Abstract
This review presents “a state of the art” report on sustainability in construction materials. The authors propose different solutions to make the concrete industry more environmentally friendly in order to reduce greenhouse gases emissions and consumption of non-renewable resources. Part 1—the present paper—focuses on the use of binders alternative to Portland cement, including sulfoaluminate cements, alkali-activated materials, and geopolymers. Part 2 will be dedicated to traditional Portland-free binders and waste management and recycling in mortar and concrete production.
Introduction
With the dawn of the 21st century, the world has entered an era of sustainable development. As a consequence, the concrete industry has to face two antithetical needs: how to feed the needs of the growing population, while being—at the same time—sustainable?
Sustainability in the construction industry can be achieved through three different routes (Figure 1), with reductions in consumption of gross energy, polluting emissions, and non-renewable natural resources. Different strategies can be identified along these three routes to make the concrete sector more environmentally friendly (Figure 2): (a) using alternative fuels and raw materials to reduce CO2 emissions to produce Portland cement; (b) replacing Portland cement with low-carbon supplementary cementitious materials (SCMs); developing alternative low-carbon binders (alkali-activated materials, geopolymers, and calcium sulfoaluminate cements); and (c) reducing natural resource consumption through to waste management and recycling.1–3,4
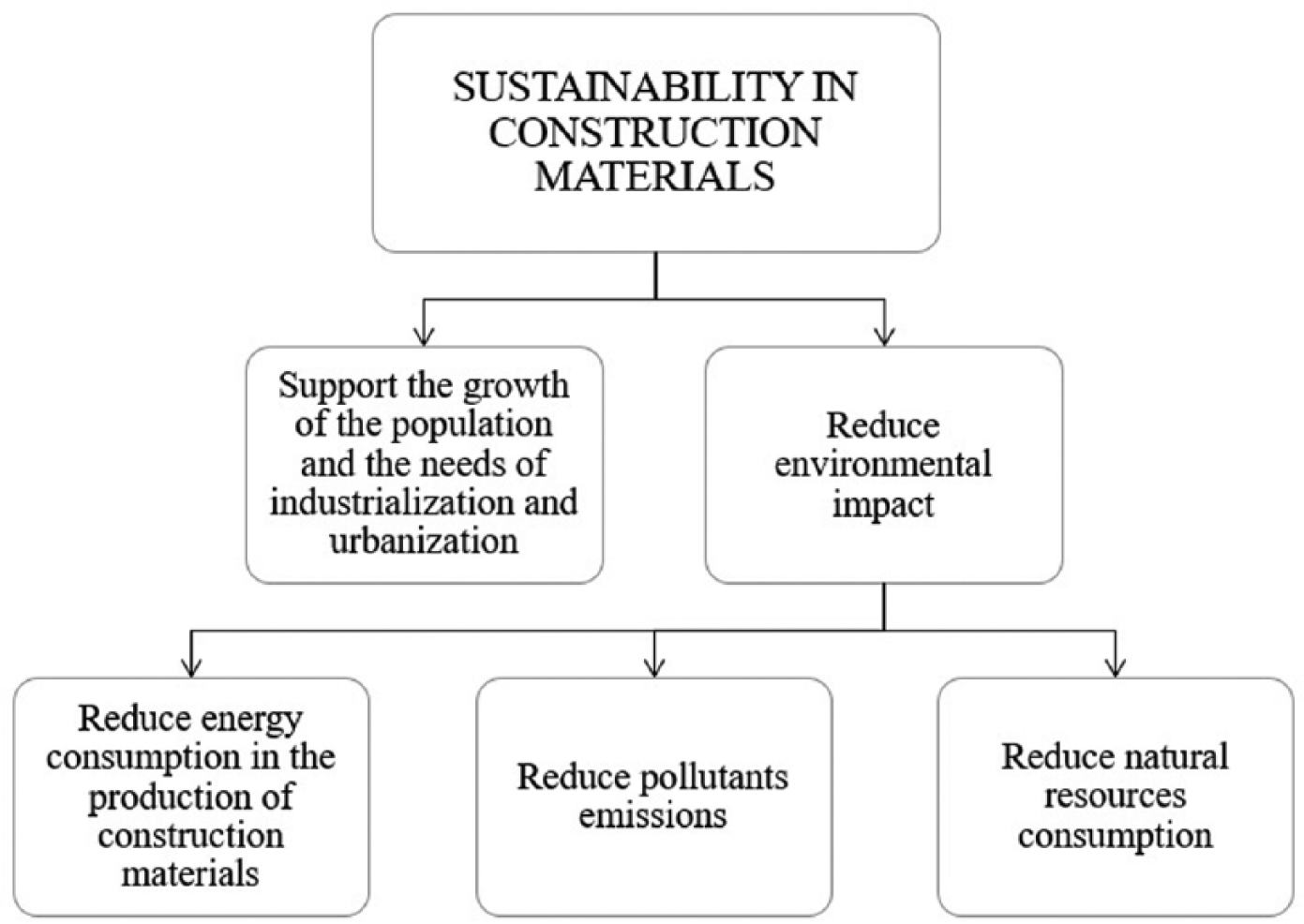
Main strategies to make the concrete sector more environmentally friendly.
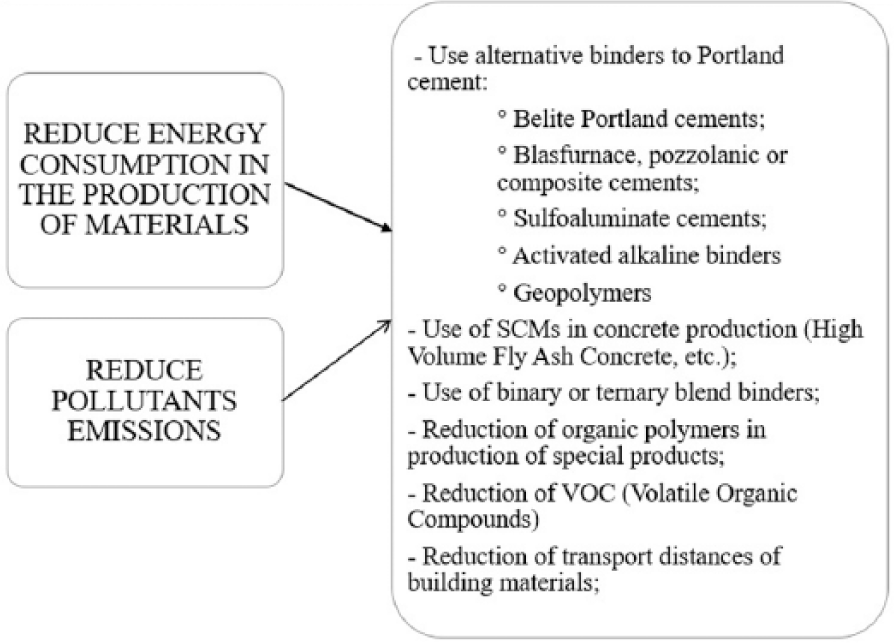
Reduction of energy and pollutants emission in construction materials production.
Part 1 of this review—the present paper—deals with use of binders other than Portland cement. Part 2 will be dedicated to waste management and recycling in mortar and concrete production.
Alternative binders to Portland cement
Different alternative binders to traditional Portland cement have been proposed, for example, sulfoaluminate cements, activated alkaline binders, and geopolymers.
Sulfoaluminate cements
Calcium sulfoaluminate cements (CSA) have been applied since the end of the 1950s.
5
In the mid-1970s, CSA cement was produced on an industrial scale in China by burning limestone, bauxite, and gypsum at 1300–1350°C.
6
In China, CSA cements are treated as a special binder with rapid setting, shrinkage compensation, and high early-age strength. The main phase of CSA is tetracalcium trialuminate sulfate or ye’elimite
The hydration process of CSA cement has been studied.18–22 The first hydration reaction in the presence of gypsum
The formation of ettringite
Study performed on CSA cement paste have revealed that a bimodal pore distribution develops at a very early stage; lower porosity is dominant, but not connected with higher porosity. 28 Moreover, the average pore size of CSA concrete is smaller than that of OPC. The porosity of the CSA mortar decreases with the increase in anhydrite content and the decrease in w/c ratio. 27 A series of factors can influence the expansion of CSA concrete,8,29 but ye’elimite content plays an important role. If the proportion of ye’elimite is more than 50% in CSA cement, expansion, cracking, and loss of strength appear at later stage; the appropriate content of ye’elimite seems to range from 30% to 40%. 30
Limited results are reported in the literature on the durability of concrete and mortars manufactured with CSA cement.27,31–33 In particular, Quillis showed that CSA-based concretes exhibit excellent sulfate resistance but higher diffusion coefficient in Cl-rich environments with respect to OPC. 13 Moreover, despite conflicting results on the carbonation rate of mixtures containing CSA, it is evident that, similarly to OPC-concretes, carbonation depth is directly proportional to the water/binder ratio.31,34
Alkali activated materials
Alkali-activated materials (AAMs), which were developed starting from the 1940s, 35 are obtained by reaction of an alkali metal source with amorphous or vitreous calcium-aluminosilicate precursors. The former is used to increase the pH of the reaction mixture thus accelerating the dissolution of the powders, while the composition of the latter determines the physical-chemical processes that produce hardening.36,37 Microstructures, workability, and strength and durability of AAMs can be tuned by the proper combination of activators and precursors. Mix design of AAMs includes materials from both natural sources (metakaolin (MK) and pozzolans (P)) and by-products (slag (GGBS), fly ash (FA), and paper sludge (PS)). AAMs can be classified according to the nature of the precursor (CaO–SiO2–Al2O3 system) into two main categories: (a) high calcium and (b) low calcium. When alumino-silicate sources (MK, FA) are used, a (Na,K)2O–Al2O3–SiO2–H2O system is generated. This can be considered a subset of AAMs usually referred as geopolymers, characterized by a peculiar pseudo-zeolitic network structure.38–40 When slag is used as precursor, a (Na,K)2O–CaO–Al2O3–SiO2–H2O system is produced. It is activated under moderate alkaline conditions,41,42 and hardening is produced by the formation of a C–A–S–H gel. A combination of the preceding two systems is also possible,43,44 where hardening is due to the formation of a C–A–S–H and (N,C)–A–S–H gels network. 45
The reactive powder used to produce the calcium-rich binder is blast furnace slag originating from the purification process of iron ore to iron. 46 Ground granulated blast furnace slag (GGBFS) is a mixture highly glassy phases with a composition close to that of gehlenite and akermanite: (31–38%) SiO2, (38–44%) CaO, (9–13%) Al2O3 and (7–12%) MgO, and S, Fe2O3, MnO, and K2O with percentages of less than 1%. When it is used to produce AAMs, parameters affecting GGBFS reactivity are the vitreous phase content (85–95%wt), its degree of depolymerization (DP, from 1.3 to 1.5), and its specific surface (400–600 m 2 /kg). 36
Slag alkaline activation consists of dissolution of the glassy particles,35–37 nucleation and growth of the initial solid phases, interactions and mechanical binding at the boundaries of the phases formed, ongoing reaction via dynamic chemical equilibria, and diffusion of reactive species through reaction products formed at advanced times of curing.47,48 At the early stages, the alkaline solution reacts with dissolved species generating the outer C–A–S–H. At later stages, the inner C–A–S–H gel is produced by ongoing reactions of the undissolved portions of the slag particles through a diffusion mechanism. 49
The cations and anions of the activator play a specific role in the activation process. When hydroxides are used, the OH– acts as a catalyst and is responsible for the pH increase, thus allowing precursor dissolution and the formation of stable hydrates. 50 Slag-based binder can be prepared using 2–4 M solution with Na2O content less than 5% slag weight to guarantee mechanical properties and reduce efflorescence.51–54 When sodium silicate is used, the gel is characterized by lower Ca/Si and a less ordered structure. In both the cases, the gel is comprises coexisting 11 and 14 Å desordered tobermorite-like phases, 48 with a Ca/Si ratio (0.9–1.2) lower than in hydrated Portland cement system. AFm type phases or strätlingite are formed when NaOH or silicate, respectively, are used.55,56 If raw materials contain high amounts of MgO,57,58 hydrotalcite (Mg6Al2CO3(OH)16·4H2O) is produced, while in the presence of low MgO (<5 %) and high Al2O3 contents, zeolites are often found in the reaction products. 59
MK- and FA-based geopolymers
As a general statement, MK can be said to be the “model system” for studying the activation process of AAMs.60–68 MK (Al2O3:2SiO2) is a natural pozzolanic material obtained by the calcination of kaolin at 500–900°C.69,70 MK consists of plate-like particles with a specific surface area generally between 9 and 20 m2/g. 71 MK pastes usually require a liquid/MK > 0.6 by mass, 72 and MK mortars need ∼1.0.73,74 In general, MK geopolymers set within 24 h. Conversely, MK geopolymers have a higher reaction rate and a faster strength gain with respect to FA geopolymers,75,76 because of the presence of secondary minerals in the kaolinite clay,77,78 the fineness of the particles, 79 and the reaction temperature.80,81 Hydrothermal ageing (95°C) because of major formation of crystalline zeolite, is responsible for strength loss. 76 Moreover, the thermal treatment of MK mixtures at 80°C accelerates strength development, but the final strength is lower than that of specimens cured at ambient temperature. 73 In geopolymers, SiO2/Al2O3, Na2O/Al2O3 and Na2O/H2O influence mechanical properties. Compressive strength and Young’s modulus were found to be dependent on alkali type (Na or K) and Si/Al ratio. 82 However, at the same compressive strength, the authors found that the modulus of elasticity is lower in geopolymers than in OPC mortars due to the large number of small pores that promote the formation of micro-cracks.75,83 K-based geopolymers produce a higher compressive strength than Na-based ones, and the increase of SiO2/Al2O3 also increases mechanical strength. 84 Davidovits indicated that the optimum Na2O/Al2O3 and SiO2/Al2O3 ratios are 1 and 4, respectively, 85 while most researchers reported an optimum SiO2/Al2O3 of 3–3.8,54,86 The increase in Si/Al ⩾ 3 leads to chemical instability in air, with efflorescence formation on the surface attributed to the high residual free alkali cations. 87 Usually, increasing Na2O/H2O leads to improved dissolution ability and mechanical strength development of clay-based geopolymers.74,84,88,89 Geopolymers prepared only with MK are highly susceptible to shrinkage both at room and elevated temperatures,72,90–94 because of their high water requirement.
Concerning the effect of aggressive/pollutant substances on MK concretes durability,95–98 Palomo et al. found that MK geopolymers were stable if immersed in seawater, Na2SO4 solution (4.4 %), and H2SO4 solution (0.001 M) for up to 90 days. 99 On the contrary, Mobili et al. noticed crack formation on MK geopolymers exposed to Na2SO4 solution (14 %), not present in FA geopolymers with the same activators. 75 Gao et al. found that MK geopolymers remain sound after 28 days in HCl solution (pH 2).100–102 The capillary water absorption of MK-based geopolymers is higher than blended blast furnace slag,103,104 FA, or OPC geopolymers. 75 Currently, researchers are studying MK-based geopolymers also to produce non-structural plasters with lightweight aggregates for thermal insulation,105–110 and to be used as mortars able to adsorb volatile organic compounds (VOCs).111–115
Another trend is to produce geopolymers suitable for refractory applications, adding a foaming agent, H2O2, or Al powder.106,116,117 Results show that only Al-geopolymers are successfully converted to crack-free ceramics upon heating. 116 The partial substitution of MK with FA also has a positive influence on both thermal resistance and compressive strength,106,118 thanks to the lower water demand and thus the lower free water evaporation. Foams have much lower thermal conductivity (0.15–0.4 W/m·K) than the solid geopolymer (0.6 W/m·K). 119 The thermal conductivity increases with increased Si/Al ratio, because of the increased connectivity, reduced porosity, and finer pore size distribution.120,121 Moreover, increasing the K/Al ratio also increases the foaming efficiency (final volume/initial volume). 122 Geopolymers derived from a K-based activator dissolve or degrade more readily compared with Na-based geopolymers whether foamed or not.123,124–126
Researchers have investigated “one-part” MK geopolymers, obtained by adding only water to the dry materials, avoiding the use of caustic solutions, by the calcination of the clay material with a powdered activator, such as NaOH or KOH, 127 soluble sodium silicate, 128 sodium carbonate, 129 or by using an alkali-rich by-product, such as potassium-rich biomass ash. 130
FA is an industrial byproduct derived from coal-fired power stations with a highly variable composition, dependent on the coal source and burning conditions. 131 Particle size distribution, chemical composition and crystalline/glassy phases of the precursor are key factors that need to be understood, since they control the precursor reactivity and solubility in alkaline solutions. 132 It was found that the geopolymer microstructure is highly influenced by particle fineness, amorphous phase composition, and oxide content (particularly Fe2O3 and CaO).133,134 The lower Na2O/SiO2 molar ratio, the higher the performances in terms of water absorption and mechanical properties. 135 Geopolymers showed good thermal stability after firing due to the formation of new crystalline phases. 136 Developing a comprehensive knowledge of precursors is a fundamental and critical step in commercializing geopolymer products. For example, a preliminary study demonstrated the use of geopolymer mortars for strengthening of concrete structures. 137 As workability is one of the main requirements, research on superplasticizers suitable for fly ash geopolymers needs to be emphasized.138–145,146
Clayey sediments and sludge for geopolymers
Geopolymers are attractive because natural and industrial silico-aluminates wastes may be used as precursors. The exploration for alternative low cost and easily available materials led among others to “normal clays”. Clayey sediments consist of different clay minerals; these are widely available all over the world, and offer significant reactivity after a thermal activation process. 147 Among silico-alumina wastes, reservoir sediments are worthy of consideration. Sediments should be removed periodically to avoid reduction in reservoir capacity. There are more than 7000 large reservoirs in the European Union (EU), of which 564 are in Italy. These data show that regular dredging operations can produce huge amounts of sediment. In this regard, some possibilities have been explored as raw materials in production of artificial aggregates, bricks and cement.148–153 Studies aimed at extending these possibilities in the field of geopolymer materials have been reported by several authors.154–156
SiO2 and Al2O3 are the main components in sediments, while CaO and Fe2O3 are present in lower concentrations; K2O, MgO, and Na2O are present in minimum percentages. The main mineralogical phases detected by X-ray diffraction analysis are quartz, calcite, clay phases, and feldspars. A pre-treatment of the sediment is always necessary in order to enhance the reactivity in alkaline environments. Messina et al. showed that 750°C is the optimal pre-treatment temperature for the production of concrete blocks and geopolymer mortars. 157 Indeed, the 27Al NMR peak at 0 ppm related to octahedral Al (Figure 3) and absorbance FT-IR peaks at 3697, 3620, and 3415 cm–1 were absent or greatly dampened, evidencing the collapse of the ordered clay structure.
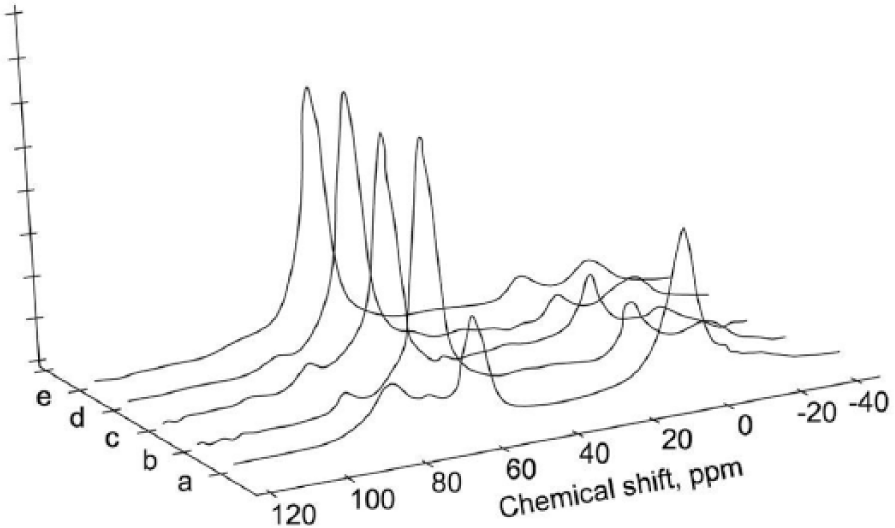
27 Al NMR resonance spectra of Occhito sediment before thermal treatment (a), after thermal treatment at 650°C for 1 h (b), at 650°C for 2 h (c), at 750°C for 1 h (d), at 750°C for 2 h (e).
The prevailing chemical components of the sediments are silica and alumina, thus making sediments good geopolymer precursors. However, within the wide range of natural and artificial silico-aluminates, the SiO2/Al2O3 ratio in this case is quite high, such to make the alkali aluminate activation and/or the addition of alumina-rich additives an interesting alternative. Aluminate activation was studied in the manufacture of precast building blocks, with encouraging results. 158 Regarding alumina-rich additives, water potabilization sludge is another key residue produced by reservoir management activities. These wastes are based on flocculation-clarification processes using alumina coagulants. 159 The amount of sludge generated, and its chemical composition, depend mainly on the chemical and physical characteristics of the water, the efficiency of the removal process, and the type and dose of coagulant. The amount of sludge can be roughly estimated in the range of 1–5% of the total amount of untreated water. 159 This waste has been studied in only a few studies, mainly with regard to potential reuse in the construction industry.157,160–166
Management of the huge amounts of sediments coming from dredged activities is an important issue to be solved in many countries worldwide. Clean dredged materials can be used for construction fill, brick or asphalt manufacturing, topsoil and marine projects. Recently, Lirer et al. proposed dredged sediments with FAs in the production of geopolymers. 165 Regarding the environmental impact, the values of hazardous elements classify geopolymers as non-dangerous materials. Therefore, these preliminary results suggest that this methodology could represent a starting point for the investigation of possible beneficial uses of polluted sediments in geopolymeric matrices.
Corrosion behavior in alternative binders-based matrix
Replacement of Portland cement with alternative binders, especially CSA cements and AAMs, open the theme of protection of reinforcements in these new concretes.168–170 Data seem to indicate that the durability of CSA concretes is at least comparable to that of traditional Portland cement mixtures, but they also evidence the need to perform long-term tests in order to recognize the corrosion protection mechanism. 24 The protective capacity of the CSA-matrix has been confirmed by positive experiences of these structures in China, in which no rebar corrosion occurred after 14 years of exposure. 171 However, little information is given about the actual aggressive environmental conditions. Most works available are devoted to the study of hydration products in the very early period but only a few papers have addressed the corrosion behavior of reinforcements by means of electrochemical techniques. Potential measurements performed in a few experimental works evidence difficulties in achieving proper values of passive rebars due to the low alkali content of the pore water. 172 Studies on the durability of mixtures manufactured with such binders address only a few aspects—carbonation and chlorides—neglecting relevant aspects governing the corrosion process.13,28,173,174
The main hydration product of CSA cement is ettringite, which does not provide OH–. The pH values of two pure CSA concretes with 0.5 w/c were 10.23 and 10.53, respectively, after 90 days. 15 In another case, w/c 0.45 CSA mortar showed low pH values (around 6) of the pore solution at 7 days after casting. 172 However, a high pH, around 13, within first 60 days has been observed by using a CSA cement paste with w/c 0.8. 175 An exhaustive investigation on two CSA cement pastes with w/c 0.72 and 0.8 revealed that, in the case of w/c 0.72 CSA cement paste, within the first hours, the pH was as low as 10.3–10.7 due to the fact that the initial saturated pore solution was dominated by aluminate, calcium, and sulfate; after 16 h, calcium and sulfate concentrations decreased noticeably due to the depletion of gypsum, thus the pH was around 11.8; after 28 days, the pH value reached 12.7 due to the ongoing release of alkali ions of CSA clinker and the increase of alkali concentrations caused by the consumption of the pore fluid by the formation of hydrates; while w/c 0.8 CSA cement paste showed a similar trend, but a slight higher pH at each stage. 19
Ettringite is susceptible to carbonation.13,14 It seems that the carbonation resistance of CSA concrete is weaker (Figure 4) than that of OPC concrete. 176 However, an investigation on two CSA concrete samples suggested that the carbonation resistance of CSA concrete is comparable with that of OPC concrete; high-strength CSA concrete has excellent carbonation resistance. 32 It was found that the carbonation resistance of CSA mortar increased along with the anhydrite content, as well as the decrease of w/c; meanwhile it was also found that carbonation changed the strength performance of CSA mortar due to the modification of porosity caused by carbonation. 27
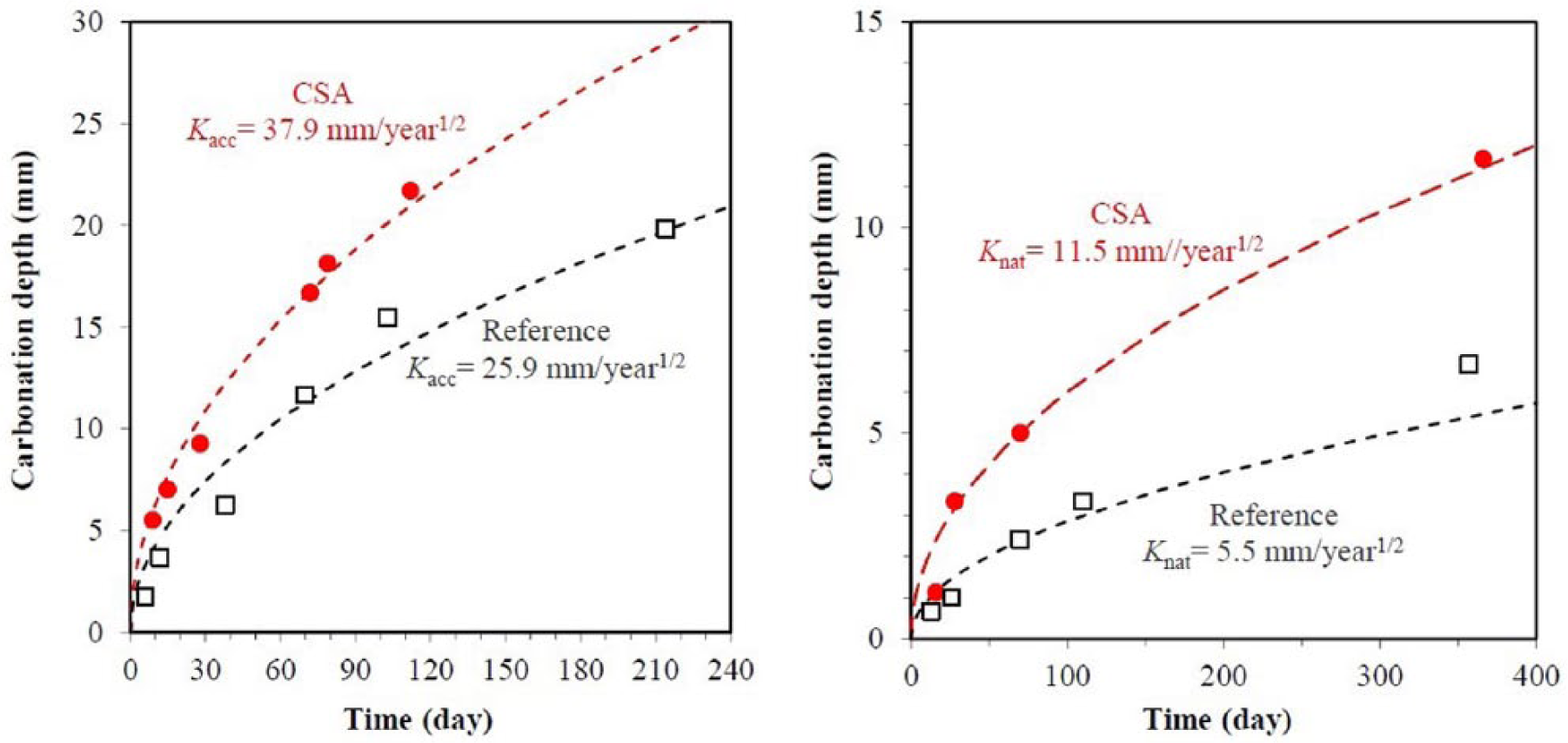
Time evolution of carbonation depth of CSA and reference OPC concrete under accelerated test (on the left) and indoor exposure (on the right). CSA: calcium sulfoaluminate cements; OPC: ordinary Portland cement.
Lower chloride penetration resistance of CSA concrete was observed when compared with OPC concrete. 13 Conversely, low chloride diffusion coefficients of CSA concretes with different strengths were obtained when compared with their OPC counterparts. 177 To enhance the chloride penetration resistance of CSA concrete, modifying AFm/AFt through varying the gypsum content with the hope to let more AFm bind chlorides, was carried out. 178 Besides, good sulfate resistance of CSA concrete has been reported.13–15 In the case of blended CSA cement, it was shown that increasing OPC in blended CSA cement (15%–85%) can improve the pH. 179
Currently, there are very few publications dealing with the passivation of steel embedded in CSA concrete. Steel in CSA mortar showed a higher corrosion rate than steel in OPC mortar exposed to 3.5% NaCl solution. 172 Half-cell potential measurement showed that steel embedded in the CSA mortar was depassivated, showing a high corrosion rate in 3.5% NaCl solution, due to the low pH (around 6) of the pore solution of CSA mortar. However, the corrosion potential and corrosion rate of steel embedded in w/c ratio 0.55 CSA concrete with a pH value of 11.5 showed the passivation of embedded steel, even in concrete exposed to an environment at 95% relative humidity and 40°C, or immersed in water. 176 Mortar made with 100% CSA cement (pH 11.88) was not capable of passivating steel; however, CSA cement blended with 15% OPC (pH 11.32) was enough to guarantee the passivation of steel. 15
The pH of alkali activated binders is very high at initial stages due to the presence of activators, leading to the common conclusion that no corrosion issues can occur. However, the pH tends to decrease under endogenous conditions to values well below the limits for steel passivation in absence of chlorides because these types of binders consume alkalinity during the hydration process. 180 In addition, very scattered pH values are reported, and several doubts have still to be solved in terms of corrosion behavior of reinforcement due to the very different mineralogical composition of precursors. 181 In addition, the role of alkalinity reservoir should be well taken into account for CSA and AAMs binders, which are generally prone to consume calcium hydroxide rather than produce it, as does Portland cement. 13 The protectiveness is attributable not only to the pH, but also to the ability of OPC concrete to bind its own chlorides, leading to a lower amount of free chlorides. The main factors influencing the critical chlorides content for pitting initiation are alkalinity and the concrete-reinforcement interface characteristics.182–185 The effect of alkalinity on localized corrosion initiation can be described in terms of chlorides-hydroxyl ions critical molar ratio, usually assumed equal to 0.6.168,184–193 The critical chloride threshold in OPC concretes is much higher due to oversaturation of calcium hydroxide. 194 This lead to an increase in critical molar ratio at values exceeding 2.183,195 This difference can be ascribed to the buffer ability by calcium hydroxide. The presence of this phase directly in contact with the carbon steel surface represents a reservoir of alkalinity, which contrasts the pH drop due to localized corrosion initiation.
On the contrary, much attention should be paid to innovative binders due to the great compositional variability of the raw materials, usually industrial by-products. Chloride contamination can be non-negligible, leading to an increased risk of localized corrosion, especially in the first period when alkalinity has not yet reached a sufficient level to maintain stable passivity.
Mobili studied also the corrosion behavior of carbon and galvanized bars embedded in pure FA and MK geopolymers with the same strength class compared to OPC mortars.75,196,197 During the curing period, geopolymers prolong the active state of rebars, but, after 10 days, corrosion rates (vcorr) decreased to moderate values (around 10 μm/year) in all mortars. 75 During wet/dry (w/d) cycles in 3.5% NaCl solution, 198 MK geopolymers showed the greatest corrosion of embedded rebars and the highest consumption of the galvanized coating because of the higher porosity compared to FA and OPC geopolymers.199,200 Aguirre-Guerrero studied the chloride-induced corrosion in OPC concrete coated with an alkali-activated mortar (90% MK (or FA) and 10% OPC); the MK geopolymer coating exhibited the best performances. 201
Accelerated carbonation (CO2 = 3 vol.%) on slag/MK geopolymers shows that carbonation occurs faster as MK content increases and leads to a reduction in compressive strength. 202 Moreover, accelerated carbonation at 50% CO2 on MK-based geopolymers forms large amounts of sodium bicarbonate, leading to a lower pH of the pore solution; while the formation of sodium carbonate in natural conditions does not lead to a pH below 10.5 after 1 year. 203
Reinforcement less sensitive to corrosion
Carbonation or chloride-induced corrosion are the main issues in reinforced concrete structure manufactured with different types of binders. In carbonated concrete without chlorides, stainless steel rebars are passive.204,205 For galvanized steel, the presence of an external layer of pure zinc and its thickness is of primary importance to form a passive film; in contact with alkaline solutions, if the pH does not exceed 13.3, a layer of calcium hydroxyzincate is formed and zinc is passivated. 206
In chloride-contaminated concrete, the onset of corrosion occurs if a chloride threshold is exceeded. Even though the measurement of this threshold is not easy, some major factors have been identified: the pH, the potential of the steel and voids at the steel/concrete interface; for carbon steel in aerated concrete at 20°C, critical chloride threshold is generally in the range 0.4–1% by cement mass. 206 In the case of stainless steels, chemical composition is also important: corrosion resistance is improved by increasing Cr and Mo content, while probably the role of Ni is beneficial in alkaline environments and Mn appears to have worsening effect: for example, critical chloride threshold at 20°C in aerated alkaline concrete for typical 18%Cr and 8% Ni AISI 304L steel (1.4307 according to EN) is a minimum 5% of cement mass for pickled surface, and lowers in the presence of a welding scale.206–211 Galvanized steels has a good resistance to chloride-induced corrosion, even if not comparable to stainless steels: in aerated concrete the critical chloride content is maximally 1–1.5%. 206
Few papers have been published about the performance of stainless or galvanized steels in new binders matrix.75,139,199,212,213 Moreover, results are not always consistent. Most researchers agree that the chloride concentration in alkali activated slag mortars is lower than in traditional mortars.139,199 This effect has been attributed to the lower porosity and the different chloride binding capacity: while in Portland cement mortars chlorides form low solubility calcium-containing compounds, in geopolymers, since calcium content is very low, the chloride binding effect is negligible.
The pH of the pore solution is a matter of discussion. Some authors stated, without indicating any practical measurement, that pH is highly alkaline, 212 or more alkaline than traditional mortars. 75 On the contrary, other authors report pH values, measured by the leaching method, for alkali-activated mortars similar to that of CEM II A-L 42.5 R-based mortars (between 12.8 and 13.2), but after exposure to 11 cycles of wetting with chloride solution and drying, the pH of alkali-activated mortars was found to be 10.5–10.7 against 12.2 for cement-based mortars. 139
Corrosion of low nickel (4.3%) manganese (7.2%) austenitic stainless steel with 16.5% Cr is compared with traditional stainless steel AISI 304 (1.4301 according to EN 10088-1) in alkali-activated FA mortars characterized by high alkalinity (the authors reported pH higher than 13, although few details are provided). 212 Both stainless steels exhibited passive behavior up to 2% chloride content, while carbon steel suffered corrosion in 0.4% chlorides. In another report, stainless steels (traditional type AISI 304 and low nickel) in carbonated mortars subjected to accelerated chloride exposure suffered localized corrosion. 213 Analysis of rebars after a 2-year exposure showed that in alkali activated slag mortars the behavior was better than in OPC mortars. The authors attributed the improvement to the higher concentration of inhibiting bicarbonate/carbonate ions present in these binders. 213 The results are promising but not conclusive: on the one hand, chloride content 2% in alkaline mortar is not high enough to evaluate the long-term performance of stainless steel rebars, 212 since the chloride content is well below than the critical chloride threshold in alkaline concrete. On the other hand, the results of the other group show a little improvement of corrosion behavior in alkali-activated mortars vs traditional ones. 213 Concerning galvanized rebars, it has been mentioned that geopolymeric mortars can have two opposite effects: a delay in the passivation due to the higher pH (potentially negative), and a reduction in corrosion rate after some cycles of wetting with 3.5% NaCl.75,199 Nevertheless, corrosion rate in alkali-activated slag mortars was found to be 50 µm/year. This value would lead to the consumption of the zinc layer (typically 150 µm) within a few years, so these results do not guarantee long-term performance of the galvanized rebar in geopolymeric mortars.
Alkali-activated materials in repair and conservation
The issue of retrofitting and seismic upgrade of existing masonry buildings and reinforced concrete structures has become of primary interest due to the huge architectural heritage all over the world. This topic is extremely complex, especially because of many compatibility issues between existing structures and Portland cement repair mortars.214–217,218 In fact, use of Portland cement mixtures on masonry structures can cause damage due to the presence of sodium and potassium ions, which can promote an alkali-aggregate reaction, 219 or, in the presence of wet environments and sulfur-rich natural stones, 220 it could determine the development of thaumasite and secondary ettringite, with expansion and cracking phenomena. Another key parameter for repair mortars is the elastic compatibility221,222: if Young’s modulus of repair material is different from the substrate, it may create detachments and cracks. Finally, it is not possible to overlook the aesthetic compatibility between the original areas and those involved in maintenance works. 223
Currently, natural hydraulic lime (NHL) represents the only binder that can be used in these contexts due to their high compatibility with the substrates.224–226 However, due to their low mechanical strength, NHL-based mortars often do not meet elasto-mechanical requirements, and, for this reason, are very often mixed with Portland cement.
The use of cement-free alkali-activated materials (AAM), such as GGBFS, could also be a suitable alternative to Portland cement mixtures.227–230
The key parameter that regulates most of properties of alkali-activated compounds is the precursor/activator ratio.231–236
A key aspect for use in maintenance is the possibility to tailor the strength and stiffness with the activator/precursor237–239; in particular, both the compressive strength and elastic modulus increased due to the high alkali-activator dosage in the mixture. Specifically (Figure 5), weakly alkali-activated GGBFS-based mortars can be used for plasters or masonry mortars while, in presence of high activator/precursor ratios, can be employed for seismic retrofitting or for reinforced concrete structures restoration.
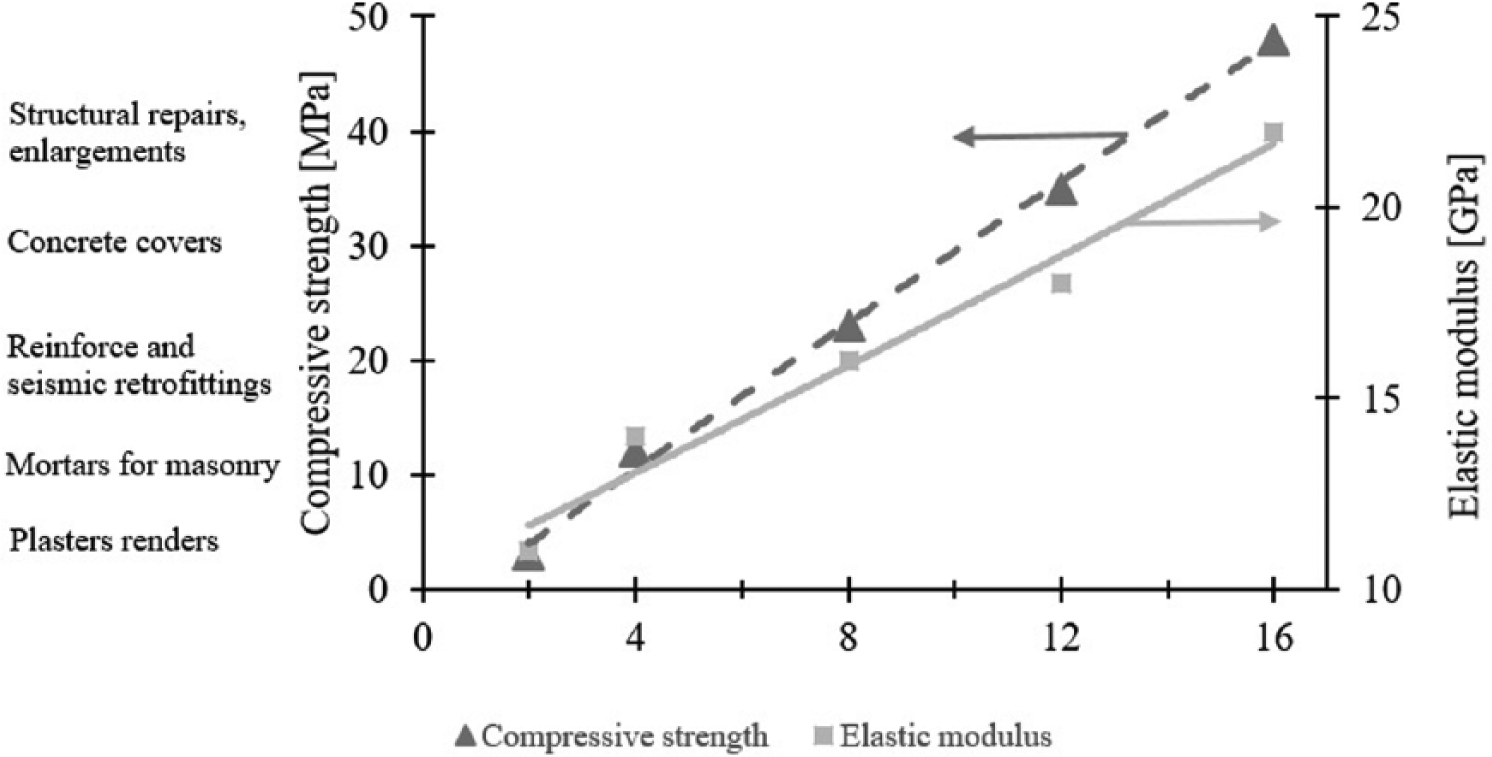
Compressive strength and elastic modulus of GGBFS-based mortars manufactured with blend of sodium metasilicate, potassium hydroxide, and sodium carbonate (7:3:1) at different activator/precursor ratios. GGBFS: Ground granulated blast furnace slag.
Another key parameter of alkali-activated mortars is elastic modulus (Figure 5); several authors showed less rigidity of GGBFS-based matrix respect to reference mixtures with OPC,36,55,240,241 at equal strength class due to the high shrinkage of AAM mortars that promote the formation of microcracks. 83 In particular, low activator/precursor ratios determine Young’s modulus ranging from 10 to 15 MPa, while higher alkaline powders dosages cause an increase in GGBFS-based matrix-stiffness, and, consequently, elastic modulus increases up to 20 MPa. In the presence of substrates restraining the dimensional contraction of repair mortar, this property determines the development of low internal tensile stresses, and thus, a lower cracking risk.
In general, alkali-activated mortars and concretes show very high free shrinkage compared to conglomerates manufactured with traditional binders. 242 These problems are caused by the large amount of water not involved in the hydration reaction, which, by evaporating, creates dimensional contraction and markedly porosity of matrix. Furthermore, alkali-activated slag pastes have a much higher range of pore sizes within mesopore region than OPC pastes. The radius of pores where the meniscus forms seems to be a key parameter for shrinkage.243–245
Researchers note that, by increasing the water/binder, there is a growth in shrinkage due to two factors: the large amount of water able to evaporate, and the increase in binder paste/aggregates. 246 In addition, it is possible to note that shrinkage is also influenced by type and contents of alkaline activators. 247 Reduction of shrinkage can be achieved by optimizing the mix with ethylene glycole SRA or calcium oxide expansive agents. In addition, methyl cellulose and starch ether (M.S.) can also be added in order to reduce water evaporation at the fresh state.41,248,249 In particular, the addition of blends based on ethylene glycol and calcium oxide can reduce free shrinkage by about 40% compared to reference GGBFS-mortars without admixtures (Figure 6).
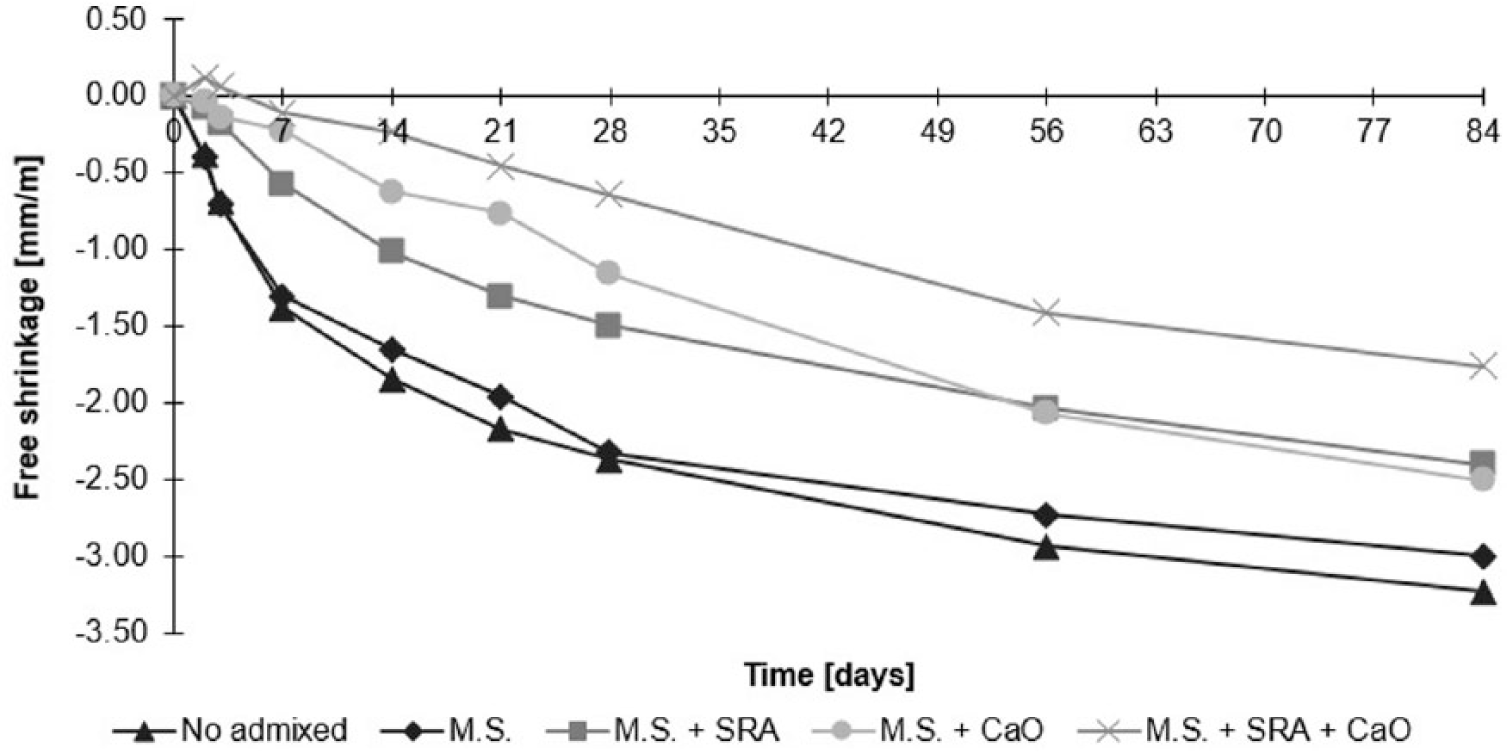
Free shrinkage of GGBFS-based mortars manufactured with blend of sodium metasilicate, potassium hydroxide, and sodium carbonate (7:3:1) at activator/precursor ratio equal to 0.12 with different type of admixtures. M.S.: methyl cellulose and starch ether; SRA: glycole ethylene-based SRA; CaO: calcium oxide-based expansive agent.
Another issue with AAMs is the efflorescence caused by excess of sodium oxide remaining unreacted in the material due to a disequilibrium in the mix towards the sodium-based activators. The parameter that influences the quantity of efflorescence is the Na/Al molar ratio; conglomerates with higher Na/Al molar ratios show a higher extent of alkali leaching, indicating a stronger tendency towards efflorescence.250,251
In conclusion, from analysis of the strengths and weaknesses of AAMs, it turns out that alkali-activated mortars and concretes can be a reasonable alternative to traditional Portland cement-based mixtures or NHL-based conglomerates for restoration of ancient buildings.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.