Abstract
Diabetes mellitus (DM) is a metabolic disorder in which blood glucose levels remain abnormally high (fasting plasma glucose ≥126 mg/dL). Type 2 diabetes mellitus (T2DM) represents the most common form of diabetes which frequently causes kidney inflammation that results in severe complications. Probiotics, which are live beneficial microbes, have been recognized for their supportive role in human health. This work examined three Thai probiotic strains, Limosilactobacillus reuteri TF314, Lacticaseibacillus paracasei MSMC39-1, and Bifidobacterium animalis TA1, for their ability to protect the kidneys in Wistar rats fed a high-fat diet and treated with streptozotocin (STZ) to induce T2DM. Experimental animals were randomly divided into six groups: control, T2DM, TF314, MSMC39-1, TA1, and a mix group (TF314, MSMC39-1, and TA1). All the experimental rats received oral probiotic supplementation for four months. The probiotic treatments given to STZ-induced rats reduced fasting blood glucose and HbA1c levels and improved glucose tolerance. They also helped to reduce kidney injury by decreasing blood urea nitrogen (BUN) and creatinine concentrations and by suppressing pro-inflammatory markers such as TNF-α while raising IL-6. Moreover, changes in gut microbiota were observed, particularly the restoration of gut symbionts Akkermansia muciniphila and an increase in L. reuteri that may underlie better glycemic control and renal protection. The histological examination revealed that probiotic-treated rats experienced reduced kidney tissue damage. This study demonstrates that Thai probiotic strains show potential to minimize kidney problems which occur in T2DM patients.
Introduction
Diabetes mellitus (DM) represents a long-term medical condition which prevents the body from managing blood glucose levels properly, thus causing elevated blood sugar levels. This condition arises from either insufficient insulin production due to pancreatic dysfunction or the body’s resistance to insulin’s effects.1,2 Globally, the prevalence of diabetes was estimated at 10.5% (536.6 million people) in 2021, with projections indicating a rise to 12.2% (783.2 million) by 2045. 3 Type 2 diabetes mellitus (T2DM) is a form of diabetes and a key driver of kidney complications. 4 The condition known as kidney disease (DKD) develops from extended periods of high blood glucose exposure. Persistent hyperglycemia initiates a cascade of events that ultimately injure the kidney through inflammation and fibrosis. A cascade of events plays out across kidney compartments—the glomeruli, the vasculature, the tubules, and the surrounding interstitial tissue—culminating in podocyte loss, glomerulosclerosis, a thickening of the glomerular basement membrane (GBM), the appearance of albumin in the urine (albuminuria), tubulointerstitial fibrosis, and a slow progressive decline in renal function. Together these pathological alterations form the core of both the onset and the advancement of kidney disease.5–7
Probiotics, defined as live micro-organisms with health-promoting properties, benefit the host when consumed in adequate amounts. Research into probiotics for diabetes management has gained increasing scientific attention because of their potential health benefits. Studies suggest that probiotics can help regulate blood sugar, reduce inflammation, and support insulin release.8,9 Probiotics provide renoprotective advantages to patients with DKD that go beyond their ability to control blood sugar levels. Studies indicate that Lactobacillus spp. and Bifidobacterium spp. help treat diabetic nephropathy symptoms through their ability to decrease blood glucose levels, blood urea nitrogen (BUN), creatinine, and urinary protein which indicate kidney damage. 10 Lactobacillus strains have shown evidence of improving blood glucose control and insulin sensitivity, reducing inflammation, and enhancing antioxidant enzyme activity in diabetic nephropathy patients. 11 The strains work to protect kidney tissue through tumor necrosis factor-alpha (TNF-α) suppression which leads to decreased inflammation and oxidative stress caused by high blood sugar levels. 12 Research studies have investigated multi-strain probiotic combinations of Lactobacillus spp. and Bifidobacterium spp. in obese patients who have T2DM. The treatment of T2DM patients with these interventions resulted in substantial BUN level decreases during 12 weeks of therapy. 13 The supplementation of Lactobacillus spp. leads to major decreases in albuminuria and serum creatinine and interleukin-18 (IL-18) levels which serve as essential markers for renal function, inflammation, and disease progression. 14
Research findings demonstrate that gut microbiota modification plays a significant role in producing these beneficial outcomes. The microbial community in T2DM and DKD patients shows decreased diversity and lower numbers of Roseburia spp. and other short-chain fatty acid (SCFA)-producing bacteria while showing elevated levels of harmful bacteria. 15 Unbalanced gut microbiota leads to damaged intestinal barriers which enable lipopolysaccharides (LPS) to enter the bloodstream and create persistent inflammation and oxidative stress that speeds up kidney damage. 16 The beneficial bacteria Lactobacillus and Bifidobacterium increase in number through probiotic treatment which leads to enhanced SCFA production and improved gut barrier integrity. 17 The therapeutic effects of probiotics include lowering endotoxin levels in blood while decreasing inflammatory cytokines and enhancing insulin sensitivity which result in protective effects for the kidneys. Experimental DKD models that received L. rhamnosus and B. longum probiotics showed changes in gut microbiota, simultaneously reduced albuminuria and serum creatinine levels, and decreased renal inflammation which indicates that probiotic-induced microbiota changes drive their therapeutic effects.18,19
The present research has employed three probiotic strains which included Limosilactobacillus reuteri TF314 (previously known as Lactobacillus reuteri TF314) from traditional Thai foods, Bifidobacterium animalis TA1 from raw milk, and Lacticaseibacillus paracasei MSMC39-1 (previously named Lactobacillus paracasei MSMC39-1) from healthy human infants. The strains demonstrate two key functions: They produce bile salt hydrolase to lower cholesterol levels, and they exhibit blood sugar lowering effects. The probiotics boost antioxidant defenses through superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx) enzyme activation while simultaneously decreasing malondialdehyde (MDA) levels which indicate oxidative stress. 20 The strains show anti-inflammatory effects because they lower inflammatory cytokine production in human subjects and laboratory animals.21–24 Given these beneficial properties, the selected probiotic strains were considered appropriate candidates for this study. Accordingly, the present research aimed to evaluate the effects of both individual and combined probiotic strains on reducing kidney complications in a rat model of T2DM.
Materials and Methods
Probiotic Strains and Culture Condition
L. reuteri TF314, B. animalis TA1, 22 and L. paracasei MSMC39-1 25 were obtained from the Center of Excellence in Probiotics, Faculty of Medicine, Srinakharinwirot University, Thailand. The probiotic strains were retrieved from storage at –80°C and subcultured on de Man, Rogosa and Sharpe (MRS) agar (HiMedia, India) at 37°C under anaerobic conditions for 48 hours. Subsequently, the colonies were subcultured three times and then inoculated into MRS broth, followed by incubation at 37°C for 48 hours. The bacterial cells were harvested by centrifugation at 4,000 g for 5 minutes, 4°C, washed to remove the MRS broth and resuspended in phosphate-buffered saline (PBS, 0.1 M, pH 7.2) at a final concentration of 2 × 10¹⁰ CFU/ml for oral administration.
Preparation of High-fat Diet
The research employed the high-fat diet (HFD) formulation which Puttarat et al. (2023) described. The HFD contained 487.05 kcal of energy per 100 grams of diet. The diet contained 17.2% (w/v) beef tallow, 40% (w/v) fresh egg yolk, 16.25% (w/v) egg yolk powder, 12.5% (w/v) sucrose, 5% (w/v) cholesterol powder, and 0.5% (w/v) sodium cholate. 20 The rats were fed with this HFD for four weeks to induce insulin resistance.
The T2DM Rat Model Induced by HFD/STZ
Forty-eight healthy male Wistar rats: WIST@Jcl (GALAS), aged seven weeks and weighing 200–250 g, were purchased from Nomura Siam International Co. Ltd., Thailand. The animal experiment protocol was approved (approval letter no. COA/AE-012-2566) by the Animal Ethics Committee, Srinakharinwirot University. The Medical Center Animal Care Laboratory at Srinakharinwirot University Faculty of Medicine provided controlled environmental conditions for all animals (12-hour light/dark cycle, temperature 22 ± 2°C, and humidity 55 ± 10%). Throughout the 16-week experimental period, the rats had free access to sterilized water and standard chow, which was obtained from the National Laboratory Animal Center, Mahidol University. The basal diet contained 12% (w/v) moisture, 24% (w/v) crude protein, 4.5% (w/v) fat, and 5% (w/v) fiber, with a metabolizable energy value of 146.5 kcal/100 g. Rats were acclimatized for 7 weeks prior to random allocation into six groups (n = 8 per group; three rats per cage). The sample size (n = 8 rats per group) was determined a priori using power analysis with the pwr package in R Studio (version 4.3.1). This analysis yielded an estimated effect size of f ≈ 0.523, with α = 0.05, providing a statistical power of approximately 0.75. The research included three experimental groups which were the control group (1 mL PBS) and T2DM group (1 mL HFD for four weeks followed by a streptozotocin (STZ) injection at 40 mg/kg); probiotic treatment groups consisting of TF314 group (T2DM rats treated with L. reuteri TF314); MSMC39-1 group (T2DM rats treated with L. paracasei MSMC39-1); TA1 group (T2DM rats treated with B. animalis TA1); and a mixed strain group (T2DM rats treated with L. reuteri TF314, L. paracasei MSMC39-1, and B. animalis TA1 at 1:1:1 ratio). The researchers gave the rats in the probiotic groups 2 × 10¹⁰ CFU/mL probiotic suspension through daily gavage starting from day 1 until week 16. The researchers monitored the body weight, food intake, and water consumption of the rats which were measured once per week. The researchers monitored probiotic preparation viability through MRS agar (for Lactobacillus) and Bifidobacterium selective agar (for Bifidobacterium) plating of gavage suspension samples each day during the 16-week study. The presence of live colonies was confirmed each day. The experimental layout is presented in Figure 1.
Experimental Design of the T2DM Rat Model. Rats Received Daily Oral Gavage of Either PBS (Control), a HFD with Streptozotocin (STZ) Injection to Induce T2DM, or T2DM Induction Followed by Probiotic Treatment (L. reuteri TF314, L. paracasei MSMC39-1, and B. animalis TA1) at a Concentration of 2 × 10¹° CFU/ml (n = 8/Group). Rats with FBG Levels ≥7.8 mmol/L (≥140 mg/dL) at Week 5 were Considered Diabetic.
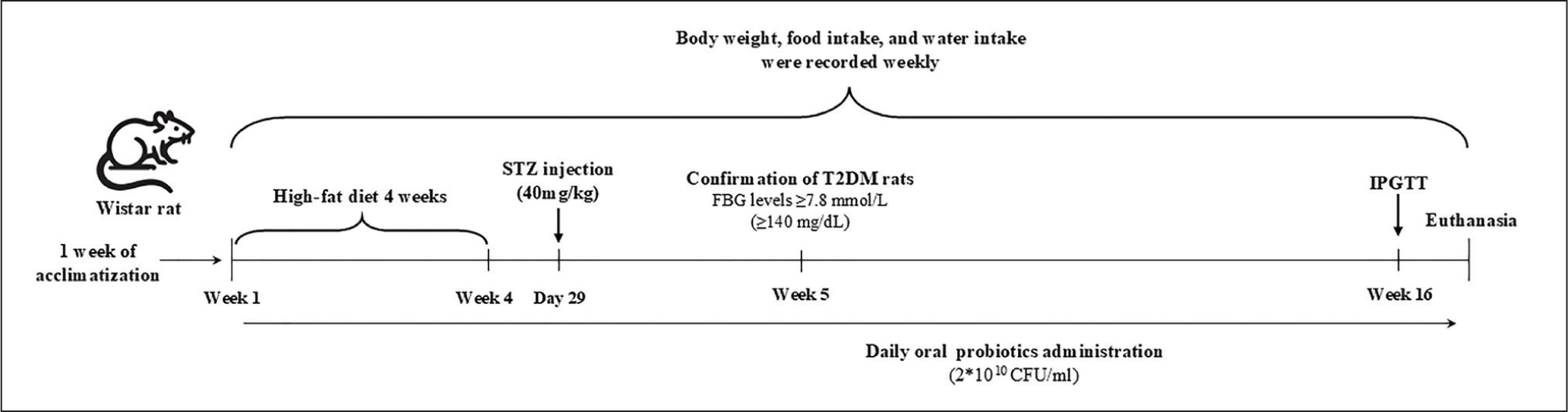
The rats received a HFD for four weeks before undergoing 12 hours of fasting followed by STZ injection at 40 mg/kg body weight through intraperitoneal administration. The solution remained on ice under light protection until immediate use after preparation. The OneTouch glucometer (Contour Next EZ) measured fasting blood glucose (FBG) from tail vein samples one week after the STZ injection. The rats developed diabetes when their FBG reached 7.8 mmol/L (140 mg/dL) or higher.
Intraperitoneal Glucose Tolerance Test (IPGTT)
The IPGTT procedure took place at week 16 of the study when researchers obtained blood samples through tail vein punctures. The rats received 2 g/kg body weight intraperitoneal glucose injections after scientists made them fast for 12 hours. The OneTouch glucometer measured blood glucose levels at five time points: 0 minutes and then at 30 minutes, 60 minutes, 90 minutes, and 120 minutes.
Collection and Determination of Serum Biochemical Parameters
At the end of week 16, all rats were fasted for 12 hours and euthanized under isoflurane anesthesia. Blood samples were collected and immediately centrifuged at 4,000 g, for 10 minutes at 4°C. The plasma samples underwent storage at −80°C temperatures until researchers conducted their analysis. The Professional Laboratory in Thailand (PROLAB) performed tests on plasma samples to determine FBG, hemoglobin A1C (HbA1c), BUN, and creatinine levels.
Evaluation of Inflammatory Cytokines in Kidney Tissue
After euthanasia, kidneys were collected and homogenized (10% w/v) with RIPA lysis buffer. The lysed tissue samples were sonicated on ice for 5 minutes using an ultrasonic homogenizer (Sonoplus, Bandelin, Germany). Samples were then centrifuged at 10,000 g, for 15 minutes at 4°C, and the supernatants were stored at −80°C. The kidney supernatants were used to measure TNF-α and IL-6 using enzyme-linked immunosorbent assays (ELISA, R&D systems, Minneapolis, MN, USA), following the manufacturer’s instructions.
Histopathological Observation of Kidney Tissue
At the end of week 16, kidney tissues from each experimental group were fixed in a 10% paraformaldehyde. The fixed tissues were then embedded in paraffin, sectioned at a thickness of 3–5 μm, and stained with hematoxylin and eosin (H&E). Histological analysis was performed under a light microscope (Olympus UC50, Tokyo, Japan) at 200× and 400× magnification, with ten randomly selected fields evaluated per sample. Bowman’s space (μm²) was quantified from H&E-stained kidney sections using ImageJ (NIH, USA). Images were captured at 200× magnification, and intact, round-shaped glomeruli were randomly selected to minimize sectioning bias. A total of n = 10 glomeruli/fields were analyzed per group, and values were expressed as mean ± standard error of the mean (SEM).
Analysis of Intestinal Microbiota in Fecal Samples
At week 16, fecal samples were obtained for gut microbiota profiling via next-generation sequencing (NGS). DNA was extracted using the nucleic acid extraction kit model S-Bin. DNA concentration and purity were assessed with a DS-11 Series Spectrophotometer (DeNovix, USA). The full-length 16S rRNA gene was then amplified and sequenced using the Oxford Nanopore platform (Oxford Nanopore Technologies, Oxford, UK). This report was produced using the EPI2ME-labs/wf-16s.
Statistical Analysis
All statistical analyses were performed using GraphPad Prism (version 10). Data are presented as mean ± SEM. Each experimental group consisted of eight rats (n = 8). For outcomes measured at a single time point (e.g., body weight, FBG at the endpoint, HbA1c, IPGTT, BUN, creatinine, cytokines, histological parameters, and microbiota relative abundance), group differences were analyzed using one-way analysis of variance (one-way ANOVA). For repeated measurements of metabolic behavior (food intake and water intake), group and time effects were evaluated using two-way repeated measures ANOVA (factors: treatment group × time). When an overall significant effect was detected, Dunnett’s multiple comparisons post hoc test was applied to compare each probiotic-treated group against the DM group. A p value <.05 was considered statistically significant.
Results and Discussion
Probiotic Intervention Improved Glycemic Control and Modulated Metabolic Behaviors in T2DM Rats
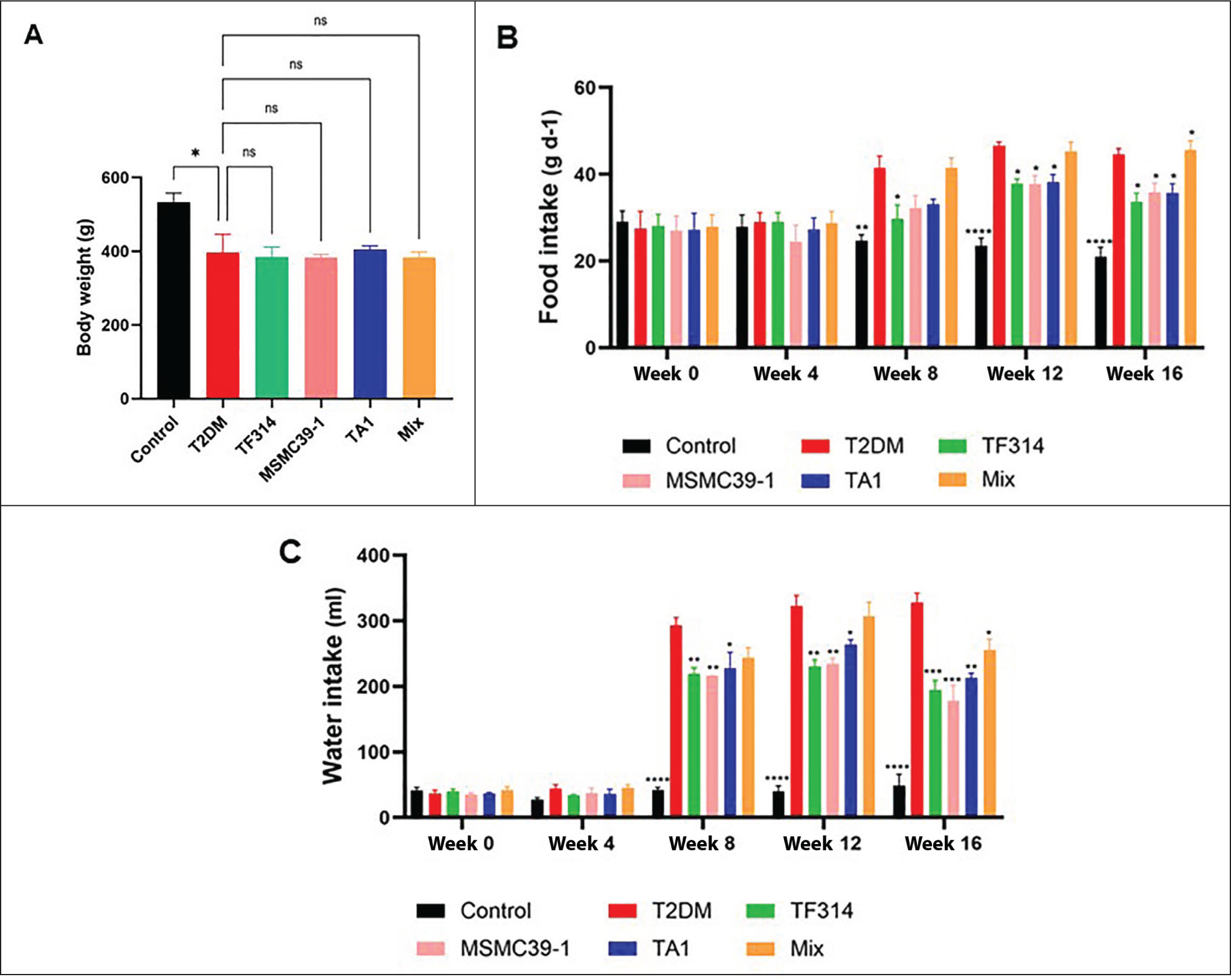
T2DM the most prevalent form of diabetes, is increasing globally. It is characterized by pancreatic β-cell and insulin resistance, resulting in chronic hyperglycemia that affects multiple organs, especially the kidneys. Diabetic nephropathy represents the most common microvascular complication and is a leading cause of chronic kidney disease (CKD). 26 Long-term high blood sugar levels may accelerate kidney damage by causing oxidative stress and problems with mitochondria. These problems lead to more damage to kidney cells and the progression of CKD.27,28 In this study, the diabetic group exhibited classic symptoms of diabetes, including significant weight loss and increased food and water intake (Figure 2), indicative of polyphagia, polydipsia, and cachexia. These metabolic abnormalities may result from impaired cellular glucose uptake due to insulin resistance, resulting in increased lipolysis and proteolysis, as well as modified hypothalamic appetite signaling. The body shows these signs because insulin resistance blocks glucose cell entry which leads to fat and muscle tissue breakdown and stronger signals for hunger in the hypothalamus. The body develops polydipsia as a response to osmotic diuresis which occurs because of polyuria. The probiotic-treated groups kept their body weight at about the same level as the T2DM group. However, the TF314, MSMC39-1, TA1, and mixed formulation groups ate and drank less than the T2DM group. This reduction may indicate enhanced metabolic efficiency and appetite regulation facilitated by gut-derived metabolites, including SCFAs, which have been demonstrated to affect energy homeostasis and appetite-related hormones (e.g., glucagon-like peptide-1 (GLP-1) and peptide YY (PYY)).29,30 The mixed formulation may offer enhanced metabolic advantages due to the synergistic effects of multiple strains, which can complement each other’s roles in modulating gut microbiota, strengthening barrier integrity, and optimizing metabolic regulation, consequently alleviating the overall diabetic burden associated with DKD risk.
Effect of Probiotics on Body Weight, Food Intake, and Water Intake in DM Rats. (A) Body Weight at Week 16; (B) Food Intake; and (C) Water Intake, Measured Every 4 Weeks Throughout the 16-week Experimental Period. Statistical Analysis is by One-way ANOVA Followed by Dunnett’s Post Hoc Test in A. Data were Analyzed Using Two-way Repeated Measures ANOVA (Factors: Group × Time) in B-C. When Significant Main Effects or Interactions were found, Dunnett’s Post Hoc Test was Applied as a Post-hoc Test to Identify Differences Between Groups at Each Time Point. Error Bars Show the Mean ± SEM (n = 8/group). *p < .05, **p < .01, ***p < .001, ****p < .0001 Compared with the T2DM Group. “ns” Indicates no Significant Difference.
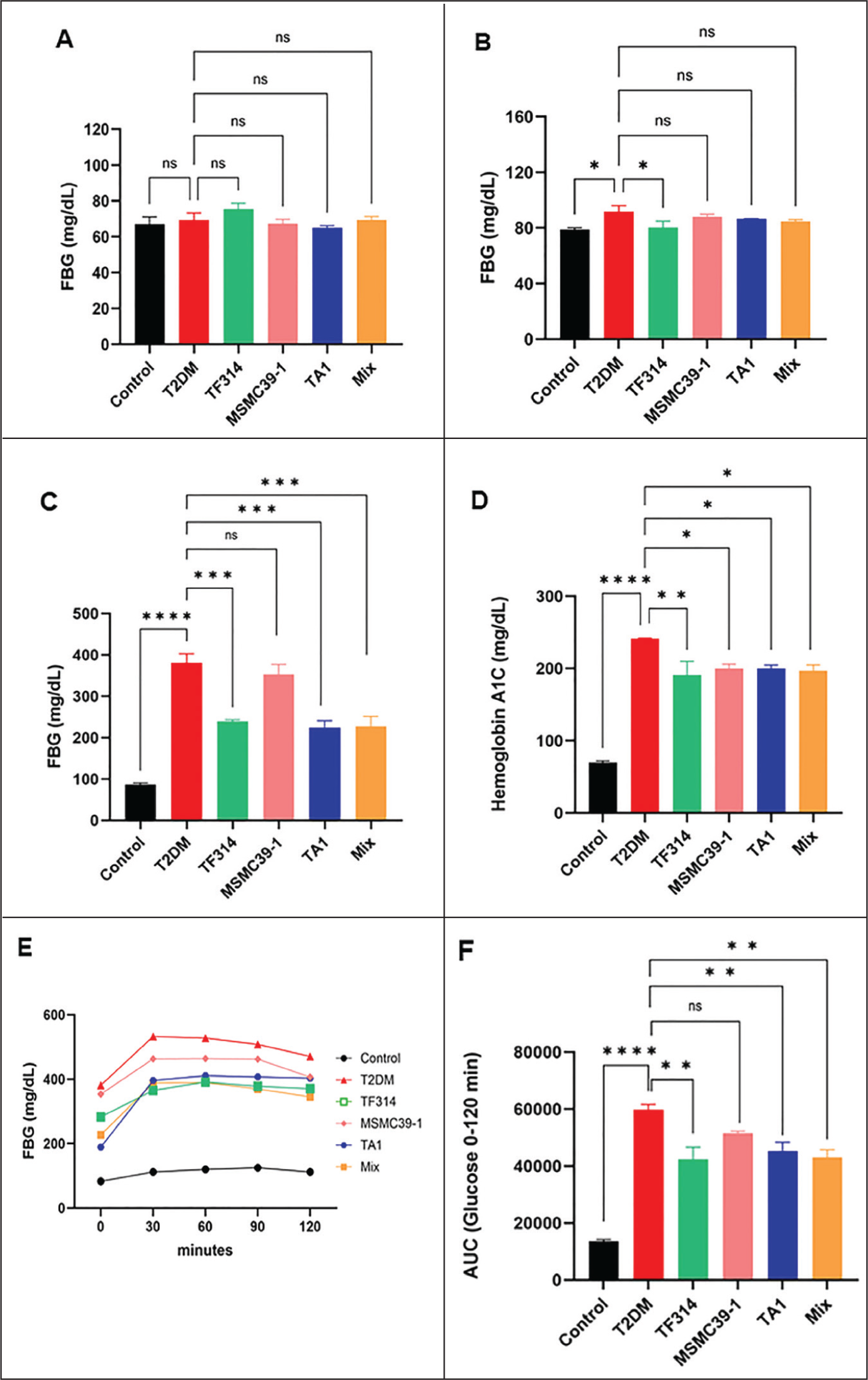
The prolonged high blood sugar levels cause sodium-glucose cotransporter 2 (SGLT2) to absorb more glucose which reduces the amount of NaCl that reaches the macula densa. The renin-angiotensin-aldosterone system (RAAS) system becomes activated because of disrupted tubuloglomerular feedback which causes efferent arteriole constriction while increased endothelial nitric oxide production enhances afferent blood flow. The resulting changes in blood pressure and kidney damage occur because of glomerular hypertension.31,32 In addition to hemodynamic dysregulation, RAAS activation is a crucial mediator of renal disease progression and is intricately associated with pro-fibrotic signaling pathways, including those related to transforming growth factor-beta (TGF-β), which facilitate extracellular matrix accumulation and renal fibrosis in DKD.33,34 The FBG measurements at week 0 showed no group differences which proved that all subjects started with equivalent blood glucose levels before diabetes development. The DM group developed higher FBG levels than the control group after consuming the HFD which confirmed the success of diabetes induction. The probiotic supplement L. reuteri TF314 effectively reduced blood glucose levels in HFD-fed subjects by producing better glucose-lowering effects than the DM group (p = .0457) (Figures 3A and 3B). The DM group showed elevated FBG and HbA1c levels during week 16 which confirmed their successful development of diabetes (Figures 3C and 3D). The probiotic treatment reduced both parameters which indicates probiotics help control essential metabolic factors linked to DKD development. Overall, probiotic supplementation improved glycemic control by significantly reducing FBG levels in the TF314, TA1, and mixed formulation groups compared with the DM group (p = .0005, p = .0002, and p = .0003, respectively) (Figure 3C). HbA1c responses varies among strains; however, TF314, MSMC39-1, TA1, and the mixed formulation groups exhibited significantly lower HbA1c levels compared to the DM group (p = .0089, p = .0321, and p = .0194, respectively), indicating enhanced long-term glycemic control (Figure 3D). In contrast, MSMC39-1 did not significantly affect FBG relative to the DM group, indicating that its effects may be strain-specific and not directly associated with glycemic regulation in this model. Improved control of blood sugar levels may protect the kidneys by lowering oxidative stress caused by glucose and limiting damage to kidney cells that happens later on, both of which are known to speed up the progression of DKD.27,28 The mixed group exhibited relatively stable glucose control, which may be advantageous, as persistent glycemic variability correlates with poorer renal outcomes in diabetes. The researchers conducted oral glucose tolerance tests to validate their findings. The T2DM group demonstrated impaired glucose elimination through blood tests which showed decreased blood glucose levels only during the 90 and 120-minute post-glucose injection period. The control and probiotic-treated groups, on the other hand, showed faster glucose clearance (glucose disposal), which means that their bodies were better able to control glucose levels than untreated T2DM rats. The control group and probiotic-treated subjects demonstrated faster glucose absorption rates. The L. reuteri TF314 group demonstrated improved insulin sensitivity through enhanced glucose utilization starting at the 60-minute mark (Figures 3E and 3F). Over the 0–120 min period of the IPGTT, glucose levels were markedly reduced in the TF314, MSMC39-1, TA1, and mixed formulation groups compared to the DM group (p = .0019, p = .0075, and p = .0025, respectively), which indicates enhanced glucose clearance (Figures 3E and 3F). The MSMC39-1 group, on the other hand, did not show a statistically significant drop in glucose levels over the 0–120 minutes compared to the DM group. This result probably shows that different strains work differently in terms of function. MSMC39-1 has a weaker effect on glucose-clearing mechanisms than TF314, TA1, and the mixed formulation, which is why the IPGTT improvement is not significant. The mixed group also showed an overall improvement in glucose tolerance, which suggests that combining strains may help the body respond more consistently to glucose challenges. Probiotics may enhance insulin resistance and glucose metabolism by modulating the microbiota and restoring gut barrier function, thereby decreasing metabolic endotoxemia (circulating LPS) that fosters chronic low-grade inflammation and insulin resistance.35,36 Furthermore, SCFAs can modulate host metabolism via receptors such as G-protein coupled receptor 43 (GPR43/GPR41), affecting both immune and metabolic pathways associated with T2DM and DKD. 37 The study indicates that probiotic supplements help control blood sugar levels and affect how the body handles energy while influencing appetite signals in the hypothalamus. These findings collectively indicate that probiotic-mediated nephroprotective effects may transcend inflammatory markers, involving integrated mechanisms such as enhanced glycemic regulation, diminished oxidative stress and mitochondrial dysfunction, fortified gut barrier integrity, reduced endotoxin translocation, and SCFA-associated metabolic signaling, which may collectively alleviate principal factors contributing to DKD progression.
The Effect of Probiotic Intervention Improves Glycemic Control and Modulates Metabolic Behaviors in T2DM Rats. (A) FBG Levels Week 0 (B) FBG Levels After HFD (C) FBG Levels Week 16 (D) Hemoglobin A1C Levels (E) Glucose Tolerance and (F) Area Under the Curve (AUC). Data were Analyzed Using One-way ANOVA Followed by Dunnett’s Post Hoc Test. Error Bars Show the Mean ± SEM (n = 8/group). *p < .05, **p < .01, ***p < .001, ****p < .0001 Compared with the T2DM Group. “ns” Indicates no Significant Difference.
Probiotic Supplementation Improved Kidney Function and Modulated Inflammation in T2DM Rats
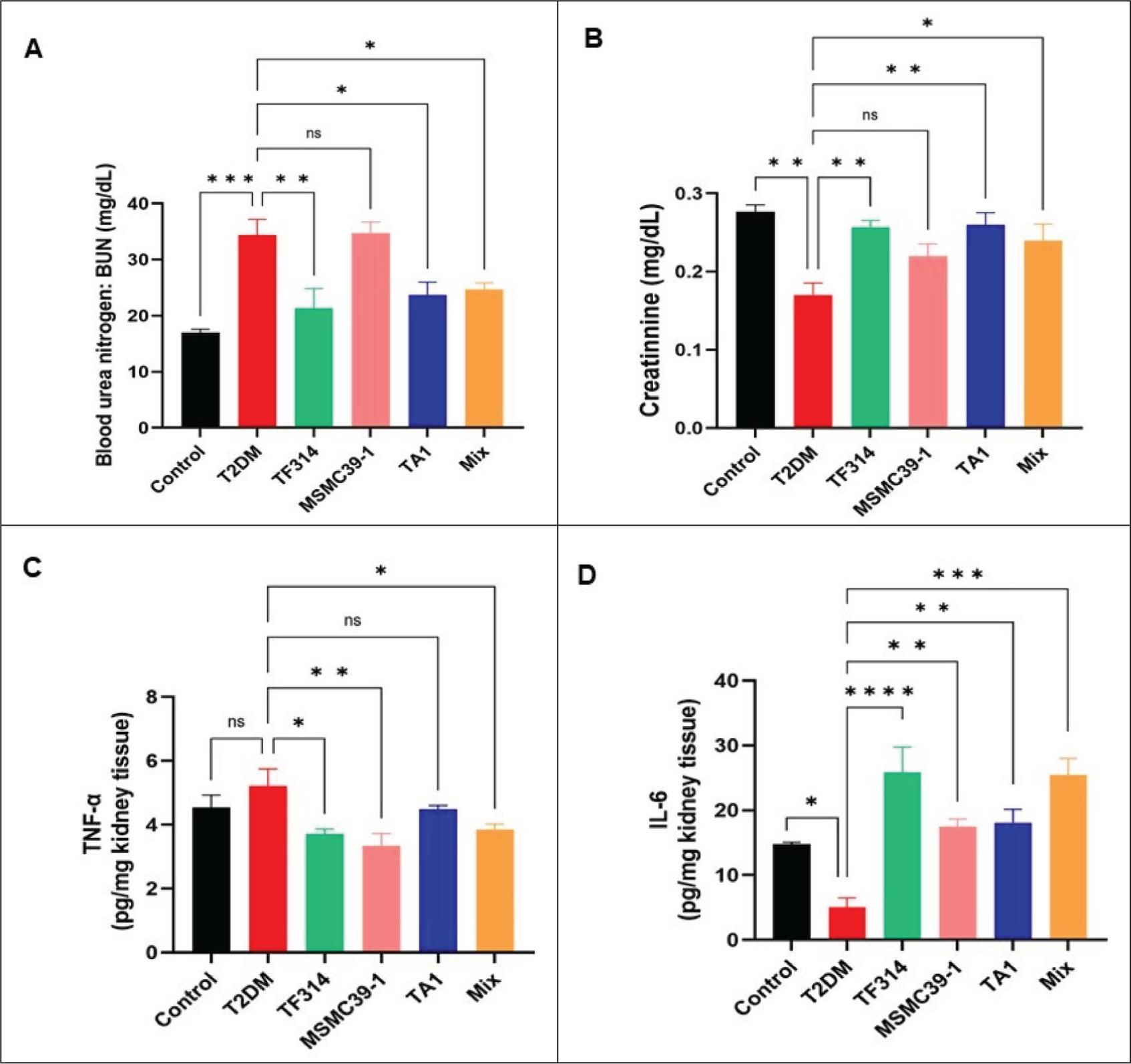
The T2DM group showed elevated BUN levels which indicated poor renal clearance according to kidney function tests. The probiotic-treated rats demonstrated better BUN results which indicated their kidneys functioned better (Figure 4A). The BUN levels were significantly decreased in the TF314, TA1, and mixed formulation groups compared to the DM group (p = .0071, p = .0252, and p = .0435, respectively), suggesting enhanced renal waste clearance in diabetic conditions. MSMC39-1, on the other hand, did not show a significant change in BUN when compared to the DM group. This suggests that the renoprotective effects are different for each strain. The mixed group may have had a stronger effect because multiple strains can work together to change the gut microbiota, strengthen the gut barrier, and control immunometabolism pathways. This gives them more protection against diabetic renal dysfunction than single-strain supplementation. Compared to the DM group, the TF314, TA1, and mixed formulation groups exhibited modestly higher creatinine levels (p = .0052, p = .0040, and p = .0215, respectively) (Figure 4B). The MSMC39-1 group, on the other hand, did not show significant change compared to the DM group. The DM group’s creatinine level is lower than average, which could be because they lost weight and muscle (cachexia) due to diabetes, which can lower creatinine production. On the other hand, the higher creatinine levels in the probiotic-treated groups may be due to better nutrition and the preservation of lean mass, which would lead to more creatinine being made and not worse kidney damage. Consequently, alterations in creatinine must be analyzed in conjunction with BUN levels and histopathological findings to more accurately represent the overall renal condition. The development of DKD heavily depends on inflammatory processes. The T2DM group showed elevated TNF-α levels but IL-6 expression decreased substantially. The probiotic treatment decreased TNF-α levels while it restored IL-6 expression (Figures 4C and 4D) which suggesting a context-dependent immunomodulatory effects. The TF314, MSMC39-1, and mixed formulation groups showed TNF-α levels that were significantly lower than those of the DM group (p = .0257, p = 0.0059, and p = .0413, respectively) (Figure 4C). Conversely, the TA1 group demonstrated no statistically significant variation in TNF-α compared to the DM group. In parallel, IL-6 expression was markedly reinstated in the TF314, MSMC39-1, TA1, and mixed formulation groups in comparison to the DM group (p < .0001, p = .0075, p = .0053, and p = .0001, respectively) (Figure 4D). The mixed group consistently displayed a favorable inflammatory profile, indicating that the combination of strains may augment the overall anti-inflammatory effect by simultaneously targeting multiple immune-regulatory pathways. The strain-specific anti-inflammatory effects of L. paracasei MSMC39-1 and L. reuteri TF314 became evident through their ability to decrease TNF-α and increase IL-6 expression. The combination of high blood sugar in T2DM patients leads to advanced glycation end-products (AGEs) and reactive oxygen species (ROS) accumulation which activates nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) and mitogen-activated protein kinases (MAPKs) pathways to produce fibrosis and kidney damage.38,39 TNF-α causes additional damage to the kidneys through its ability to harm podocytes and produce more albumin in the urine. The effects of IL-6 on the body remain complex because they depend on specific situations. IL-6 activates two different signaling pathways which include membrane-bound IL-6 receptors (mIL-6R) that function in immune cells and podocytes and soluble IL-6 receptors (sIL-6R) that enable IL-6 signaling in tubular epithelial cells and other cells without mIL-6R.40,41 Research now shows that IL-6 trans-signaling may exert protective effects during metabolic stress even though it typically causes inflammation. The signal transducer and activator of transcription 3 (STAT3)/lactate dehydrogenase A (LDHA)/sirtuin 2 (SIRT2) pathway activated by IL-6 trans-signaling enhances glycolysis while simultaneously activating autophagy and nuclear factor erythroid 2-related factor 2 (NRF2) pathway to protect pancreatic β-cells from oxidative damage. 42 IL-6 functions to decrease neutrophil adhesion to glomerular endothelial cells which helps control localized inflammation. 43 The T2DM group showed decreased IL-6 expression but probiotic-treated rats maintained IL-6 levels at control group levels. The research indicates that probiotics could direct IL-6 signaling pathways toward protective mechanisms in kidney tissue. Research on hepatic ischemia/reperfusion injury shows that IL-6 deficiency leads to worse tissue damage but IL-6 supplementation decreases TNF-α, c-reactive protein (CRP) levels, and tissue necrosis. 44 The simultaneous decrease in TNF-α and normalization of IL-6 in probiotic-treated groups—especially in the mixed formulation—may help reduce the inflammatory and oxidative damage that causes kidney problems in DKD, which would explain the positive effects on kidney health seen in this study.
The Effect of Probiotics Improves Kidney Function and Inflammation in T2DM Rats After 16 Weeks of Treatment. (A) Blood Urea Nitrogen (B) Creatinine Levels in Blood (C) TNF-α in Kidney Tissues, and (D) IL-6 Levels in Kidney Tissues. Data were Analyzed Using One-way ANOVA followed by Dunnett’s Post Hoc Test. Error Bars Show the Mean ± SEM (n = 8/group). *p < .05, **p < .01, ***p < .001, ****p < .0001 Compared with the T2DM Group. “ns” Indicates no Significant Difference.
Renal Protective Effects of Probiotics in T2DM Rats Via Histological Analysis
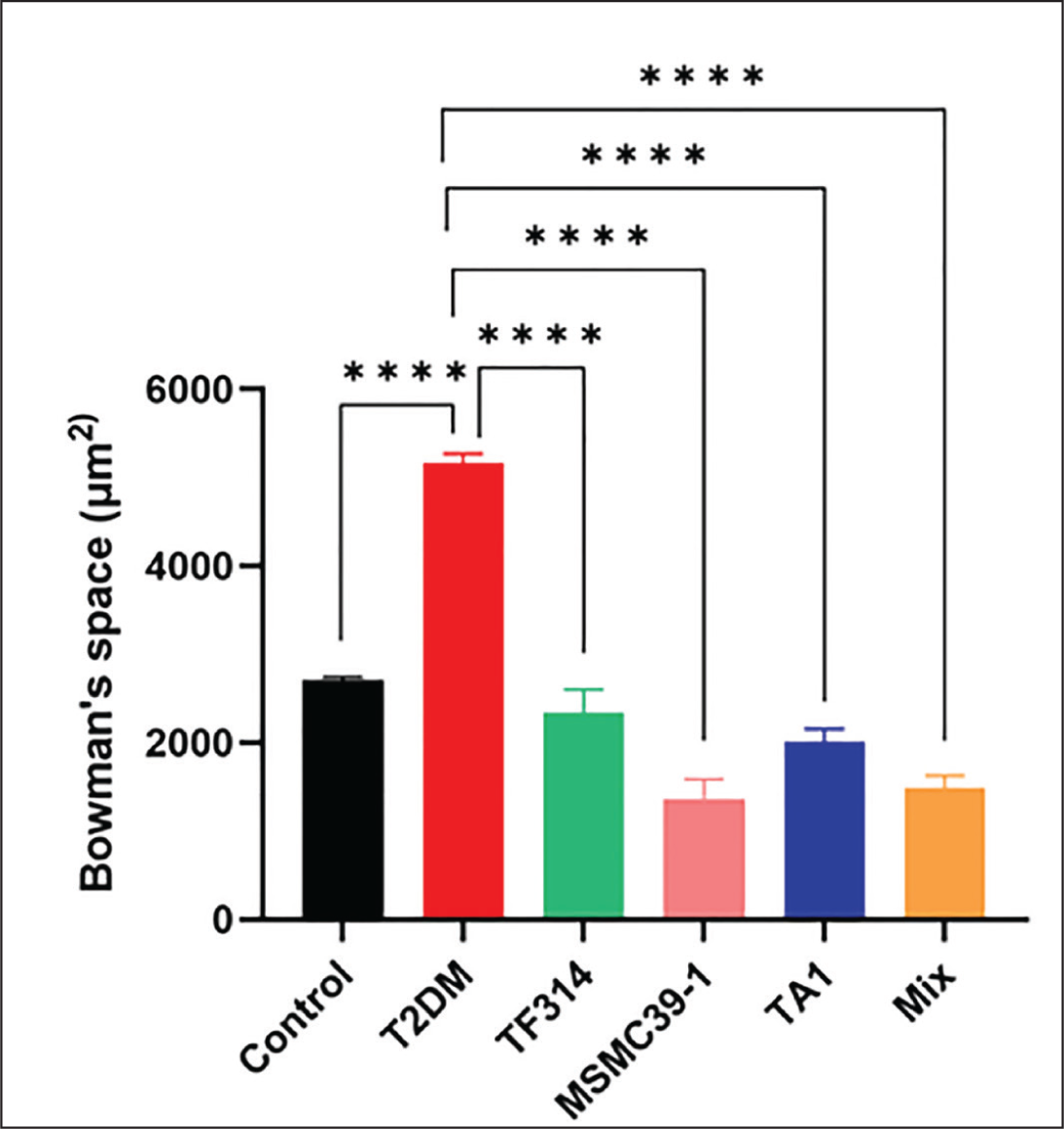
The histological evaluation of kidney tissues from T2DM rats showed multiple severe pathological changes which resulted from long-term high blood sugar levels and inflammatory responses. The kidney tissue showed four main histopathological changes which included tubular epithelial cell necrosis and hemorrhage and tubular dilation due to hypertrophy and epithelial detachment. The glomeruli displayed severe changes which included thickened Bowman’s capsule with partial scarring, damaged filtration membrane, expanded mesangial matrix, collapsed capillaries that indicated worsening kidney function, and filtration problems. These lesions are indicative of progressive DKD pathology, characterized by chronic hyperglycemia and inflammatory stress that lead to tubular injury and glomerular structural remodeling, ultimately compromising filtration efficiency. Mechanistically, persistent hyperglycemia facilitates the accumulation of AGEs and induces oxidative stress, which can trigger downstream inflammatory signaling pathways (e.g., NF-κB and MAPK pathways), consequently exacerbating renal cellular injury and structural deterioration in diabetic nephropathy.45,46 Simultaneously, pro-fibrotic mediators, especially TGF-β/mothers against decapentaplegic (TGF-β/Smad) signaling, promote extracellular matrix accumulation and mesangial enlargement, leading to glomerulosclerosis and advancing renal fibrosis.47,48 In contrast, rats treated with Thai probiotic strains showed substantial preservation of renal architecture. The proximal tubules displayed minimal epithelial damage (Figure 5), and the glomeruli largely retained normal morphology, with significantly reduced Bowman’s space widening and mesangial expansion (Figures 6 and 7). Similarly, parameters associated with glomerular damage, such as the widening of Bowman’s space, were significantly superior in the TF314, MSMC39-1, TA1, and mixed formulation groups compared to the DM group (p < .0001, p < .0001, p < .0001, and p < .0001, respectively). There was little evidence of necrosis, inflammatory cell infiltration, or interstitial damage. The mixed group exhibited a more consistently maintained renal architecture in both tubular and glomerular compartments, indicating that the combination of strains may offer enhanced histological protection relative to single-strain supplementation. These findings indicate that probiotic supplementation effectively mitigated T2DM-associated renal damage and preserved nephron integrity, suggesting a renal protective role for the Thai probiotic strains under diabetic conditions. This histological preservation corresponds with the enhancements noted in metabolic regulation and inflammatory modulation, reinforcing the hypothesis that probiotics may mitigate renal injury by alleviating glucotoxicity stress and curtailing inflammatory damage that promotes mesangial expansion and glomerular scarring in DKD. Moreover, probiotic-driven increases in gut microbiota-derived metabolites such as SCFAs have been shown to directly exert anti-inflammatory actions in renal cells and ameliorate experimental diabetic nephropathy, partly via GPR43-mediated suppression of oxidative stress and inflammatory responses, providing a plausible mechanistic explanation for the reduced tubular necrosis and preserved glomerular structure observed in probiotic-treated groups, particularly in the mixed formulation group.49–51
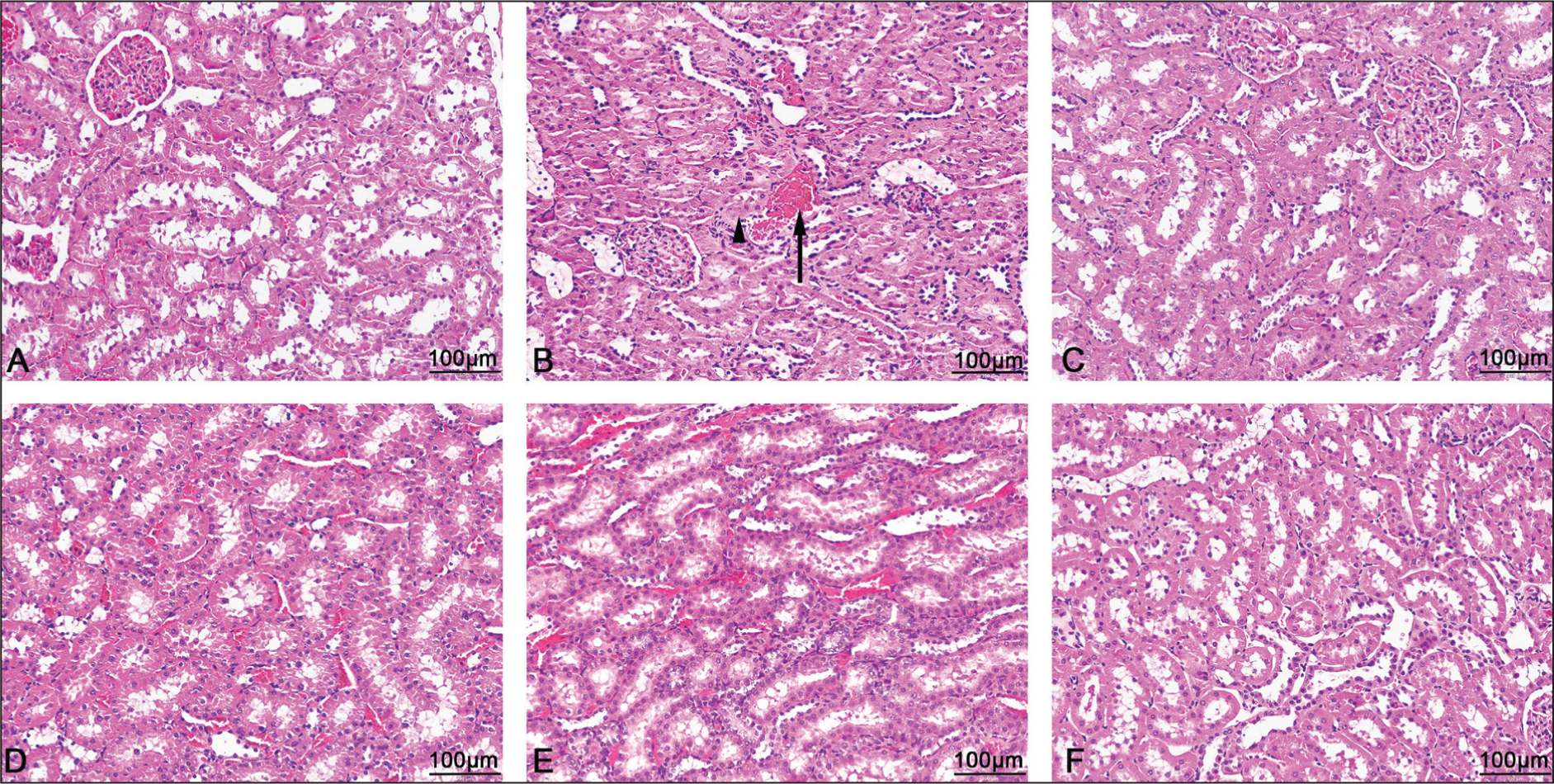
The Researchers Performed Histological Examinations of Renal Tubules from all Experimental Groups Through H&E Staining. The Control Group Renal Tissue (A) Displayed Typical Tubular Architecture Without any Evidence of Hemorrhage or Cellular Damage. The T2DM Group (B) Presented with Severe Tubular Damage that Included Black Arrow Pointing to Interstitial Hemorrhage and Black Arrowheads Indicating Epithelial Cell Necrosis. These Abnormalities were Markedly Reduced in T2DM Rats Treated with L. reuteri TF314 Group (C), L. paracasei MSMC39-1 (D), B. animalis TA1 (E), and the Mixed Strain Group (F). Images were Obtained at 200× Magnification.
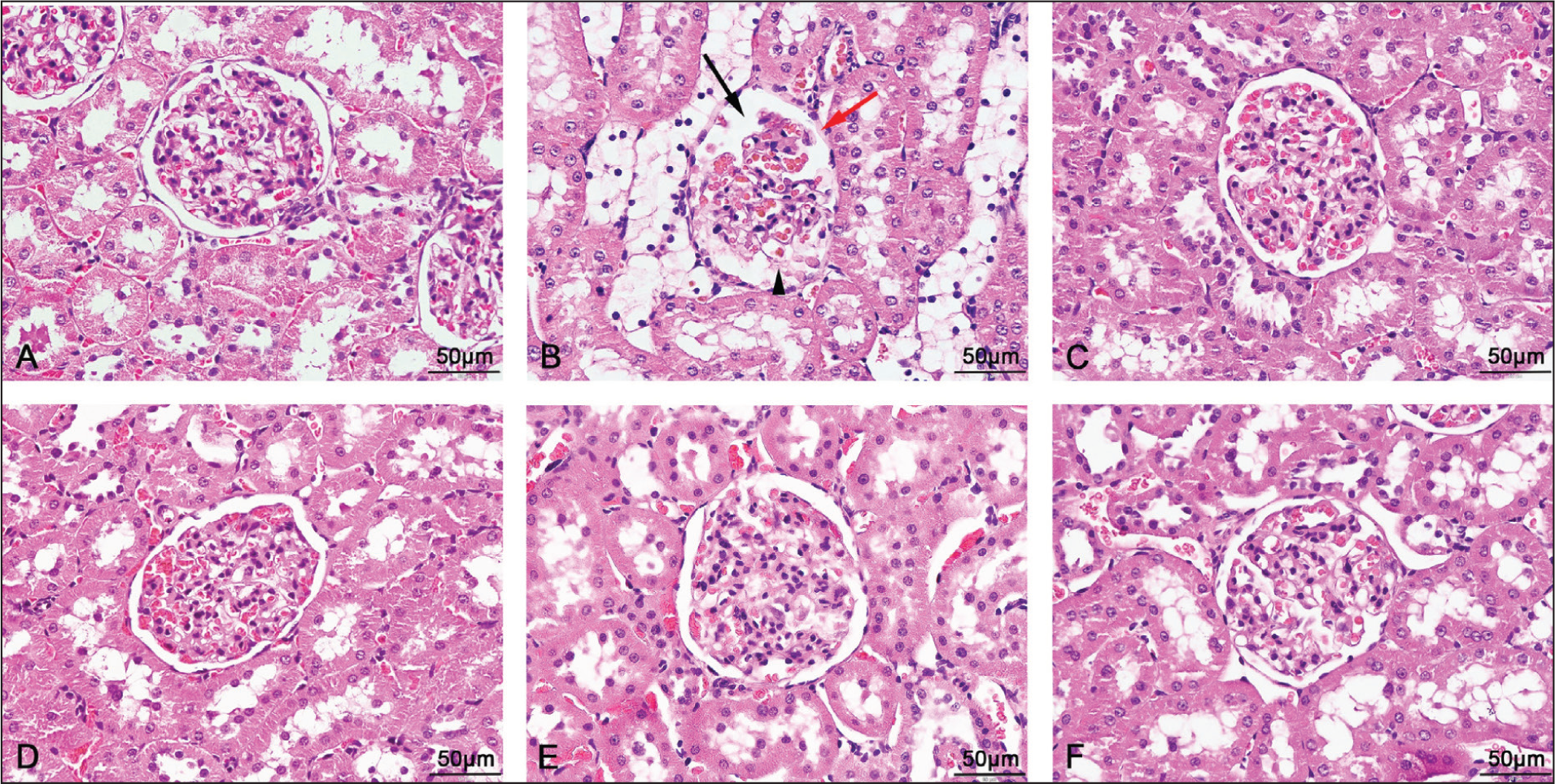
The Examination of Glomerular Structures Through H&E Staining at 400× Magnification Revealed Normal Glomerular Structure in Control Samples (A) with Unaltered Filtration Membranes and Small Bowman’s Space. The T2DM Group Kidneys (B) Displayed Distinct Pathological Changes Which Included Expanded Bowman’s Space (Black Arrows) and Thickened Bowman’s Capsule (Red Arrows) and Damaged Filtration Membranes (Black Arrowheads). These Abnormalities were Markedly Reduced in T2DM Rats Treated with L. reuteri TF314 Group (C), L. paracasei MSMC39-1 (D), B. animalis TA1 (E), and the Mixed Strain Group (F). Images were Obtained at 400× Magnification.
The Study Measured Glomerular Area in Kidney Tissue Sections from T2DM Rats Who Received Probiotic Strains. The Bar Graph Shows the Average Glomerular Area (µm²) with SEM for n = 10 Glomerular Fields in Each Group. The T2DM Group Showed Larger Glomerular Areas than Controls which Indicated Glomerular Hypertrophy. The Glomerular Area Measurements in T2DM Rats Receiving TF314, MSMC39-1, TA1, and Mix Probiotics Showed Lower Values than the T2DM Group. Data were Analyzed Using One-way ANOVA Followed by Dunnett’s Post Hoc Test. *p < .05, **p < .01, ***p < .001, ****p < .0001 Compared with the T2DM Group.
Gut Microbiota Composition and Diversity were Analyzed at Both the Genus and Species Levels
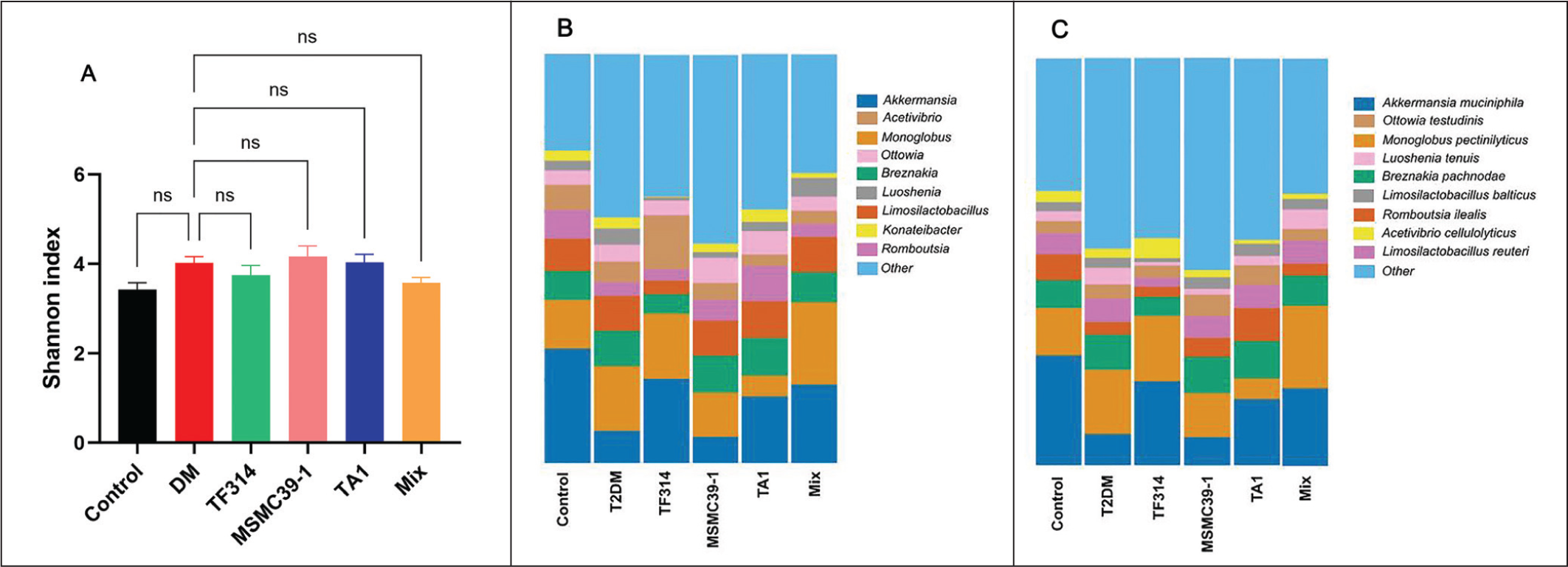
The gut microbiota functions as a vital element in T2DM development and DKD progression because it affects host metabolic processes, energy management, and immune system control.52–54 The Shannon diversity analysis showed no significant differences in α-diversity between the different groups (Figure 8A). The results indicate that diabetes induction and probiotic treatment failed to produce significant changes in the microbial community’s total diversity and evenness. Research indicates that diabetic dysbiosis occurs through specific taxonomic changes rather than major diversity shifts.55,56 The control rats in this study contained Akkermansia as their primary genus because this bacterium specializes in mucin breakdown and supports both gut barrier function and metabolic wellness. 57 The diabetic rats exhibited decreased Akkermansia populations while their microbial community became more dominated by the Monoglobus, Breznakia, and Romboutsia species (Figure 8B). The probiotic strains L. reuteri TF314, B. animalis TA1, and the mixed formulation group demonstrated effectiveness in restoring microbial balance through increased Akkermansia and Limosilactobacillus populations. The mixed group exhibited a more reliable restoration of these advantageous taxa, indicating that multi-strain supplementation may offer more extensive microbiota support compared to single-strain intervention. Research indicates that Akkermansia muciniphila exists at low levels in obese people with insulin resistance and systemic inflammation which makes it a promising candidate for future probiotic development.58,59 The T2DM group displayed lower A. muciniphila numbers than control rats (Figure 8C) which resulted in damaged mucus layer structure and elevated intestinal permeability that leads to body-wide inflammation and kidney damage. 60 The probiotic treatment groups containing L. reuteri TF314, B. animalis TA1, and the mixed formulation achieved A. muciniphila levels similar to controls which indicate better gut barrier integrity. The probiotic treatment led to increased levels of Monoglobus pectinilyticus which are known to ferment carbohydrates and generate short-chain fatty acids. 61 The probiotic treatment led to increased L. reuteri populations because this bacterium has a proven ability to regulate immune responses and reduce inflammation.62,63 The three SCFAs, acetate, propionate, and butyrate, work together to control glucose and lipid metabolism and boost regulatory T cells (Treg) cells and decrease oxidative stress which helps prevent DKD progression. 48 Mechanistically, the restoration of A. muciniphila may improve the integrity of the mucus layer and the function of tight junctions, thereby decreasing gut permeability and the translocation of endotoxin (LPS) into systemic circulation. Lower levels of endotoxemia may help stop chronic low-grade inflammation and inflammatory signaling that leads to kidney damage in DKD. At the same time, more SCFAs may activate host receptors like GPR43/GPR41, which could make insulin more sensitive, boost Treg-mediated immune regulation, and lower oxidative stress. These combined effects may lessen inflammation and fibrosis in the kidney, which would help keep nephrons healthy and slow down the progression of DKD. The results indicate that probiotics maintained stable overall diversity but specifically modified important microbial populations which protect the intestinal barrier, generate SCFAs, and control immune responses thus explaining their protective effects against DKD. Collectively, these findings support a gut–kidney axis mechanism in which probiotic-driven restoration of A. muciniphila and SCFA-associated taxa may strengthen intestinal barrier integrity, reduce gut permeability and endotoxin translocation, and thereby attenuate systemic inflammation, ultimately contributing to renal protection and delayed DKD progression.
Gut Microbiota Diversity and Composition in Control, Diabetic, and Probiotic-Treated Rats After 16 Weeks of Treatment. (A) Shannon Diversity Index. (B) Relative Abundance of Gut Microbiota at the Genus Level. (C) Relative Abundance of Gut Microbiota at the Species Level. Data are Presented as Mean ± SEM (n = 8/group). Statistical Analysis was Performed Using One-way ANOVA Followed by Dunnett’s Post Hoc Test; “ns” Indicates no Significant Difference.
Conclusion
The research stands as the first assessment of Thai probiotic strains native to indigenous populations when used to treat DKD. The three probiotic strains Limosilactobacillus reuteri TF314, Lacticaseibacillus paracasei MSMC39-1, and Bifidobacterium animalis TA1 showed positive effects through their ability to lower FBG and HbA1c levels, enhance glucose tolerance, and partially reduce BUN and creatinine levels while controlling inflammation through TNF-α reduction and IL-6 elevation. The histopathological results showed reduced kidney damage which proved renoprotective effects at the tissue level. The probiotic treatment led to specific beneficial microbial growth while Akkermansia muciniphila levels returned to normal which established a connection between probiotic treatment and improved metabolic health through microbiota modification. The research demonstrates both the new discovery and practical uses of specific probiotic strains which show promise for treating DKD complications. The study establishes a base for future research into mechanisms and clinical uses of indigenous probiotics for diabetes treatment.
Footnotes
Acknowledgements
This research was funded by Faculty of Medicine and HRH Princess Maha Chakri Sirindhorn Medical Center, Srinakharinwirot University (grant number 288/2567) and the Center of Excellence in Probiotics (grant number 142/2568).
Authors’ Contribution
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Data Availability Statement
All the data is available with the authors and shall be provided upon request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study protocol was approved by the Animal Ethics Committee of Faculty of Medicine, Srinakharinwirot University, Thailand (Approval No. COA/AE-012-2566).
Informed Consent
Not applicable.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of the publisher, the editors and the reviewers. This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
Use of Artificial Intelligence-assisted Tools
The authors declare that they have not used artificial intelligence (AI)-tools for the writing and editing of the manuscript, and no images were manipulated using AI.
