Abstract
Atherosclerosis is a complex disease characterized by the accumulation of plaques in arterial walls. Understanding its pathogenesis remains incomplete, with factors like inflammation, oxidative stress, and hypertension playing critical roles. The disease exhibits preferential localization of plaques, with variability observed even within the same individual. Genetic, environmental, and lifestyle factors contribute to its heterogeneity. Histological plaque phenotypes vary widely, prompting classification schemes focusing on systemic and local factors deteriorating fibrous caps. Recent research highlights differences in plaque histology among arterial systems, suggesting unique pathophysiological mechanisms. This study reports on multiple atherosclerotic plaques detected at autopsy in various vascular sites of a single subject, emphasizing their histological diversity and underscoring the systemic nature of atherosclerosis.
Introduction
Atherosclerosis, a complex and multifaceted disease, continues to be shrouded in a multitude of mysteries regarding its pathogenesis.1–3 In particular, our understanding of the pro-inflammatory and anti-inflammatory factors that play a critical role in shaping the atherosclerotic landscape within an individual remains incomplete. 4 Oxidative stress, hypertension, inflammation, and hyperlipidemia are recognized as factors that contribute to the buildup of low-density lipoprotein in the neo-intima of arteries. 5 These elements, along with other biological processes, significantly impact the development, severity, distribution, and advancement of atherosclerotic plaques.6,7
Atherosclerosis presents another layer of complexity with the preferential localization of plaques in specific arterial systems within the same individual. This selective vulnerability remains puzzling and suggests a range of genetic, environmental, and lifestyle factors, including fetal programing, which interact in ways that are not fully understood yet.8–11 Continued investigation into these complexities is crucial for developing better therapeutic and preventive strategies for atherosclerosis.
A diverse range of histological plaque phenotypes have been identified, leading to various classification schemes for atherosclerotic lesions. 12 These models consider both systemic and local factors that may contribute to fibrous cap deterioration, which predisposes plaques to instability and potential rupture, a key event in atherosclerosis progression. 13 Recent studies have focused on understanding the interplay between systemic and local factors in promoting fibrous cap weakening, shedding light on plaque stability and rupture mechanisms.14–16 Differences in plaque histology among different vascular systems suggest unique pathophysiological mechanisms at play, potentially influencing plaque structural characteristics.17,18 Studies have revealed variations in plaque histology based on their location, implying that local characteristics unique to specific areas of the vascular tree may impact the development of more stable or more vulnerable plaques. In the setting of carotid and coronary atherosclerosis, the following elementary lesions have been identified as possible differential markers between stable and unstable atherosclerotic plaques: plaque erosion, fibrous cap thickness, macrophage accumulation inside the fibrous cap, lipid-rich necrotic core dimensions. 19 Vulnerable carotid plaques are characterized by thicker fibrous cap, higher incidence of intra-plaque hemorrhage (IPH), lower incidence of plaque erosion, and higher incidence of calcified nodules compared to coronary plaques.20,21 Plaques in superficial femoral arteries exhibit more fibrotic elements, fewer degenerative features, smaller lipid-rich necrotic core, less severe inflammatory changes, and lower density of foam cells. 22
The aim of this study was to report the histological characteristics of atherosclerotic plaques detected at autopsy in different vascular sites of the same individual, focusing on plaque structure across various vascular territories.
Patient and methods
A 52-year-old man experienced sudden death due to aortic dissection complicated by hemopericardium. During autopsy, multiple atherosclerotic plaques were observed in the aorta, spanning from the thoracic to the abdominal tract. Several samples of aortic plaques were obtained, formalin-fixed, routinely processed, and paraffin-embedded. Additionally, multiple samples were collected from the coronary arteries, iliac arteries, femoral arteries, mesenteric arteries, and carotid arteries. Histological analysis was performed on five-micron-thick sections stained with hematoxylin and eosin.
Results
1. Aortic plaques. The histological study of aortic atherosclerotic lesions evidenced multiple different patterns, often observed even in adjacent plaques. a. Fibroatheroma with a thick fibrous cap, rich in fibroblasts, and a small basal necrotic core (Figure 1(a)); b. Fibroatheroma with a large lipid-rich necrotic core and a thin fibrous cap infiltrated by foam cells, lymphocytes, and monocytes (Figure 1(b)); c. Fibroatheroama with diffuse spot-like calcium deposits (Figure 1(c)); d. Fibroatheroma with IPH (Figure 1(d)); e. Fibroatheroma with a small lipid-rich core enveloped by inflammatory cells (Figure 1(e)); f. Fibroatheroma with predominance of fibrotic elements, with a necrotic core completely calcified (Figure 1(f)); g. Thin fibrous cap atheroma with a large necrotic core, intra-plaque hemorrhage, spot-like calcifications and plasma cells.
2. Carotids. a. Both carotids were characterized by the absence of protruding plaques upon macroscopy. Histologically, multiple crescent-shaped, eccentric pathological intimal thickenings (PITs) were observed in both the right and left carotid arteries, which were rich in fibroblasts
23
(Figure 2). This change is generally considered among the pre-stage lesions of atherosclerosis.
24
3. Subclavian arteries. a. In the right subclavian artery, a PIT was detected, characterized by the presence of foam cells, fibroblasts, and lipid pools. Additionally, plasma cells were also detected within the same lesion.
4. Coronary arteries a. In the descending tract of the left coronary artery, multiple PITs were observed (Figure 3(a)). At higher magnification, it was possible to evidence the migration of smooth muscle cells from the tunica muscularis into the intima, passing through holes in the elastic internal lamina (Figure 3(b)); b. In the left coronary artery, a PIT was found, characterized by a high presence of fibroblasts, along with periarterial adventitial inflammation and fibrosis (Figure 3(c)).
5. Iliac arteries. a. In the right iliac artery, we found a fibroatheroama with a lipid-rich thin necrotic core and a thick fibrous cap (Figure 4(a)); b. Adjacent to the fibroatheroma in the same iliac artery, two adaptive intimal thickenings (AITs) were found, often associated with “holes” in the internal elastic lamina (Figure 4(b)); c. In the left iliac artery, we observed a fibroatheroma with inflammation, a large lipid-rich necrotic core, and a thin fibrous cap with plaque rupture (Figure 4(c)); d. In the same artery, a PIT with lipid pools and abundant foam cells was detected (Figure 4(d)).
6. Femoral arteries a. In the left femoral artery, we identified an intermediate lesion displaying characteristics between a PIT and a fibroatheroma, exhibiting a high presence of fibroblasts. b. Adjacent to the PIT, an AIT was observed, characterized by abundant fibroblasts, in the absence of lipid pools.
7. Mesenteric arteries a. In the superior mesenteric artery, a fibroatheroma with a thick fibrous cap and a basal necrotic core surrounded by foam cells was identified (Figure 5(a)). At higher magnification, an IPH was detected within the necrotic core (Figure 5(b)); b. In the inferior mesenteric artery, we observed a fibroatheroma with a highly inflamed necrotic core (Figure 5(c)). Adjacent to the fibroatheroma, a highly inflamed PIT was also detected (Figure 5(d)).
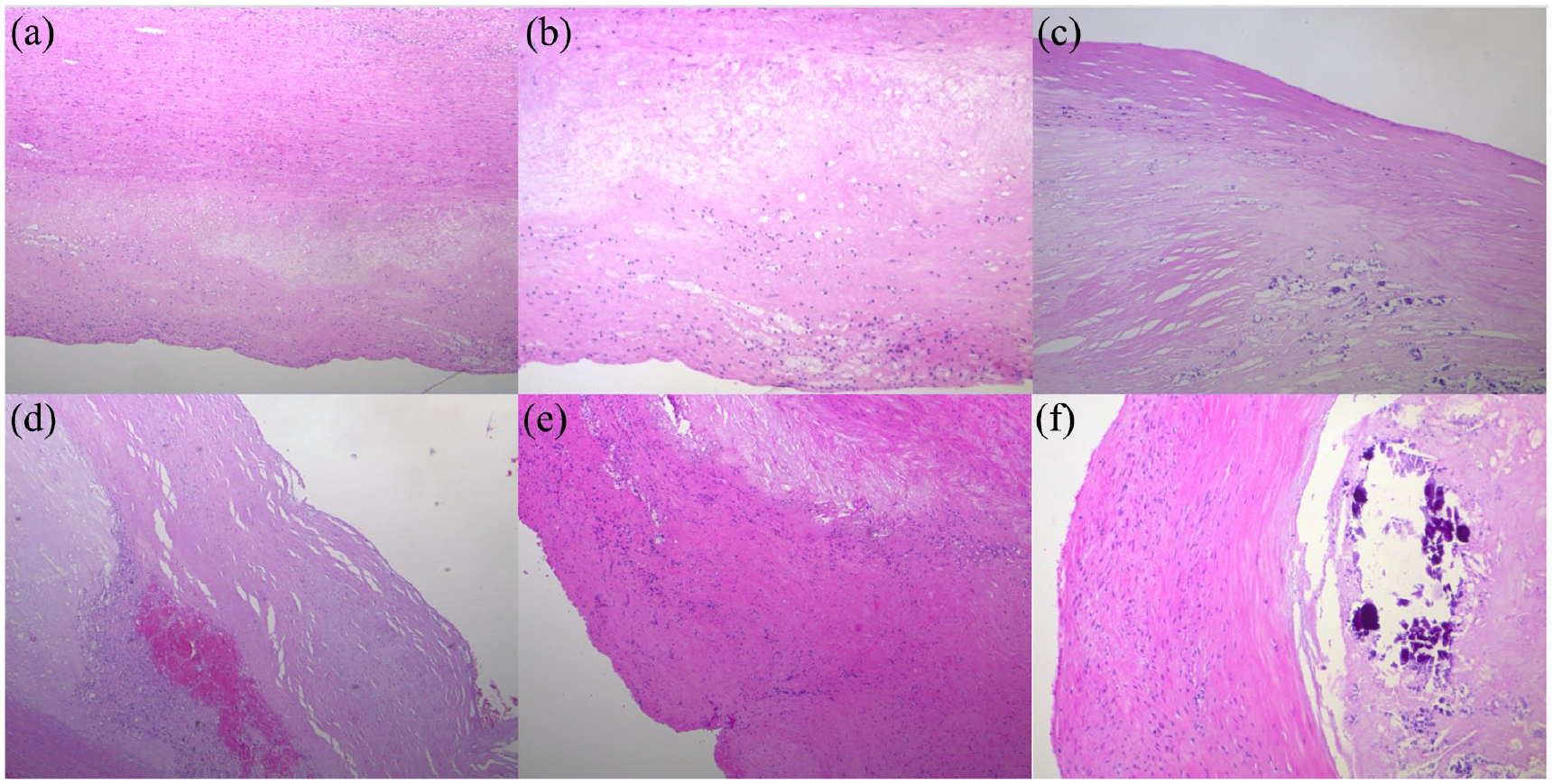
Aortic plaques. (a) Fibroatheroma with a thick fibrous cap, rich in fibroblasts, and a small basal necrotic core. Hematoxylin and Eosin (H&E). X50. (b) Fibroatheroma with a large necrotic core and an inflamed irregular thin fibrous cap. H&E. X100. (c) Fibroatheroma with a predominant fibrocellular component. On the right, micro- and macro-calcifications. H&E. X100. (d) Fibroatheroma with intra-plaque hemorrhage localized at the basis of the fibrous cap. H&E. X100. (e) Aortic fibroatheroma with a small lipid-rich necrotic core enveloped by inflammatory cells which extend into a thick fibrous cap. H&E. X50. (f) Fibroatheroma with a voluminous necrotic core with macrocalcifications. The thick fibrous cap shows abundant activated fibroblasts. H&E. X100.

Carotid. Pathological intimal thickening with signs of endothelial dysfunction. Note the duplications and focal interruptions of the internal elastic lamina. H&E. X200.

Coronary plaques. (a) Left coronary artery. Pathological intimal thickening with interruption of the internal elastic lamina and migration of smooth muscle cells into the intimal layer (arrow). H&E. X200. (b) Pathological intimal thickening of the left coronary artery. At higher power, smooth muscle cell migration from the muscular layer into the intima is clearly shown (arrows). H&E. X400. (c) Left coronary artery. Pathological intimal thickening with peri-arterial and peri-neural adventitial inflammation and fibrosis. The inflamed peri-arterial fat is characterized by a marked variability of the volume of adipocytes. H&E. X100.

Iliac-femoral plaques. (a) Fibroatheroma with a thick fibrous cap covering a thin lipid-rich necrotic core. H&E. X100. (b) Adaptive intimal thickening with a fragmented internal elastic lamina, showing “holes” allowing migration of smooth muscle cells. H&E. X200. (c) Iliac artery with a vulnerable plaque characterized by a thin fragmented fibrous cap and a large lipid-rich necrotic core. The plaque is undergoing rupture. Microcalcifications are present at the basis of the necrotic core. H&E. X50. (d) Iliac artery with a pathological intimal thickening characterized by a prominent fibrocellular component, scattered lipid pools and small aggregates of foam cells. H&E. X100.

Mesenteric plaques. (a) Mesenteric superior artery. Fibroatheroma with a very thick fibrous cap covering a large lipid-rich necrotic core, including multiple discrete intra-plaque hemorrhages. H&E. X100. (b) Superior mesenteric artery. Fibroatheroma. At higher power, intra-plaque hemorrhages are clearly shown within the necrotic core. H&E. X200. (c) Mesenteric artery. Unstable fibroatheroma with a large lipid-rich necrotic core and abundant foam cells. The fibrous cap is thick but highly inflamed. H&E. X100. (d) Inferior mesenteric artery. Pathological intimal thickening with a prevalent fibroblast component associated with lymphocytes. H&E. X100.
Table 1 summarizes the atherosclerotic plaque detected in histological specimens from different vascular systems.
Summarizes the composition of atherosclerotic plaque detected in histological specimens from different vascular systems.
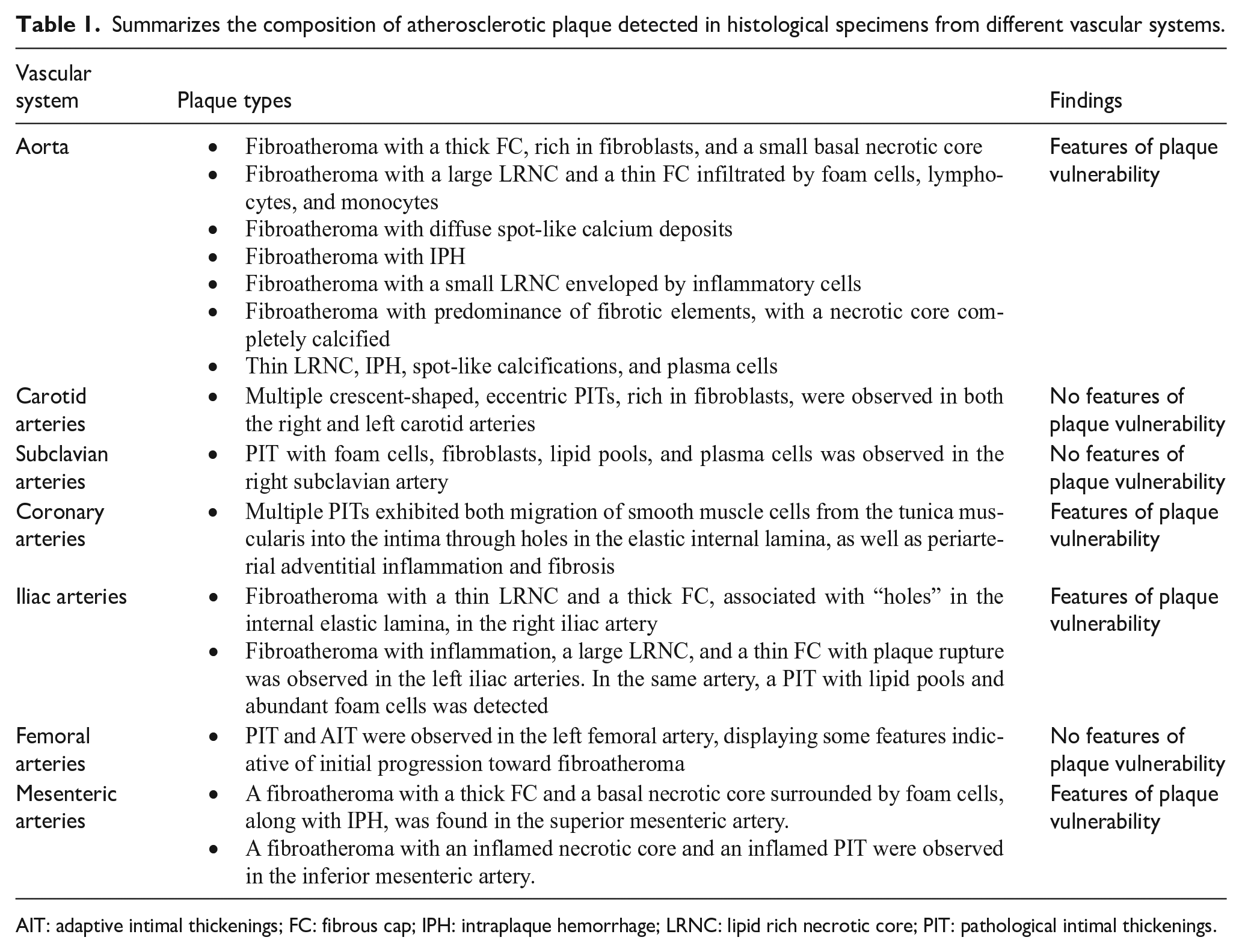
AIT: adaptive intimal thickenings; FC: fibrous cap; IPH: intraplaque hemorrhage; LRNC: lipid rich necrotic core; PIT: pathological intimal thickenings.
Discussion
The primary finding of our study is the significant variability in the histological structure of atherosclerotic plaques within the same individual. Previous research has associated this heterogeneity with the location of the plaques. 18 Specifically, coronary plaques have been suggested to differ from plaques found in peripheral arterial systems. 17 This discrepancy is partially attributed to the distinctive geometry of coronary arteries, which influences the architecture and cellular composition of coronary plaques compared to those in other vascular districts. 8 According to this hypothesis, plaques in peripheral arteries are expected to exhibit more fibroproliferative structure, characterized by a thicker fibrous cap, a smaller lipid-rich necrotic core, lower macrophage infiltration, fewer lymphocytes or plasma cells, a higher content of vascular smooth muscle cells, a lower density of vasa vasorum, infrequent intra-plaque hemorrhages, a high incidence of medial calcification, low levels of foam cells, and rare erosions. This differs from plaques observed in coronary arteries. 18
With the caveat that our observations are based on a single autopsy case, our findings reveal a significant variability in the histopathological lesions of atherosclerosis, which, to the best of our knowledge, has not been clearly demonstrated previously. We observed marked inter-plaque variability across all vascular districts analyzed. Specifically, within the multiple plaques detected in the aortic wall, we noted the coexistence of stable and vulnerable plaques in close proximity.
Among the six aortic plaques analyzed, two were characterized by a dominant fibrous component, featuring a thick fibrous cap and, in one case, a calcified necrotic core. These features led us to classify them as stable plaques. In contrast, the remaining four adjacent aortic plaques exhibited characteristics indicative of vulnerability. These included a thin fibrous cap, an extensive lipid-rich necrotic core, abundant foam cells, lymphoplasmacytic infiltrates, spot-like calcifications, and intra-plaque hemorrhage. 13 Taken together, these findings indicate that the latter atherosclerotic lesions were identified as plaques at medium to high risk of rupture. 25
Significant differences in the extent of atherosclerotic involvement and plaque burden were observed between the aortic and carotid/subclavian districts. In both the right and left carotid, despite the absence of macroscopic plaques, histological examination revealed the presence of crescent-shaped eccentric AIT, characterized by a thicker but un-diseased intima in the absence of extracellular lipid storage.23,25 In contrast, the subclavian artery exhibited a PIT, featuring extracellular lipid accumulation in the form of lipid pools. The presence of foam cells, monocytes, and plasma cells within the lesion indicated the progression of PIT toward fibroatheroma. 26
The histological examination of the coronary district revealed the presence of a fibroatheroma rich in vasa vasorum within the left coronary artery, accompanied by an erosion and initial thrombosis. Adjacent to fibroatheroma, a PIT was also found. Another PIT was detected in a branch of the left coronary artery along the posterior wall of the heart. While the overall plaque burden along the entire coronary tree was not high, the discovery of a fibroatheroma with erosion and signs of initial thrombosis in the left coronary artery led to the diagnosis of a destabilized plaque. This atherosclerotic lesion is associated with a high risk for major adverse cardiac events. 27
In this study, significant variability was also noted in the pathobiology of peripheral atherosclerotic disease. A stable fibroatheroma was identified in the right femoral artery, characterized by a thick fibrous cap and a modest lipid-rich necrotic core. In contrast, the histology of the fibroatheroma in the left iliac artery revealed a thin fibrous cap, inflammatory signs, and a plaque rupture.
In essence, within the same iliac vascular system, a stable and clinically inactive lesion was discovered in the immediate vicinity of a considerably more severe and destructive atherosclerotic lesion. Of low clinical relevance is the finding of a PIT, with signs of possible evolution toward a fibroatheroma, in a femoral artery. Significant variations in plaque development, progression, and destabilization were also observed in the mesenteric system. The superior mesenteric artery exhibited a fibrotic fibroatheroma with a thick fibrous cap, indicative of a stable plaque. In contrast, the inferior mesenteric artery displayed a highly inflamed, unstable fibroatheroma with a large lipid-rich necrotic core, suggesting increased vulnerability to rupture.
These findings collectively confirm previous studies that have highlighted the marked heterogeneity of atherosclerotic involvement across various arterial districts.1,17,18,22 In our case, the aorta was the vascular district predominantly affected by the atherosclerotic process, while the carotids showed only mild involvement. Furthermore, significant inter-plaque variability was observed in this study, even within the same vascular district. Stable, predominantly fibrotic lesions were found in close proximity to unstable, lipid-rich, and inflamed plaques. This finding indicates that the structure and histology of atherosclerotic plaques vary not only between different vascular territories but also within each territory and across every vascular district. 1 According to our findings, atherosclerosis emerges as a systemic disease, manifesting distinct characteristics in each plaque. This suggests that each plaque should be viewed as a unique lesion with potential clinical significance, differing from other atherosclerotic lesions even within the same vascular district. This observation underscores the significant influence of local factors, such as wall shear stress—the frictional force exerted by flowing blood on the vessel wall—on plaque composition and evolution. 28 Therefore, atherosclerosis is a dynamic multi-step process that affects different vascular systems differently. Indeed, our results confirm that there is not a vulnerable patient, but rather vulnerable plaques, highlighting the heterogeneity of atherosclerotic plaques. The advancement of knowledge in biological processes, coupled with the development of non-invasive technological modalities,29,30 could enable earlier diagnosis with tailored cardiovascular risk stratification and patient management.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
