Abstract
Background
Plants from the genus “Terminalia” are well known for their various pharmacological and medicinal properties. Terminalia bellirica is considered a vital medicinal plant from the genus Terminalia. It has been reported for its astringent, antipyretic, and purgative effects. It is also used to treat dyspepsia, tropical pulmonary eosinophilia, allergic eruptions, bronchitis, upper respiratory tract infections, and leprosy. The present research study aimed to investigate the antidiabetic activity of T. bellirica in an experimental model of type 2 diabetes in experimental rats.
Materials and Methods
In rats, type 2 diabetes was induced by the alteration in the diet by administration of a high-fat diet for 15 days, followed by the administration of streptozotocin (35 mg/kg, i.p.). The experimental animals were given 500 and 1,000 mg/kg of T. bellirica aqueous fruit extract for six weeks. Liver enzymes and lipid parameters, homeostatic model assessment–insulin resistance, insulin sensitivity index, glycohemoglobin, and oral glucose tolerance tests were performed at the end of the study. Histopathology and immunohistochemical analysis for the SIRT1 expression of pancreatic tissue were also performed.
Results
The selected doses of the aqueous extract of T. bellirica (500 and 1,000 mg/kg) significantly reduced blood glucose (p < 0.05). The 1,000 mg/kg dose of T. bellirica notably decreased glucose tolerance (p < 0.001) in experimental animals. A high dose of T. bellirica significantly reduced resistance (p < 0.001). The lipid profile was improved significantly (p < 0.01) at both the selected dose levels. Glycohemoglobin concentration was significantly reduced in experimental animals (p < 0.001) at both dose levels. The extract treatment increased the expression of SIRT1 in the pancreas.
Conclusion
The results of the present study indicate that the extract has significant effects on the management of type 2 diabetes.
Keywords
Introduction
Diabetes is one of the major metabolic disorders prevailing at the global level because of lifestyle changes (International Diabetes Federation Atlas, 2019). As per the data mentioned in the factsheet by the International Diabetes Federation (IDF) for Southeast Asia, 1 in 11 adults has diabetes, and 1 in 5 with type 2 diabetes in the world belongs to Southeast Asia (International Diabetes Federation, 2019). Type 2 diabetes accounts for around 90%–95% of all confirmed populations of diabetes. The life expectancy of a person who has diabetes (type 2) is reduced by 15 years primarily because of the development of complications at the vascular level (Gillies et al., 2007). Many medicinal plants have been reported to have antidiabetic effects. T. bellirica is one of the important medicinal plants. The important class of constituents present in T. bellirica fruit is tannins. They are a group of phenolic compounds which is clearly distinguished from the other secondary phenolic metabolites in their biological activity and chemical reactivities (Hagerman, 2002). Reportedly, tannins possess an antidiabetic and antioxidant effect. T. bellirica has reported pharmacological activities such as anti-cancer, antioxidant, anti-bacterial, wound healing, hepatoprotective potential, and anti-inflammatory (Hagerman, 2002; Torres-León et al., 2017). T. bellirica has been reported for its antidiabetic effect in a type 1 diabetes animal model (Gupta et al., 2020). There are no scientific reports on type 2 diabetes. Therefore, the current research work was designed to study the effect of T. bellirica in high-fat, low-dose streptozotocin-induced type 2 diabetes in experimental animals.
Experimental Section
Crude Drugs and Chemicals
T. bellirica fruits were procured from the D.G. Ayurvedic Sangrah, Andheri (W), Mumbai, Maharashtra, India. The fruits were authenticated by the scientists at the Herbal Drug Administration at Agharkar Research Institute, Pune, Maharashtra, India. A voucher copy of dried fruits has been submitted to the research laboratory for further reference (Voucher No. 3/187/2018/adm. 252, December 3, 2018). Casein was purchased from Clarion Casein Limited, Gujarat, India. Streptozotocin was purchased from Sigma Aldrich (St. Louis, MO, USA). DL-methionine and yeast extract powder were procured from Molychem, India. Cholesterol was purchased from Loba Chemie Pvt. Ltd, India, and sodium chloride was procured from S.D. Fine Chemicals, Mumbai, Maharashtra, India. Ellagic acid was purchased from Merck, and gallic acid was purchased from Sigma Aldrich. Diagnostic kits were purchased from Transasia Biomedicals Ltd, India. A rat insulin ELISA kit was procured from Abbkine, USA. SIRT1 primary antibody was procured from Santa Cruz Biotech, USA.
Preparation of an Aqueous Extract of Fruits of T. bellirica
Kernels of dried fruits of T. bellirica were removed, powdered, and used for the preparation of an aqueous extract by using the double maceration technique. Powdered fruits (500 g) were macerated with 5,000 mL of distilled water for seven days.
At the end of the seventh day, the extract was filtered through muslin cloth, followed by filter paper, and the filtrate was stored in the refrigerator till further use. The marc was again kept for maceration with fresh distilled water for seven days. The extract was filtered through muslin cloth, followed by filter cloth after seven days. The filtrate obtained in this step was mixed with the filtrate obtained in the first step. The extract was concentrated in a water bath and was stored in a refrigerator until further use (Kulkarni et al., 2013).
Standardization of the Aqueous Extract of Fruits of T. bellirica by HPTLC
The ellagic acid and gallic acid contents in the prepared aqueous extract of T. bellirica were determined by an HPTLC method. The chromatographic analysis was carried out on Camag HPTLC (Muttenz, Switzerland) equipped with visionCATS HPTLC Software, CAMAG Linomat V applicator, CAMAG Hamilton Micro syringe (100 µL), CAMAG TLC Scanner 4 (4.6 mm × 25 cm, 5 µm), CAMAG UV Cabinet (254 & 366 nm) CAMAG Twin Trough Chambers (20 × 10 cm). The separation was performed using a solvent system of methanol:water:formic acid (4:6:0.5) v/v/v. The stationary phase was Merck, HPTLC plate silica gel 60 RP-18. The scanning was done at 254 nm.
Preparation of Standard Solutions
The standard solutions of ellagic acid and gallic acid of 100 µg/mL were prepared in methanol and applied to the HPTLC system.
Preparation of Extract Solution
To determine the content of ellagic acid and gallic acid present in the aqueous extract of T. bellirica, 50 mg of extract was weighed and diluted with 5 mL of methanol. From the resulting solution, 1 mL was diluted to 10 mL of methanol. The prepared solution was used for further analysis. The above-prepared standard and extract solutions were applied to the HPTLC system. The peak area and retention factor of tannins in the extract were compared for standardization of the contents of ellagic acid and gallic acid in the extract.
Experimental Animals
From the National Institute of Biosciences, Pune, Maharashtra, India, male Sprague Dawley (SD) rats (160–170 g) were procured. The male SD rats were housed in the central animal house at relative humidity (75 ± 5%), standard temperature (22 ± 2°C), and a light/dark cycle of 12 hours during the study. The housed rats were given a normal pellet diet obtained from Nutrimix Laboratory Animal Feed, Maharashtra, India, along with purified water ad libitum. The Institutional Animal Ethics Committee approved the research protocol-wide number CPCSEA/IAEC/P-25/2020 dated 29.02.2020. The normal diet (chow) consists of 5% fats, 53% carbohydrates, and 23% protein. The composition of the high-fat diet was as per Srinivasan et al. (2005).
Induction of Type 2 Diabetes
Streptozotocin (35 mg/kg, i.p. in 0.1 M cold citrate buffer, pH 4.4) was given to experimental animals after 15 days of diet with a high-fat content. The high-fat diet was continued till the end of the study. Experimental animals with blood glucose levels above 250 mg/dl were considered for study. The diabetic animals were divided into four groups. One group was kept as a diabetic control group, and animals in this group received water. Two groups containing diabetic animals received an aqueous extract of T. bellirica fruits 500 and 1,000 mg/kg, respectively, by the oral route. Another group containing diabetic animals received glipizide at a dose of 5 mg/kg by the oral route. One group of normal animals was also part of the study, and animals in this group received water. All treatment was given for six weeks (Kulkarni & Garud, 2016; Srinivasan et al., 2005).
Parameters Measured
Body Weight
The body weight of the animals from different treatment groups was recorded at the end of the study.
Determination of Plasma Glucose, Lipids, and Glycohemoglobin Content
Blood samples of animals were collected at the end of the study using ethylene diamine tetra acetic acid (EDTA) as an anticoagulant. Plasma glucose and lipid markers such as high-density lipoproteins (HDL), triglycerides (TG), total cholesterol (TC), and low-density lipoproteins (LDL) were determined using commercial diagnostic kits supplied by Transasia Biomedicals Ltd., India, by Biochemistry Analyzer (Erba Chem7, Germany). Glycohemoglobin was determined in freshly collected whole blood samples using a commercially available kit (Transasia Bio-Medicals Ltd., India).
Determination of Liver Enzymes
Liver enzymes such as aspartate aminotransferase (AST) and alanine aminotransferase (ALT) were estimated using diagnostic kits supplied by Transasia Biomedicals Ltd., India.
Measurement of Glucose Tolerance and Insulin Resistance
Determination of Oral Glucose Tolerance Tests
An oral glucose tolerance test (OGTT) was performed at the end of the 42nd day. After eight hours of fasting, the blood sample was withdrawn by retro-orbital plexus to obtain baseline blood glucose levels (0 min). Subsequently, all the rats were administered with 20% D-glucose (2 g/kg body weight) solution orally. Blood was withdrawn at 0, 30, 60, 90, and 120 min from all the groups, and the plasma glucose level was measured. The remaining plasma of 0 min was stored at −80℃ for measurement of plasma insulin level (Oza & Kulkarni, 2018).
Measurement of Plasma Insulin, Insulin Sensitivity Index, and Homeostatic Model Assessment–Insulin Resistance
Insulin level was determined in rat plasma samples using enzyme-linked immunosorbent assay (ELISA) kits (Abbkine, USA). Homeostatic model assessment-insulin resistance (HOMA-IR) and insulin sensitivity index (ISI) were calculated using the following formula: ISI = Ln (1/FINS × F.B.G.)
HOMA-IR = (glucose × Insulin)/22.5
Here, the concentration of insulin is expressed in mIU/L and that of glucose in mmol/L.
Determination of Oxidative Stress Parameters
At the end of the study, animals from different treatment groups were humanely sacrificed, and the pancreas was isolated to determine oxidative stress parameters, histopathological studies, and immunohistochemistry.
For oxidative stress parameters, the tissue homogenate was prepared in 10% w/v phosphate buffer (50 mM, pH 7.4). Oxidative stress parameters such as superoxide dismutase (SOD), catalase (CAT), reduced glutathione (GSH), and malondialdehyde (MDA) were estimated in pancreatic tissue. MDA and GSH levels were measured in the homogenate of pancreatic tissue. CAT was evaluated in the post-nuclear fraction of pancreatic homogenate, whereas SOD was estimated in the post-mitochondrial fraction of homogenate (Ellman, 1959; Ohkawa et al., 1979; Paoletti et al., 1990).
Histopathology of Pancreatic Tissue
For histopathology, pancreatic tissue was fixed in 10% buffered formalin. Formalin-fixed tissues of the pancreas were trimmed longitudinally and routinely processed. Tissue processing was done to dehydrate in ascending grades of alcohol, clearing in xylene and embedding in paraffin wax. Paraffin-wax-embedded tissue blocks were sectioned into 3-mm thickness with a rotary microtome. All the slides of the pancreas were stained with hematoxylin and eosin (H & E) stain as per a previously reported method for the determination of tissue necrosis under a Motic microscope (Oza & Kulkarni, 2018).
Immunohistochemistry
IHC staining of pancreatic tissue was carried out to determine SIRT1 expression, as reported earlier (Oza & Kulkarni, 2018).
Statistical Analysis
The obtained data are expressed as mean ± SEM (n = 6). GraphPad Prism ver. 5.00 for Windows was used to evaluate the results. Significant differences between the study groups were assessed by one-way analysis of variance (ANOVA) tests followed by Bonferroni’s multiple comparison tests to determine the significance level. A p value less than 0.05 was considered significant.
Results
Standardization of ellagic acid and gallic acid contents in the aqueous extract of fruits of T. bellirica was done by HPTLC.
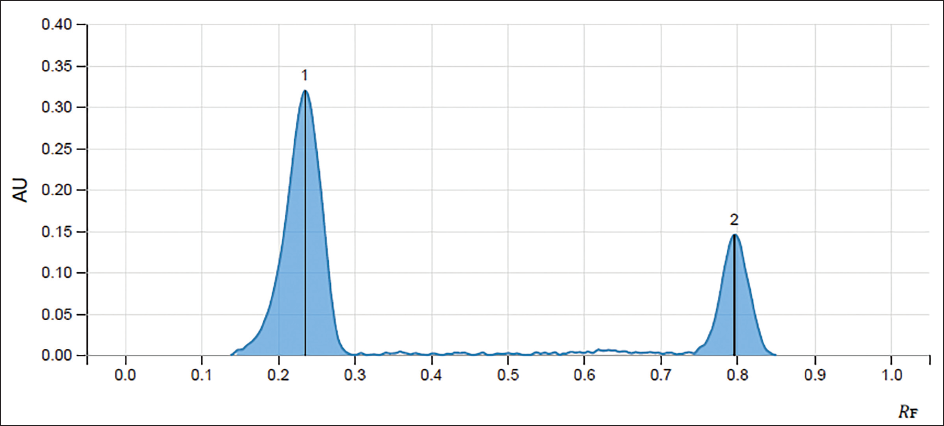
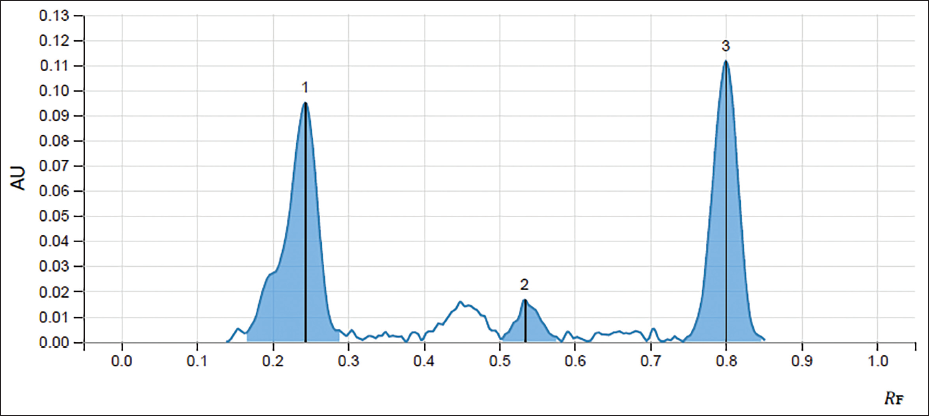
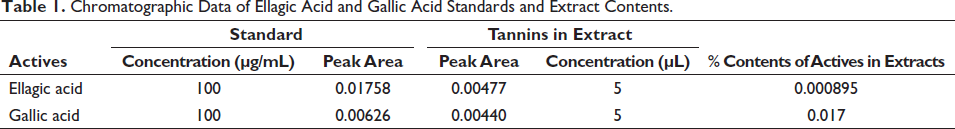
The retention factor (Rf) of standards of ellagic acid and gallic acid was obtained at 0.235 and 0.797, respectively, whereas the peaks of ellagic acid and gallic acid in the extract were obtained at a similar retention factor as that of standards, confirming the presence of desired tannins in the extract. The percentage content of ellagic acid and gallic acid in the T. bellirica aqueous extract was found to be 0.00089531% and 0.017%, respectively (Figures 1–3 and Table 1).
Chromatogram of Standard Ellagic Acid (1) and Gallic Acid (2).
Chromatogram of the Aqueous Extract of Fruits of T. bellirica Showing the Presence of Ellagic Acid (1) and Gallic Acid (3).
HPTLC Plate Under a Visualizer Showing the Presence of Ellagic and Gallic Acid in the Standard and in the Extract.
Chromatographic Data of Ellagic Acid and Gallic Acid Standards and Extract Contents.
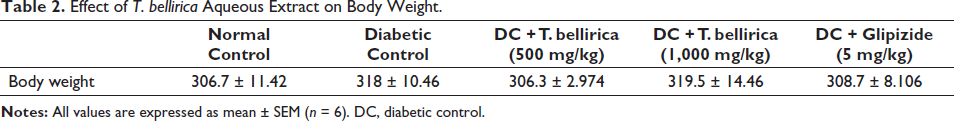
Body Weight
No significant differences were observed in normal control, diabetic control, and treatment group animals (Table 2).
Effect of T. bellirica Aqueous Extract on Body Weight.
Determination of Plasma Glucose and Lipid Parameters
Plasma Glucose
A significantly high plasma glucose level was reported in the diabetic control group compared with normal animals (p < 0.001). T. bellirica aqueous extract-treated animals showed a significant decrease in PG at both doses. At the end of the study, PG levels at both the selected doses are comparable with that of the standard glipizide (Table 3).
Effect of T. bellirica Extract on Plasma Glucose.

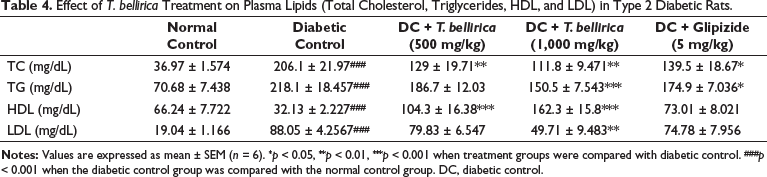
Lipid Parameters
Elevated levels (p < 0.001) of LDL-C, total cholesterol, and triglycerides and reduced levels (p < 0.001) of plasma HDL were reported in diabetic experimental animals when compared with the normal control rats. T. bellirica-treated animals showed a significant decrease in plasma LDL-C, TG, and TC at higher dose levels when compared with the disease-control rats. T. bellirica-treated animals showed improvement in the HDL-C level significantly (p < 0.001) when compared with the disease-control rats (Table 4).
Effect of T. bellirica Treatment on Plasma Lipids (Total Cholesterol, Triglycerides, HDL, and LDL) in Type 2 Diabetic Rats.
Effect of T. bellirica on Liver Enzymes
Diabetic control animals showed a significant increase in the levels of AST and ALT when compared with normal animals (p < 0.001). In the extract treatment group, the AST level significantly (p < 0.05) reduced with the higher dose when compared with the diabetic control group. The aqueous extract also showed a significant reduction in ALT at 500 mg/kg (p < 0.05) and 1,000 mg/kg (p < 0.001) dose levels when compared with diabetic control animals. Glipizide treatment significantly decreased the level of ALT when compared with disease-control animals (p < 0.01) (Table 5).
Effect of T. bellirica on Liver Enzymes (AST and ALT) in Type 2 Diabetic Rats.

Effect of T. bellirica on Glycohemoglobin
Glycohemoglobin concentration was significantly increased in the untreated diabetic group when compared with animals in the normal group. T. bellirica reduced glycohemoglobin levels significantly (p < 0.001) at 500 and 1,000 mg/kg when compared with the diabetic control group (Table 6).
Effect of T. bellirica Treatment on HbA1c (%Hb) in Type 2 Diabetic Rats.

Determination of Glucose Tolerance and Insulin Resistance Measurement
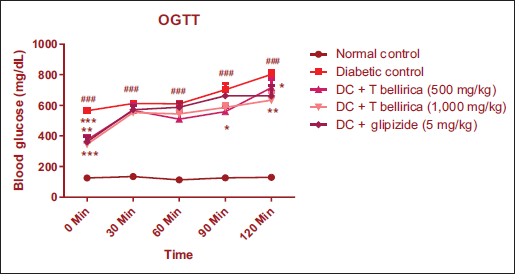
Effect of T. bellirica on Oral Glucose Tolerance Tests
OGTT was performed at the end of the study. The plasma glucose level was found to remain significantly high in diabetic animals (*p < 0.05, **p < 0.01, and ***p < 0.001), while diabetic animals treated with an aqueous extract of T. bellirica significantly improved glucose tolerance when compared with untreated diabetic animals (Figure 4).
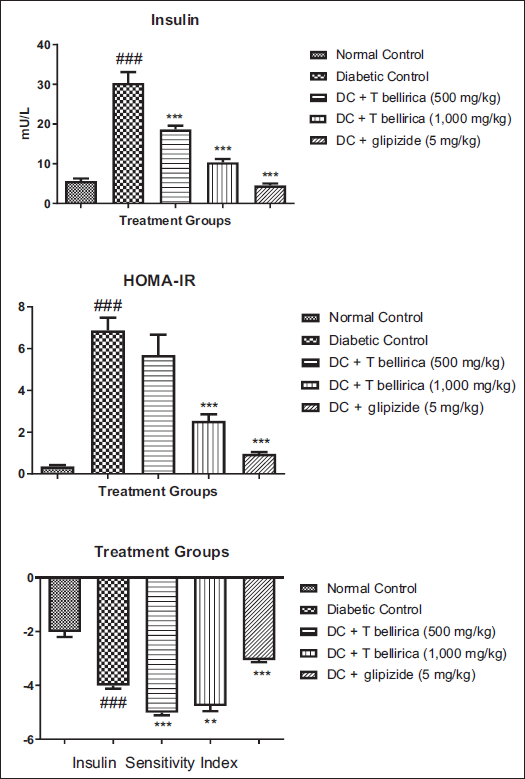
Measurement of Plasma Insulin, Insulin Sensitivity Index, and Homeostatic Model Assessment–Insulin Resistance
Untreated diabetic animals showed a significant increase in the plasma insulin level at the end of the study of diabetic induction when compared with normal animals. At the same time, diabetic animals treated with the T. bellirica aqueous extract showed significant reduction (p < 0.001) in plasma insulin levels. The treatment also showed a significant reduction in insulin resistance and improved insulin sensitivity at all selected doses (Figure 5).
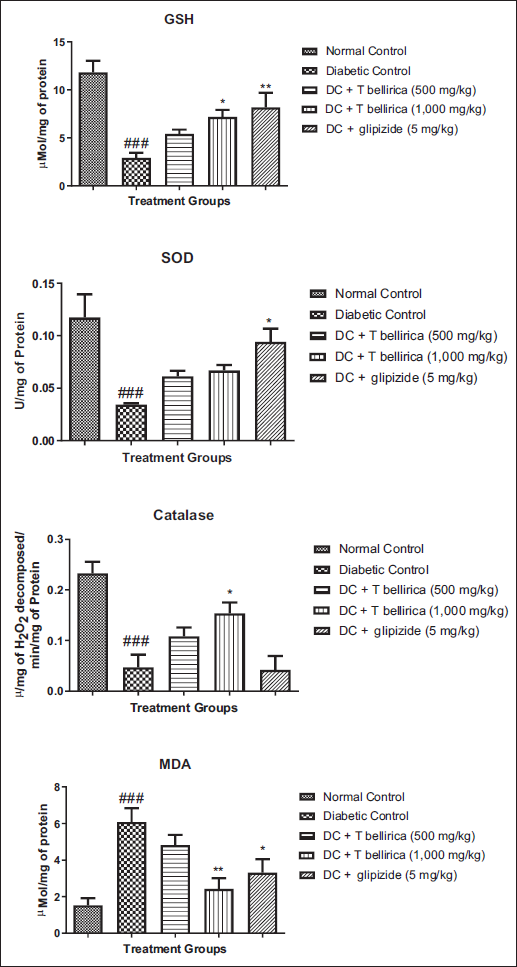
Effect of T. bellirica on Oxidative Stress Parameters
Animals in the diabetic group showed a significant increase in MDA and a decrease in SOD, CAT, and GSH when compared with animals in the normal control group. Treatment with an aqueous extract of T. bellirica at a dose of 1,000 mg/kg significantly increased (p < 0.05) GSH and CAT when compared to animals in the diabetic control group (Figure 6).
Effect of T. bellirica on Histopathology of Pancreatic Tissue
Pancreatic tissue of the diabetic animals showed hyperplastic acini in the exocrine region while degeneration and atrophy of islets of Langerhans as compared to normal animals’ pancreatic tissue. Adipose tissue deposition at the acini lobe, hyperplastic acini, and necrosis of peri-parenchymal adipose tissue was observed in diabetic animals treated with 1,000 mg/kg of aqueous extract. The severity of the lesions is shown in Figure 7.
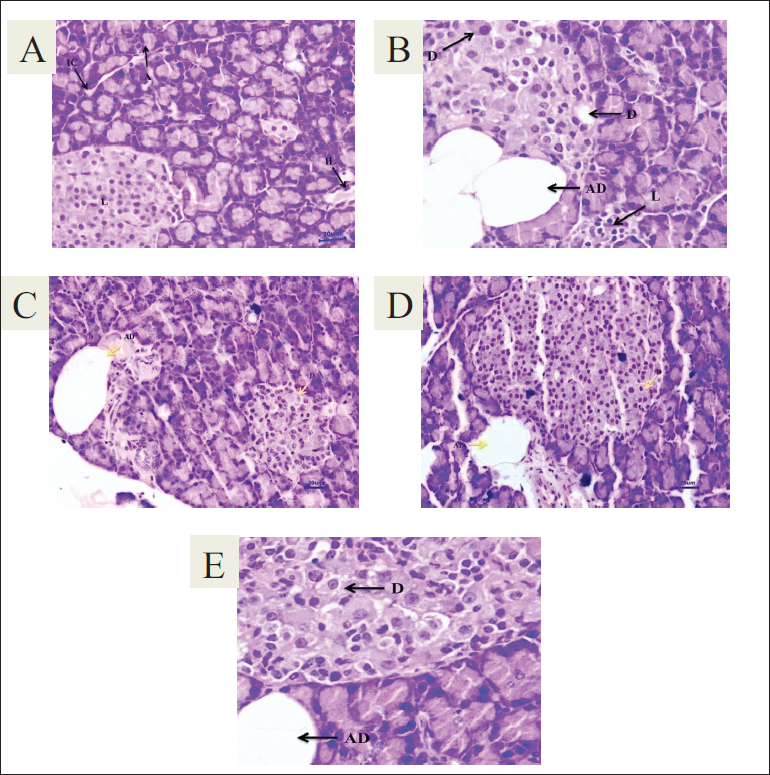
(A) Normal control, (B) Diabetic control, (C) DC + T. bellirica (500 mg/kg), (D) DC + T. bellirica (1,000 mg/kg), (E) DC + Glipizide (5 mg/kg).
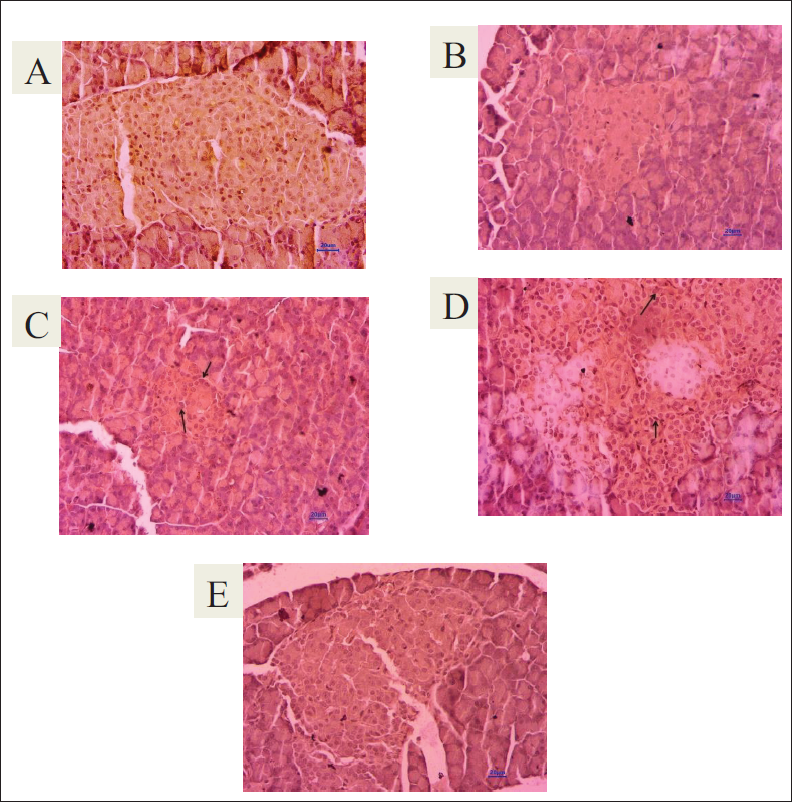
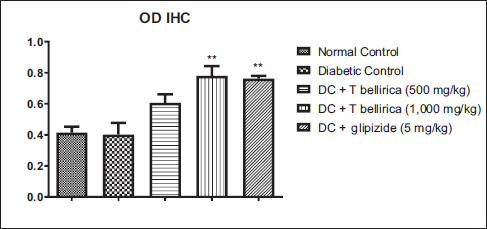
Effect of T. bellirica Extract on SIRT1 Expression in Pancreatic Tissue
Untreated diabetic animals showed a reduction in the optical density of immune-stained pancreatic tissue compared to normal animals, showing a reduction in SIRT1 expression in pancreatic tissue. Treatment with 500 and 1,000 mg/kg of the extract showed a significant increase in SIRT1 expression in pancreatic tissue (Figures 8 and 9).
Immunohistochemical Staining of the Pancreas (400×). Arrows show expression of SIRT1 in the islet of Langerhans. (A) Normal control. (B) Diabetic control. (C) DC + T. bellirica (500 mg/kg). (D) DC + T. bellirica (1,000 mg/kg). (E) DC + Glipizide (5 mg/kg).
Discussion
Pancreatic β-cells’ defective production of insulin and comparative reduction in insulin sensitivity results in type 2 diabetes mellitus. The HFD was given for a substantially extended period, resulting in obesity and insulin resistance as per the evidence in the standard literature. The content of HFD is mainly responsible for the increase in the level of free fatty acid in plasma. As a result, an increase in β-oxidation disrupts glucose metabolism and insulin action (Jornayvaz et al., 2010). To induce diabetes, streptozotocin was used in a dose-dependent manner by the alkylation of DNA, which promotes β-cell destruction. Streptozotocin in low-dose produces mild impairment in the level of secretion of insulin, which is similar to the late phase of type 2 diabetes mellitus (Zhang et al., 2008).
During the investigation, it was observed that increased body weight was reported for the rats fed with lard in the form of HFD, which is saturated fat that will accumulate in the body fat pads (Srinivasan et al., 2004). The imbalance in glycemic control showed a decrease in the body weight of the diabetic group. T. bellirica aqueous extract treatment showed improvement in body weight but did not significantly indicate its effect on body weight by controlling hyperglycemia. In type 2 diabetes mellitus, an elevation in glucose production has been reported considering improper insulin utilization and secretion, coupled with a decrease in splanchnic glucose uptake. This showed that secretion of hepatic insulin action has a vital role in the homogenous maintenance of the concentration of glucose in the body’s circulatory system (Basu et al., 2004). Our findings showed that T. bellirica aqueous extract treatment mainly controls type 2 hyperglycemia in the diabetic group because of an elevated sensitivity of insulin at the cellular level and a decrease in insulin resistance. The results from the OGTT reported that a high dose of T. bellirica also reduces glucose intolerance. The plasma insulin concentration was reportedly elevated in the diabetic group and showed hyper-insulinemic condition. In the initial stage, it has been observed that insulin secretion is regular, but tissues have not utilized it properly. In the diabetic control group, it has been observed that HOMA-IR was increased and ISI was significantly decreased in comparison with the non-treatment group, resulting in the situation of insulin resistance at the tissue levels. Insulin resistance was reduced with the treatment of T. bellirica aqueous extract, as concluded from the increase in ISI and reduction in HOMA-IR data. One of the best parameters to measure type 2 diabetic conditions is the measurement of glycohemoglobin as it gives the average concentration of blood glucose level of 4–6 weeks’ duration of an individual. In the glycated haemoglobin (HbA1c), where glucose molecule is present on the N-terminal of the valine residue of Hb, the degree of glycated Hb depends on the blood glucose level (Alqahtani et al., 2013). In the current study, glycohemoglobin content was measured at the end of the study. The excess lipid deposition occurs in skeletal muscles, liver, and beta cells of the pancreas in an obese person, which will impart insulin resistance. This will ultimately result in an abnormal lipid profile. Insulin resistance may cause a higher free fatty acid production, resulting in more secretion of TG and VLDL levels. Reduction in HDL and increase in LDL level have also been reported in hyperinsulinemia (Mooradian, 2009). In the present research study, the diabetic group showed significant improvement in HDL and a reduction in LDL, TG, and TC levels because of a reduction in insulin resistance at the cellular level. In the histopathological studies, the diabetic group showed alteration in pancreatic tissues, such as degenerative, inflammatory lesions and hyperplastic all over endocrine and exocrine pancreatic tissues. In the islet of Langerhans, necrosis was observed in diabetic animals. In most animals, in acinar lobules, fat deposition was observed. T. bellirica aqueous extract treatment results are comparable with standard drugs as they showed maximum protection to the pancreatic tissues at a higher dose level. In pancreatic tissues, T. bellirica extract showed increased expression of SIRT1. “SIRT1 is an NAD-dependent class III histone deacetylase that participates in the regulation of glucose homeostasis through regulating hepatic glucose production, lipid metabolism, insulin production, and sensitivity” (Oza & Kulkarni, 2018). It also decreases hepatic glucose production via deacetylation and activation of the AMPK kinase LKB1. It regulates glycolysis, gluconeogenesis, and fatty acid b-oxidation by activating PGC-1α transcription coactivator and stimulating insulin secretion. In the metabolism of glucose and mitochondrial function, SIRT1 is one of the important regulators. An increase in insulin sensitivity and secretion is linked with an elevation in the SIRT1 expression. A pancreatic SIRT1 immunohistochemical study indicates that SIRT1 expression in the diabetic group was remarkably decreased in comparison with normal groups. Also, it showed considerable SIRT1 expression enhancement in pancreatic tissues.
Conclusion
This study attributes antidiabetic effect of the aqueous extract of T. bellirica, in SD rats fed a high-fat diet and low-dose streptozotocin-induced type 2 diabetes. The results would be more prominent in experimental rats if the isolated phytoconstituents would be used for antidiabetic study.
Abbreviations
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Statement of Informed Consent and Ethical Approval
The Institutional Animal Ethics Committee approved the research protocol-wide number CPCSEA/IAEC/P-25/2020 dated February 29, 2020.
