Abstract
A series of bioactive glass scaffolds doped with SrO or ZnO (0, 5, and 10 mol%) were synthesized by the foam replica and melting method. The thermodynamic evolution, phase composition, microstructure, ion release, in vitro bioactivity, and oxygen density of the scaffolds were characterized. The proliferation of murine long bone osteocyte Y4 cells was studied by cell culture. The survival rate of the BGs evaluated the antibacterial activity and
Introduction
Bones, as structure, are composed of bone cells (osteoblasts, osteoclasts, and so on), bone matrix, and inorganic substance. 1 Large bone defects or functional disorders were caused by injuries, tumors, congenital diseases, sports accidents, osteoporosis, and arthritis. 2 The growth rate of bone tissue was abnormally slow, and it was challenging to treat bone defects and other related diseases through self-repair. 3 The major challenges of the clinical field are damage caused by bone defects and subsequent infection. Current treatments based on autogenous bone grafts have some limitations. Therefore, it is urgent to develop artificial bone graft materials with bone tissue repair funtion. 4
In bone repair, bioactive glasses (BGs) have been developed as bone-filler materials since they are bioactive and biodegradable. 5 The first commercial bioactive glass (BG), Bioglass® 45S5, invented by Hench et al. 6 have been developed and successfully applied in orthopedics bone defect repair, scaffolds, small bone grafting, bioactive coatings, and periodontal reconstruction. Due to their amorphous structure, bioactive glasses allow greater flexibility in composition, unlike crystals that are dependent on specific stoichiometry. This makes it possible to make bioactive glass with different physiological activity and therapeutic properties by incorporating varying concentrations and species of ions. 7 Since ion release, glass degradation, and apatite formation are the critical indicators for the clinical use of bioactive glass, researchers have conducted much research on them. 8 The relationship between the ion release, glass structure, and apatite formation was revealed by the analysis of bioactive glasses in various dissolution media (Tris buffer, 9 simulated body fluids (SBF), 10 or cell culture medium 11 ) combined with a variety of test methods.
Bone repair materials should not only promote osteocyte formation and osteogenic differentiation but also can inhibit bacterial growth. The clinical risk of bacterial infection with open bone injuries is high, up to 20%, while the risk of infection with closed fractures is approximately 3%–8%. The bacteria can cause serious bone infections that can even require amputation. Therefore, bone infection has always been a challenge in clinical treatment. 12 Making bioactive glasses have antibacterial properties is an effective method to solve this problem. 13
The element strontium (Sr) and calcium (Ca) have similar chemical and biological properties. 14 Strontium found in the bone, muscle, liver, and physiological fluid has antibacterial, augment osseointegration, and stimulating osteoblastic proliferation. 15 Extensive researches on strontium in bioactive glasses have been conducted by many researchers, confirming the benefits of strontium doped bioactive glasses. Many studies indicated the positive influence of strontium on antibacterial efficiency, 16 stimulating bone formation, 17 treating osteoporosis, 18 improving bone density, and reducing the risk of fracture. 19 Other reports indicated that strontium reduces biological activity by inhibiting or delaying calcium phosphate layer formation.20 –22
Zinc (Zn) is necessary for bone growth in the human body. Zinc can promote the growth, proliferation and differentiation of bone cells, and it also promotes DNA replication, protein production, and enzyme production.23,24 Zinc is ideal for developing bioactive glass because of its antibacterial and angiogenesis properties. 25 Adding zinc element to bioactive glass matrix can produce higher chemical stability and densification. 26 Zinc can inhibit the transformation of amorphous apatite to crystalline hydroxyapatite, hindering the nucleation rate of apatite in SBF solution. 27
The present study’s objective was to create porous bioactive glasses scaffolds using either strontium and zinc in place of calcium in the bioactive glass and study the effects of substitution elements on physicochemical properties, ion release, and bioactivity bioactive glass. The cell viability and proliferation were detected by Thiazolyl Blue Tetrazolium Bromide assay (MTT). The antimicrobial activity of the scaffolds was evaluated by the inhibition of E. coli (ATCC 25922).
Materials and experiments
Materials
Na2CO3, K2CO3, MgCO3, CaCO3, SiO2, H3BO3, NaH2PO4, SrCO3, ZnO (analytical grade, Sinopharm Chemical Reagent Co., Ltd, China) were used for the synthesis of BGs as a source of Na2O, K2O, MgO, CaO, B2O3, SiO2, P2O5, SrO, ZnO in BGs composition. NaCl, KCl, K2HPO4, MgCl2.6H2O, CaCl2, Na2SO4, (HOCH2)3CNH2, and HCl (analytical grade, Sinopharm Chemical Reagent Co., Ltd, China) were used to prepare the SBF. Polyurethane foam was provided by Hangzhou Hangmei Foam Co., Ltd, China.
Preparation of bioactive glass scaffolds
The elemental composition of bioactive glass (BG) is shown in Table 1. The mixture was heated and melted in a muffle furnace in a platinum crucible for 2 h at 1500°C. Uniform and transparent glass were obtained and then crushed and ground to obtain particles with particle size less than 30 μm.
Elemental compositions of the various BGs (mol%).

The slurry was prepared by stirring bioactive glass particles (53 wt%), anhydrous ethanol (44 wt%), and ethyl cellulose (3 wt%) at room temperature. Polyurethane foam with a porosity of 50 holes per inch was dipped into the slurry. The foam was dried at room temperature for 24 h, and heated at 400°C for 1.5 h to decompose the template foam, then heated at 570°C for 2 h to form a dense 3D scaffold network structure.
Preparation of SBF
The SBF immersion method was used to evaluate the synthesized bioactivity scaffolds in vitro. According to Kokubo’s et al. 28 method, the SBF solution was prepared by chemical reagents.
Degradation and mineralization of bioactive glass scaffolds in SBF
The degradation of scaffolds in SBF was used to characterize the weight loss of the scaffolds.
29
The scaffolds were immersed in SBF at 37°C (0.001 g\mL) and completely replaced with a fresh medium at a fixed time point. After the scaffolds were removed from the SBF solution, they were rinsed with anhydrous ethanol and deionized water three times, dried at 90°C for 24 h, and weighed to calculate the mass loss. The reacted scaffolds’ weight loss was defined as (Wo − Wt)/Wo, where Wo was the initial (unreacted) mass and Wt was the mass at an immersion time
Density measurements and calculation
Density (
Oxygen density (


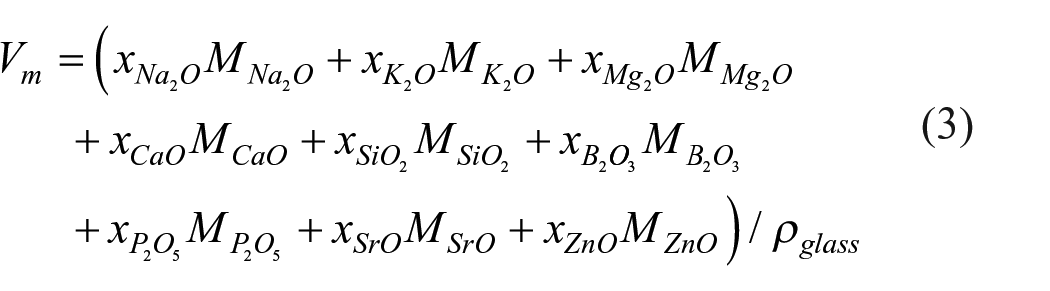
where
Characterization
Simultaneous Differential Scanning Calorimetry (DSC, Netzsch STA449F5) was carried out from room temperature to 1000°C (10°C/min) under Ar flow (20 mL/min) in platinum crucibles. The crystalline phases of the powder samples were analyzed by X-ray diffraction (XRD, Bruker D8). Scanning electron microscopy (SEM, Hitachi S4800) was used to examine the surface morphology of the specimens. Elemental analysis was performed by Energy Dispersive Spectrometer (EDS, Oxford INCA X-MAX).
MTT assay
Biocompatibility of the bioactive glass in vitro was investigated by MLO-Y4 (murine long bone osteocyte Y4) cells. The initial density of MLO-Y4 was 1 × 105 cells/scaffold, and they were cultured on 5 × 5 × 5 mm scaffolds placed in 96-well culture plates. MLO-Y4 cells were grown in Dulbecco’s modified Eagle’s Medium (DMEM) containing L-Glutamine 2 mM, 1 mM sodium pyruvate, streptomycin, and 10% (v/v) FBS (Fetal Bovine Serum-Invitrogen). The cells for the MTT assay were cultured in 96-well plates containing the sample eluates. The cell culture was conducted at 37°C ± 1°C, 90% ± 5% humidity, and 5% ± 1% CO2/air in an incubator. The cells were mixed with eluent for 1 and 3 days, and each sample was repeated three times. The scaffolds and 0.5 mg/mL of MTT (Sigma-Aldrich) were cultured at 37°C for the collection of formazans. The MTT medium was discarded after the incubation, and dimethyl sulfoxide (DMSO) was used to dissolve formazan. The outcomes were demonstrated as absorbance per well (minus the optical density of the blank ones) at 495 nm.
In vitro antibacterial activity of the BGs
The antibacterial activities of BG were evaluated by the spread plate method. E. coli (ATCC 25922) was used to evaluate the antibacterial activity of the BG. Disinfect all samples with ultraviolet light lamp for 2 h prior to use. Before the experiment, E. coli were cultured in liquid lysogeny broth (LB) medium and diluted approximately to 1 × 105 mL−1 at 37°C. BG powder and LB medium (10 mg, 0.9 mL) were added to the Eppendorf tube and stirred for 5 min. Then, 0.1 mL bacterial suspension was added to each Eppendorf tube, and cultured for 24 h at 37°C. The solution was diluted continuously 1000 times, then coated with 100 μL suspensions on LB-agar plates, and incubated overnight at 37°C in the dark. 32 The bactericidal effect was calculated by calculating the final colony-forming units. The tests were repeated three times.
Statistical analyses
The data (
Results and discussion
Physical-chemical characterization results
DTA determined the temperature of glass transition (Tg) and the crystallization temperature (Tc) to study the thermodynamic properties of BGs. BG, 5Sr-BG, 10Sr-BG, 5Zn-BG, and 10Zn-BG showed exothermic peaks at 690°C, 693°C, 695°C, 768°C, 770°C, respectively as shown in Table 2 and Figure 1(a). The Tg and Tc increased with the increase of the substitution amount of Sr and Zn. As a result, the Sr series’ processing window (Tc-Tg) decreased from 143°C to 135°C. By contrast, the processing window of the Zn series increased from 143°C to 209°C. This can be due to the disorder of glass structure caused by the substitution of Sr for Ca. 33 Since ZnO formed [ZnO4] in bioactive glasses structure entered the glass network, the resulting Tg and Tc of bioactive glass containing ZnO increased. 34 Thus, bioactive glass needs a suitable processing window for reprocessing that requires heat treatment, such as coatings, sintered samples, and porous scaffolds.
Characteristic temperatures of BGs.


(a) DTA thermograms curves of the different samples, (b) comparative XRD diagrams of the as-synthesized BGs, and (c) SEM images of BG scaffold.
XRD patterns of the BGs powder are presented in Figure 1(b). The results showed a typical glass amorphous peak. The introduction of Sr and Zn has not affected the amorphous character of bioactive glasses samples.
The SEM micrographs in Figure 1(c) display the microstructure of the BG scaffold. As can be seen from SEM analysis, there was no significant difference in microstructures among the five scaffold groups. Consequently, only SEM images of the BG scaffold are shown in Figure 1(c). The scaffolds possess a significantly porous structure with pore sizes of 200–400 μm. High magnification SEM images show that the scaffold’s surfaces are smooth (except particles).
Ions released and mineralization of BGs scaffolds
The weight loss of the reacted scaffolds in SBF at different points is shown in Figure 2. BG’s weight loss was significantly higher, while the weight loss of the remaining samples was smaller. When the tests were over at 28 days, the final weight loss was 24.20% ± 2.1%, 17.57% ± 3.2%, 12.55% ± 2.5%, 14.63% ± 1.8%, and 8.18% ± 1.6%, respectively, for the BG, 5Sr-BG, 10Sr-BG, 5Zn-BG, and 10Zn-BG scaffolds. Assuming that all the calcium was converted to HA and all the boron was released into SBF in the BG sample, the scaffold’s theoretical weight loss is 25.32%. After 28 days of SBF immersion, the BG scaffolds were almost completely converted to HA. The rest of the scaffolds can further convert into HA with prolonged soaking time. SrO can be used as only network modification oxide in the glass structure. Since the charge of SrO is balanced, its introduction does not change the network structure of the glass, forming two tetrahedrons connected by an ionic bond and leading to an increase in the glass’s chemical stability. ZnO can be used as network modification oxide and intermediate oxide in the glass network. 35 Part of the function of Zn2+ is to make [BO4] or [SiO4] replaced by [ZnO4] charge balance in the glass structure. The other part, Zn2+, can be used as a network modifier to improve the chemical stability of glass. 36 Incorporating Sr and Zn into the glass will enhance the chemical stability of glass, causing the degradation rate of glass to slow down.

Weight loss of five groups of scaffolds as a function of immersion time in SBF.
As the soaking time increases, more B, Si, Sr, and Zn ions are released into the SBF solution. When the HA was formed, the concentration of P ions in SBF declined. Figure 3(a) to (f) illustrates the change curves in the concentration of B, Ca, P, Si, Sr, and Zn in the SBF with soaking time up to 28 days. Figure 3(a) shows that the total amount of B released into the SBF solution were 579.3, 547.77, 465.3, 501.37, and 442.4 ppm for BG, 5Sr-BG, 10Sr-BG, 5Zn-BG, and 10Zn-BG. Figure 3(b) shows that Ca concentrations in SBF solution for BG, 5Sr-BG, 10Sr-BG, 5Zn-BG, and 10Zn-BG increased significantly from 60 ppm in the SBF solution to 242.81, 226.83, 191.38, 214.82, and 210.91 ppm on day 1, 3, and 7, as a result of cationic release from the scaffold. These values rapidly decrease on day 7 for BG, 5Sr-BG, 10Sr-BG, 5Zn-BG, day 14 for 10Zn-BGs to about 140.12, 157.35, 159.64, 176.23, and 170.51 ppm, indicating HA was formed on the surface of the scaffold at different growth rates. The trend of Ca for BGs changed due to the formation of the hydroxyapatite layer on the BG scaffolds surface. 37 The formation of the hydroxyapatite layer consumed large amounts of Ca ions. Additionally, ion concentration was also related to the formation rate of the hydroxyapatite layer. 38

The variation of elemental concentration in the SBF with soaking time for boron (a), calcium (b), phosphorus (c), silicon (d), strontium (e), zinc (f).
From Figure 3(d), Sr-BGs shows an increase in the amount of Si releases into the solution. The trend was more evident with the increase of Sr content; the opposite was observed for Zn-BGs where Si concentration was lower than BG and decreased with an increase in Zn content. The release amount of Si ions was related to Sr and Zn ionic radius. The ionic radius of Sr2+ (118 pm) is larger than that of Ca2+ (100 pm), which leads to the looser structure of Sr2+ network, thus increasing the ions release of the Sr-BGs. The lower ionic radius of Zn2+ (74 pm) concerning Ca2+ has the opposite effect.
The release amount of Sr and Zn was related to Sr and Zn’s content in the scaffolds. The Sr and Zn release in the SBF solution was almost two times higher for 10Sr-BG and 10Zn-BG in comparison with 5Sr-BG and 5Zn-BG, respectively (Figure 3(e) and (f)).
Figure 4 shows the XRD patterns of the scaffolds soaked in the SBF solution at different time. After 1 day of soaking in the SBF solution, the XRD pattern indicated the presence of HA on the surface of the BG scaffold (Figure 4(a)) by the appearance of a peak at 33.09° corresponding to (300) plane (according to a reference HA, Ca10(PO4)6OH2 (JCPDS 47-1758)). The peak became more pronounced over soak time. The rest scaffold exhibited an amorphous phase on the day. 5Sr-BG, 10Sr-BG, and 5Zn-BG scaffolds (Figure 4(b)–(d)) are observed a diffraction peak of (300) at 33.09° after soaking 3 days. 10Zn-BG scaffold (Figure 4(e)) is observed a diffraction peak of (300) at 33.09° after soaking 7 days. 10Sr-BG and 5Zn-BG scaffolds appear a diffraction peak of (112) at 31.92°. The XRD results indicated that the scaffolds’ bioactivity reduced with increasing Sr and Zn content in BGs. Zn has a more pronounced effect on the formation of HA compared to Sr.

The XRD patterns of synthesized BGs after 1, 3, 7, 14, and 28 days soaking in SBF, (a) BG, (b) 5Sr-BG, (c) 10Sr-BG, (d) 5Zn-BG, and (e) 10Zn-BG.
SEM and EDS analysis are carried out for BG (Figure 5(a1)–a6), 5Sr-BG (Figure 5(b1)–(b6)), 10Sr-BG (Figure 5(c1)–(c6)), 5Zn-BG (Figure 5(d1)–(d6)), and 10Zn-BG (Figure 5(e1)–(e6)) scaffolds soaked in the SBF solution for 1, 3, 7, 14, 28 days. The surface of all samples was completely covered with a hydroxyapatite layer after soaking with SBF, which was significantly different from the relatively smooth surface before soaking. Some spherical nuclei of HA were observed on the BG scaffold surface (Figure 5a1). As shown in Figure 5(a2) to (a5), the apatite assembles into sub-micro-spherical aggregates, which further form a dense layer covering the entire scaffold. The rest scaffolds need to be soaked for a longer period for this phenomenon to happen. EDS analysis of the surfaces after SBF solution immersion detected prominent characteristic peaks of Ca, P elements; the Ca/P atomic ratio of 1.60–1.70 was comparable to that for HA.
SEM images for the BG (a1–a5), 5Sr-BG (b1–b5), 10Sr-BG (c1–c5), 5Zn-BG (d1–d5), and 10Zn-BG (e1–d5) scaffold before soaking in SBF; EDS analysis for the BG (a6), 5Sr-BG (b6), 10Sr-BG (c6), 5Zn-BG (d6), and 10Zn-BG (e6).
The results of ion release, XRD, and SEM confirmed each other. When the Ca ions concentration began to decrease, the hydroxyapatite phase appeared between 1 and 7 days in XRD, and spherical particles appeared in SEM images in the same period. SrO and ZnO could be used as network modifiers to balance charge. This removed cations from the silica network, reduced non-bridged oxygen, and decreased the glass’s biological activity by reducing its dissolution. 39 The degradation process of bioactive glass first involved the rapid exchange of Na+ and H3O+ ions in the surrounding solution, but this process was hindered by the doping of Sr and Zn ions. The increase of Sr and Zn ions can decrease the release of alkaline earth metal ions (Ca2+, Mg2+) and alkaline metal ions (Na+, K+).27,40 Therefore, the spheroidal particle structure on the sample surface was less evident, and the crystallinity was reduced compared to the BG sample.
Bioactive glass density and oxygen density
Figure 6 shows the variation of the molar volume (a) and oxygen density (b) as a function of CaO substitution by SrO or ZnO. The values showed an increasing molar volume with increasing Sr substitution and a decreasing molar volume with increasing Zn substitution. The trend of oxygen density is opposite to that of molar density as SrO and ZnO substitute CaO.

The variation of molar volume (a) and oxygen density (b) as a function of CaO substitution SrO or ZnO in Sr-BGs and Zn-BGs (mol%), respectively.
The change of ion radius of substituted elements significantly affects the packing of the silicate network (molar volume and oxygen density) of the glass. 41 When smaller ions are replaced by larger ions (Sr2+ instead of Ca2+), the glass network expands with a larger molar volume and a decreased oxygen density. On the contrary, when Ca2+ replaced Zn2+, the glass network becomes denser, the molar volume becomes smaller, and the oxygen density increases.
Oxygen density is the mass of oxygen atoms per unit volume and is comparable for different glass systems where the number of oxygen atoms is constant.
42
In this study, strontium oxide or zinc oxide was used instead of calcium oxide on a molar basis, and thus the number of oxygen atoms remained the same. The results showed that a more disrupted silicate network contributes to ion release, then the Si ion release rate of strontium-replaced glass was faster because the network was more extensive (caused by larger modified ions), making it easier to permeate water and ion exchange. On the other hand, the Si ion release rate from zinc-substituted glasses was slow because the network was more compact, impeding water molecules’ penetration.
43
This indicated that Si ion release in the bioactive glass is directly controlled by the packing of silicate networks
In vitro evaluation of cell proliferation and antibacterial properties
To make a constant comparison in MLO-Y4 cells’ proliferation on various BGs scaffolds, MTT assay is conducted (Figure 7). As can be seen from the results, the growth of cells increased with the change of culture time. MTT results revealed that 5Sr-BG had a higher effect on cell proliferation than BG, while the remaining samples had no significant effect on cell proliferation. A small amount of strontium doping can promote cell proliferation. Zinc doping significantly inhibited cell proliferation. Inhibition of cell proliferation depends not only on the amount of zinc oxide added but also on the composition of glass and its degradation rate.
44
None of the scaffolds was obvious toxicity to cells. The cell proliferation was no apparent differences (

MTT after 1 or 3 days of MLO-Y4 cells cultured.
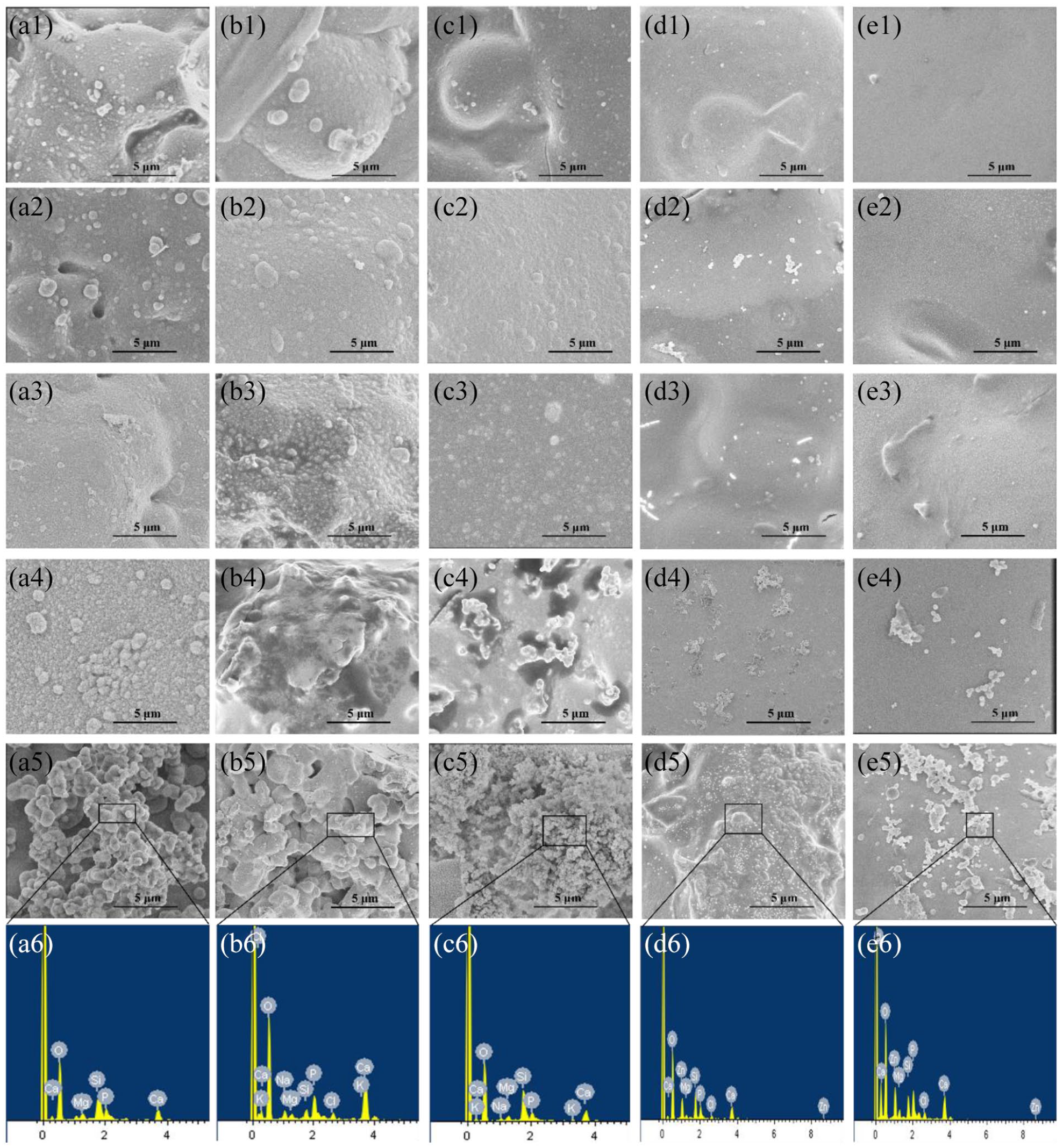
The spread plate method was used to evaluate the antibacterial activity of BGs against E. coli (ATCC 25922). Twenty-four hours later, the number of viable bacteria colony-forming units was numbered. Figure 8 shows that the BGs inhibited the proliferation of E. coli. Fewer CFUs of 10Sr-BG (16 ± 3) and 10Zn-BG (12 ± 5) were recorded than those of BG (363 ± 12), 5Sr-BG (68 ± 7), 5Zn-BG (110 ± 10), and blank (400 ± 14). Figure 9 demonstrates the histograms of the antibacterial activity of the BGs powders against E. coli bacterial strains. Sr series and Zn series have an apparent bactericidal effect. Compared with blank, the bactericidal efficiency of BG was statistically significant (*
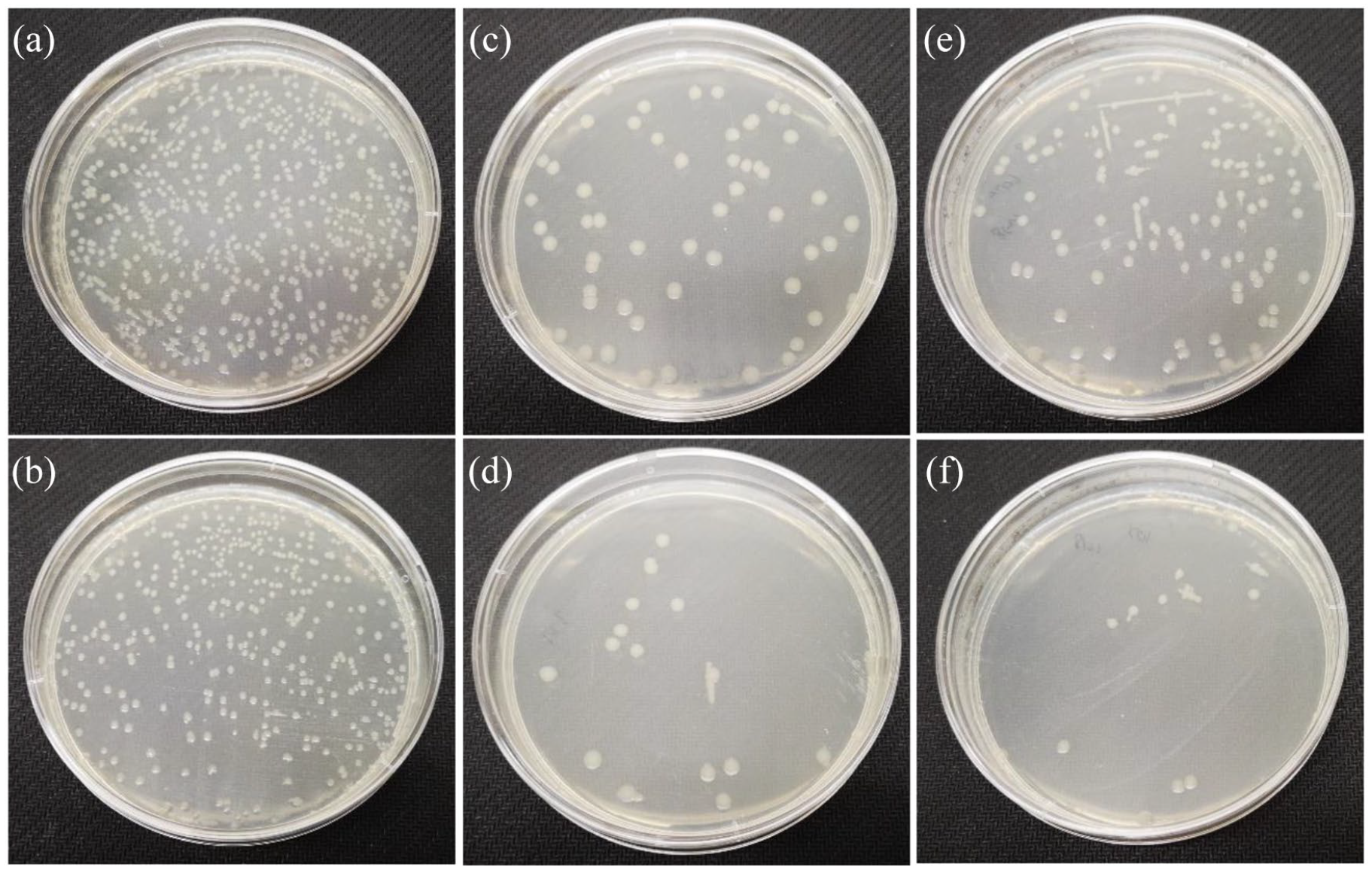
Representative images of E. coli bacteria grown, (a) blank, (b) BG, (c) 5Sr-BG, (d) 10Sr-BG, (e) 5Zn-BG, and (f) 10Zn-BG.

Histograms of the antibacterial activity of the BGs powders against E. coli bacterial strains.
About 30% of proteins contained metal ions in their molecules. 45 Half of the proteins require metal ions to perform their functions. The most important ions for bacteria are iron, manganese, and magnesium, but trace amounts of transition metals are also needed. As the concentration of the transition metal rises, the bacteria try to adapt, and toxic phenomena appear when the optimal concentration is exceeded. 46
Potential pathways by which metal ions impede bacterial proliferation include: (i) damaging the bacterial cell membrane 47 ; (ii) enzyme modification 48 ; (iii) oxidative stress (production of reactive oxygen species (ROS) and intercellular reactive oxygen species) 49 ; (iv) DNA damage, the regulatory role of DNA and RNA. 50
The antibacterial mechanisms of Sr and Zn ions are different. Sr ions can cause enzyme blockage and loss of function, leading to bacteria’s death. 51 The cell membrane contains electronegative molecules with a high affinity for metal ions, and Zn2+ binds to them, thereby inhibiting the normal function of the cell. Zinc can prevent the entry of essential ions in bacteria, accumulate in the body, and produce toxic effects, ultimately leading to death. 52
Conclusion
The bioactive glass scaffolds with porous structure (200–400 μm) were prepared. A systematic investigation was performed on BG, Sr-BGs, and Zn-BGs scaffolds with different SrO or ZnO (5 and 10 mol%) contents in vitro bioactivity and antibacterial activities. The result of DTA indicated that 10Zn-BG has the largest processing window. When immersed in SBF, the Sr and Zn in glass retarded the degradation process of bioactive glass but did not affect hydroxyapatite (HA) formation after 28 days of soaking. The ion release, glass dissolution, and surface degradation were controlled by the change of ion radius and variations in network packing. MTT results revealed that 5Sr-BG had the most obvious effect on cell proliferation. Sr-BGs and Zn-BGs exhibited significant antimicrobial efficiency against E. coli bacterial strains. Also, the experimental results indicated that 5Sr-BGs could be used as a potential candidate material for bone tissue engineering.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the Anhui Emphatic Research and Developmental Projects (201904a05020080); Key Natural Science Project of Bengbu Medical College (2020byzd170).
