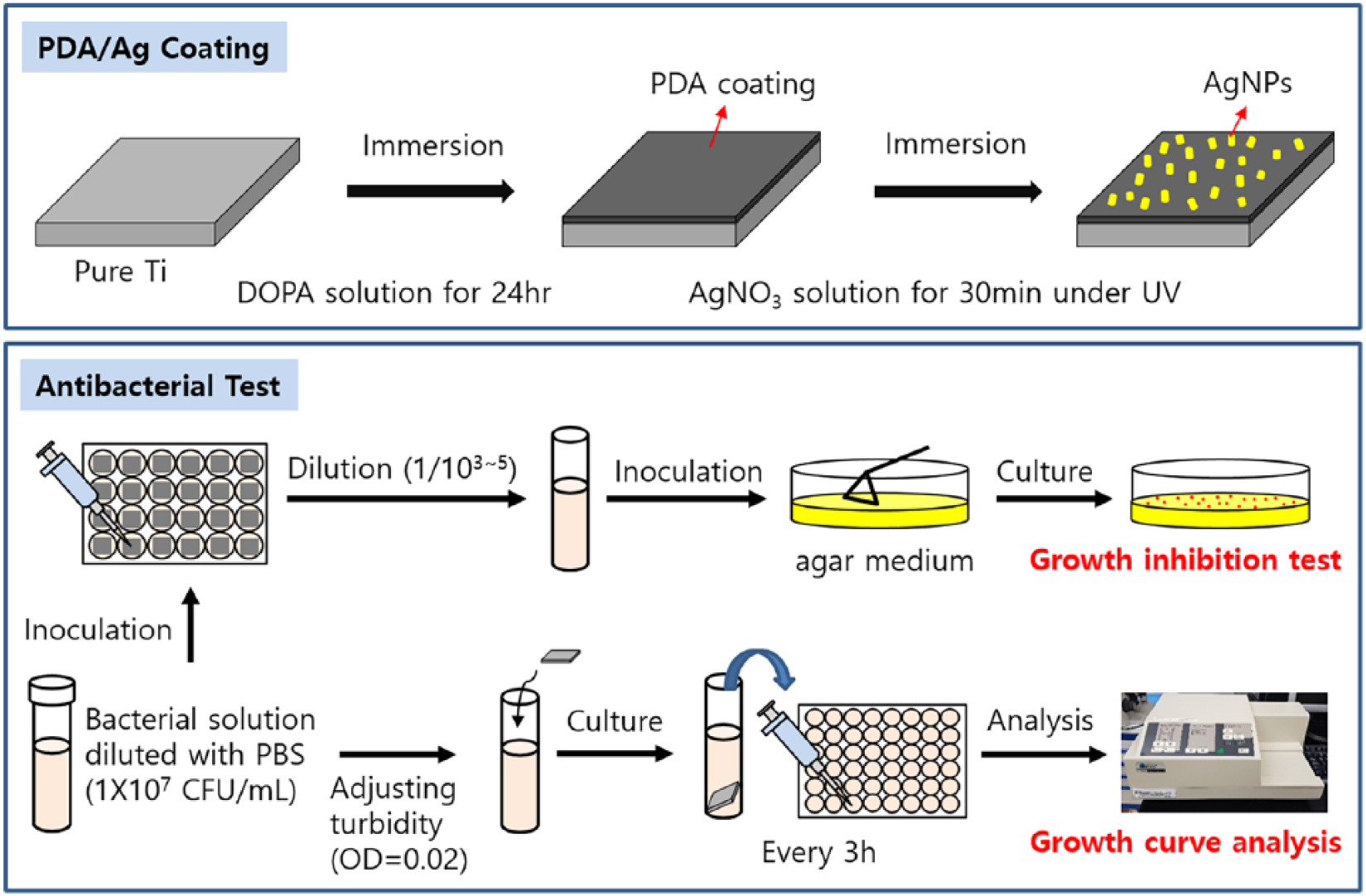
Schematic image for the experimental process.
Results
Surface analysis
Figure 2 shows the surface morphologies and the chemical composition on the surface of PDA and silver-coated Ti specimens, which were analyzed using FE-SEM and EDX. It was identified that silver nanoparticles (AgNPs) with a size of between 100~200 nm were uniformly incorporated in AgNPs-PDA coating layer on the Ti surfaces, and 2.4 wt.% silver was detected on the surface by EDX analysis.
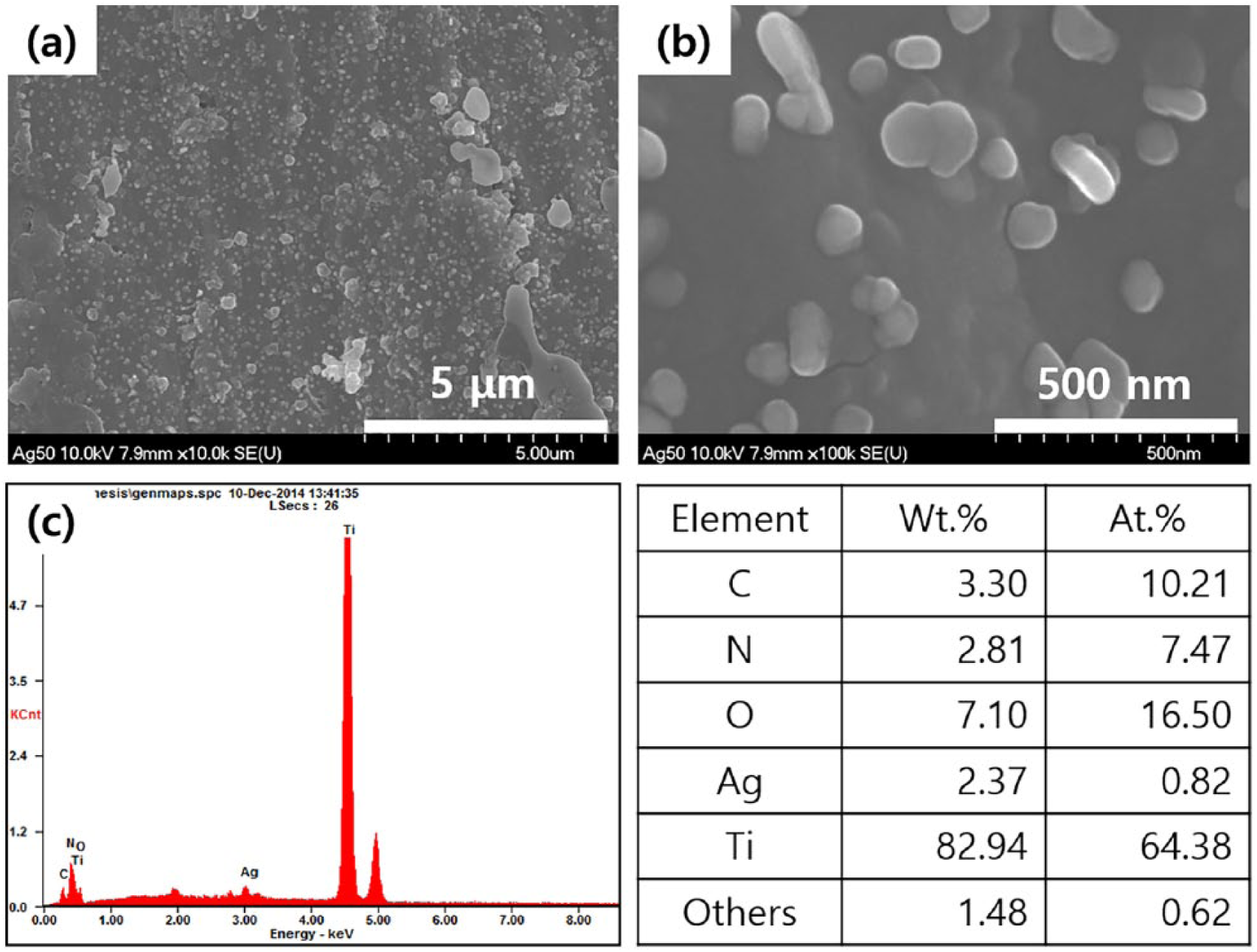
Scanning electron microscope (SEM) images at (a) low magnification (× 10,000) and (b) high magnification (× 100,000), and (c) energy dispersive X-ray spectroscopy (EDX) results for the PDA and silver-coated Ti specimens with silver concentration 50 g/L (Ag50).
Microbial growth inhibition test by serial dilution assay
Figure 3 and Figure 4, respectively, show the growth inhibition effect of

Growth inhibition effect of
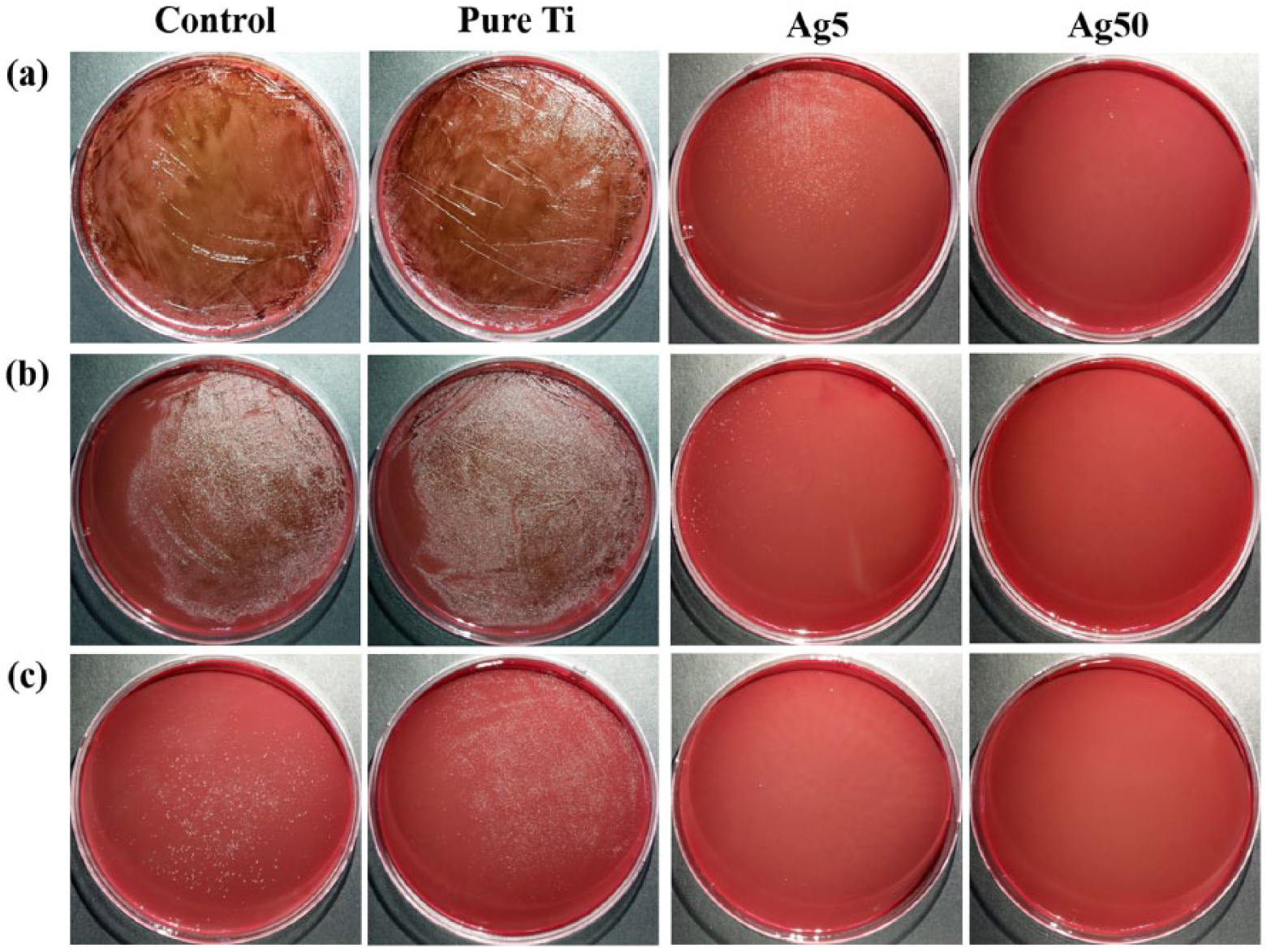
Growth inhibition effect of
Microbial growth curve analysis
Figure 5 shows the results of microbial growth curve analysis to verify the growth rates of
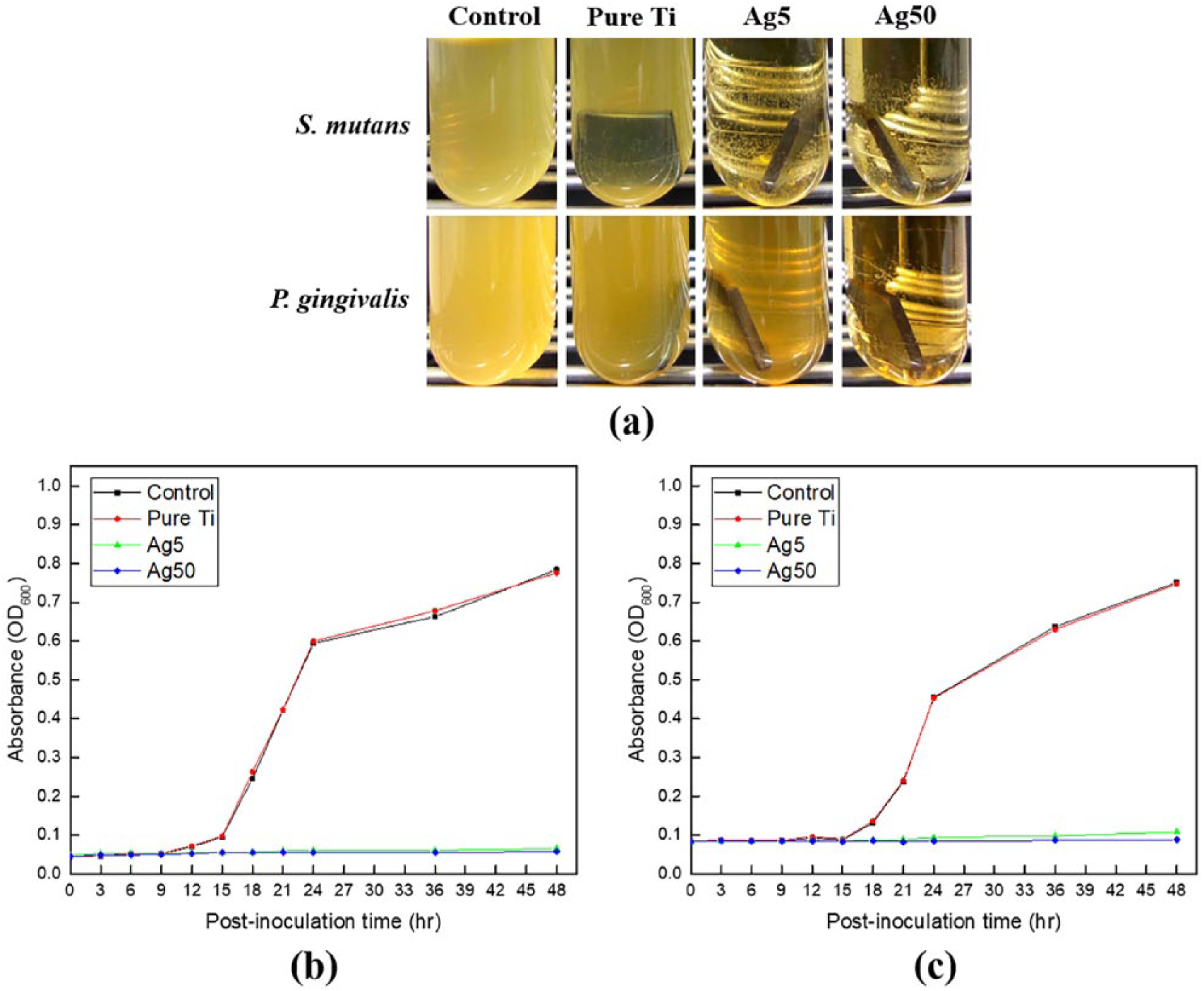
(a) A photograph for culture mediums in which the turbidity was changed after culturing
Discussion
Operations using hip and knee orthopedic implants and as well as dental implants are steadily increasing as the world is rapidly becoming an aging society.
39
In particular, dental implants have been used in a variety of applications such as recuperation of masticatory functions and aesthetic treatment. Titanium and its alloys are widely used as implant materials in dentistry and orthopedics due to their excellent mechanical properties, high corrosion resistance, and biocompatibility.
40
However, failures of Ti implantation due to bacterial infection have also been increasing.1,2 About 10% premature failures of Ti implant have resulted from the bacterial infection within a year after implantation, and especially it has been found that the major cause for the failures of dental implant were the tissue inflammation and bone resorption by some bacteria such as
Silver is widely used in medicine because it is less harmless to the human body, has excellent antibacterial properties, and is stable with regard to decomposition and volatilization. In the previous studies, some forms of silver have been shown to be effective in burns, chronic osteomyelitis, urinary tract infections, and central nervous system inflammation. It was also reported that the hydroxyapatite coating layer containing silver on the alumina substrate exhibits excellent antibacterial properties against the bacteria such as
Mussels produce and secrete adhesive proteins to adhere strongly on the rock against tidal current and rough waves of the sea. Using such properties, bio-friendly adhesives using mussel-derived materials have been actively developed since adhesives produced from natural organisms generally have excellent mechanical strength and durability. In previous research, lower cytotoxicity and better viability for the osteoblastic MC3T3-E1 and MG-63 human osteosarcoma cells were confirmed, and the surface was changed to more hydrophilic when PDA was coated on the pure Ti.23,24,26 Also, even though the significant differences in antibacterial activities between Ti and PDA-coated Ti were not confirmed in previous studies,49,50 the antibacterial activities for both gram-negative and gram-positive bacteria were increased and biofilm formation was inhibited for a long time with good biocompatibility when antibiotics or silver nanoparticles were incorporated in PDA coating on Ti, and new bone formation by encouraging biomineralization was promoted when using bioactive materials such as hydroxyapatite and collagen in PDA coating on Ti.27,28,49,51
Table 1 shows a brief summary of previous studies for the effects of the silver-loaded PDA coating on the antibacterial activities and biocompatibility of Ti. When the silver nanoparticles (AgNPs) were incorporated in PDA coating on the bare Ti or anodized Ti with micro or nano-structure compared with PDA coated and Ti, the antibacterial activities were maintained for much longer, and the proliferation of osteoblast cells was improved in comparison with PDA-coated or bare Ti, since the release of silver ions from the coating was retarded and the intrinsic cytotoxicity of Ag nanoparticles was compensated by the mineralization, which can promote the osseointegration of Ti implant.35,37,38,50,52–54
Summary of existing researches for the silver-loaded polydopamine (PDA) coating of titanium.

In this study, antibacterial activity against
Results of this study show that the growth of
Bacterial cells induce division processes for growth through active metabolism. The growth curve of bacteria can generally be divided into four phases: the lag phase, the exponential phase, the stationary phase, and the death phase. The lag phase is the period when bacteria adapt to a new environment. As shown in Figure 5, the Ti specimens coated with PDA and silver inhibited the metabolism of
Conclusions
The antibacterial characteristics for
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study has reconstructed the data of Soo-Hyoen Choi’s Master’s dissertation. This work was financially supported by ‘National Research Foundation of Korea’ (NRF) grants funded by the Korea government (MSIP) (No. 2014R1A4A1005309). Soo-Hyoen Choi and Yong-seok Jang contributed equally to this work and is considered as joint first author.
