Abstract
The current study aimed to investigate the therapeutic effects of 5,7-dihydroxy-6-oxoheptadecanoic acid (DHA) from Tiliacora triandra on rat models of type 2 diabetes mellitus (T2DM). T2DM was induced with a combination of high-fat diet/streptozotocin (HFD/STZ), and diabetic rats were treated with DHA (25 mg/kg) for 30 days. The body weight, fasting blood glucose (FBG), serum, and liver biochemical parameters, as well as histological evaluations of the liver and pancreas, were evaluated. Diabetic rats displayed a significant increase in FBG, serum lipid profiles (triglycerides, total cholesterol, and low-density lipoprotein cholesterol), liver function enzymes (aspartate transaminase, alkaline phosphatase, and alanine transaminase), creatinine, liver malondialdehyde (MDA), and myeloperoxidase (MPO) contents. Furthermore, insulin level and liver antioxidant enzyme activities (catalase [CAT], superoxide dismutase [SOD], and glutathione peroxidase [GSH-Px]) were significantly reduced in the diabetic rats. Whereas, treatment with DHA significantly reduced FBG, serum lipids, liver function enzymes, serum creatinine, liver MDA, and MPO contents. In addition, treatment with DHA significantly increased serum insulin level and liver SOD, CAT, and GSH-Px activities. In addition, DHA alleviated histopathological changes in the pancreas and liver caused by T2DM. These results portray the antidiabetic and antioxidative properties of DHA and can be considered as a potential treatment for T2DM.
Diabetes mellitus is a lifelong group of metabolic diseases that has an unpleasant impact on the quality of life of the patient, caregivers, and the general society. Around 463 million adults have diabetes at present, and it has been projected that 700 million adults will have the disease by 2045. Furthermore, approximately 4.2 million people died from diabetes in 2019. All these alarming figures have earned diabetes as the third most common chronic and life-threatening disease after cancer and cardiovascular disease. 1,2 Type 2 diabetes mellitus (T2DM) is the most common type of diabetes, and it accounts for almost 90% of the global diabetic population. T2DM is associated with severe complications that affect the kidney, brain, nerves, eyes, and reproductive organs. 2 -4 T2DM is characterized by pancreatic β-cell impairment, insulin resistance (IR), and reduced insulin secretion resulting in chronic elevation of blood glucose level. Quite a number of factors, such as unhealthy diet, sedentary lifestyle, as well as genetic predisposition, have been determined to play a significant role in the development of the disease. 5 -7
Oral antidiabetic drugs, such as biguanides, sulfonylureas, meglitinides, thiazolidinediones, dipeptidyl peptidase IV inhibitors, glucagon-like peptide-1 receptor agonists, and α-glucosidase inhibitors, are the most common antidiabetic drugs available in the market. These drugs primarily function by curtailing severe hyperglycemia and enhancing insulin sensitization through various mechanistic pathways. 8,9 However, the success stories of these synthetic antidiabetic agents have been limited due to several side effects, including hypoglycemia, excessive weight gain, gastrointestinal discomforts, lactic, acidosis among others. Furthermore, these drugs are not broad in their therapeutic approach. 9 Therefore, a replacement therapy that significantly eases off these drawbacks is urgently needed. Medicinal plants and their bioactive ingredients have been extensively reported as effective antidiabetic agents, due to their multiple therapeutic actions and their ability to attenuate the underlying pathological mechanisms involved in T2DM. In addition, to the safety profile, the world health organization has also advocated for the use of medicinal plants for the management of several diseases including diabetes. 10,11
Tiliacora triandra (family Menispermaceae) is a common vegetable that is also traditionally used as a herbal medicine in Southeast Asia, and it has several pharmacological effects such as neuroprotection, antimalarial, and antibacterial effects. 12 Previous studies have shown that the crude ethanol extract of the plant displayed antidiabetic properties, 13 -15 without any report on the effect of the active constituents of the plant. 5,7-Dihydroxy-6-oxoheptadecanoic acid (DHA) is one of the major bioactive fatty acid derivative isolated from the aerial parts of T. triandra. A previous study indicated that DHA displayed a significant inhibitory effect against alphaamylase and alphaglucosidase enzymes. 16 Due to the seemingly potent in vitro antidiabetic effect of DHA, 16 we envisaged that DHA may also have significant antidiabetic activity in vivo. As such, this present study investigated the effect of DHA on hyperglycemia, dyslipidemia and other abnormalities in high-fat diet/streptozotocin (HFD/STZ)-induced diabetic rats.
Results and Discussion
A growing number of studies have demonstrated that the combination of HFD with fructose along with a low dose of STZ mimics the metabolic disturbances observed in human T2DM, and this model has been widely used for inducing T2DM in animal models. 16,17 Consequently, the rats treated with HFD/HF-STZ showed obvious and characteristic signs of T2DM, including polydipsia, polyphagia, hyperglycemia, body weight loss, marred insulin secretion, and glucose intolerance. Due to insufficient insulin levels experienced in T2DM, there is a decline in the supply of energy to cells because of loss in the utilization of glucose. The kidney excretes additional amount of water to dilute the excessive concentration of glucose in the blood which ultimately results in loss of water creating polydipsia and frequent urination. 18 Previous studies have demonstrated that food and water consumptions are increased in diabetic animal models. 19,20 As shown in Table 1, the body weight gain was significantly decreased, while food and water intake in the diabetic control group (DCG) was significantly higher compared with the normal control group (NCG) (Table 1). Whereas, diabetic rats administered with a daily dose of 25 mg/kg DHA showed a significant increase in body weight gain and marked decrease in their consumption of food and water when compared with DCG (P < 0.05).
Effect of DHA on Body Weight Gain, Food, and Water Intake in Diabetic Rats.

DCG, diabetic control group; D + DHA, diabetic rats treated with 25 mg/kg of DHA; DHA, 5,7-dihydroxy-6-oxoheptadecanoic acid; NCG, normal control group.
Data were expressed as mean ± SD (n = 6).
a P < 0.05 when compared with NCG.
b P < 0.05 when compared with DCG.
IR is one of the major hallmarks of T2DM, and it significantly contributes to the development and severity of diabetic complications. The secretion of insulin by the pancreatic beta cells is very paramount for the maintenance of blood glucose level, and a number of antidiabetic drugs’ primary target is reducing blood glucose level by enhancing insulin secretion. 21 Table 2 shows the effect of DHA on fasting blood glucose (FBG) levels at different periods during the study. The diabetic rats showed a significant and continuous increase in FBG level when compared with the non-diabetic rats (normal control group [NCG]). In the DHA-treated diabetic rats, we observed an obvious decrease in FBG in comparison to the DCG (P < 0.05). Furthermore, the effect of DHA on the intraperitoneal glucose tolerance test (IPGTT) at different time intervals (0, 30, 60, and 120 minutes) is shown in Table 3. Significant increase in the blood glucose level was observed at all time points (0 minutes = 506.8 mg/dL; 30 minutes = 567.4 mg/dL; 60 minutes = 600 mg/dL; 120 minutes = 601.0 mg/dL) in DCG when compared with NCG (0 minutes = 131.6 mg/dL; 30 minutes = 175.2 mg/dL; 60 minutes = 152.4 mg/dL; 120 minutes = 117.6 mg/dL). In the DHA-treated diabetic group, the blood glucose level was observed to be markedly decreased at all time intervals when compared with DCG (0 minutes = 114.5 mg/dL; 30 minutes = 212.25 mg/dL; 60 minutes = 187.0 mg/dL; 120 minutes = 128.5 mg/dL) (Table 3).
Effect of DHA on Fasting Blood Glucose Level in Diabetic Rats.
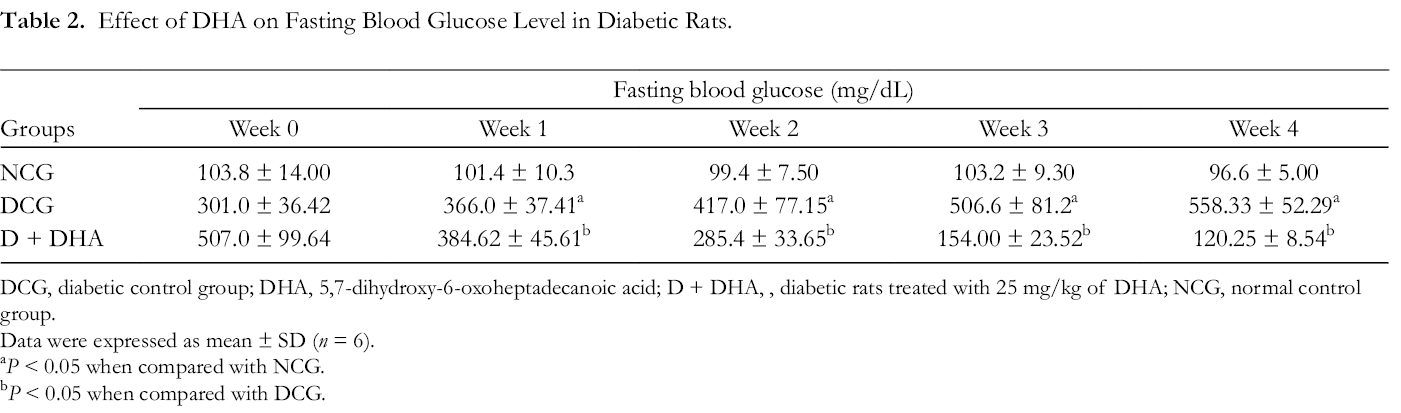
DCG, diabetic control group; DHA, 5,7-dihydroxy-6-oxoheptadecanoic acid; D + DHA, , diabetic rats treated with 25 mg/kg of DHA; NCG, normal control group.
Data were expressed as mean ± SD (n = 6).
a P < 0.05 when compared with NCG.
b P < 0.05 when compared with DCG.
Effect of DHA on Intraperitoneal Glucose Tolerance Test in Diabetic Rats.

DCG, diabetic control group; DHA, 5,7, dihydroxy-6-oxoheptadecanoic acid; D + DHA, diabetic rats treated with 25 mg/kg of DHA; NCG, normal control group.
Data were expressed as mean ± SD (n = 6).
a P < 0.05 when compared with NCG.
b P < 0.05 when compared with DCG.
Dyslipidemia is a very common feature associated with T2DM, and it is evidently characterized by aberrant decline in serum high-density lipoprotein cholesterol (HDL-C), increase in serum total cholesterol (TC), triglyceride (TG), and low-density lipoprotein cholesterol (LDL-C), which predisposes diabetic patients to the risk of cardio-cerebrovascular and coronary heart diseases. 22,23 In T2DM, the reduction in lipoprotein lipase activity involved in the hydrolysis of triglycerides due to IR enhances hypertriglyceridemia in diabetic patients. 24 The liver is a very important organ in the body, and it is involved in the metabolism of protein, carbohydrate, fats, and glucose homeostasis, and it is also one of the organs affected by T2DM. A number of liver aberrations, such as liver fibrosis, lipid accumulation, deposition of glycogen, non-alcoholic fatty liver disease, cirrhosis, and elevated levels of liver function enzymes, have been associated with T2DM. In addition, hyperglycemia destroys liver hepatocytes and also enhances the infiltration of inflammatory cells into the liver. 25 -27 The serum levels of TG (100.0 mg/dL), TC (97.78 mg/dL), LDL-C (69.67 mg/dL), aspartate transaminase (AST, 836.0 U/L), alkaline phosphatase (ALP, 235.0 U/L), alanine transaminase (ALT, 330 U/L), blood urea nitrogen (BUN, 41.25 mg/dL), glycated hemoglobin (Hb1Ac, 5.68%), total bilirubin (0.45 g/dL), and globulin (2.44 g/dL) were significantly increased in the DCG when compared with the NCG (Table 4). In addition, insulin (3.49 µIU/mL), total protein (4.97 g/dL), and albumin (2.44 g/dL) levels were observed to be reduced in the DCG when compared with the NCG. Treatment with DHA resulted in an obvious decrease in the levels of all these altered biochemical parameters in the treated diabetic rats when compared with the untreated diabetic rats. Furthermore, DHA increased insulin, total protein, and albumin levels in comparison with the DCG (Table 4).
Effect of DHA on Serum Biochemical Parameters in Diabetic Rats.
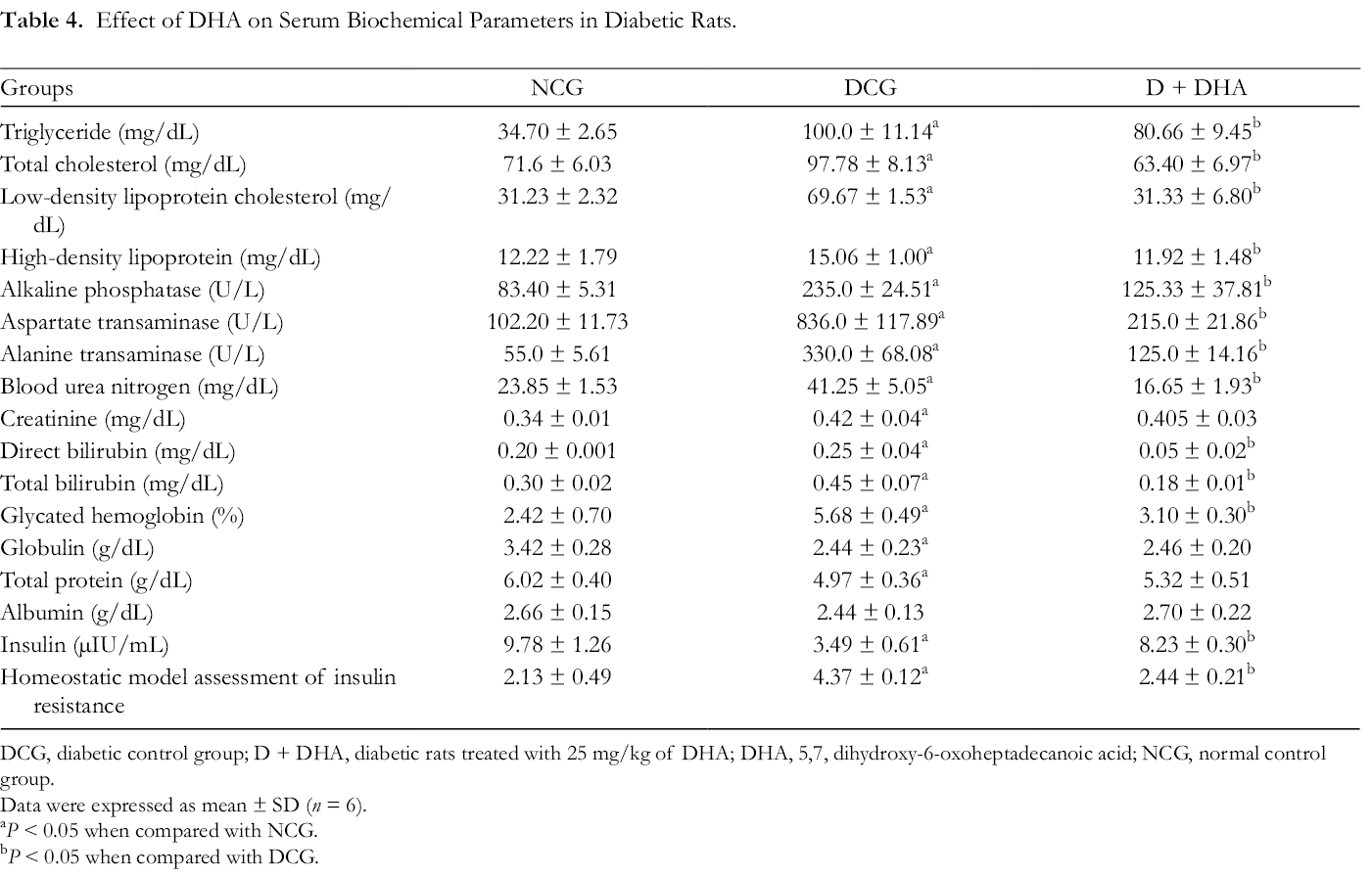
DCG, diabetic control group; D + DHA, diabetic rats treated with 25 mg/kg of DHA; DHA, 5,7, dihydroxy-6-oxoheptadecanoic acid; NCG, normal control group.
Data were expressed as mean ± SD (n = 6).
a P < 0.05 when compared with NCG.
b P < 0.05 when compared with DCG.
Accumulating evidences from several studies have continued to support the striking relation between hyperglycemia, T2DM, and oxidative stress. Hyperglycemia can initiate several oxidative stress-related apoptotic pathways which cause damages to several organs in the body. The most common evidence of diabetes-induced oxidative stress is visualized as an increased level of lipid peroxidation and a decline in antioxidant enzyme activities, including superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px). 28,29 Malondialdehyde (MDA), a product of lipid peroxidation is an important biomarker that indicates the extent of oxidative damage. On the other hand, SOD and CAT are enzymatic antioxidants that living cells employ to counteract the damaging effect of oxidative stress and free radicals. SOD and CAT inactivates and scavenges free radicals by catalyzing the removal of superoxide radicals and eliminating harmful metabolites in the living cells. 29,30 In the DCG rats, the activities of SOD, CAT, and GSH-Px in the liver tissues were significantly decreased, while MDA and myeloperoxidase (MPO) levels were markedly increased when compared with the NCG (P < 0.05). Whereas, the administration of DHA restored the activities of liver antioxidant enzymes and reduced MDA and MPO levels toward the level observed in the NCG rats (Table 5). These results were coherent with findings of other researchers. 28,31
Effect of DHA on Liver Antioxidant Enzymes Activity and Lipid Peroxidation in Diabetic Rats.

CAT, catalase; DCG, diabetic control group; GSH-Px, glutathione peroxidase; MDA, malondialdehyde; MPO, myeloperoxidase; D + DHA, diabetic rats treated with 25 mg/kg of DHA; DHA, 5,7, dihydroxy-6-oxoheptadecanoicacid; NCG, normal control group.
Data were expressed as mean ± SD (n = 6).
a P < 0.05 when compared with NCG.
b P < 0.05 when compared with DCG.
The pancreatic β-cells play a vital role in insulin secretion and blood glucose regulation because they are specialized cells that produce insulin needed for proper glucose homeostasis. Malfunctioning of these cells can result in impairment of glucose metabolism and diabetes. 32 As shown in Figure 1, the hematoxylin and eosin (H&E) staining pictures of the pancreas tissues of diabetic rats showed gross degeneration, including reduced pancreatic beta cell mass and deterioration of pancreatic islets (Figure 1(B)), when compared with the normal control (Figure 1(A)). The DHA-treated group showed marked regeneration and reinstatement of pancreatic beta cell islets to almost normal architecture (Figure 1(C)). Furthermore, the histology of the liver of the untreated diabetic rats (DCG) showed distorted architecture, lipid accumulation, fatty degeneration in hepatocytes, and degeneration of the liver vacuole, while the histology of the liver tissues of the DHA-treated group showed attenuation of the liver architecture (Figure 2(A)–(C)).

Effect of DHA on the pancreas of diabetic rats (magnification: 400×). Arrows indicate the size of the pancreatic beta cell. DCG, diabetic control group; D + DHA, diabetic rats treated with 25 mg/kg of DHA; DHA, 5,7-dihydroxy-6-oxoheptadecanoic acid; NCG, normal control group.

Effect of DHA on the hepatic histology in diabetic rats (magnification: 400×). DCG, diabetic control group; D + DHA, diabetic rats treated with 25 mg/kg of DHA; DHA, 5,7-dihydroxy-6-oxoheptadecanoic acid; NCG, normal control group.
In conclusion, in this study, we explored and presented the efficacy of DHA on dysglycemia, dyslipidemia, and other abnormalities in diabetic rats. Our findings indicated that DHA attenuated hyperglycemia, IR, and hyperlipidemia in diabetic rats. Further, DHA also mitigated altered liver function enzymes and oxidative stress in the liver tissues. These results present a probable rationale for the use of DHA in the treatment of T2DM.
Experimental
Chemicals and Reagents
STZ and fructose were purchased from Santa Cruz Biotechnology Inc. (Dallas, TX, USA) and Sigma Aldrich (St. Louis, MO, USA), respectively. All other chemicals used were of analytical grade.
Preparation of DHA
DHA was isolated from the aerial parts (leaves and twigs) of T. triandra (Colebr.) Diels. The isolation protocol and the structural elucidation of the compound have already been previously described. 15
Experimental Animals
The animal experiment was conducted in line with the principles of laboratory animal care of the National Institute of Health (NIH publication the 8th edition, revised 1985), and approval was obtained from the Animal Ethics Committee of the Prince of Songkla University, Hat Yai, Thailand (approval number MOE 0521.11/1231). Male Sprague Dawley rats (7-week-old) (Nomura Siam International, Bangkok, Thailand) were housed in stainless steel cages in a room with environmental conditions of 23 ± 2 °C (temperature), 55 ± 5% (relative humidity), and a light/dark cycle of 12/12 hours. Before the commencement of the experiment, the animals were allowed 7 days of acclimatization, and thereafter, the rats were indiscriminately divided into normal control and experimental groups. The NCG was fed with normal diet and water only, while rats in the experimental groups were fed with a diet high in fat and fructose solution (15%) for 4 weeks. The composition of the HFD was based on the previous report. 33 Thereafter, the rats in the experimental group were administered STZ (35 mg/kg, dissolved in freshly prepared citrate buffer; pH 4.5) via intraperitoneal injection to induce diabetes. 34,35 The rats were confirmed diabetic after 3 days of injection by measuring the FBG in overnight fasted rats. Rats with FBG above 250 mg/dL were taken as diabetic and further divided into 2 groups with six rats in each group: the DCG and diabetic rat treated with DHA at a dose of 25 mg/kg (D + DHA). The treatment was administered on a daily basis and lasted for 30 days. DHA was dissolved in 1% tween 80 and orally administered. Rats in the NCG and DCG groups were administered with the vehicle (1% tween 80). Throughout the period of experiment, FBG and the body weight changes of the animals were measured on a weekly basis. For the FBG determination, blood was obtained using the tail-pricking method and measured with a glucometer (Accu Chek Performa Roche Diagnostics, Germany).
IPGTT was performed on the 30th day, 30 minutes after the last DHA, or 1% tween 80 administration; the blood glucose was determined and indicated as time zero (0 minutes), and thereafter, the rats were administered with 2 g/kg of glucose solution via the intraperitoneal route, and blood glucose level was determined at 30, 60, and 120 minutes after the injection.
After the experiment, the animals were anesthetized with sodium thiopental (150 mg/kg), and blood samples were obtained through cardiac puncture for the estimation of biochemical parameters. For the biochemical analysis, blood samples were centrifuged (2500 rmp, 4 °C, 10 minutes), and the serum obtained was used for analyzing the following parameters TG, TC, LDL-C, HDL, ALP, AST, ALT, BUN, creatinine, total protein, total bilirubin, globulin, albumin, direct bilirubin, glycated hemoglobin, and insulin level. Homeostatic model assessment of insulin resistance was calculated according to this equation:

The pancreas and liver were quickly harvested, washed with ice-cold phosphate-buffered saline, fixed in 10% buffered formalin solution, and embedded in paraffin. The paraffin-embedded sections of the pancreas and liver were stained with H&E. The stained sections were visualized under a light microscope.
Another portion of the liver tissues was homogenized in phosphate buffer to obtain 10% homogenate and centrifuged at 2500 rmp, 4 °C for 10 minutes. The supernatant obtained was used for estimating the antioxidant enzyme level (MDA, MPO, SOD, CAT, and GSH-Px) in the liver with the aid of enzyme-linked immunosorbent assay kit following the manufacturer’s instruction (Nanjing Jiancheng Bioengineering Institute, China).
Statistical Analysis
Data are presented as mean ± SD. Data analysis was performed using GraphPad Prism 7, and differences were evaluated using a one-way analysis of variance followed by Tukey’s multiple comparison test. The level of statistical significance was set at P < 0.05.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the authors disclosed receipt of financial support for the research from the Grants for a General Researcher, Annual Income Budget, Prince of Songkla University (TTM610424S; 2017-2019).
