Abstract

In an era when “doing more with less” has become a way of life for clinical laboratories it is urgent that all opportunities to realize economics of scale and scope be implemented.
CLS is a large independent clinical laboratory in New York City which performs an extensive menu of serological tests using microtiter plate assays (MPAs), immunofluorescent assays (IFAs) and a flocculation tests (FT) for syphilis screening. In mid-1997 in an effort to reduce personnel requirements while maintaining test turn-around time (TAT), CLS began implementation of an automated MPA processing station. The key requirement for such a system in the clinical laboratory setting is the capability of performing MPAs with varying protocols during the course of a shift. This requirement limited the possible choice of systems. CLS eventually selected the Omni (Bio Tek; Winooski, VT). This system uses an internal robot and track configuration to automate the washing/dispensing/incubation process for bar-coded microtiter plates and strips presented to the instrument in plate carriers of uniform dimension. The instrument can accommodate 3 different wash buffers and reagents for up to 10 assays. The software is capable of storing 99 distinct protocols for the wash/dispense/incubation cycle. Calibration data is fitted to one of 20 possible functions (the specific choice is defined by the assay manufacturer) and, when required, test results are blanked against a well filled with diluent. At CLS, the Omni is linked to an autopipettor with barcode reading capabilities (Packard; Downers Grove, IL) and connected to the laboratory information system [LIS] (Cerner; Kansas City, MO). Work list and plate map data are downloaded to the Omni and patient results transmitted to the LIS. Other than the loading of patient specimen tubes, reagents and wash solution, the only manual intervention required is the removal of the plate from the autopipettor and its placement in the Omni. The possibility of using a robotic arm for the latter function is currently being investigated. Although throughput depends on assay mix, at CLS an output of 20 microtiter plates (96 wells) per 8-hour shift can be achieved.
Over the course of the ensuing twelve months, CLS moved 23 assays (refer to Table 1) to the automated system. Total volume for these assays in 1997 was 182, 225 billable test results. These included procedures previously performed using IFA and FT as well as MPA. Two workstations were eliminated and total personnel requirements for these assays were reduced by 50% (from 4.6 to 2.3 full time employees) without deterioration of TAT.
ASSAYS ON THE OMNI SYSTEM

Interfacing of the Omni system to the LIS reduced the possibility of transcription error and made possible successful implementation of testing algorithms for Epstein-Barr virus, syphilis and autoimmune serology. The projected pay-back period, using a cost accounting system developed at CLS, was estimated at 1.6 – 2.1 years. Ancillary but real advantages from this transition include simplification of reagent delivery schedules and inventory needs, and reduction of cross-training requirements in the Immunology department.
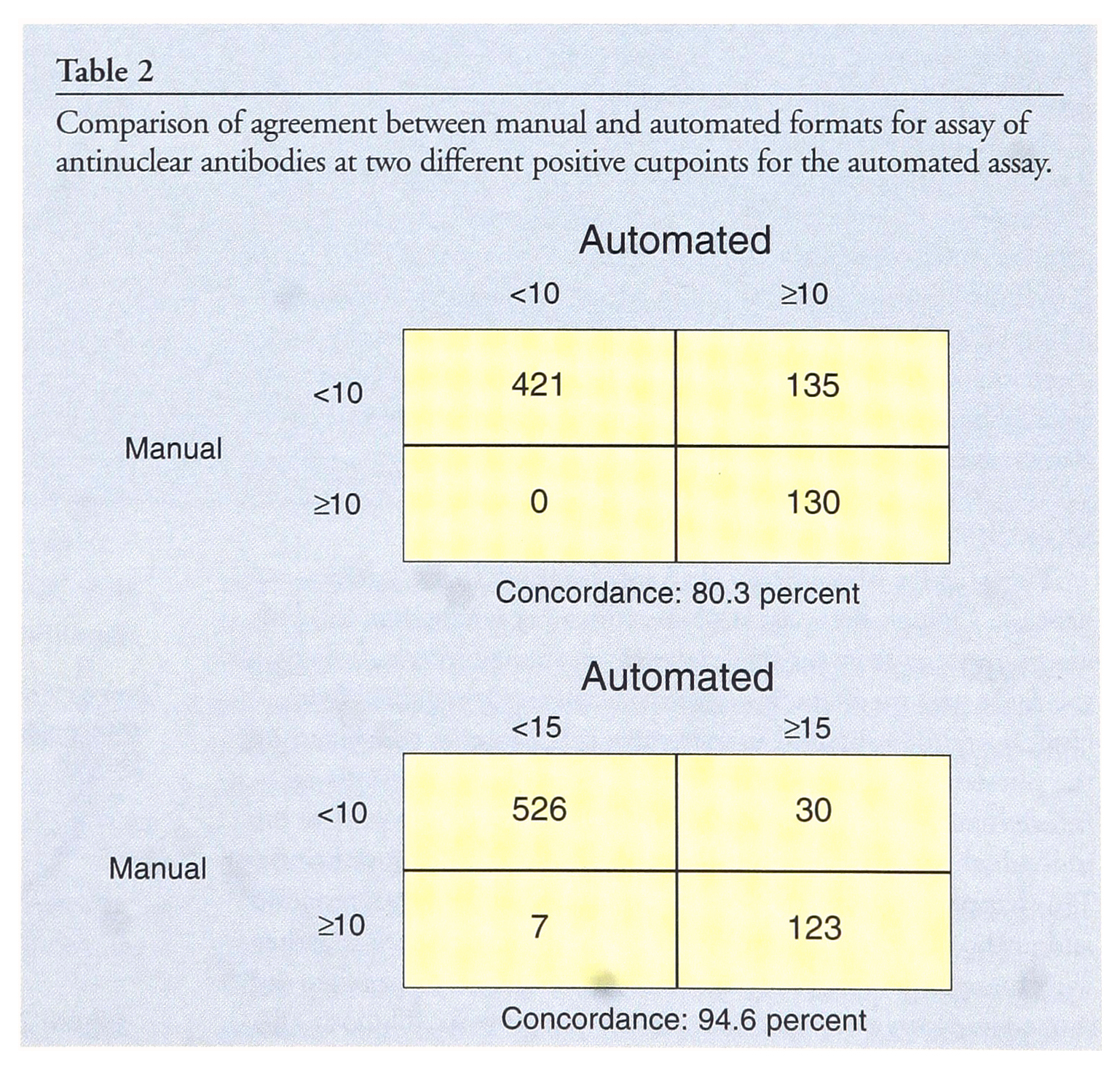
Potential users however, should be cautioned that manufacturers' recommended reference ranges and negative/positive cut points are frequently based on manual techniques and may require re-investigation when an automated approach is put into place. At CLS, this was the case with the assay for anti-nuclear antibody (Incstar; Stillwater, MN), When both the manual and automated assay were performed according to manufacturers' instructions, there was a considerably higher positive rate when the ANA assay was performed in an automated format. In a run of 686 consecutive specimens, 265 were positive (38.6%). This, in our experience, was more than twice the usual positive rate when the assay was run manually. When the 265 automation positive specimens were run manually, only 130 were positive. Of the 265 automation positive specimens, 105 were also confirmed positive for antibodies against an 8-antigen panel (Incstar). These antibodies are considered significant for the diagnosis of specific autoimmune disorders. Of these 105, there were 83 among the 130 ANA positive specimens when the latter assay was performed in the manual format. Studies (n=2) in which 3 high positive specimens were placed in-between 2 negative specimens ruled out specimen carryover. Although the solution containing the second (enzyme labeled) antibody was viscous, reagent carryover was eliminated by incorporating a detergent wash step. Also, there was no spatial pattern of higher readings suggesting a carryover problem. A study in which microtiter plates were processed manually and in the Omni after pipetting indicated higher results for the latter suggesting that the problem was in the Omni. We found that temperature readings inside the ambient temperature incubation chamber in the Omni however, were consistently 3°C higher than temperature readings in the laboratory (confirmed by the manufacturer). This was apparently a consequence of heat generated by the instrument. Assuming activation energy of 11.1 kcal/mole for the HRP catalyzed oxidation of TMB (1) and using the Arrhenius equation which relates reaction rate to temperature, a 21.2 percent increase in rate constant at the higher temperature is predicted. This possibly accounts for the increase in the number of positive ANA results. CLS resolved this problem by increasing the value of the assay result required to define a positive outcome (refer to Table 2).
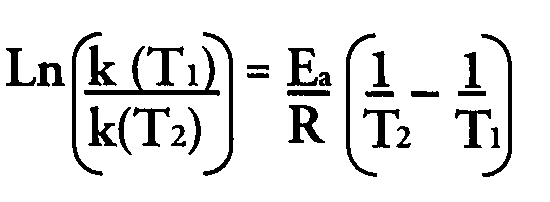
Comparison of agreement between manual and automated formats for assay of antinuclear antibodies at two different positive cutpoints for the automated assay.
Economists recognize various means by which enhanced productivity and decreased costs can be realized (2,3). The ability to transfer 23 assays performed at three workstations using different methodologies to one automated workstation realized significant “economies of scope” and “economies of scale”. Specifically, several mechanisms appear to be operative in explaining the re-instrumentation results obtained at CLS. Among these are included workstation consolidation, learning curve effects, volume discounts on immunoassay reagents, simplification of delivery schedules and inventory needs, and reduction of cross-training requirements in the immunoassay section.
The results from the CLS reconfiguration in its immunoassay area strongly indicate that substantial cost savings can be achieved through appropriate re-instrumentation. This conclusion has, in our view, wide-ranging implications with respect to both the organization of clinical laboratory testing (e.g., central versus hospital laboratories for non-STAT testing) and the training of those responsible for making choice-of-technology decisions. With respect to the former issue, the CLS experience suggests that “core” laboratories that can bring together a larger volume of tests would be more likely to enjoy the types of economies described above. Such facilities also would be able to distribute fixed costs among a larger volume of billable results. If the CLS experience has any larger relevance, it would be expected that hospitals would move toward outsourcing their non-STAT laboratory work to higher volume facilities.
It is not our purpose to discuss at any length the implications of the CLS redesign of its immunoassay area for the training of future laboratorians. The CLS experience does however suggest that those responsible for assessment of new technology in the clinical laboratory must be able to evaluate multiple technical, financial, and medical necessity issues.

