Abstract
Carotid atherosclerosis-related stenosis, marked by atherosclerotic plaque formation in the carotid artery, significantly increases ischemic stroke risk. Its prevalence varies across ethnic groups, reflecting racial disparities. Epidemiological studies have highlighted different susceptibilities to carotid stenosis among racial groups. Native Americans and Whites show greater vulnerability, indicating genetic and environmental influences. The impact of carotid stenosis is more severe in Hispanic and Black populations, with a higher incidence of related brain injuries, underscoring the need for targeted interventions. Comparative imaging studies between Chinese and White individuals reveal unique patterns of carotid stenosis, enhancing understanding of its pathophysiology and management across ethnicities. This review also categorizes risk factors, distinguishing those with racial disparity (such as genetic loci, sleep apnea, and emotional factors, socioeconomic status) from those without. In summary, racial disparities affect carotid stenosis, leading to varying susceptibilities and outcomes among ethnic groups. Recognizing these differences is essential for developing effective prevention, diagnosis, and management strategies. Addressing these disparities is critical to reducing ischemic stroke’s burden across populations. Continued research and targeted interventions are crucial to improve outcomes for individuals at risk of carotid stenosis and its complications.
Introduction
Symptomatic carotid stenosis accounts for 8%–15% of ischemic strokes, while asymptomatic carotid stenosis affects 1%–2% of adults. 1 A comprehensive global mortality study reveals that the principal causes of death in 1990 remained predominant in 2010. However, during the intervening period, there has been a notable rise of 26%–35% in the incidence of ischemic heart disease and stroke. 2 Health disparity is defined as variations in health outcomes among different populations influenced by racial, ethnic, gender, educational, socioeconomic, and geographical factors. This review focuses on the epidemiology and risk factors of carotid stenosis, particularly examining racial disparities among Black, non-Hispanic, Hispanic, Native, and Asian Americans, and other nationalities. It will further explore how risk factors, both related and unrelated to racial disparities, influence carotid atherosclerosis.
Review
Methodology
PubMed/Medline, Science Direct, Google Scholar, and Embase were used to search original articles and review articles published in the English language with the following keywords “carotid artery stenosis,” “Carotid Artery Narrowing,” “plaque vulnerability,” “Unstable Plaque,” “plaque burden,” “Vulnerable plaque,” “Racial disparity,” “Ethical disparity,” “Western and Chinese,” “Chinese/Asian versus Western,” “Asian and Western,” “American and European,” “Multi-Ethnic Study of Atherosclerosis (MESA).” Keywords in the results obtained were further used to expand the search strategy and included “plaque characteristic,” “plaque feature,” “plaque characterization,” “Morphologic Features of Plaque,” “plaque composition,” “Plaque Components,” “plaque layout,” “plaque structure,” “plaque constitution,” “plaque construction,” “plaque morphology,” “Carotid Intima-Media Thickness,” “Diagnostic Techniques and Procedures” from January 2017 to October 2023.
Epidemiology and clinical presentation of carotid stenosis
Higher incidence of carotid stenosis in White and Native American populations compared to other racial groups
There are ambiguous findings across racial groups in the epidemiology of carotid stenosis. Some studies found no significant racial differences. In 2018, in the Women’s Health Initiative (1993–2010), African-American, White, and Hispanic women with or without baseline risk factors were compared to estimate heart failure mortality risk or all-cause mortality. Breathett et al. found no marked disparities among African-Americans, Whites, and Hispanics in carotid disease. 3 Similar trends were noted in Asian populations. Beyaz et al. reported comparable rates of moderate and severe carotid artery stenosis (CAS) between Syrian immigrants and Turkish natives in 2022. 4 These patterns may be attributed to common risk factors such as hypertension, hyperlipidemia, diabetes, stress, obesity, and genetic similarities (Table 1).
The summary of findings without racial disparity.

However, some scholars revealed that White and Native American populations have higher rates of carotid stenosis compared to other racial groups. In 2018, Fernandez-Jimenez et al. found that Non-Hispanic Blacks are more prone to early subclinical atherosclerosis than Hispanic/Latino blacks. 5 Research comparing Asian, European, and American populations found significant differences. Vishnu et al. reported that in the United States, White men have double the carotid plaque of Korean men and quadruple that of Japanese men. 6 Later, Rockman et al. noted that African-American, Asian, and Hispanic individuals have lower risks of carotid stenosis than Caucasians, with Native Americans exhibiting the highest risk, as cited by Shaw et al.7,8 In 2019, Steffen et al. suggested that race might modulate the risk of carotid plaque associated with Lp(a), with a more pronounced effect in Whites than Blacks. 9 In 2020, den Brok et al. found internal CAS more prevalent in White individuals and men, irrespective of stenosis severity. 10 Hicks et al. discovered that, compared to Whites, Blacks, and Hispanics, most Asian age groups show lower high-grade stenosis incidence, while Native Americans have higher frequencies. 11 In 2022, Renedo et al. indicated that Black and Hispanic participants had lower CAS rates than Whites (Table 2). Additionally, Black individuals’ lower likelihood of undergoing carotid revascularization was attributed to disparities in care access. 12
The summary of findings with racial disparity.

In conclusion, White and Native American populations exhibit higher rates of carotid stenosis than Hispanics, Blacks, and Asians, with Asians demonstrating the lowest prevalence among these races. Further research is necessary to explore the disparities between Hispanic/Black and Asian populations.
Comparative analysis of carotid atherosclerosis: Characteristics and location in Chinese and White populations
Cardiovascular disease (CVD) prediction using ultrasound carotid plaque features varies among races. Mitchell et al. 13 identified the carotid plaque area (TPA) which is a significant indicator of CVD and acute coronary heart disease (CHD) events in middle-aged patients without prior CVD history. While not as predictive as coronary artery calcium (CAC) scoring, TPA still effectively forecasts CHD, akin to the carotid plaque score. Compared to Whites, Chinese race/ethnicity showed a negative correlation with TPA, and Black participants had a lower grayscale median. 13 This suggests that both Chinese and Black populations exhibit less carotid atherosclerosis than Whites, aligning with previous discussions.
Furthermore, vascular imaging studies have revealed racial differences in plaque location and characteristics of carotid atherosclerosis. In 2021, Watase et al. found that Caucasian individuals more frequently exhibited plaques with luminal surface disruption and hallmarks of high-risk plaque (HRP) but not lipid-rich necrotic core (LRNC), intraplaque hemorrhage (IPH), or calcifications in the ipsilateral extracranial carotid artery. This study included US Caucasian and Chinese participants who had recently experienced an anterior circulation ischemic stroke. While Caucasian participants less commonly developed intracranial plaques with LRNC compared to Chinese individuals, there were no significant differences in other traits like IPH or HRP between the groups for intracranial incidence. 14 Consequently, the Caucasian population tends to have more extracranial plaques with HRP characteristics and fewer intracranial plaques with LRNC than the Chinese.
Imaging techniques can identify differences in carotid artery characteristics and plaque location. Ultrasound analysis of carotid plaque features has shown that Chinese and Black populations exhibit patterns distinct from those of the White population. Among individuals with recent anterior circulation ischemic strokes, the Chinese population is likelier to have intracranial plaques with an LRNC and fewer extracranial plaques with HRP characteristics than Whites (Table 3).
Characteristics and location of carotid atherosclerosis between Chinese and White people.

HRP: the hallmark of high-risk plaque; LRNC: lipid-rich necrotic core.
Increased susceptibility to carotid atherosclerosis-related brain injury in Hispanic and Black populations
Multiethnic research has focused on elucidating ethnic disparities in carotid-related brain injuries, encompassing stroke, white matter hyperintensities (WMHs), and cognitive function. It appears that Hispanic and Black populations are more closely associated with symptomatic carotid stenosis. In a 2018 cohort study, Breathett et al. evaluated the likelihood of developing heart failure or experiencing all-cause mortality, discovering that African women exhibited a higher incidence of stroke and myocardial infarction. 3 However, their methodology, reliant on the history of carotid disease and stroke, was limited in its accuracy, specifically in differentiating the symptomatic types of carotid stenosis and assessing stenosis severity.
Recent studies have focused on racial disparities in carotid stenosis-related WMH. In 2017, Rundek et al. found a stronger correlation between increased diastolic diameter (DD), reduced STRAIN ((systolic diameter − DD)/DD), and elevated WMH in Hispanics, a pattern not observed in Black or White individuals. 15 They evaluated carotid artery disease based on stiffness and diameter via ultrasound of the common carotid artery, not accounting for stenosis severity or plaque presence in individual carotid segments. Subsequently, the 2018 Northern Manhattan Study, involving participants without stroke, identified a notable link between larger carotid intima-media thickness (cIMT), increased WMH volume in Hispanics, and a heightened WMH burden in both Black and Hispanic groups. 16 This racial variation between cIMT and WMH could be attributed to ethnic differences in vascular risk factors, including genetic predispositions to cIMT and WMH.
The association between carotid stenosis and cognitive function varies among racial groups. Gardener et al.’s 2017 study on carotid atherosclerosis and Alzheimer’s disease revealed a correlation between cognitive performance and cIMT but not with cognitive decline. They found that age or race/ethnicity did not significantly influence the relationship between carotid plaque presence and cIMT. 17 In a 2021 randomized control trial, Lazar et al. observed that patients with severe carotid stenosis (⩾70% in CREST-2) exhibited reduced baseline cognitive function, a trend consistent across different racial groups, even after controlling for demographic and cardiovascular risk factors. 18
Carotid stenosis is a critical factor influencing WMH and cognitive function. Studies have shown that Black and Hispanic populations experience higher WMH burdens than White populations, with Hispanics particularly demonstrating a stronger correlation between cIMT and WMH volume. Furthermore, African women are reported to have a higher incidence of stroke and myocardial infarction, highlighting the need for more refined research methodologies. Additionally, while patients with severe carotid stenosis exhibit cognitive impairments compared to healthy controls, these impairments are consistent across racial/ethnic groups. Notably, cIMT has been found to correlate significantly with cognitive function.
The minority population has worse postoperative outcomes
Carotid endarterectomy (CEA) and carotid artery stenting (CAS) are recommended for patients with moderate to severe carotid atherosclerotic stenosis.19,20 The role of racial disparities in carotid revascularization postoperative outcomes remains unclear.
In 2018, the perioperative risks of stroke and death, as well as unadjusted survival rates, were similar between White and Black patients despite a higher incidence of vascular complications in Black individuals and racial disparities in surgical treatment. Additionally, Black patients who underwent CEA demonstrated better-adjusted long-term survival. 21
Hospital selection and racial disparities may influence the frequency of CEA, potentially leading to less favorable outcomes for minority individuals. In 2019, a study using the ARIC trial cohort revealed that, even after adjusting for risk, Black participants consistently underwent fewer CEAs than White participants. 22 Additionally, the frequency of CEA after ischemic stroke varied depending on the hospital, with significant differences noted between minority-serving and White-serving hospitals. Faigle et al. found that hospital preference contributed to racial disparities in carotid revascularization, with lower CEA rates in minority-serving hospitals compared to those primarily serving White populations. 23 These findings suggest that the differences in CEA outcomes for minority patients may be partly due to both lower incidence of the procedure and variations in hospital practices.
In the context of emergent carotid endarterectomy (eCEA), non-White patients are more frequently assigned to the eCEA group, potentially resulting in adverse outcomes. According to Faateh et al., in 2019, non-White individuals were predominantly placed in the eCEA group, while White patients more often received non-eCEA treatment. Patients presenting with stroke or amaurosis fugax showed similar outcomes in both eCEA and non-eCEA groups. However, for those presenting with a transient ischemic attack (TIA), the adjusted analysis revealed that eCEA significantly increased the risk of stroke and stroke/death, leading to worse outcomes compared to non-eCEA treatment. 24 Consequently, minority patients undergoing emergent CEA for TIA have an elevated risk of perioperative stroke and death.
Postoperative outcomes of CAS exhibit racial variations. A 2017 study from the American College of Surgeons National Surgical Quality Improvement Program, led by Bennett et al., demonstrated a significant correlation between race and the incidence of serious adverse events post-CAS in a nonclinical trial setting. Notably, black patients exhibited a higher association with major adverse cardiovascular events (MACEs) following CAS, as identified in multivariable analysis. 25 Furthermore, Aronow et al. reported an increased likelihood of stroke and mortality due to neurological conditions in black patients within 30 days post-CAS. 26
In summary, hospital choice and racial discrimination combine to result in worse stroke/death rate in minorities considering carotid revascularization. The procedure of hospitals serving minority populations may contribute to poorer outcomes in minority population with CEA. Specifically, non-White patients are more prone to the eCEA group, leading to adverse outcomes. In the context of postoperative outcomes following CAS, the black race is significantly associated with the development of MACEs, leading to substantial adverse outcomes (Figure 1).

The flowchart shows the progress of the Black experience during the period of carotid revascularization.
Risk factors with racial disparity
Candidate gene loci
Research encompassing diverse racial groups, including Mexican Americans, European Americans, Chinese, East Asians, and multiethnic populations, has been instrumental in identifying gene loci pertinent to subclinical atherosclerosis and CVD risk assessment. Notably, a study focusing on Mexican Americans and European Americans with rheumatoid arthritis (RA) has shed light on potential new risk loci for subclinical atherosclerosis. Arya et al.’s work revealed novel associations between cIMT and genetic variants in SLC17A2 and PPCDC and between arterial plaque and variants in COL4A1 and SLC4A13 in these populations. 27 These findings offer valuable insights into candidate gene loci for RA-associated subclinical atherosclerosis.
Investigations into the potential correlations between carotid plaque, CVDs, and genetic predispositions in Chinese and other East Asian populations have yielded significant findings. Liu et al. focused on the Chinese Han population, known for its heightened susceptibility to type 2 diabetes mellitus (T2DM). Their research centered on the role of oxidative stress-related genes, including ALOX5, ALOX5AP, GPX1, GPX3, and MPO, in the predisposition to T2DM within this demographic. Notably, they uncovered new insights regarding the MPO gene’s influence on the likelihood of developing T2DM. Moreover, their study bolstered the theory that variants in oxidative stress-related genes (GPX1 and MPO) are linked to carotid plaques in T2DM patients. 28 This evidence supports the notion that genetic factors may contribute to the vulnerability to T2DM-related cardiovascular complications. Concurrently, Song et al. pursued an investigation to evaluate the interaction between the ABCA1 gene variant rs2066715 and egg consumption, examining their impact on ischemic stroke, carotid plaque, and cIMT in the Chinese population. Their primary goal was to ascertain any significant correlations between egg consumption and these cardiovascular risk factors. Their findings revealed that the genetic variant rs2066715 interacts with egg intake in the Chinese population, influencing the prevalence of carotid plaque. 29
Tam et al. developed genomewide polygenic risk scores tailored explicitly to East Asian populations, highlighting the potential for early genomic screening and personalized risk assessment in CVD. This innovation reflects a focused concern for the unique genetic characteristics of East Asian individuals. 30 Moreover, the study on the enzyme catechol-O-methyltransferase (COMT) and its three genetic variants has revealed predictive capabilities for CVD or plaque load in a broad and diverse population cohort. Specifically, the rs4818G allele of COMT, crucial in estrogen and catecholamine metabolism, is associated with a decreased risk of CVD and lower fibrinogen levels. However, it does not correlate with intima-media thickness (IMT) or CAC. This evidence suggests COMT’s role in the advanced stages of CVD. 31 Additionally, Dueker et al.’s research identified genetic variants associated with total carotid plaque area (TCPA) that traditional vascular risk factors do not explain. They pinpointed three genes within the GO:0050913 gene set—TAS2R20, TAS2R50, and ITPR3—that are significantly linked with high TCPA. 32
The studies identified novel associations between specific gene loci and cIMT or plaque formation in Mexican Americans and European Americans with RA. Additionally, in the Chinese Han population, genetic predispositions to type 2 diabetes were correlated with an increased likelihood of carotid plaques. Moreover, specific gene variants were found to modulate the relationship between egg consumption and the development of carotid plaque. Furthermore, the COMT enzyme and three genetic variants predicted CVD and were significantly associated with the TCPA across different racial groups.
Sleep apnea
Advanced atherosclerosis may significantly contribute to the health risks associated with sleep apnea. In the Multi-Ethnic Study of Atherosclerosis (MESA) study, a marked correlation was observed between sleep apnea and carotid plaque in individuals below 68 years. When evaluating race-specific outcomes, the association between carotid plaque and sleep apnea was pronounced in white participants compared to other racial or ethnic groups, and notably, increased hypoxemia correlated with augmented cIMT in black individuals. Meanwhile, white participants exhibited a robust association between carotid plaque and sleep apnea. This carotid plaque formation might stem from the detrimental impacts of repeated respiratory disruptions and potential sympathetic surges associated with arousal events. 33
Conversely, increased cIMT may result from hypoxemia-induced stressors, including inflammation, endothelial dysfunction, hypertension, and dyslipidemia. 33 Additionally, arousal-related sympathetic surges might contribute to carotid plaque formation. In a related context, Shah et al. explored the association between obstructive sleep apnea (OSA) and carotid plaque inflammation using 18-F-fluorodeoxyglucose (FDG) positron emission tomography and magnetic resonance imaging (PET/MRI) in the MESA. Their research indicated moderate to severe OSA correlates with increased carotid-wall thickness and elevated plaque inflammation. Significantly, the relationship between OSA and inflammation is moderated by obesity, with more significant carotid plaque inflammation observed in nonobese individuals. Notably, obesity is a known risk factor for OSA development. 34 Furthermore, Deol et al. examined the link between heightened OSA risk and subclinical atherosclerosis in South Asians residing in the United States. Their findings revealed that a high OSA risk, encompassing comorbidities like hypertension and obesity, was not associated with the duration of residence in the United States. 35 Additionally, markers of subclinical atherosclerosis, such as CAC and cIMT, were related to the length of stay in the country.
Furthermore, short sleep duration (SD) is linked to an elevated risk of CVD. Kundel et al. investigated this relationship between SD and subclinical atherosclerosis, employing hybrid PET/MRI and FDG as the tracer in the MESA cohort. Their findings indicate that individuals sleeping fewer than 6 h per night are more likely to exhibit pathologic vascular inflammation. Specifically, those with an average sleep duration of less than 6 h showed increased vascular inflammation levels. 36 A positive correlation was also observed between carotid wall thickness and the sleep fragmentation index. This index reflects the degree of restlessness experienced during sleep and is expressed as a percentage. 37
In summary, sleep apnea and sleep deprivation suggested an increased risk of atherosclerosis in the carotid arteries. In white people, sleep apnea was related to an increased presence of carotid plaque, but in black people, it was associated with an increased cIMT. Furthermore, sleep deprivation was associated with an increased incidence of pathologic vascular inflammation.
Emotional factors
Various emotional stressors, including traumatic experiences such as sexual assault and the loss of loved ones, significantly influence atherosclerosis in the carotid artery. Research has shown that African-American women are more likely to experience severe carotid atherosclerosis, potentially linked to distressing losses during midlife. In a comparative study of women from diverse racial backgrounds, it was observed that African-American women were more frequently affected by the loss of friends or family members during middle age. Moreover, these losses had a more substantial impact on the progression of carotid atherosclerosis in African-American women than in women from other racial groups. 38 Furthermore, in 2021, African-American women exhibited a higher incidence of CVD compared to women of different races or ethnicities. The relationship between distressing losses and cIMT was found to be more pronounced in women of African-American, Chinese, and Hispanic descent, suggesting that such midlife losses may be a significant factor through which race and ethnicity influence the risk of CVD. 39
Partner-related emotional stress, notably from sexual assault, has been implicated in increasing the risk of atherosclerosis in carotid arteries. A landmark 2021 study involving nonsmoking, late perimenopausal, and postmenopausal women, previously participants in the MsHeart project (2012–2015), focused on menopausal symptoms and cardiovascular health. This research uniquely identified a correlation between a history of sexual assault and elevated levels of carotid atherosclerosis in midlife women. Traditional cardiovascular risk factors were inadequate to fully account for this association. 40 This study represents the first of its kind to establish a link between a history of sexual assault and the progression of carotid plaque over time, in a longitudinal context involving middle-aged women.
Emotional factors, particularly distressing midlife losses, and sexual assault, are significant contributors to the development of carotid atherosclerosis. This association is notably more pronounced in African-American women compared to other racial groups. Future research should prioritize including emotional factors as critical risk elements influencing carotid atherosclerosis.
Socioeconomic status
Low socioeconomic status (SES) and elevated racial discrimination have been linked to increased carotid atherosclerosis, subsequently leading to poorer clinical outcomes. A retrospective study conducted in 2018 involving 3,696,778 white and black participants revealed that lower-income socioeconomic environments are correlated with a higher incidence of peripheral arterial disease (PAD) and CAS. This correlation may be an independent predictor, surpassing traditional cardiovascular risk factors. 41 Miller et al., 42 discovered that residing in socioeconomically disadvantaged neighborhoods escalates the risk of atherosclerosis-related CVD. This heightened risk is associated with preclinical manifestations of atherosclerosis in the carotid artery. 42 Castro et al., 43 observed that Hispanic patients hospitalized for PAD often have a more vulnerable socioeconomic profile compared to non-Hispanic white patients. These patients typically present with more advanced stages of PAD and a more intricate risk profile. Consequently, the development of preventative and treatment strategies tailored to the unique risk profiles of minority populations is imperative to mitigate the pronounced disparities in PAD risk, which vary according to ethnic background. 43 Furthermore, among African Americans, those reporting high levels of racism and depressive symptoms exhibit increased carotid atherosclerosis and may face a higher risk for clinical endpoints compared to those reporting fewer or none of these risk factors. This increased risk is particularly pronounced in individuals experiencing both high levels of prejudice and depressive symptoms. 44
Moreover, Wendell et al. discovered that African Americans with high-SES exhibited significantly thicker IMTs and accelerated pulse wave velocities compared to other subgroups, including low-SES African Americans, low-SES whites, and high-SES whites. This unexpected finding could be attributed to disparities in healthcare access and intensified discrimination experienced by African Americans upon attaining higher SES. 45
Consequently, living in a low socioeconomic environment combined with racial discrimination can lead to severe physical conditions and depressive symptoms. These factors may culminate in more advanced carotid and peripheral arterial atherosclerosis stages.
Risk factors without racial disparity
Calcium artery calcium
The CAC score is now a fundamental component of preventive cardiology. Its role as a robust tool for assessing cardiovascular risk, especially when combined with established metrics like the Framingham risk score, has been thoroughly researched and validated. 46 In 2008, Detrano et al. demonstrated that the CAC score is a dependable predictor of acute coronary artery disease, offering additional prognostic value across four major racial and ethnic groups in the United States. The study also noted the absence of significant disparities in the predictive accuracy of calcium scores among different ethnic backgrounds. 47
CAC serves as both a risk factor and a biomarker, providing an early, asymptomatic indication of various CVDs, including carotid artery diseases, atherosclerotic cardiovascular disease, CHD, and CVD, with its relevance consistent across different ethnic groups. Moreover, combining CAC and cIMT enhances CVD risk assessment in patients with T2DM. The significance of a CAC score of zero (CAC = 0) in multiethnic studies is well established, reliably excluding the likelihood of severe cardiovascular events or CHD. These insights are instrumental in identifying individuals who may not substantially benefit from lifelong preventive pharmacotherapy. Additionally, a CAC score of zero is crucial for re-evaluating cardiovascular risk and guiding drug therapy decisions.48–51
In summary, a zero CAC (CAC = 0) score in multiethnic studies has been utilized to rule out the risk of severe CHD, albeit not ischemic stroke. Conversely, the role of a CAC = 0 score in assessing overall cardiovascular risk remains a subject of ongoing debate. Furthermore, this score can be pivotal in reclassifying cardiovascular risk and is an essential criterion in guiding decisions regarding drug therapy.
Carotid intima-media thickness
cIMT is a pivotal surrogate marker in identifying subclinical atherosclerosis and is instrumental in risk assessment and monitoring the progression of atherosclerosis in therapeutic contexts. Its role extends beyond merely indicating the presence of subclinical atherosclerosis; it also offers critical insights into the passage of the disease, thereby guiding therapeutic interventions.52–54
Current research indicates no significant racial disparities in utilizing cIMT for predicting subclinical atherosclerosis. Furthermore, integrating cIMT with CAC scoring could enhance risk evaluation for ischemic stroke or TIA. Independent of traditional cardiovascular risk factors, Tschiderer and colleagues established a roughly linear relationship between cIMT and the probability of developing an initial carotid plaque. A study involving 95 participants revealed no substantial variation in cIMT measurements. 55 Additionally, Osawa and the team reported that the combination of cIMT and CAC scoring might improve risk assessments for ischemic stroke and TIA prevention, potentially guiding the targeted use of statin therapy to reduce risk in the most vulnerable individuals, irrespective of racial background. 56
cIMT is broadly recognized as a marker for predicting subclinical atherosclerosis. However, according to some studies, its efficacy in forecasting CVD appears limited. The predictive utility of cIMT shows no significant racial variation in detecting subclinical atherosclerosis. Combined with CAC scoring, this approach amplifies its predictive capacity, particularly in preventing ischemic strokes and TIAs.
Lipid protein
Lipoproteins, often called apolipoproteins (apo), comprise triglycerides, phospholipids, and both esterified and unesterified cholesterol. Elevated levels of blood lipids are substantially linked to the development of atherosclerosis across all racial and ethnic groups. This relationship encompasses increased concentrations of low-density lipoprotein (LDL), lipoprotein(a) (Lp(a)), apolipoprotein(a) (apo(a)), non-high-density lipoprotein cholesterol (non-HDL-C), and high-density lipoprotein cholesterol (HDL-C).57,58
Lp(a), allele-specific apo(a), non-HDL-C, and HDL-C are crucially implicated in the development of carotid atherosclerosis. 59 Enkhmaa et al., 59 reported that elevated levels of Lp(a) and allele-specific apo(a) in human immunodeficiency virus (HIV)-infected young Black women suggest these markers may predict carotid artery IMT in this demographic. Additionally, Ma et al. identified a unique association between non-HDL-C and carotid atherosclerosis in Chinese women with normal blood pressure and glucose levels. 60 Generoso et al., in their 2018 study in Brazil, demonstrated that HDL2-C, HDL3-C, and the HDL2/HDL3-C ratio inversely correlated with carotid IMT, a finding consistent across various ethnicities, including White, Brown, Black, Asian, and Indigenous populations. 61 Hou et al. highlighted that total cholesterol, LDL-C, and non-HDL-C have superior predictive capacities for IMT and carotid plaque presence compared to other lipid parameters in the Chinese population, with triglyceride content showing the highest predictive value for carotid plaque presence. However, the predictive ability of non-HDL-C for carotid IMT and plaque was not found to be inferior to that of LDL-C. 62 Regarding HDL and HDL-mediated cholesterol efflux, studies within the MESA found no association with subclinical atherosclerosis.63,64 Furthermore, no correlation was observed between abnormal cholesterol distribution (CD) and Lp(a), although traditional cardiovascular risk factors were linked with abnormal CD. 65
In multiracial studies, various lipoproteins, except HDL, have been significantly associated with the progression of carotid atherosclerosis. However, HDL-C demonstrates an inverse relationship with carotid atherosclerosis. The nature of specific associations between lipoproteins and atherosclerosis warrants further exploration. In the MESA, neither HDL subtypes nor HDL-mediated cholesterol efflux were found to correlate with subclinical atherosclerosis. The only observed positive correlation was between Lp(a) levels and CAC scores in asymptomatic Caucasian individuals.
Insulin resistance
Glucose is a critical component of the human body’s energy supply, making maintaining glucose homeostasis vital for health. Disruptions in glucose homeostasis are the primary cause of diabetes mellitus type 2. 66 Insulin resistance is a complex condition where the body’s response to insulin is impaired, disrupting normal glucose homeostasis, chiefly regulated by insulin and glucagon. 67 Furthermore, diabetes mellitus is linked with an elevated risk of CVDs, and the prediction of cardiovascular events is increasingly focused on factors like insulin sensitivity and glucose homeostasis. 68
Clinical indicators of carotid atherosclerosis encompass insulin resistance, as determined by the Homeostatic Model Assessment for Insulin Resistance (HOMA-IR), along with elevated insulin levels. The HOMA-IR method, formulated by Matthews et al., assesses insulin resistance and beta-cell function. 69 A 2017 study by Santos et al. in Brazil, involving a large cohort of nondiabetic individuals, identified a direct relationship between HOMA-IR scores and cIMT. This correlation was consistent across all racial groups, including white, brown, and black participants. 70 However, research by Fowokan et al. indicated that ethnic variations can alter the predictive relationship between insulin and glucose levels and subclinical signs of carotid atherosclerosis, 71 highlighting that this relationship does not uniformly extend to insulin resistance and glucose homeostasis.
Diabetes is believed to contribute to accelerated atherosclerosis, and various glycemic markers have been associated with the preclinical stages of CVD. 72 In a 2018 study, Wilcox et al. demonstrated that diabetes, particularly when combined with other risk factors, increased the risk of peripheral artery disease (PAD) and CAS in a multiracial cohort. 73 Subsequent research by Echouffo-Tcheugui et al. revealed that in African-American populations, glycemic indicators such as hemoglobin A1C, fasting plasma glucose, and insulin resistance showed variable associations with different aspects of subclinical CVD. It is advisable for individuals with diabetes and abnormal glycemic markers to consider screening for PAD and to take preventive measures. 74 Furthermore, the prevalence of multisite atherosclerosis is notable in groups with risk factors linked to cardiovascular and cardiac events.
In conclusion, ethnicity does not alter the predictive correlation between insulin resistance and subclinical carotid stenosis. Nonetheless, specific glycemic indicators demonstrate varying associations with multiple aspects of subclinical CVD, particularly in individuals of Black descent. Furthermore, the presence of multisite atherosclerosis is indicative of cardiovascular heart disease across all ethnic groups.
Obesity
Research indicates a normative association between obesity, ethnic differences, and cIMT, though this association does not extend to atherosclerotic carotid disease. Studies have demonstrated that African-American adolescents exhibit a higher cIMT compared to their White counterparts. 75 In a Chinese cohort, a significant correlation was observed between lean body mass and cIMT, rather than plaque load, in healthy individuals. This finding suggests a normative relationship rather than an indicator of atherosclerotic disease. 76
Menopausal
A multitude of studies have explored various aspects of the relationship between menopausal status and carotid atherosclerosis. Investigations have focused on determining the potential involvement of genetic mutations and high-density lipoprotein (HDL) in mediating this association. On the one hand, Chu et al. found a significant intensification in the association between two scavenger receptor single nucleotide polymorphisms (SNPs) (CD36 rs1761667 and MARCO rs12998782) and carotid atherosclerosis postmenopause in a Chinese Han population. 77 Conversely, research suggests that the cardioprotective effect of large high-density lipoprotein particles may diminish near menopause. This underscores the importance of investigating how the menopausal transition impacts HDL quality and subsequent CVD risk later in life. 78 Nevertheless, Barinas-Mitchell et al. demonstrated that race and ethnicity do not markedly influence the relationship between traditional CVD risk factors and late midlife subclinical vascular disease during the menopausal transition. 79
Consequently, there is a pressing need for research exploring the association between SNPs and carotid atherosclerosis in postmenopausal women across various racial groups. Furthermore, the shift from reproductive to postmenopausal stages may influence HDL quality and CVD risk in later life. It appears that race or ethnicity does not significantly mediate the relationship between traditional CVD risk factors and changes during the menopausal transition.
Human immunodeficiency virus
Individuals infected with the HIV might face an increased risk of CVD, even with the widespread use of antiretroviral therapy (ART). 80 Research is being conducted to elucidate the connection between HIV and atherosclerosis across diverse ethnic populations, including Black, Asian, and multiethnic groups.
African Americans and Asians with HIV infection are found to be particularly vulnerable to atherosclerosis, influenced by distinct risk factors. 59 Furthermore, the interplay of age and ethnicity or race in this context has been clarified through studies of various racial groups. The impact of HIV on carotid artery structure appears to be age-dependent, with traditional CVD risk factors assuming greater significance in older adults compared to younger individuals (aged 6–29 years) and children. This was demonstrated by findings from five cohorts in the National Heart, Lung, and Blood Institute HIV-CVD Collaborative. 81 Msoka et al. conducted a meta-analysis of 20 studies (5 cohorts, 15 cross-sectional, and 2 combining cohort and cross-sectional data), encompassing 7948 participants, to assess the effect of age on HIV-positive individuals (4656 HIV-infected; 3292 controls). The research indicated that both HIV infection and ART influence cIMT, potentially contributing to cardiovascular events. However, it was inconclusive in determining the effects of gender and ethnicity/race disparities on cIMT within HIV-infected populations. 82 Thus, while age is a significant factor in the influence of HIV infection on atherosclerosis, the specific roles of ethnicity and race in HIV-infected individuals regarding carotid atherosclerosis have yet to be established.
In summary, age appears to be a significant factor in the risk of atherosclerosis among HIV-infected populations. However, current evidence does not indicate any racial or ethnic disparities in the prevalence of carotid atherosclerosis in HIV-positive individuals. Additionally, the presence of circulating nonclassical monocytes may influence the clinical outcomes in those with HIV infection (Table 4).
The risk factors with or without racial disparity.
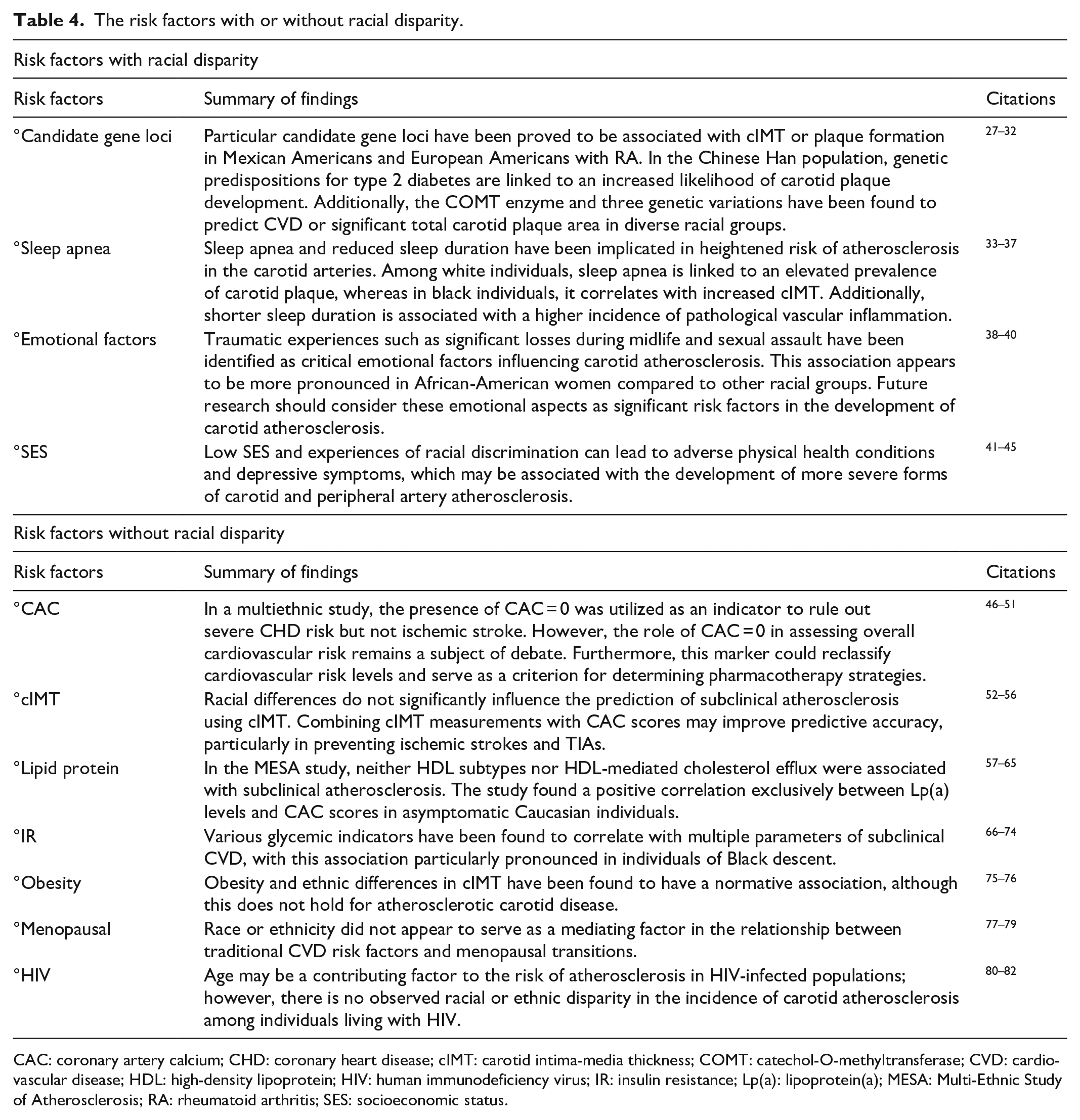
CAC: coronary artery calcium; CHD: coronary heart disease; cIMT: carotid intima-media thickness; COMT: catechol-O-methyltransferase; CVD: cardiovascular disease; HDL: high-density lipoprotein; HIV: human immunodeficiency virus; IR: insulin resistance; Lp(a): lipoprotein(a); MESA: Multi-Ethnic Study of Atherosclerosis; RA: rheumatoid arthritis; SES: socioeconomic status.
Limitations
This review consolidates data from various studies with varying designs, populations, and analytical approaches, including epidemiology and racial disparities of carotid atherosclerosis. Consequently, additional large prospective observational cohorts are necessary to offer a rigorous evaluation of the crucial issues surrounding racial disparities. Additionally, the mechanisms by which risk factors impact carotid atherosclerosis across different racial groups remain uncertain. A significant limitation of this review is its inability to fully address the intricate nature of racial disparities in carotid stenosis and the related risk factors.
Discussion
Based on findings from the MESA study, it is evident that whites and Native Americans exhibit the highest rates of carotid stenosis among different racial groups. In contrast, Hispanics and Blacks are more prone to experiencing brain lesions associated with carotid atherosclerosis compared to other racial groups. Utilizing vascular imaging technology, carotid ultrasound confirms that the Chinese and Black populations have lower rates of carotid stenosis in contrast to Whites. Concurrently, MRI scans ensure that the Chinese population has a higher prevalence of intracranial plaque with LRNC and a lower prevalence of extracranial plaque with HRP relative to the White population, especially in subjects with recent anterior circulation ischemic stroke. Moreover, carotid revascularization procedures have demonstrated the potential to alleviate carotid stenosis. Nevertheless, the Black population experiences less favorable postoperative outcomes, primarily attributable to hospital preferences and racial discrimination.
A series of risk factors play a crucial role in the development of carotid atherosclerosis with or without racial disparities. On the one hand, specific risk factors contribute to carotid stenosis with racial variations. Sleep apnea, for instance, is linked to an increased presence of carotid plaque in White individuals. At the same time, it is associated with an elevated cIMT in Black individuals. Midlife loss and incidents of sexual assault have been found to disproportionately impact the health outcomes of African-American women compared to individuals of other ethnicities. Specific candidate gene loci have been newly associated with cIMT or plaque formation in Mexican and European Americans with RA. Additionally, genetic susceptibility to type 2 diabetes has been linked to a higher likelihood of developing carotid plaques in the Chinese Han population. The COMT gene and three genetic variations have demonstrated predictive capabilities for CVD and extreme TCPA across different racial groups.
On the other hand, some factors influence carotid artery atherosclerosis without ethnic disparities. For example, CAC is a valuable biomarker that can predict cardiovascular events when combined with cIMT. When CAC equals zero, it can effectively rule out severe cardiovascular events, though it may not apply to ischemic strokes and guide drug therapy. During the perimenopausal phase, the transition from reproductive to postmenopausal life can alter the quality of HDL and affect the risk of future CVD. However, HDL subtypes and HDL-mediated cholesterol export do not appear to be associated with subclinical atherosclerosis, necessitating further exploration. Furthermore, factors such as a low socioeconomic environment and experiences of racial discrimination can lead to adverse physical conditions and depressive symptoms. These, in turn, may manifest as more advanced forms of carotid and peripheral artery atherosclerosis. Insulin resistance also significantly influences the development of carotid atherosclerosis, particularly in HIV-infected individuals, where age is a contributing factor.
In conclusion, there is an urgent need for more clinical trials and research studies focused on ethnicity to analyze the specific locations and reasons behind the existence of racial disparities in carotid atherosclerosis. Such efforts will contribute to developing procedures to reduce these disparities and improve overall health quality.
Conclusion
White and Native American populations exhibit a higher prevalence of carotid stenosis compared to other racial groups. Nevertheless, Hispanics and Blacks demonstrate more pronounced brain lesions associated with carotid stenosis. Chinese and Black individuals display less susceptible carotid plaque compared to White individuals. However, disparities in carotid revascularization outcomes persist among minority patients, attributable to racial inequalities and hospital choice. Furthermore, we offer an overview of risk factors associated with racial disparities, encompassing genetic loci, sleep apnea, and emotional aspects, alongside risk factors unaffected by racial inequality.
Footnotes
Author contributions
Huang Yijun proposed that race and ethnicity may have a certain influence on carotid atherosclerosis and wrote the initial draft of the paper. Shi Weihao reviewed the relevant literature, focusing on the influence of risk factors such as racial differences on the incidence of carotid atherosclerosis, and comparing the incidence of carotid atherosclerosis between white and non-white individuals. He Qing reviewed the relevant literature, focusing on the effects of nonracial differences and other risk factors on the incidence of carotid atherosclerosis. These factors include arterial calcium, cIMT, lipid proteins, insulin resistance, and other related factors. Tan Jinyun visualized the collected literature, drew all the tables and figures in the article, and provided writing ideas. Tong Jingdong organized discussions to explore the limitations of the potential impact of race and ethnicity on carotid atherosclerosis. He summarized the risk factors into two categories: those with and those without racial disparities. Yu Bo revised the first draft of the article, focusing on clarifying the content and enhancing its readability. The final manuscript has been read and approved by all authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was sponsored by grants from Medical Key Subspecialty Department of Shanghai Pudong Health System (Grant No. PWZy2020-13) and the Shanghai Sailing Program (Grant No. 21YF1404900).
Ethical approval
This review did not require ethical approval because no human or animal experiment was performed during the process.
Informed consent
Informed consent was unnecessary for this review as it involved no human or animal experiments.
