Abstract
Since 2016/17, three disease modifying therapies for spinal muscular atrophy (SMA) have been translated into clinical practice. This has driven the implementation of newborn screening to transform health outcomes and clinical practice. SMA registries have provided important sources of data on the evolution of novel phenotypes within the therapeutic era, treatment patterns, epidemiology, genotype-phenotype correlations, care and lived experiences of people living with SMA, to enrich knowledge and learnings of the condition in this changed landscape. In this state-of-the-art review, we consider the utility and outcomes of SMA registries and evaluate their role and importance. In 2024 there are more than 35 national registries cataloguing over 8000 individuals with SMA. Additional registries are operated by advocacy groups and pharmaceutical companies, compiling data for more than 10,000 individuals with this condition. This review highlights the essential role of registries in supporting clinical trial recruitment, defining the changing incidence and prevalence of SMA in an age of reproductive carrier and newborn screening, establishing natural history data, contributing to post market drug surveillance, assessing real world clinical and cost effectiveness and capturing patient-reported outcome measures (PROMS) and experience measures (PREMS). Whilst their utility is broad, barriers to effective data curation and management are evaluated including challenges of data curation and fragmentation, quality and sharing, alongside resource constraints and sustainability. Strategies to enhance the value of registries include the imperative to collaborate across clinical research networks and the value of interoperability, enabled by standardization of data collection and entry, sharing procedures and public and patient involvement. As new phenotypes and unmet needs emerge in the post therapeutic era, registries remain central tools in generating new insights now and into the future and are poised to respond meaningfully to the priorities of individuals living with SMA.
Introduction
With the advent of newborn screening (NBS) and disease modifying therapies for spinal muscular atrophy (SMA), there has been intensified focus on clinical outcomes, phenotypes, treatment patterns and lived experiences to inform care, research and policy. Real world evidence and longer-term studies are necessary to extend findings from pivotal clinical trials, to understand the utility of paradigms of early diagnosis and use of treatments in broader, heterogenous populations, inform personalised care, harness opportunities in SMA therapeutics and identify future directions and priorities. Data curation, synthesis and evaluation remains complicated by the condition's rare disease status, the clinical, functional, and genotypic heterogeneity of the population, and variability in access to newborn screening and high-cost disease modifying therapies across the international landscape. 1
Registries provide a consistent approach to data collection and are recognised as key and effective tools for supporting translational research and improving outcomes, including quality of care, quality of life and survival. 2 With collection of accurate and high-quality data, registries enable clinical trial recruitment, epidemiological research, health service planning and post marketing drug surveillance, to inform evidence driven best care practice, support regulatory approvals of novel therapeutics based on long-term efficacy and safety data, and support clinicians, researchers, industry, the patient community and government.
In the pretreatment era, SMA registry data was considered pivotal to interrogate the natural history of the condition. 3 Data was leveraged to enable clinical trial readiness, (in parallel with research and development of disease modifying treatments), to generate evidence to inform standards of care, identify a research ready population and to understand the role of prognostic modifiers of disease severity such as SMN2 copy number. 4 At this time there was a move from laboratory-based, private databases to national neuromuscular diseases registries and internationally shared datasets. The development of new database tools and multistakeholder engagement was also considered a priority for the integration of real-world research to inform clinical best practice. 5
In 2007, the Translational Research in Europe for the Assessment and Treatment of Neuromuscular Diseases (Treat-NMD), a global academic network with a focus on advancing neuromuscular disease research was established. The consequent TREAT-NMD registry model and guidelines enabled development of a number national SMA registries, harmonised data collection, standardised common data elements and definitions. Pivotally, this model provided a framework of governance and custodianship to reduce fragmentation of data and to support interoperability. A survey in 2012 identified that the most frequent (95%) use of registry data was for clinical research and recruitment into clinical trials. 6 Expansion of their role in the post therapeutic era has included the ability to forge collaborative links across clinical and research networks, 6 to foster stakeholder engagement 7 and opportunities to participate in research for affected individuals and their families and to examine the long-term impact of newborn screening and the clinical sequalae of therapeutic interventions. 8 The development of additional therapeutics is expected to further bolster the importance of registries, thus consolidating their place in data-driven research.
The acceleration of registry development in SMA has been underpinned in part by international directives that have also sought to streamline clinical research, patient care and health planning in the field of rare diseases and elevate the status of registries as a priority. The European Organisation for Rare Diseases (EURORDIS), the National Organization for Rare Disorders (NORD) and the Canadian Organization for Rare Disorders (CORD) released a joint declaration on common principles regarding Rare Disease Patient Registries in 2012. Similarly, the first National Strategic Action Plan for Rare Diseases in Australia (2020) identified ‘research and data collaboration’ as one of three core pillars of priority in rare diseases. Aligning with this, the United Nations’ Resolution (2021) on Persons Living with Rare Diseases also acknowledged rare diseases as a global health priority and advocated for a system of global collaboration and establishment of networks to strengthen health systems dedicated to addressing rare diseases.9,10
Within the context of a new and changing proactive paradigm of diagnosis, treatment and care, a comprehensive contemporary review evaluating the current status of SMA registries is pertinent and timely. The purpose of this state-of-the-art review is to consider the most recent and relevant contributions from SMA registries to current knowledge in the disease modifying era and highlight priorities for future investigations and research in SMA using registry data.
Methods
We conducted a search of three databases, PubMed, Google Scholar, clinical trials.gov and the websites from key organisations identified in the literature from 01 January 2007 to 20 August 2024 to identify publications and research activities related to SMA and rare disease registries. We used a combination of MeSH terms and keywords, including: “spinal muscular atrophy"[All Fields], “muscular atrophy, spinal"[MeSH Terms], “5qSMA"[All Fields]. Registry-related terms encompassed “registries"[MeSH Terms], “registry"[All Fields], “registers"[All Fields]. Subsequently, we updated the search to include relevant articles published up to November 2024. This time interval was chosen to best reflect the evolution of SMA registries during a period of diagnostic and therapeutic advancement. Studies in non-English languages were translated using Google translate. Studies that described how registries were developed and organised (including governance, operations and/or data collection methodologies), and those with outputs were selected based on relevance and significance to a contemporary landscape (natural history and epidemiological studies, drug effects and side effects, original registry reports relevant to SMA). Conference extracts were excluded. A narrative synthesis was carried out, organised to characterize their scope and purpose, how data was used, identify gaps and limitations and provide recommendations for registry development in SMA, as an exemplar of data curation for other rare neurological conditions. Findings were evaluated according to the EURORDIS-NORD-CORD Joint Declaration of 10 Key Principles for Rare Disease Patient Registries (Box 1). 11
be acknowledged as a leading international priority to inform the scientific and clinical landscape in rare diseases. utilise the broadest geographical scope possible. focus on conditions or groups of conditions and not on treatment(s). be developed and have governance structures that facilitate interoperability and harmonization of data sets. be developed to contain a minimum set of Common Data Elements. be linked with biobank data. include patient and caregiver meaningful outcomes and including data directly reported by affected individuals and their families, concomitant with data derived from clinician reports. be modelled to encourage Public-Private Partnerships to facilitate implementability and sustainability. equally involve stakeholders in registry governance. be developed as a tool to serve consumers and facilitate knowledge and empowerment within rare disease communities.
Results
The literature search identified 37 registry-based publications that were included in this review, encompassing about 10,000 individuals living with SMA across more than 35 health jurisdictions.
Mapping SMA registries across the international landscape: International priority setting to inform the scientific and clinical landscape
To date, SMA registries have usually begun within health jurisdictions that have established or emerging diagnostic and treatment capabilities for the condition, and also within countries that have national directives for rare conditions, such as Singapore and Thailand. 12 This includes registries across Europe, North America, parts of Asia/Oceania and Latin America (Figure 1). National registries developed through academic endeavours exist in the Indian subcontinent as a foundation on which to develop the diagnostic and therapeutic capacity for SMA within the country, 13 whilst outputs from the registries within the Middle East are intrinsically linked with implementation of new standards of care. 14
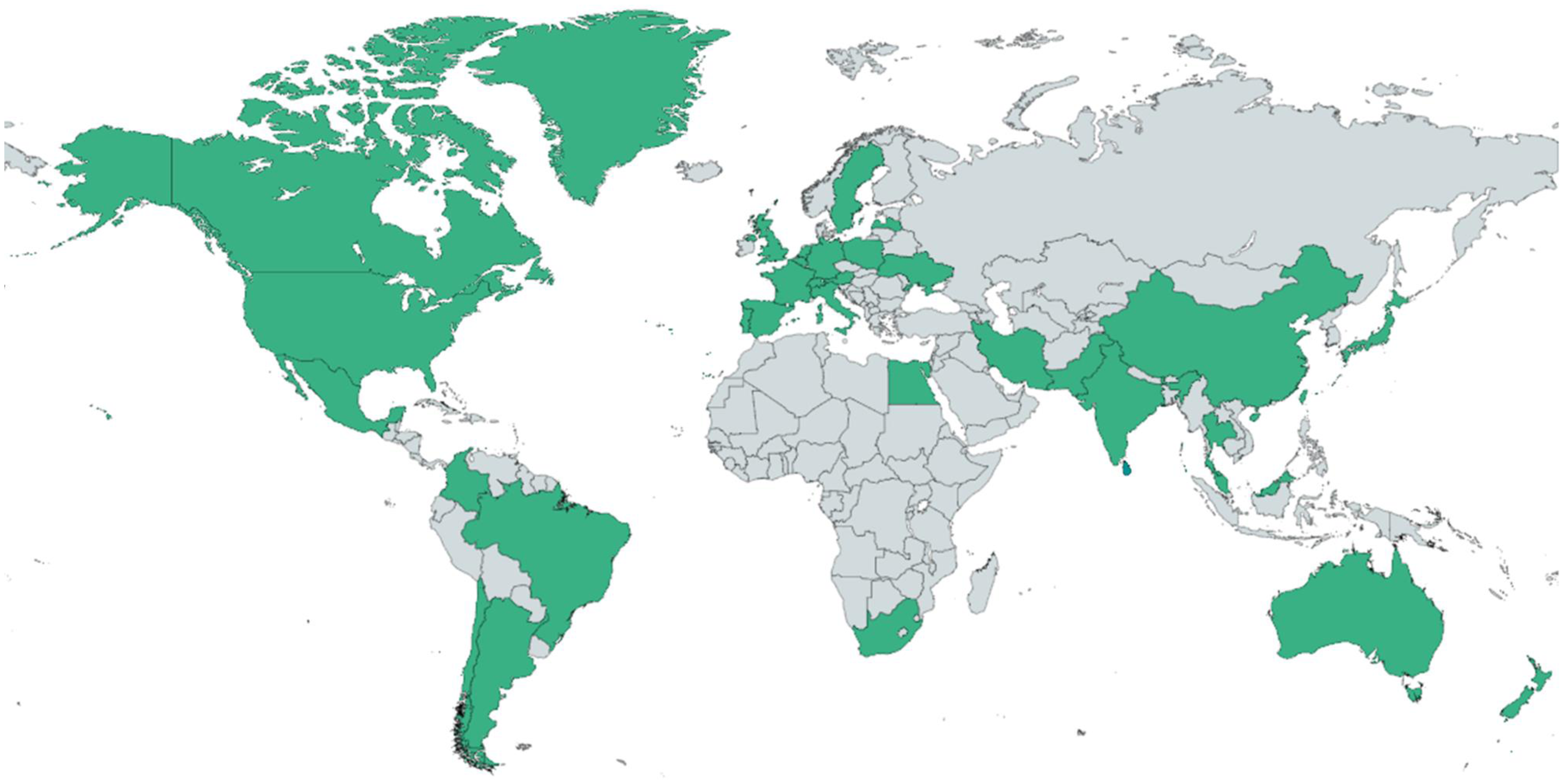
Global distribution of SMA registries, 2024.
The organisation of SMA registries: Development of governance structures to facilitate harmonisation and interoperability
Registries have evolved from less structured, simple databases, with incomplete data capture to organised and structured databases with formal data elements and data dictionary. The former, with open data fields, limited data analyses and interpretation, contrasts with a structured registry of the present day, which uses formal data elements and data dictionary. This facilitates interoperability and merging of data through use of common minimal datasets.
The development, methodology, and operations of SMA registries, whether operating individually and/or through established networks, aim to increase awareness and strengthen registry science.15–23 These may be led by academics, industry or advocacy groups and rarely by co-developed partnerships established between stakeholders across scientific, clinical, industry and consumer domains. Many are national neuromuscular diseases registries15–19,21,23,24 that include SMA, whilst others are SMA specific and extend beyond national boundaries.22,25
SMA registries employ a range of patient recruitment methods, including through healthcare professional and clinical networks, self-referral and patient organisations. Recruitment strategies have been enhanced with promotion and dissemination of information on public websites, social media, newsletters, mailouts and public forums, co-design, patient involvement and collaboration with partners. Informed consent has been obtained in writing or electronically. Data has been principally clinician reported and/or self-reported, with updates occurring during clinic or by phone. Data entry may be undertaken by registry staff, volunteers, advocacy organisations or clinicians. Registries are increasingly showing the value of engagement and participation of stakeholders in management with clear roles and responsibilities defined, for example through steering committees to supervise project progress, approve amendments and create reports. 22
A pivotal initiative in the SMA research landscape has been the Pediatric Neuromuscular Clinical Research (PNCR) Network, established in the United States, and funded by the SMA Foundation since its inception. With its aim to standardise outcome measures and create a comprehensive understanding of SMA's natural history, 26 it played a foundational role in creating frameworks for comprehensive data capture (including motor and non-motor functions, phenotype-genotype correlations) and the development of outcome measures used in seminal clinical trials.
The efforts of PNCR Network's efforts in standardising data collection are complemented by TREAT-NMD, established in 2007. The TREATNMD Global Registry Network is an example of diversity in registry organisation, function and scope and currently includes 65 members, forming a federated alliance of individual, independent, national (or regional) neuromuscular diseases registries, including SMA. Within TREAT-NMD there are more than 8524 individuals who have an SMA phenotype, of whom 7303 have a genetic diagnosis. 27 The TREAT-NMD Core Dataset supports registries to collect robust longitudinal data with the Global Data systems Oversight Committee (TGDOC) reviewing requests for data or recruitment support from the global registries network. 28 TREAT-NMD has also established the Care and Trial Site Registry (CTSR), an online self-registration database that is open to any centre internationally. 29 The data is held for the benefit of the neuromuscular community and is not ‘owned by’ or ‘affiliated with’ any one organisation. The CSTR has a core aim of providing the infrastructure for clinical trial feasibility and site selection, and as such has provided data to facilitate the opening of trial sites in six countries and recruitment of patients supported by registries in these countries. 29 Flexibility of the registry benefits users by enabling the addition of supplemental quantitative and qualitative data to the core sets from other sources such as audit activities, professional and patient questionnaires, and surveys, thereby providing a holistic and multidimensional view of neuromuscular diseases.
Patient advocacy organisations have been foundational in the organisation of SMA specific registries. Cure SMA (previously Families of SMA) founded the international SMA registry in 1986 to support research and people living with SMA. It is the largest self-reported data repository on individuals with SMA worldwide, with 9789 individuals as of 2023.7,30 In 2008 the Registry joined TREAT-NMD in a global collaboration and continues to recruit from the Cure SMA care centre network across the United States, incorporating more than 150 healthcare services. This Clinical Data Registry (SMA Registry) is central to the work of Cure SMA to improve standards of care and generate new knowledge to inform the community of the changing landscape and unmet needs of people living with SMA within the therapeutic era. As such it has been leveraged to provide pivotal epidemiological data on SMA in the United States, within an era where newborn, carrier and prenatal screening for SMA has been integrated as public health directives.7,30 The most recent initiative is the Cure SMA Newborn Screening registry, an online registry solely governed by Cure SMA, with the purpose of improving clinical care and supporting the development of new therapies.
Academic collaborations such as the international SMA consortium (iSMAc) have also formed registries for multicentre prospective natural history studies. This partnership of Italian, United States and United Kingdom researchers links the iSMAc registry with retrospective data from previous studies across the network. 31
The SMArtCARE registry is a joint initiative of 65 paediatric and adult neurology centres that has been operational since 2017 in the German speaking countries of Germany, Austria and Switzerland and collects clinician entered longitudinal data on all SMA patients, 25 to complement the German patient entered registry. The registry has been funded through industry partnerships; however, the academic SMArtCARE network owns the data sovereignty with all responsibilities for data protection, data analysis and the right to publish results of the study, with this balance mitigating the risk of conflicts of interest and issues of data integrity whilst ensuring feasibility and sustainability. Regular training of physiotherapists aimed at enhancing interrater reliability of functional assessments is a notable feature, particularly within the context of research evaluating functional outcomes.
A further model is the multinational and multicentre RESTORE observational registry, sponsored by Novartis Gene Therapies, Inc., the manufacturer of onasemnogene abeparvovec. It has partnered with existing registries such as iSMAC, TREAT-NMD, Cure SMA and SMArtCARE, and recruits de novo from SMA treatment centers, for transfer of data related to people with SMA treated with gene therapy. 32 Notably, registries such as RESTORE and SMArtCARE are designed to meet reporting requirements for European Medicines Agency (EMA), with focus on longitudinal safety data of DMTs. 33 The Biogen pregnancy registry, embedded within the SMArtCARE (Germany, Austria, Switzerland) and iSMAc registries (United Kingdom, United states, Italy), aims to assess safety and outcomes of nusinersen use in pregnant women with SMA, birth outcomes and adverse effects in infants born to participants with SMA exposed to nusinersen (NCT05789758). These post-authorisation requirements are key to adding to the current evidence base, identifying evolving safety signals and ensuring optimal benefit-risk balance, especially with the expansion of treatment into broader, and understudied populations, such as adults and pregnant women.
The outcomes and impacts of SMA registries: Using common data elements as a tool to serve consumers and facilitate knowledge and empowerment within the SMA community
The outputs from registries in the past decade have yielded insights into several aspects of SMA (Table 1). Registry participation has notably increased since the availability of disease-modifying therapies.21,51 The disease ranks among the top five most frequent neuromuscular diseases in certain regions.16,20,52 They have facilitated the observation of natural history in the age before treatments 37 and of shifted natural history in the therapeutic era,23,24 provided insights into drug effects and toxicities,39,42,43 and clinical characteristics,14,19,23,24 frequencies and patterns of comorbidities.6,14,19,24 Furthermore, registries have been leveraged to provide essential epidemiological and demographic information6,7,24,53 and determinants of disease such as SMN2 copy numbers, age and motor function 36 and sex. 38 The application of this information has highlighted the substantial impact of SMA and informed best practice care and delivery of healthcare services.
Insights and outcomes facilitated by SMA registries as model of proactive treatment paradigm.
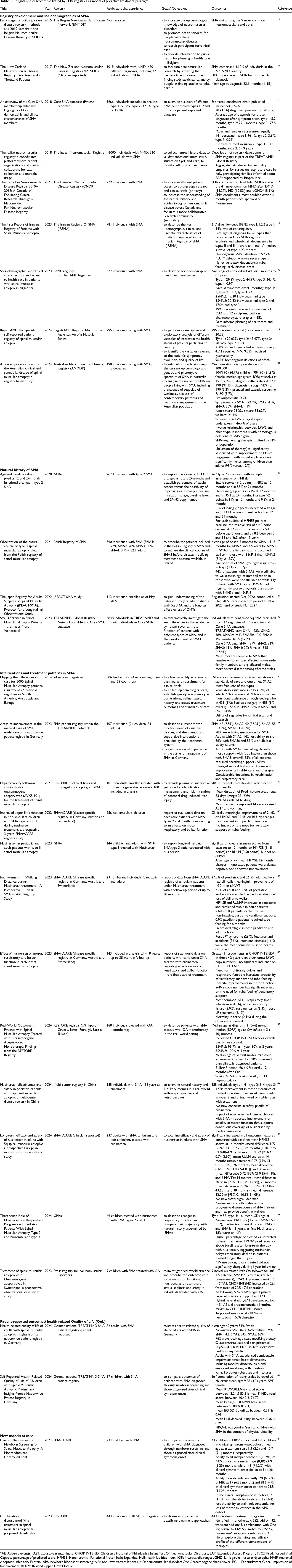
*AE: Adverse event(s); AST: aspartate transaminase; CHOP INTEND: Children's Hospital of Philadelphia Infant Test Of Neuromuscular Disorders; EAP: Expanded Access Program; FVC% Pred: Forced Vital Capacity percentage of predicted score; HFMSE: Hammersmith Functional Motor Scale Expanded; HUI: health Utilities Index; IQR: Interquartile range; LGMD: Limb girdle muscular dystrophy; NAIP: neuronal Apoptosis Inhibitory Protein; NBS: newborn bloodspot screening; NIV: non-invasive ventilation; NMD: neuromuscular disorder; OA: Onasemnogene abeparvovec; PGI-I: Patient/Parent Global Impression of Improvement; RULM: Revised Upper Limb Module.
Despite an international consensus for early treatment in children with 4SMN2 copies, 54 variability in practice abounds internationally due to the presence of evidence gaps associated with the optimal therapeutic window for this genotype. The SMArtCARE registry has leveraged large data sets to describe the natural history in 268 individuals with 4SMN2 copies highlighting the phenotypic heterogeneity, 55 with a median age at disease onset of 3.0 years and mean of 6.4 years, supporting a proactive approach to their management.
Whilst clinical trials provide clinical effectivity data for homogenous populations across discrete timelines, registry data has been invaluable in ascertaining durability of effect, safety and utility in heterogenous SMA populations. For example, the effectiveness and safety of nusinersen in 385 people less than 18 years at enrolment in China was demonstrated, supporting continuous coverage by medical insurance. 44 The RESTORE registry has demonstrated clinical trial findings of efficiency and safety in up to 168 children treated with onasemnogene abeparvovec-xioi, highlighting the durability of effect. 32 Registries have also been leveraged to understand the outcomes of combined or sequential treatment, bridging the data gap that exists due to the paucity of head to head clinical trials. The outcomes of these registry studies will inform therapeutic decision making and help to define sub-cohorts who would benefit from additional treatment, whilst also providing evidence for a changing classification system of intervention. 50
A further four studies from the SMArtCARE Registry report the positive effect of nusinersen on disease progression and survival in 143 individuals with early-onset spinal muscular atrophy (37), upper limb function in 256 non-ambulant children with SMA types 2 and 3 (44), walking distance in 231 ambulant individuals (38), and longer term (38 months) efficacy and safety in adults with SMA (45). Registry-based data has also included the description of clinical effectiveness of newborn screening (46), consolidating previous findings.
The value and capacity of registries to continue to apply knowledge is also demonstrated in the domain of quality of life and health economics. Pharmacoeconomic and quality of care outcomes, including healthcare utilization and caregiver burden were included in RESTORE, which analysed data from 385 individuals between 2018 and 2022, to provide 37 months of post-surveillance data, that assisted with regulatory approvals, and commercial interests. A cross-sectional study of 82 adults living with SMA in Germany showed considerable impairment across health dimensions, with variability across subgroups and measures. 48 Another study provided preliminary insights in 17 German children, suggesting health related quality of life was good, despite physical disability. 49 The economic burden of SMA, healthcare resource utilisation and direct medical costs, was reported to be significant following analyses of 290 SMA records within the Swedish national registry. 56
Taken together, this collective information has directly supported regulatory approvals in some jurisdictions 57 and informed the construct of consensus therapeutic guidelines based on SMN2 copy numbers and time to treatment.54,58 Registry data has provided vital evidence regarding efficacy and safety of disease modifying treatments,32,40,42,43 and survival. 14 Increasingly, patient reported outcomes including quality of life and functional independence measures are incorporated into datasets, to offer valuable insights into the impact of the condition within a biopsychosocial model of care. Registries have facilitated clinical trials by serving as valuable resources of recruitment and trial design.15,16,34,59 Furthermore, collaboration between registries has helped leverage larger sample sizes with enhanced statistical power, allowing for examination of disease variations amongst different populations.6,7
Future outputs from registries are expected to provide vital insights into treatment paradigms, evolving phenotypes and support in healthcare policies. For example, with focus on the emergent dynamics of an expanding population of adults (an under-represented population within the clinical trials realm), the Japan Registry (jREACT-SMA) has instigated a model to explore the natural history of SMA and the long-term effectiveness of disease modifying treatments. 21 Additionally, the Biogen clinical trials centre is undertaking a study in pregnant women with SMA treated with nusinersen within the SMArtCARE and International SMA Registries. Another expected output is comparison of patient-reported data with clinician-reported information, that would provide validity to patient-reported data (embedded in several national registries), while also highlighting any gaps in communication between the two groups. 30
Barriers, facilitators and future directions for SMA registries: Governance structures, sustainability and forging links with biobanks
Whilst progress is being made in the field of registry development, operational, ethical, legal, and social (ELSI) barriers continue to arise in the curation and use of collected data. Questions arise as to whether self-regulation of SMA registries is sufficient as they broaden in scope and purpose, or whether additional legislative safeguards need to be developed to ensure legal and ethical clarity on participation, data protection, access and commercialisation of intellectual properties generated through the use of data. 60
From an operational perspective, funding of registries for SMA is from diverse sources and integral to their implementability and sustainability. Funding sources include government grants, 51 fund-raising initiatives, 61 support from non-profit organisations and patient advocacy groups, 18 pharmaceutical industry, institutional resources, 15 research foundation trusts34,52 and grants. 20 Conflicts of interest within these funding streams pertaining to data ownership, access and use require strong governance frameworks.
Whilst cohesive data registries are ideal to inform key scientific and clinical evidence gaps, strategies to ensure the integrity of data storage, privacy and access to sensitive patient information is vital, to ensure that trust and acceptance of registries is maintained within SMA communities. In addition to how registry data is collected, reviewed and verified, the utility and applicability of registry data also depends on the quality of the data analysis plan and completeness of the dataset. The latter is often constrained by missing data due to incomplete acquisition, incomplete reporting, loss to follow-up, thus introducing bias and less robust analyses. Standard methods such as inverse probability weighting and multiple imputation are common strategies employed to address missing data and improve analytical robustness. 24 The utility can be further enhanced by incorporation of new elements as they emerge in post marketing surveillance or along the clinical course. This is exemplified by thrombotic microangiopathy (TMA) which was identified as a newly reported adverse event in infants treated with onasemnogene. 62 Its addition as a data element to registries will enable further assessment of its epidemiology, aetiology and outcomes.
Data privacy is a particularly pertinent issue in healthcare models where there is a strong privately funded or insurance-based component, with legal regulations key to safeguarding individuals against misuse of condition specific information. As a facilitator of this, registries have embraced a co-design and development approach, engaging a broad spectrum of stakeholders, including experts in Ethical, Legal, and Social issues (ELSI) principles and data management/governance, consumers and their representatives, and clinical and scientific professionals. This collaborative effort helps ensure that the risks and benefits of data sharing are evaluated from multiple perspectives, ensuring a comprehensive understanding of their impact. 63
Inclusivity of under-represented populations remains a significant research challenge, limiting generalisability of outcomes. Adults with SMA, who in some jurisdictions have a non-centralised model of management, individuals within culturally and linguistically diverse communities and those with variable health literacy are often not enrolled in research initiatives, including registry databases. Providing fair and transparent opportunities to be enrolled in registries is fundamental to improving representation. Bridging this gap, hub and spoke models are now being established to devolve recruitment strategies away from centralised (often research centres) (Cure SMA), to non-specialist and community environments to improve representation (TREAT NMD).
The data collected in SMA registries are often used for multiple purposes, across a spectrum of studies. Aligning with the changing landscape of SMA registries and their role, alternative consent models are being reviewed that fit the purpose of the defined registry. These include, but are not limited to, broad consent (individuals prospectively agree to their samples and information being used in any future research), tiered consent (individuals opt into specified areas of research, for example, biobank sample collection or future ethically approved research or sharing of data) and dynamic consent (digital platforms support preference setting in new or additional research).
Concomitant with consent processes, issues surrounding transparency of data collection, management and analysis are integral to providing robust outputs, that have the scientific rigor required to inform research driven care. Adhering to the principles defined for best practice in the formation of data registries ensures that the data is navigable, accessible, interoperable and reuseable. Whilst sharing of data and data availability to researchers is essential to facilitate effective clinical research, safeguards are mandatory to ensure the confidentiality of clinical information and maintain trust and continued public engagement in registry data collection. Data registries thus employ specific consenting processes in Iine with jurisdictional legal frameworks. The General Data Protection Regulation (GDPR) in Europe alongside country specific laws preserve data sovereignty from third parties including insurance and pharmaceutical companies. Emphasis on data security, privacy and purpose limitation, particularly for sharing with pharmaceutical companies, are common to laws within countries such as United States, Australia, Canada, Germany, Italy, etc,. For highly identifiable data such as genomic data in longitudinal registries, renewed consent for ongoing data collection is provided as an ethical safeguard. 64
There are both benefits and limitations to patient/surrogate parent and investigator collected data in registries. While patient data provides a unique, real-world perspective on lived experience and can thereby inform an evidence-base as a foundation to patient centred treatment and care, introduction of biases can occur. These include recall bias, non-specific data that does not reflect clinical change and over/under reporting. Investigator collected data, particularly conducted within a structured visit, can capture clinical measures objectively and report outcomes with improved accuracy and clinical relevance. However, this is resource-intensive, can impact longitudinal data collection in large populations of patients and preclude the collection of a wide range of patient experiences within daily life. Thus, adopting a hybrid approach, combining the patient/surrogate parent and investigator-collected data within registries, can facilitate a more comprehensive data capture, leveraging the strengths of each while mitigating their limitations.
Additionally, the SMA community must endeavour to support these processes by establishing the nomenclature and definitions used to define patient characteristics and disease status within a new treatment era65,66 and to hone research questions that are important to the community and that may generate improved health and psychosocial outcomes for affected patients and their families. Collection of data for registries should be adequately resourced and considered as ‘core business’ within clinical-research activities, to ensure their longevity and use over the lifetime of an individual.
Sustainability is a major issue for registries at multiple levels, including funding, exponential data collection and curation requirements and patient fatigue, especially with patient entered data. This may be facilitated in the future using machine learning and artificial intelligence (AI). Potential benefits include the ability to link large data sets, assist in stratification of individuals to guide targeted interventions and alter data sets to account for the rapidly evolving clinical and therapeutic landscape. However, the paucity of standardised validation methodology and heterogeneity of performance outcome reporting suggests that the application of AI to clinical registries is still in its infancy, limited by poor accessibility of registry data and reflecting the need for focussed expertise of domain experts in the future development of clinically deployable AI.
Data ownership historically has been with the registry sponsor, which, in the case of pharma-sponsored registries can create a point of attrition where the concept of data ownership is concerned. Where a drug/intervention does not meet preestablished endpoints, registries often close, precluding further access to valuable data that can inform stakeholders of the risks, benefits and implications of new and emerging interventions. Thus, a responsible sharing of data to advance scientific knowledge and benefit of the broader community represents a balance of proprietary interest with ethical imperative. Federated registries, which allow for decentralized data ownership, offer a model for selective sharing including data availability for research purposes but restricted for external commercial use.
Whilst individual registries grow rapidly, there is further scope to increase the power of real-world data by connecting data sets from various sources across a harmonised ecosystem. 67 One example is a deliberative move towards integration of data from biobanks, a step seen as pivotal in discovering novel biomarkers and defining surrogate endpoints for this condition. 63 A federated data ecosystem can further enable a virtual cohort in SMA, allowing for secure and streamlined access to repositories of data, with each jurisdiction adhering to its own consent processes, data security and privacy procedures, and legal requirements. In order to achieve this, multidisciplinary actions that leverage global infrastructure for rare disease are being developed. Examples include the development of streamlined electronic platforms to connect and integrate existing databases, registries, biobanks and clinical bioinformatics for conditions including but not limited to SMA, mitigating issues of incompatibility and variabilities in data collection, ensuring that core data sets are robust, cohesive and applicable to the population of interest. 68
Conclusions
Registries have evolved in parallel with the dynamic clinical landscape in SMA, supporting the approval and commercialisation of three orphan drugs and implementation of newborn screening. The new proactive paradigm of diagnosis and treatment has amplified heterogeneity in the condition, with different populations evident, and new phenotypes emerging. Registries are effectively positioned to generate new insights into the evolving needs of a treated population and fast-track real world data to inform contemporary standards of care and respond meaningfully to the priorities of people living with SMA.
As a rare condition, cross-border collaborations are vital to facilitate data pooling and harmonisation in SMA to answer key clinical questions and optimise the power of individual data repositories. Global guidelines for the development of data registries have provided a framework on which to start this process, however future work includes overcoming barriers related to data standardization, access, governance, storage and representation, sustainability and addition of data sets that reflect and are meaningful to affected individuals and their families including patient reported experiences and outcome measures. Flexibility of registries to capture the dynamic and rapid development of therapeutics and evolution of novel outcomes measures in SMA is also essential. This state-of-the-art review has identified SMA registries as an effective and essential research tool for knowledge generation, and optimisation of clinical outcomes for affected individuals, supporting the need for investment of resources for the inception and continuance of SMA registry networks globally.
Footnotes
Ethics approval and consent to participate
Not applicable.
Author contributions
Lakshmi Balaji: Conceptualization; Literature search, Writing – original draft; Writing – review & editing.
Didu Kariyawasam: Conceptualization; Writing – review & editing.
Michelle Farrar: Conceptualization; Writing – review & editing.
Eppie M Yiu: Writing – review & editing.
Statements and declarations
MAF is a site principal investigator for Biogen, Roche and Novartis Gene Therapies, Inc., clinical trials, and the institution has received funds for contract research related to the conduct of these trials. MAF has received honoraria for advisory board participation and participation in educational events from Biogen, Novartis, and Roche, speaker's fees from Biogen and Novartis and serves as a medical director on the board of Muscular Dystrophy NSW (not-for-profit). EMY is the principal investigator of the Australian Neuromuscular Disease Registry (ANMDR) which has received research support from TREAT-NMD, MD-NSW, The Daniel Ferguson Foundation, Save our Sons Duchenne Foundation, the Western Australian government, Biogen, Roche, PTC therapeutics and Pfizer. EMY is a site principal or sub-investigator for Biogen and Novartis clinical trials, and has received honoraria for advisory board participation from Biogen and Roche, and speaker fees and/or honoraria paid to their institution from Biogen and Roche for advisory board participation. DK has received honoraria for advisory board participation from Biogen and speaker's fees from Biogen, Novartis and Roche.
Funding
LB acknowledges the Australian Government's Research Training Program (RTP) Scholarship. MAF and DK acknowledge support by the National Health and Medical Research Council under Grant numbers 1194940 and 2026317, respectively.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
